These Highlights Do Not Include All The Information Needed To Use Twyneo Safely And Effectively. See Full Prescribing Information For Twyneo.
27208dff-e376-4c18-b56e-0a260f685a39
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
TWYNEO is indicated for the topical treatment of acne vulgaris in adults and pediatric patients 9 years of age and older.
Indications and Usage
TWYNEO is indicated for the topical treatment of acne vulgaris in adults and pediatric patients 9 years of age and older.
Dosage and Administration
Apply a thin layer of TWYNEO to the affected areas once daily on clean and dry skin. Avoid contact with the eyes, lips, paranasal creases, and mucous membranes. Wash hands after application. TWYNEO is for topical use only. TWYNEO is not for oral, ophthalmic, or intravaginal use.
Warnings and Precautions
Hypersensitivity: Severe hypersensitivity reactions, including anaphylaxis and angioedema, have been reported with use of benzoyl peroxide products. ( 4 , 5.1 ) Skin Irritation: Pain, dryness, exfoliation, erythema, and irritation may occur with use of TWYNEO. Avoid application of TWYNEO to cuts abrasions, eczematous or sunburned skin. ( 5.2 ) Photosensitivity : Minimize unprotected exposure to sunlight and sunlamps. Use sunscreen and protective clothing when sun exposure cannot be avoided. ( 5.3 )
Contraindications
TWYNEO is contraindicated in patients with a history of hypersensitivity reaction to benzoyl peroxide or any components of TWYNEO [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The most common adverse reactions (incidence ≥ 1%) are pain, dryness, exfoliation erythema, dermatitis, pruritus and irritation (all at the application site). ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Galderma Laboratories, L.P. at 1-866-735-4137 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
How Supplied TWYNEO (tretinoin and benzoyl peroxide) cream, 0.1%/3% is a yellow cream and is supplied as:
How Supplied
How Supplied TWYNEO (tretinoin and benzoyl peroxide) cream, 0.1%/3% is a yellow cream and is supplied as:
Medication Information
Warnings and Precautions
Hypersensitivity: Severe hypersensitivity reactions, including anaphylaxis and angioedema, have been reported with use of benzoyl peroxide products. ( 4 , 5.1 ) Skin Irritation: Pain, dryness, exfoliation, erythema, and irritation may occur with use of TWYNEO. Avoid application of TWYNEO to cuts abrasions, eczematous or sunburned skin. ( 5.2 ) Photosensitivity : Minimize unprotected exposure to sunlight and sunlamps. Use sunscreen and protective clothing when sun exposure cannot be avoided. ( 5.3 )
Indications and Usage
TWYNEO is indicated for the topical treatment of acne vulgaris in adults and pediatric patients 9 years of age and older.
Dosage and Administration
Apply a thin layer of TWYNEO to the affected areas once daily on clean and dry skin. Avoid contact with the eyes, lips, paranasal creases, and mucous membranes. Wash hands after application. TWYNEO is for topical use only. TWYNEO is not for oral, ophthalmic, or intravaginal use.
Contraindications
TWYNEO is contraindicated in patients with a history of hypersensitivity reaction to benzoyl peroxide or any components of TWYNEO [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The most common adverse reactions (incidence ≥ 1%) are pain, dryness, exfoliation erythema, dermatitis, pruritus and irritation (all at the application site). ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Galderma Laboratories, L.P. at 1-866-735-4137 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
How Supplied TWYNEO (tretinoin and benzoyl peroxide) cream, 0.1%/3% is a yellow cream and is supplied as:
How Supplied
How Supplied TWYNEO (tretinoin and benzoyl peroxide) cream, 0.1%/3% is a yellow cream and is supplied as:
Description
TWYNEO is indicated for the topical treatment of acne vulgaris in adults and pediatric patients 9 years of age and older.
Section 42229-5
Risk Summary
Available data from published observational studies of topical tretinoin in pregnant women have not established a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. Studies conducted with topical benzoyl peroxide have not demonstrated systemic absorption and maternal use is not expected to result in fetal exposure to benzoyl peroxide. There are no data on TWYNEO use in pregnant women.
There are reports of major birth defects reported with maternal use of topical tretinoin similar to those seen in infants exposed to oral retinoids, but these case reports do not establish a pattern or association with tretinoin-related embryopathy (see Data
).
Animal reproductive studies have not been conducted with TWYNEO or benzoyl peroxide. Topical administration of tretinoin to pregnant rats during organogenesis was associated with malformations (craniofacial abnormalities [hydrocephaly], asymmetrical thyroids, variations in ossification, and increased supernumerary ribs) at doses greater than 1 mg tretinoin/kg/day, approximately 5 times the maximum recommended human dose (MRHD) based on body surface area (BSA) comparison and assuming 100% absorption. Oral administration of tretinoin to pregnant cynomolgus monkeys during organogenesis was associated with malformations at 10 mg/kg/day (approximately 100 times the MRHD based on BSA comparison and assuming 100% absorption) (see Data
).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of major birth defects, loss and other adverse outcomes. The background risk in the U.S. general population of major birth defects is 2 to 4% of miscarriage is 15 to 20% of clinically recognized pregnancies.
Section 44425-7
- 30-gram bottle with a pump, NDC 0299-5945-30
Storage and Handling
- Prior to Dispensing: Store TWYNEO between 2ºC to 8ºC (36ºF to 46ºF) until dispensed to the patient.
-
After Dispensing: Store TWYNEO at room temperature between 20ºC to 25ºC (68ºF to 77ºF). Discard 12 weeks after date of dispensing or 60 days after first opening, whichever is sooner.
- Do not freeze.
11 Description
TWYNEO (tretinoin and benzoyl peroxide) cream is a yellow cream for topical use. Each gram of TWYNEO contains 1 mg (0.1%) of tretinoin and 30 mg (3%) of benzoyl peroxide. Tretinoin is a retinoid and benzoyl peroxide is an oxidizing agent.
The chemical name for tretinoin is all-trans-retinoic acid, also known as (all-E)-3,7-dimethyl-9-(2,6,6-trimethyl-1-cyclohexen-1-yl)-2,4,6,8-nonatetraenoic acid. Tretinoin has the following structural formula:
Molecular Formula: C20H28O2 Molecular Weight: 300.44
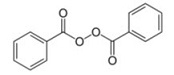
The chemical name for benzoyl peroxide is benzoyl benzenecarboperoxoate. Benzoyl peroxide has the following structural formula:
Molecular Formula: C14H10O4 Molecular Weight: 242.23
The formulation of TWYNEO uses silica (silicon dioxide) core shell structures to separately micro-encapsulate tretinoin crystals and benzoyl peroxide crystals enabling inclusion of the two active ingredients in the. TWYNEO contains the following inactive ingredients: anhydrous citric acid, butylated hydroxytoluene, carbomer homopolymer type C, cetrimonium chloride, cetyl alcohol, cyclomethicone, edetate disodium, glycerin, hydrochloric acid, imidurea, (S)lactic acid, macrogol stearate, mono and di-glycerides, polyquaternium-7, purified water, silicon dioxide, sodium hydroxide, squalane, tetraethyl ortho silicate and white wax.
8.4 Pediatric Use
The safety and effectiveness of TWYNEO for the topical treatment of acne vulgaris have been established in pediatric patients 9 years of age and older based on evidence from two multicenter, randomized, double-blind, parallel-group, vehicle-controlled, 12-week clinical trials and an open-label pharmacokinetic study. A total of 283 pediatric subjects 9 years of age and older received TWYNEO in the clinical studies [see Clinical Pharmacology (12.3) and Clinical Studies (14) ].
The safety and effectiveness of TWYNEO in pediatric patients below 9 years of age have not been established.
8.5 Geriatric Use
Clinical trials of TWYNEO did not include sufficient numbers of subjects 65 years of age and older to determine whether they respond differently than younger subjects.
14 Clinical Studies
The safety and efficacy of TWYNEO was evaluated in the treatment of acne vulgaris in two multicenter, randomized, double-blind, vehicle-controlled trials [Trial 1 (NCT03761784), Trial 2 (NCT03761810)], which were identical in design. The trials were conducted in 858 subjects 9 years of age and older, with facial acne vulgaris who were treated once daily for 12 weeks with either TWYNEO or vehicle.
Subjects were required to have a score of moderate (3) or severe (4) on the Investigator Global Assessment (IGA), 20 to 100 inflammatory lesions (papules, pustules and nodules), 30 to 150 non-inflammatory lesions (open and closed comedones) and two or fewer facial nodules.
Overall, 73% of subjects were White and 59% were female. Eighteen (18) (2%) subjects were 9 to 11 years of age, 370 (43%) subjects were 12 to 17 years of age, and 470 (55%) subjects were 18 years of age or older. At baseline, subjects had a mean inflammatory lesion count of 30.7 and a mean noninflammatory lesion count of 46.4. Additionally, 91% of subjects had an IGA score of 3 ("moderate").
The co-primary efficacy endpoints were the absolute change from baseline in non-inflammatory lesion count, and absolute change in inflammatory lesion count at Week 12 and the proportion of subjects with IGA success at Week 12, defined as an IGA score of 0 ("clear") or 1 ("almost clear"), and at least a two-grade improvement (decrease) from baseline at Week 12. The efficacy results are provided in Table 4.
| Trial 1 | Trial 2 | |||
|
TWYNEO (N = 281) |
Vehicle N = 143) |
TWYNEO (N = 290) |
Vehicle (N = 144) |
|
| IGA Success* | 33.9% | 14.3% | 26.8% | 15.1% |
| Difference from Vehicle | 25.7% | 11.6% | ||
| (95% CI) | (17.1%, 34.2%) | (3.6%, 19.7%) | ||
| Inflammatory Lesions |
|
|
|
|
| Mean‡ Absolute Change from Baseline | -21.6 | -14.8 | -16.2 | -14.1 |
| Difference from Vehicle | -6.8 | -2.1 | ||
| (95% CI) | (-9.1, -4.6) | (-3.9, -0.4) | ||
| Mean‡ Percent Change from Baseline | -66.1% | -43.5% | -57.6% | -50.8% |
| Difference from Vehicle | -22.6% | -6.8% | ||
| (95% CI) | (-29.2%, -16.0%) | (-13.1%, -0.5%) | ||
| Non-Inflammatory Lesions | ||||
| Mean‡ Absolute Change from Baseline | -29.7 | -19.8 | -24.2 | -17.4 |
| Difference from Vehicle | -9.9 | -6.8 | ||
| (95% CI) | (-13.0, -6.8) | (-9.9, -3.7) | ||
| Mean‡ Percent Change from Baseline | -61.6% | -40.9% | -54.4% | -41.5% |
| Difference from Vehicle | -20.7% | -13.0% | ||
| (95% CI) | (-27.2%, -14.2%) | (-19.6%, -6.4%) |
*Investigator Global Assessment (IGA) success was defined as an IGA score of 0 ("clear") or1 ("almost clear") with at least at two-grade reduction from baseline.
‡ Means presented in table are Least Square (LS) Means.
4 Contraindications
TWYNEO is contraindicated in patients with a history of hypersensitivity reaction to benzoyl peroxide or any components of TWYNEO [see Warnings and Precautions (5.1)].
5.2 Skin Irritation
Patients using TWYNEO may experience application site dryness, pain, exfoliation, erythema, dermatitis, pruritis, and irritation [see Adverse Reactions (6.1) ]. Depending upon the severity of these adverse reactions, instruct patients to use a moisturizer, reduce the frequency of the application of TWYNEO, or discontinue use. Avoid application of TWYNEO to cuts, abrasions, eczematous, or sunburned skin.
6 Adverse Reactions
The most common adverse reactions (incidence ≥ 1%) are pain, dryness, exfoliation erythema, dermatitis, pruritus and irritation (all at the application site). (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Galderma Laboratories, L.P. at 1-866-735-4137 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
5.1 Hypersensitivity
Hypersensitivity reactions, including anaphylaxis, angioedema, and urticaria, have been reported with the use of benzoyl peroxide products. If a serious hypersensitivity reaction occurs, discontinue TWYNEO immediately and initiate appropriate therapy.
12.2 Pharmacodynamics
Pharmacodynamics of TWYNEO in the treatment of acne vulgaris are unknown.
12.3 Pharmacokinetics
The systemic exposure of benzoyl peroxide was not assessed. Benzoyl peroxide is absorbed by skin where it is converted to benzoic acid and eliminated in the urine.
Plasma concentrations of tretinoin and its major metabolites were evaluated in 35 subjects in an open-label, randomized, pharmacokinetic (PK) study. Subjects 9 years of age and older with acne vulgaris applied a mean dose of 1.9 g TWYNEO to the skin of the face, shoulders, upper back and upper chest once daily for 14 days.
Steady-state PK characteristics were determined from samples drawn on Day 14. The mean baseline corrected Cmax and AUC0-24 of tretinoin and its metabolites after once daily application of TWYNEO for 14 days are provided in Table 3. No detectable levels of the metabolites all-trans 4-keto retinoic acid and 9-cis retinoid acid were found in subjects treated with TWYNEO.
| Age Group (years) | n | Compound | Mean(± SD) Cmax (ng/mL) |
Mean(± SD) AUC0-24 (ng-h/mL) |
| ≥ 18 years of age | 12 | tretinoin | 0.15 ± 0.17 | 0.63 ± 0.95 |
| 4-keto 13-cis RA | 0.27 ± 0.29 | 2.88 ± 3.61 | ||
| 13-cis RA | 0.21 ± 0.19 | 1.99 ± 2.90 | ||
| 12 to 17 | 15 | tretinoin | 0.19 ± 0.18 | 1.56 ± 1.97 |
|
|
4-keto 13-cis RA | 0.32 ± 0.28 | 2.39 ± 3.05 | |
| 13-cis RA | 0.28 ± 0.35 | 1.79 ± 2.79 | ||
| 9 to 11 | 8 | tretinoin | 0.18 ± 0.22 | 2.06 ± 3.96 |
|
|
4-keto 13-cis RA | 0.34 ± 0.36 | 2.89 ± 3.17 | |
| 13-cis RA | 0.13 ± 0.09 | 0.96 ± 1.36 |
5.3 Photosensitivity
TWYNEO may increase sensitivity to ultraviolet light. Minimize or avoid exposure to natural or artificial sunlight (tanning beds or UVA/B treatment) while using TWYNEO. Instruct patients to implement sun protection measures (e.g., sunscreen and loose-fitting clothes) when sun exposure cannot be avoided. Discontinue TWYNEO at the first evidence of sunburn.
1 Indications and Usage
TWYNEO is indicated for the topical treatment of acne vulgaris in adults and pediatric patients 9 years of age and older.
12.1 Mechanism of Action
Benzoyl peroxide is an oxidizing agent with bactericidal and keratolytic effects, but the precise mechanism of action is unknown. Tretinoin is a metabolite of vitamin A that binds with high affinity to specific retinoic acid receptors located in both the cytosol and nucleus. Tretinoin activates three members of the retinoic acid (RAR) nuclear receptors (RARα , RARß, RARγ) which act to modify gene expression, subsequent protein synthesis, and epithelial cell growth and differentiation. It has not been established whether the clinical effects of tretinoin are mediated through activation of retinoic acid receptors, and/or other mechanisms.
Although the exact mode of action of tretinoin in acne treatment is unknown, current evidence suggests that topical tretinoin decreases cohesiveness of follicular epithelial cells with decreased microcomedo formation. Additionally, tretinoin stimulates mitotic activity and increased turnover of follicular epithelial cells causing extrusion of the comedones.
5 Warnings and Precautions
- Hypersensitivity: Severe hypersensitivity reactions, including anaphylaxis and angioedema, have been reported with use of benzoyl peroxide products. (4, 5.1)
- Skin Irritation: Pain, dryness, exfoliation, erythema, and irritation may occur with use of TWYNEO. Avoid application of TWYNEO to cuts abrasions, eczematous or sunburned skin. (5.2)
- Photosensitivity: Minimize unprotected exposure to sunlight and sunlamps. Use sunscreen and protective clothing when sun exposure cannot be avoided. (5.3)
2 Dosage and Administration
- Apply a thin layer of TWYNEO to the affected areas once daily on clean and dry skin. Avoid contact with the eyes, lips, paranasal creases, and mucous membranes.
- Wash hands after application.
- TWYNEO is for topical use only. TWYNEO is not for oral, ophthalmic, or intravaginal use.
3 Dosage Forms and Strengths
Cream, 0.1%/3%: Each gram of TWYNEO contains 1mg (0.1%) of tretinoin and 30 mg (3%) of benzoyl peroxide in a yellow cream in a 30-gram bottle with a pump.
6.2 Postmarketing Experience
The following adverse reactions have been identified during use of benzoyl peroxide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Anaphylaxis, angioedema and urticaria
6.1 Clinical Trials Experience
The following adverse reactions are discussed in greater detail elsewhere in the labeling:
- Hypersensitivity [see Warnings and Precautions (5.1)]
- Skin Irritation [see Warnings and Precautions (5.2)]
Because clinical trials are conducted under widely varying conditions, adverse reaction rates are observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In two multicenter, randomized, double-blind, vehicle-controlled trials (Trial 1 and 2), 832 subjects 9 years of age and older with facial acne vulgaris applied TWYNEO (N=555) or vehicle (N=277) daily for 12 weeks. The majority of subjects were White (73%) and female (59%). Approximately 33% were Hispanic/Latino, and 46% were younger than 18 years of age. Adverse reactions reported in ≥ 1.0% of subjects treated with TWYNEO (and for which the rate exceeded the rate for vehicle), as well as the corresponding rates reported in subjects treated with vehicle are presented in Table 1.
|
|
TWYNEO Cream (N = 555) n (%) |
Vehicle Cream (N = 277) n (%) |
|---|---|---|
| Application Site Pain* | 59 (10.6) | 1 (0.4) |
| Application Site Dryness | 27 (4.9) | 1 (0.4) |
| Application Site Exfoliation | 23 (4.1) | 0 |
| Application Site Erythema | 22 (4.0) | 0 |
| Application Site Dermatitis | 7 (1.3) | 1 (0.4) |
| Application Site Pruritus | 7 (1.3) | 0 |
| Application Site Irritation | 6 (1.1) | 1 (0.4) |
* Application site pain defined as application site stinging, burning or pain.
Local tolerability evaluations were conducted at each study visit in the clinical trial by assessment of erythema, scaling, pigmentation, dryness, itching, burning and stinging. Table 2 presents the active assessment of the signs and symptoms of local facial tolerability at Week 12 in subjects treated with TWYNEO.
|
TWYNEO (N=494*) (%) |
Vehicle (N = 264*) (%) |
|||||
| Mild | Moderate | Severe | Mild | Moderate | Severe | |
| Erythema |
33.0 | 6.9 | 0.2 | 26.9 | 8.0 | 0 |
| Pigmentation | 27.3 | 6.3 | 0.4 | 26.5 | 4.5 | 0 |
| Dryness | 22.3 | 5.3 | 0.4 | 16.7 | 2.3 | 0 |
| Scaling | 16.4 | 2.6 | 0 | 12.9 | 0.8 | 0 |
| Burning | 5.9 | 2.2 | 0 | 3.4 | 0.8 | 0 |
| Itching | 11.1 | 1.8 | 0 | 8.7 | 2.7 | 0 |
| Stinging | 5.3 | 0.2 | 0 | 1.9 | 1.1 | 0 |
* The denominators for calculating the percentages were 494 of 555 subjects treated with TWYNEO and 264 of 277 subjects treated with vehicle in these trials who had cutaneous signs and local tolerability results reported at Week 12.
Local tolerability scores for erythema, scaling, dryness, itching, burning and stinging rose during the first two weeks of treatment and decreased thereafter.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Hypersensitivity
Inform patients that serious hypersensitivity reactions have occurred with the use of benzoyl peroxide products. If a patient experiences a serious hypersensitivity reaction, instruct patient to discontinue TWYNEO immediately and seek medical help [see Warnings and Precautions (5.1)].
Skin Irritation
Inform patients that TWYNEO may cause irritation such as erythema, dryness, stinging or burning. Advise the patient to use a moisturizer for irritation. [see Warnings and Precautions (5.2)].
Photosensitivity
Advise patients to minimize unprotected exposure to sunlight and sunlamps; recommend the use of sunscreen products and protective apparel (e.g., hat) over treated areas when sun exposure cannot be avoided. [see Warnings and Precautions (5.3)].
Administration Instructions
Advise patients to apply TWYNEO exactly as directed in a thin layer, avoiding the eyes, lips, paranasal creases and mucous membranes and to wash hands immediately after application. Inform patients that TWYNEO may bleach hair or colored fabric [see Dosage and Administration (
2
)].
Discard Instructions
Instruct patients to store TWYNEO at room temperature and to discard 12 weeks after date of dispensing or 60 days after first opening, whichever is sooner. [see How Supplied/Storage and Handling (16)].
16 How Supplied/storage and Handling
How Supplied
TWYNEO (tretinoin and benzoyl peroxide) cream, 0.1%/3% is a yellow cream and is supplied as:
Principal Display Panel 30 G Pump Box
Rx Only
NDC 0299-5945-30
GALDERMA
TYWNEO®
(tretinoin and benzoyl peroxide) cream, 0.1%/3%
For topical use only.
Not for ophthalmic, oral,
or intravaginal use.
30g
Bottle with a pump
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity, mutagenicity, and impairment of fertility studies were not conducted with TWYNEO.
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
Available data from published observational studies of topical tretinoin in pregnant women have not established a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. Studies conducted with topical benzoyl peroxide have not demonstrated systemic absorption and maternal use is not expected to result in fetal exposure to benzoyl peroxide. There are no data on TWYNEO use in pregnant women.
There are reports of major birth defects reported with maternal use of topical tretinoin similar to those seen in infants exposed to oral retinoids, but these case reports do not establish a pattern or association with tretinoin-related embryopathy (see Data
).
Animal reproductive studies have not been conducted with TWYNEO or benzoyl peroxide. Topical administration of tretinoin to pregnant rats during organogenesis was associated with malformations (craniofacial abnormalities [hydrocephaly], asymmetrical thyroids, variations in ossification, and increased supernumerary ribs) at doses greater than 1 mg tretinoin/kg/day, approximately 5 times the maximum recommended human dose (MRHD) based on body surface area (BSA) comparison and assuming 100% absorption. Oral administration of tretinoin to pregnant cynomolgus monkeys during organogenesis was associated with malformations at 10 mg/kg/day (approximately 100 times the MRHD based on BSA comparison and assuming 100% absorption) (see Data
).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of major birth defects, loss and other adverse outcomes. The background risk in the U.S. general population of major birth defects is 2 to 4% of miscarriage is 15 to 20% of clinically recognized pregnancies.
Section 44425-7 (44425-7)
- 30-gram bottle with a pump, NDC 0299-5945-30
Storage and Handling
- Prior to Dispensing: Store TWYNEO between 2ºC to 8ºC (36ºF to 46ºF) until dispensed to the patient.
-
After Dispensing: Store TWYNEO at room temperature between 20ºC to 25ºC (68ºF to 77ºF). Discard 12 weeks after date of dispensing or 60 days after first opening, whichever is sooner.
- Do not freeze.
11 Description (11 DESCRIPTION)
TWYNEO (tretinoin and benzoyl peroxide) cream is a yellow cream for topical use. Each gram of TWYNEO contains 1 mg (0.1%) of tretinoin and 30 mg (3%) of benzoyl peroxide. Tretinoin is a retinoid and benzoyl peroxide is an oxidizing agent.
The chemical name for tretinoin is all-trans-retinoic acid, also known as (all-E)-3,7-dimethyl-9-(2,6,6-trimethyl-1-cyclohexen-1-yl)-2,4,6,8-nonatetraenoic acid. Tretinoin has the following structural formula:
Molecular Formula: C20H28O2 Molecular Weight: 300.44
The chemical name for benzoyl peroxide is benzoyl benzenecarboperoxoate. Benzoyl peroxide has the following structural formula:
Molecular Formula: C14H10O4 Molecular Weight: 242.23
The formulation of TWYNEO uses silica (silicon dioxide) core shell structures to separately micro-encapsulate tretinoin crystals and benzoyl peroxide crystals enabling inclusion of the two active ingredients in the. TWYNEO contains the following inactive ingredients: anhydrous citric acid, butylated hydroxytoluene, carbomer homopolymer type C, cetrimonium chloride, cetyl alcohol, cyclomethicone, edetate disodium, glycerin, hydrochloric acid, imidurea, (S)lactic acid, macrogol stearate, mono and di-glycerides, polyquaternium-7, purified water, silicon dioxide, sodium hydroxide, squalane, tetraethyl ortho silicate and white wax.
8.4 Pediatric Use
The safety and effectiveness of TWYNEO for the topical treatment of acne vulgaris have been established in pediatric patients 9 years of age and older based on evidence from two multicenter, randomized, double-blind, parallel-group, vehicle-controlled, 12-week clinical trials and an open-label pharmacokinetic study. A total of 283 pediatric subjects 9 years of age and older received TWYNEO in the clinical studies [see Clinical Pharmacology (12.3) and Clinical Studies (14) ].
The safety and effectiveness of TWYNEO in pediatric patients below 9 years of age have not been established.
8.5 Geriatric Use
Clinical trials of TWYNEO did not include sufficient numbers of subjects 65 years of age and older to determine whether they respond differently than younger subjects.
14 Clinical Studies (14 CLINICAL STUDIES)
The safety and efficacy of TWYNEO was evaluated in the treatment of acne vulgaris in two multicenter, randomized, double-blind, vehicle-controlled trials [Trial 1 (NCT03761784), Trial 2 (NCT03761810)], which were identical in design. The trials were conducted in 858 subjects 9 years of age and older, with facial acne vulgaris who were treated once daily for 12 weeks with either TWYNEO or vehicle.
Subjects were required to have a score of moderate (3) or severe (4) on the Investigator Global Assessment (IGA), 20 to 100 inflammatory lesions (papules, pustules and nodules), 30 to 150 non-inflammatory lesions (open and closed comedones) and two or fewer facial nodules.
Overall, 73% of subjects were White and 59% were female. Eighteen (18) (2%) subjects were 9 to 11 years of age, 370 (43%) subjects were 12 to 17 years of age, and 470 (55%) subjects were 18 years of age or older. At baseline, subjects had a mean inflammatory lesion count of 30.7 and a mean noninflammatory lesion count of 46.4. Additionally, 91% of subjects had an IGA score of 3 ("moderate").
The co-primary efficacy endpoints were the absolute change from baseline in non-inflammatory lesion count, and absolute change in inflammatory lesion count at Week 12 and the proportion of subjects with IGA success at Week 12, defined as an IGA score of 0 ("clear") or 1 ("almost clear"), and at least a two-grade improvement (decrease) from baseline at Week 12. The efficacy results are provided in Table 4.
| Trial 1 | Trial 2 | |||
|
TWYNEO (N = 281) |
Vehicle N = 143) |
TWYNEO (N = 290) |
Vehicle (N = 144) |
|
| IGA Success* | 33.9% | 14.3% | 26.8% | 15.1% |
| Difference from Vehicle | 25.7% | 11.6% | ||
| (95% CI) | (17.1%, 34.2%) | (3.6%, 19.7%) | ||
| Inflammatory Lesions |
|
|
|
|
| Mean‡ Absolute Change from Baseline | -21.6 | -14.8 | -16.2 | -14.1 |
| Difference from Vehicle | -6.8 | -2.1 | ||
| (95% CI) | (-9.1, -4.6) | (-3.9, -0.4) | ||
| Mean‡ Percent Change from Baseline | -66.1% | -43.5% | -57.6% | -50.8% |
| Difference from Vehicle | -22.6% | -6.8% | ||
| (95% CI) | (-29.2%, -16.0%) | (-13.1%, -0.5%) | ||
| Non-Inflammatory Lesions | ||||
| Mean‡ Absolute Change from Baseline | -29.7 | -19.8 | -24.2 | -17.4 |
| Difference from Vehicle | -9.9 | -6.8 | ||
| (95% CI) | (-13.0, -6.8) | (-9.9, -3.7) | ||
| Mean‡ Percent Change from Baseline | -61.6% | -40.9% | -54.4% | -41.5% |
| Difference from Vehicle | -20.7% | -13.0% | ||
| (95% CI) | (-27.2%, -14.2%) | (-19.6%, -6.4%) |
*Investigator Global Assessment (IGA) success was defined as an IGA score of 0 ("clear") or1 ("almost clear") with at least at two-grade reduction from baseline.
‡ Means presented in table are Least Square (LS) Means.
4 Contraindications (4 CONTRAINDICATIONS)
TWYNEO is contraindicated in patients with a history of hypersensitivity reaction to benzoyl peroxide or any components of TWYNEO [see Warnings and Precautions (5.1)].
5.2 Skin Irritation
Patients using TWYNEO may experience application site dryness, pain, exfoliation, erythema, dermatitis, pruritis, and irritation [see Adverse Reactions (6.1) ]. Depending upon the severity of these adverse reactions, instruct patients to use a moisturizer, reduce the frequency of the application of TWYNEO, or discontinue use. Avoid application of TWYNEO to cuts, abrasions, eczematous, or sunburned skin.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions (incidence ≥ 1%) are pain, dryness, exfoliation erythema, dermatitis, pruritus and irritation (all at the application site). (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Galderma Laboratories, L.P. at 1-866-735-4137 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
5.1 Hypersensitivity
Hypersensitivity reactions, including anaphylaxis, angioedema, and urticaria, have been reported with the use of benzoyl peroxide products. If a serious hypersensitivity reaction occurs, discontinue TWYNEO immediately and initiate appropriate therapy.
12.2 Pharmacodynamics
Pharmacodynamics of TWYNEO in the treatment of acne vulgaris are unknown.
12.3 Pharmacokinetics
The systemic exposure of benzoyl peroxide was not assessed. Benzoyl peroxide is absorbed by skin where it is converted to benzoic acid and eliminated in the urine.
Plasma concentrations of tretinoin and its major metabolites were evaluated in 35 subjects in an open-label, randomized, pharmacokinetic (PK) study. Subjects 9 years of age and older with acne vulgaris applied a mean dose of 1.9 g TWYNEO to the skin of the face, shoulders, upper back and upper chest once daily for 14 days.
Steady-state PK characteristics were determined from samples drawn on Day 14. The mean baseline corrected Cmax and AUC0-24 of tretinoin and its metabolites after once daily application of TWYNEO for 14 days are provided in Table 3. No detectable levels of the metabolites all-trans 4-keto retinoic acid and 9-cis retinoid acid were found in subjects treated with TWYNEO.
| Age Group (years) | n | Compound | Mean(± SD) Cmax (ng/mL) |
Mean(± SD) AUC0-24 (ng-h/mL) |
| ≥ 18 years of age | 12 | tretinoin | 0.15 ± 0.17 | 0.63 ± 0.95 |
| 4-keto 13-cis RA | 0.27 ± 0.29 | 2.88 ± 3.61 | ||
| 13-cis RA | 0.21 ± 0.19 | 1.99 ± 2.90 | ||
| 12 to 17 | 15 | tretinoin | 0.19 ± 0.18 | 1.56 ± 1.97 |
|
|
4-keto 13-cis RA | 0.32 ± 0.28 | 2.39 ± 3.05 | |
| 13-cis RA | 0.28 ± 0.35 | 1.79 ± 2.79 | ||
| 9 to 11 | 8 | tretinoin | 0.18 ± 0.22 | 2.06 ± 3.96 |
|
|
4-keto 13-cis RA | 0.34 ± 0.36 | 2.89 ± 3.17 | |
| 13-cis RA | 0.13 ± 0.09 | 0.96 ± 1.36 |
5.3 Photosensitivity
TWYNEO may increase sensitivity to ultraviolet light. Minimize or avoid exposure to natural or artificial sunlight (tanning beds or UVA/B treatment) while using TWYNEO. Instruct patients to implement sun protection measures (e.g., sunscreen and loose-fitting clothes) when sun exposure cannot be avoided. Discontinue TWYNEO at the first evidence of sunburn.
1 Indications and Usage (1 INDICATIONS AND USAGE)
TWYNEO is indicated for the topical treatment of acne vulgaris in adults and pediatric patients 9 years of age and older.
12.1 Mechanism of Action
Benzoyl peroxide is an oxidizing agent with bactericidal and keratolytic effects, but the precise mechanism of action is unknown. Tretinoin is a metabolite of vitamin A that binds with high affinity to specific retinoic acid receptors located in both the cytosol and nucleus. Tretinoin activates three members of the retinoic acid (RAR) nuclear receptors (RARα , RARß, RARγ) which act to modify gene expression, subsequent protein synthesis, and epithelial cell growth and differentiation. It has not been established whether the clinical effects of tretinoin are mediated through activation of retinoic acid receptors, and/or other mechanisms.
Although the exact mode of action of tretinoin in acne treatment is unknown, current evidence suggests that topical tretinoin decreases cohesiveness of follicular epithelial cells with decreased microcomedo formation. Additionally, tretinoin stimulates mitotic activity and increased turnover of follicular epithelial cells causing extrusion of the comedones.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity: Severe hypersensitivity reactions, including anaphylaxis and angioedema, have been reported with use of benzoyl peroxide products. (4, 5.1)
- Skin Irritation: Pain, dryness, exfoliation, erythema, and irritation may occur with use of TWYNEO. Avoid application of TWYNEO to cuts abrasions, eczematous or sunburned skin. (5.2)
- Photosensitivity: Minimize unprotected exposure to sunlight and sunlamps. Use sunscreen and protective clothing when sun exposure cannot be avoided. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Apply a thin layer of TWYNEO to the affected areas once daily on clean and dry skin. Avoid contact with the eyes, lips, paranasal creases, and mucous membranes.
- Wash hands after application.
- TWYNEO is for topical use only. TWYNEO is not for oral, ophthalmic, or intravaginal use.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Cream, 0.1%/3%: Each gram of TWYNEO contains 1mg (0.1%) of tretinoin and 30 mg (3%) of benzoyl peroxide in a yellow cream in a 30-gram bottle with a pump.
6.2 Postmarketing Experience
The following adverse reactions have been identified during use of benzoyl peroxide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Anaphylaxis, angioedema and urticaria
6.1 Clinical Trials Experience (6.1 Clinical trials experience)
The following adverse reactions are discussed in greater detail elsewhere in the labeling:
- Hypersensitivity [see Warnings and Precautions (5.1)]
- Skin Irritation [see Warnings and Precautions (5.2)]
Because clinical trials are conducted under widely varying conditions, adverse reaction rates are observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In two multicenter, randomized, double-blind, vehicle-controlled trials (Trial 1 and 2), 832 subjects 9 years of age and older with facial acne vulgaris applied TWYNEO (N=555) or vehicle (N=277) daily for 12 weeks. The majority of subjects were White (73%) and female (59%). Approximately 33% were Hispanic/Latino, and 46% were younger than 18 years of age. Adverse reactions reported in ≥ 1.0% of subjects treated with TWYNEO (and for which the rate exceeded the rate for vehicle), as well as the corresponding rates reported in subjects treated with vehicle are presented in Table 1.
|
|
TWYNEO Cream (N = 555) n (%) |
Vehicle Cream (N = 277) n (%) |
|---|---|---|
| Application Site Pain* | 59 (10.6) | 1 (0.4) |
| Application Site Dryness | 27 (4.9) | 1 (0.4) |
| Application Site Exfoliation | 23 (4.1) | 0 |
| Application Site Erythema | 22 (4.0) | 0 |
| Application Site Dermatitis | 7 (1.3) | 1 (0.4) |
| Application Site Pruritus | 7 (1.3) | 0 |
| Application Site Irritation | 6 (1.1) | 1 (0.4) |
* Application site pain defined as application site stinging, burning or pain.
Local tolerability evaluations were conducted at each study visit in the clinical trial by assessment of erythema, scaling, pigmentation, dryness, itching, burning and stinging. Table 2 presents the active assessment of the signs and symptoms of local facial tolerability at Week 12 in subjects treated with TWYNEO.
|
TWYNEO (N=494*) (%) |
Vehicle (N = 264*) (%) |
|||||
| Mild | Moderate | Severe | Mild | Moderate | Severe | |
| Erythema |
33.0 | 6.9 | 0.2 | 26.9 | 8.0 | 0 |
| Pigmentation | 27.3 | 6.3 | 0.4 | 26.5 | 4.5 | 0 |
| Dryness | 22.3 | 5.3 | 0.4 | 16.7 | 2.3 | 0 |
| Scaling | 16.4 | 2.6 | 0 | 12.9 | 0.8 | 0 |
| Burning | 5.9 | 2.2 | 0 | 3.4 | 0.8 | 0 |
| Itching | 11.1 | 1.8 | 0 | 8.7 | 2.7 | 0 |
| Stinging | 5.3 | 0.2 | 0 | 1.9 | 1.1 | 0 |
* The denominators for calculating the percentages were 494 of 555 subjects treated with TWYNEO and 264 of 277 subjects treated with vehicle in these trials who had cutaneous signs and local tolerability results reported at Week 12.
Local tolerability scores for erythema, scaling, dryness, itching, burning and stinging rose during the first two weeks of treatment and decreased thereafter.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Hypersensitivity
Inform patients that serious hypersensitivity reactions have occurred with the use of benzoyl peroxide products. If a patient experiences a serious hypersensitivity reaction, instruct patient to discontinue TWYNEO immediately and seek medical help [see Warnings and Precautions (5.1)].
Skin Irritation
Inform patients that TWYNEO may cause irritation such as erythema, dryness, stinging or burning. Advise the patient to use a moisturizer for irritation. [see Warnings and Precautions (5.2)].
Photosensitivity
Advise patients to minimize unprotected exposure to sunlight and sunlamps; recommend the use of sunscreen products and protective apparel (e.g., hat) over treated areas when sun exposure cannot be avoided. [see Warnings and Precautions (5.3)].
Administration Instructions
Advise patients to apply TWYNEO exactly as directed in a thin layer, avoiding the eyes, lips, paranasal creases and mucous membranes and to wash hands immediately after application. Inform patients that TWYNEO may bleach hair or colored fabric [see Dosage and Administration (
2
)].
Discard Instructions
Instruct patients to store TWYNEO at room temperature and to discard 12 weeks after date of dispensing or 60 days after first opening, whichever is sooner. [see How Supplied/Storage and Handling (16)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
How Supplied
TWYNEO (tretinoin and benzoyl peroxide) cream, 0.1%/3% is a yellow cream and is supplied as:
Principal Display Panel 30 G Pump Box (PRINCIPAL DISPLAY PANEL - 30 g Pump Box)
Rx Only
NDC 0299-5945-30
GALDERMA
TYWNEO®
(tretinoin and benzoyl peroxide) cream, 0.1%/3%
For topical use only.
Not for ophthalmic, oral,
or intravaginal use.
30g
Bottle with a pump
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity, mutagenicity, and impairment of fertility studies were not conducted with TWYNEO.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:02.742047 · Updated: 2026-03-14T22:28:55.050842