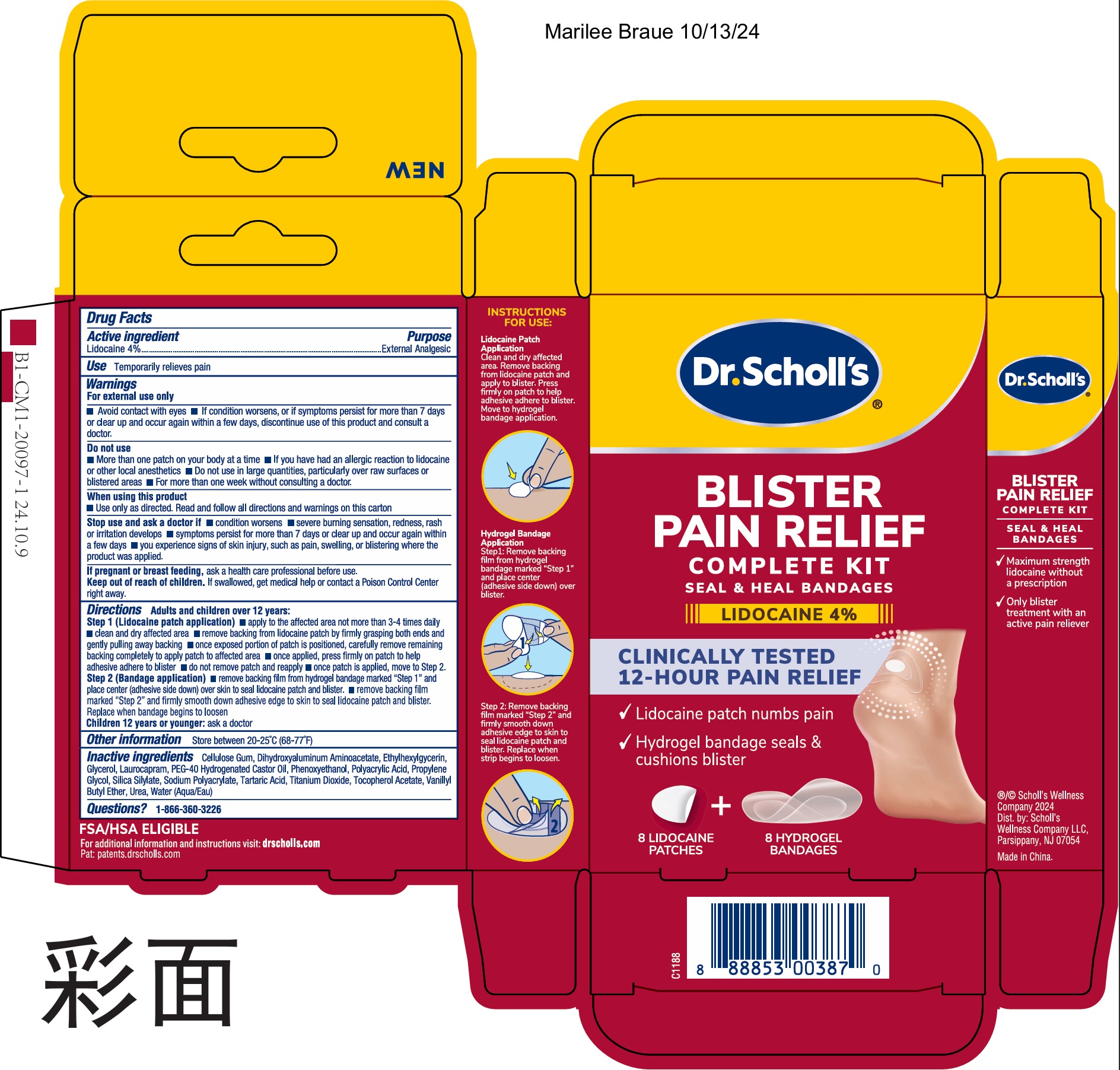
Dr. Scholl's Blister Pain Relief Complete Kit
26e3a678-b28f-588b-e063-6394a90ac025
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 4%
Purpose
External Analgesic
Medication Information
Purpose
External Analgesic
Description
Lidocaine 4%
Use
Temporarily relieves pain
Warnings
For external use only
- Avoid contact with eyes
- If condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days, discontinue use of this product and consult a doctor.
Directions
Adults and children over 12 years:
Step 1 (Lidocaine patch application) • apply to the affected area not more than 3-4 times daily
- clean and dry affected area
- remove backing from lidocaine patch by firmly grasping both ends and gently pulling away backing
- once exposed portion of patch is positioned, carefully remove remaining backing completely to apply patch to affected area
- once applied, press firmly on patch to help adhesive adhere to blister
- do not remove patch and reapply
- once patch is applied, move to Step 2.
Step 2 (Bandage application) •remove backing film from hydrogel bandage marked "Step 1" and place center (adhesive side down) over skin to seal lidocaine patch and blister.
- remove backing film marked "Step 2" and firmly smooth down adhesive edge to skin to seal lidocaine patch and blister. Replace when bandage begins to loosen
children 12 years or younger: ask a doctor
Do Not Use
- More than one patch on your body at a time
- If you have had an allergic reaction to lidocaine or other local anesthetics
- Do not use in large quantities, particularly over raw surfaces or blistered areas
- For more than one week without consulting a doctor.
Drug Facts
Questions?
1-866-360-3226
Active Ingredient
Lidocaine 4%
Other Information
Store between 20-25°C (68-77°F)
Package Labeling:
Inactive Ingredients
Cellulose Gum, Dihydroxyaluminum Aminoacetate, Ethylhexylglycerin, Glycerol, Laurocapram, PEG-40 Hydrogenated Castor Oil, Phenoxyethanol, Polyacrylic Acid, Propylene Glycol, Silica Silylate, Sodium Polyacrylate, Tartaric Acid, Titanium Dioxide, Tocopherol Acetate, Vanillyl Butyl Ether, Urea, Water (Aqua)
When Using This Product
- Use only as directed. Read and follow all directions and warnings on this carton
Stop Use and Ask A Doctor If
- condition worsens
- severe burning sensation, redness, rash or irritation develops
- symptoms persist for more than 7 days or clear up and occur again within a few days
- you experience signs of skin injury, such as pain, swelling, or blistering where the product was applied.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
If Pregnant Or Breast Feeding,
ask a health care professional before use.
Structured Label Content
Use
Temporarily relieves pain
Purpose
External Analgesic
Warnings
For external use only
- Avoid contact with eyes
- If condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days, discontinue use of this product and consult a doctor.
Directions
Adults and children over 12 years:
Step 1 (Lidocaine patch application) • apply to the affected area not more than 3-4 times daily
- clean and dry affected area
- remove backing from lidocaine patch by firmly grasping both ends and gently pulling away backing
- once exposed portion of patch is positioned, carefully remove remaining backing completely to apply patch to affected area
- once applied, press firmly on patch to help adhesive adhere to blister
- do not remove patch and reapply
- once patch is applied, move to Step 2.
Step 2 (Bandage application) •remove backing film from hydrogel bandage marked "Step 1" and place center (adhesive side down) over skin to seal lidocaine patch and blister.
- remove backing film marked "Step 2" and firmly smooth down adhesive edge to skin to seal lidocaine patch and blister. Replace when bandage begins to loosen
children 12 years or younger: ask a doctor
Do Not Use (Do not use)
- More than one patch on your body at a time
- If you have had an allergic reaction to lidocaine or other local anesthetics
- Do not use in large quantities, particularly over raw surfaces or blistered areas
- For more than one week without consulting a doctor.
Drug Facts
Questions?
1-866-360-3226
Active Ingredient (Active ingredient)
Lidocaine 4%
Other Information (Other information)
Store between 20-25°C (68-77°F)
Package Labeling:
Inactive Ingredients (Inactive ingredients)
Cellulose Gum, Dihydroxyaluminum Aminoacetate, Ethylhexylglycerin, Glycerol, Laurocapram, PEG-40 Hydrogenated Castor Oil, Phenoxyethanol, Polyacrylic Acid, Propylene Glycol, Silica Silylate, Sodium Polyacrylate, Tartaric Acid, Titanium Dioxide, Tocopherol Acetate, Vanillyl Butyl Ether, Urea, Water (Aqua)
When Using This Product (When using this product)
- Use only as directed. Read and follow all directions and warnings on this carton
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens
- severe burning sensation, redness, rash or irritation develops
- symptoms persist for more than 7 days or clear up and occur again within a few days
- you experience signs of skin injury, such as pain, swelling, or blistering where the product was applied.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding, )
ask a health care professional before use.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:33.397473 · Updated: 2026-03-14T23:07:34.278056