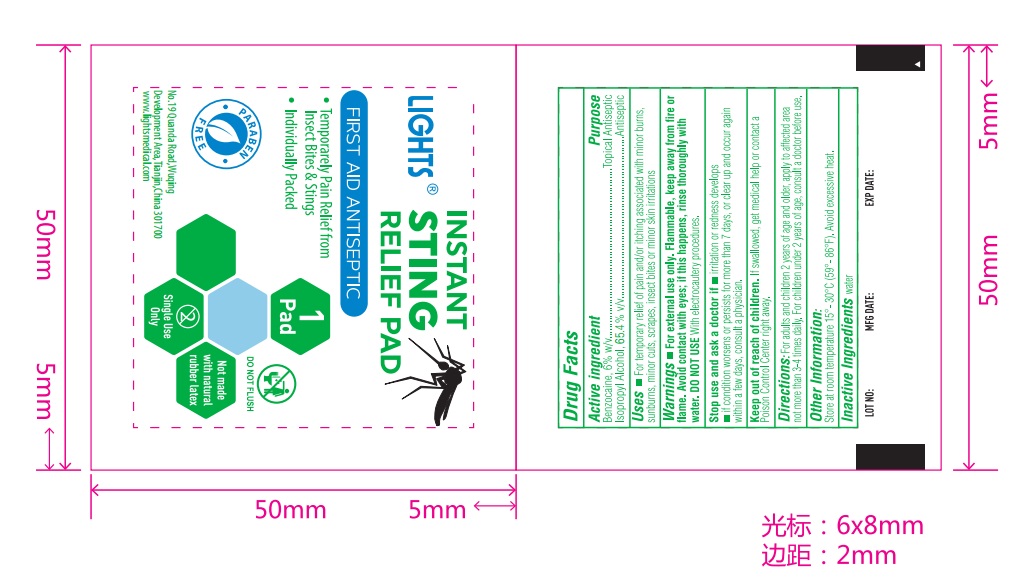
61333-215 light instant sting relief pad
28dd82a7-f360-a022-e063-6394a90a3a76
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Topical Anesthetic Antiseptic
Description
Benzocaine 6% w/v Isopropyl Alcohol: 65.4% v/v
Medication Information
Warnings and Precautions
Warnings:
For external use only. Flammable, keep away from fire or flame. Avoid contact with eyes, if this happens, rinse throughly with water. DO NOT USE with electrocautery procedures.
Indications and Usage
Uses: For temporary relief of pain and itching associated with minor burns, sunburns, minor cut, scrapes, insect bites, or minor skin irritations
Purpose
Topical Anesthetic
Antiseptic
Directions
For adults and children 2 yeas of age or older: Apply to affected area not more than 3-4 times daily
Children under 2 yeas of age: Consult a doctor before use
Other Information
Store at room temperature 15-30 C (59-86F). Avoid excessive heat.
Description
Benzocaine 6% w/v Isopropyl Alcohol: 65.4% v/v
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away
Section 50566-9
Stop use and ask a doctor if
- irritation or redness develops
- if condition worsens or persists for more than 7 days, or clear up and occur again within a few days, consult a physician
Section 51727-6
Inactive Ingredient
Purified Water
Section 51945-4
Active Ingredient
Benzocaine 6% w/v
Isopropyl Alcohol: 65.4% v/v
Structured Label Content
Warnings and Precautions (34071-1)
Warnings:
For external use only. Flammable, keep away from fire or flame. Avoid contact with eyes, if this happens, rinse throughly with water. DO NOT USE with electrocautery procedures.
Indications and Usage (34067-9)
Uses: For temporary relief of pain and itching associated with minor burns, sunburns, minor cut, scrapes, insect bites, or minor skin irritations
Purpose
Topical Anesthetic
Antiseptic
Directions
For adults and children 2 yeas of age or older: Apply to affected area not more than 3-4 times daily
Children under 2 yeas of age: Consult a doctor before use
Other Information
Store at room temperature 15-30 C (59-86F). Avoid excessive heat.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- irritation or redness develops
- if condition worsens or persists for more than 7 days, or clear up and occur again within a few days, consult a physician
Section 51727-6 (51727-6)
Inactive Ingredient
Purified Water
Section 51945-4 (51945-4)
Active Ingredient
Benzocaine 6% w/v
Isopropyl Alcohol: 65.4% v/v
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:20.321984 · Updated: 2026-03-14T23:12:32.924893