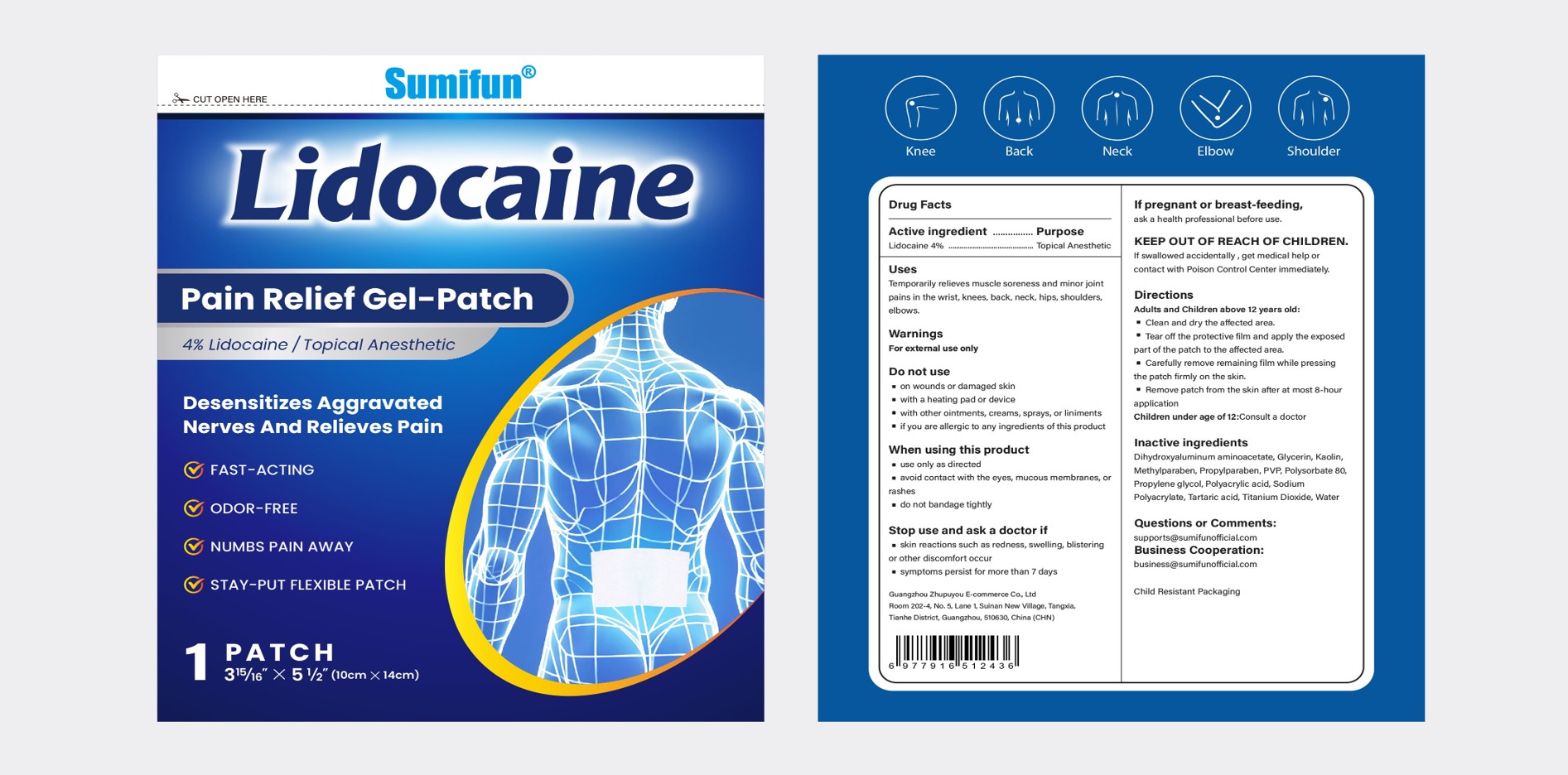
lidocaine pain relief gel-patch
296fcc36-563f-19ca-e063-6394a90ab3ae
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine Pain Relief Gel-Patch
Medication Information
Warnings and Precautions
For extemal use only
Active Ingredient
Lidocaine 4%
Indications and Usage
Temporarily relieves muscle soreness and minor jointpains in the wrist, knees, back, neck, hips, shoulders.elbows.
Dosage and Administration
Adults and Children above 12 years old:
Clean and dry the affected area.
Tear off the protective film and apply the exposed part of the patch to the affected area.
Carefully remove remaining film while pressing the patch firmly on the skin
Remove patch from the skin after at most 8-hour application
Children under age of 12:Consult a doctor
Description
Lidocaine Pain Relief Gel-Patch
Section 42229-5
Lidocaine Pain Relief Gel-Patch
Section 50565-1
lf swallowed accidentally , get medical help orcontact with Poison Control Center immediately.
Section 50566-9
skin reactions such as redness,swelling, blistering or other discomfort occur
symptoms persist for more than 7 days
Section 50567-7
use only as directed
avoid contact with the eyes, mucous membranes, or rashes
do not bandage tightly
Section 50570-1
on wounds or damaged skin
with a heating pad or device
with other ointments, creams, sprays, or liniments
if you are allergic to any ingredients of this product
Section 51727-6
Glycerin
Sodium Polyacrylate
Water
Methylparaben
Propylparaben
Dihydroxyaluminum aminoacetate
Tartaric acid
PVP
Polysorbate 80
Kaolin
Titanium Dioxide
Propylene glycol
Polyacrylic acid
Section 51945-4
Section 55105-1
Topical Anesthetic
Structured Label Content
Warnings and Precautions (34071-1)
For extemal use only
Active Ingredient (55106-9)
Lidocaine 4%
Indications and Usage (34067-9)
Temporarily relieves muscle soreness and minor jointpains in the wrist, knees, back, neck, hips, shoulders.elbows.
Dosage and Administration (34068-7)
Adults and Children above 12 years old:
Clean and dry the affected area.
Tear off the protective film and apply the exposed part of the patch to the affected area.
Carefully remove remaining film while pressing the patch firmly on the skin
Remove patch from the skin after at most 8-hour application
Children under age of 12:Consult a doctor
Section 42229-5 (42229-5)
Lidocaine Pain Relief Gel-Patch
Section 50565-1 (50565-1)
lf swallowed accidentally , get medical help orcontact with Poison Control Center immediately.
Section 50566-9 (50566-9)
skin reactions such as redness,swelling, blistering or other discomfort occur
symptoms persist for more than 7 days
Section 50567-7 (50567-7)
use only as directed
avoid contact with the eyes, mucous membranes, or rashes
do not bandage tightly
Section 50570-1 (50570-1)
on wounds or damaged skin
with a heating pad or device
with other ointments, creams, sprays, or liniments
if you are allergic to any ingredients of this product
Section 51727-6 (51727-6)
Glycerin
Sodium Polyacrylate
Water
Methylparaben
Propylparaben
Dihydroxyaluminum aminoacetate
Tartaric acid
PVP
Polysorbate 80
Kaolin
Titanium Dioxide
Propylene glycol
Polyacrylic acid
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Topical Anesthetic
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:21.047744 · Updated: 2026-03-14T23:07:01.452935