Premier Value 44-112-delisted
270e9221-d305-4148-b523-741e02fe733f
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Pseudoephedrine HCl 30 mg
Purpose
Nasal decongestant
Medication Information
Purpose
Nasal decongestant
Description
Pseudoephedrine HCl 30 mg
Uses
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- temporarily relieves sinus congestion and pressure
Directions
| adults and children 12 years and over | take 2 tablets every 4 to 6 hours; do not take more than 8 tablets in 24 hours |
| children ages 6 to 11 years | take 1 tablet every 4 to 6 hours; do not take more than 4 tablets in 24 hours |
| children under 6 years | do not use |
Do Not Use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Other Information
- each tablet contains: calcium 15 mg
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- see end flap for expiration date and lot number
Inactive Ingredients
croscarmellose sodium, dibasic calcium phosphate dihydrate, FD&C red #40 aluminum lake, FD&C yellow #6 aluminum lake, hypromellose, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, silicon dioxide, titanium dioxide, triacetin
Questions Or Comments?
1-800-426-9391
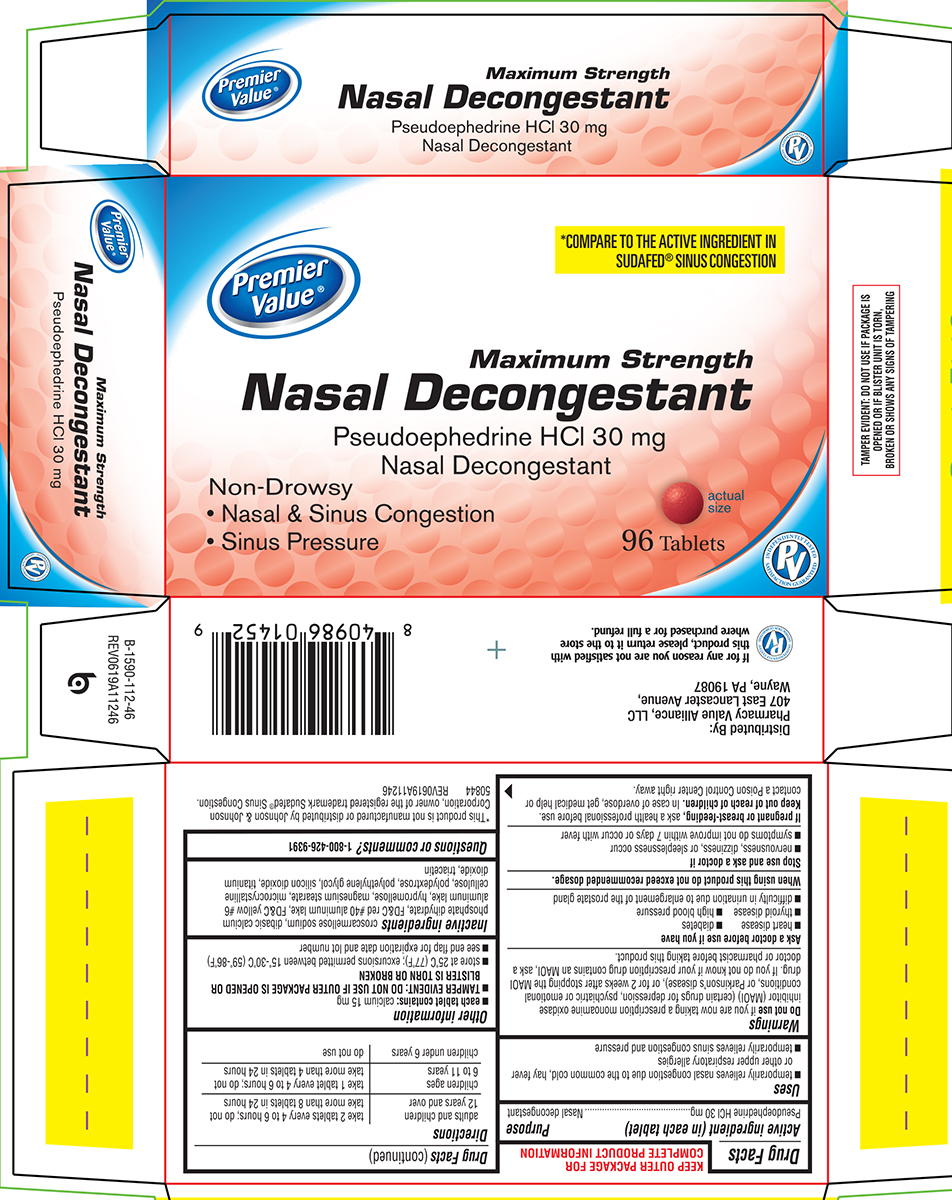
Principal Display Panel
Premier
Value®
*COMPARE TO THE ACTIVE INGREDIENT IN
SUDAFED® SINUS CONGESTION
Maximum Strength
Nasal Decongestant
Pseudoephedrine HCl 30 mg
Nasal Decongestant
Non-Drowsy
• Nasal & Sinus Congestion
• Sinus Pressure
96 Tablets
actual
size
PV
INDEPENDENTLY TESTED
SATISFACTION GUARANTEED
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS
OPENED OR IF BLISTER UNIT IS TORN,
BROKEN OR SHOWS ANY SIGNS OF TAMPERING
*This product is not manufactured or distributed by Johnson & Johnson
Corporation, owner of the registered trademark Sudafed® Sinus Congestion.
50844 REV0619A11246
Distributed By:
Pharmacy Value Alliance, LLC
407 East Lancaster Avenue,
Wayne, PA 19087
PV
INDEPENDENTLY TESTED
SATISFACTION GUARANTEED
If for any reason you are not satisfied with
this product, please return it to the store
where purchased for a full refund.
When Using This Product
do not exceed recommended dosage.
Stop Use and Ask A Doctor If
- nervousness, dizziness, or sleeplessness occur
- symptoms do not improve within 7 days or occur with fever
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet)
Pseudoephedrine HCl 30 mg
Ask A Doctor Before Use If You Have
- diabetes
- heart disease
- high blood pressure
- thyroid disease
- difficulty in urination due to enlargement of the prostate gland
Structured Label Content
Uses
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- temporarily relieves sinus congestion and pressure
Purpose
Nasal decongestant
Directions
| adults and children 12 years and over | take 2 tablets every 4 to 6 hours; do not take more than 8 tablets in 24 hours |
| children ages 6 to 11 years | take 1 tablet every 4 to 6 hours; do not take more than 4 tablets in 24 hours |
| children under 6 years | do not use |
Do Not Use (Do not use)
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Other Information (Other information)
- each tablet contains: calcium 15 mg
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- see end flap for expiration date and lot number
Inactive Ingredients (Inactive ingredients)
croscarmellose sodium, dibasic calcium phosphate dihydrate, FD&C red #40 aluminum lake, FD&C yellow #6 aluminum lake, hypromellose, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, silicon dioxide, titanium dioxide, triacetin
Questions Or Comments? (Questions or comments?)
1-800-426-9391
Principal Display Panel
Premier
Value®
*COMPARE TO THE ACTIVE INGREDIENT IN
SUDAFED® SINUS CONGESTION
Maximum Strength
Nasal Decongestant
Pseudoephedrine HCl 30 mg
Nasal Decongestant
Non-Drowsy
• Nasal & Sinus Congestion
• Sinus Pressure
96 Tablets
actual
size
PV
INDEPENDENTLY TESTED
SATISFACTION GUARANTEED
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS
OPENED OR IF BLISTER UNIT IS TORN,
BROKEN OR SHOWS ANY SIGNS OF TAMPERING
*This product is not manufactured or distributed by Johnson & Johnson
Corporation, owner of the registered trademark Sudafed® Sinus Congestion.
50844 REV0619A11246
Distributed By:
Pharmacy Value Alliance, LLC
407 East Lancaster Avenue,
Wayne, PA 19087
PV
INDEPENDENTLY TESTED
SATISFACTION GUARANTEED
If for any reason you are not satisfied with
this product, please return it to the store
where purchased for a full refund.
When Using This Product (When using this product)
do not exceed recommended dosage.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- nervousness, dizziness, or sleeplessness occur
- symptoms do not improve within 7 days or occur with fever
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet) (Active ingredient (in each tablet))
Pseudoephedrine HCl 30 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- diabetes
- heart disease
- high blood pressure
- thyroid disease
- difficulty in urination due to enlargement of the prostate gland
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:33.845623 · Updated: 2026-03-14T23:09:23.715399