These Highlights Do Not Include All The Information Needed To Use Guanfacine Extended-release Tablets Safely And Effectively. See Full Prescribing Information For Guanfacine Extended-release Tablets.
261c06ce-8708-d659-e063-6294a90ac381
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Guanfacine extended-release tablets are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) as monotherapy and as adjunctive therapy to stimulant medications [see Clinical Studies (14) ] .
Indications and Usage
Guanfacine extended-release tablets are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) as monotherapy and as adjunctive therapy to stimulant medications [see Clinical Studies (14) ] .
Dosage and Administration
Recommended dose: 1 mg to 7 mg (0.05-0.12 mg/kg target weight based dose range) once daily in the morning or evening based on clinical response and tolerability ( 2.2 ). Begin at a dose of 1 mg once daily and adjust in increments of no more than 1 mg/week ( 2.2 ). Do not crush, chew or break tablets before swallowing ( 2.1 ). Do not administer with high-fat meals, because of increased exposure ( 2.1 ). Do not substitute for immediate-release guanfacine tablets on a mg-per-mg basis, because of differing pharmacokinetic profiles ( 2.3 ). If switching from immediate-release guanfacine, discontinue that treatment and titrate with guanfacine extended-release tablets as directed ( 2.3 ). When discontinuing, taper the dose in decrements of no more than 1 mg every 3 to 7 days to avoid rebound hypertension ( 2.5 ).
Warnings and Precautions
Hypotension, bradycardia, syncope: Titrate slowly and monitor vital signs frequently in patients at risk for hypotension, heart block, bradycardia, syncope, cardiovascular disease, vascular disease, cerebrovascular disease or chronic renal failure. Measure heart rate and blood pressure prior to initiation of therapy, following dose increases, and periodically while on therapy. Avoid concomitant use of drugs with additive effects unless clinically indicated. Advise patients to avoid becoming dehydrated or overheated ( 5.1 ). Sedation and somnolence: Occur commonly with guanfacine extended-release tablets. Consider the potential for additive sedative effects with CNS depressant drugs. Caution patients against operating heavy equipment or driving until they know how they respond to guanfacine extended-release tablets ( 5.2 ). Cardiac Conduction Abnormalities: May worsen sinus node dysfunction and atrioventricular (AV) block, especially in patients taking other sympatholytic drugs. Titrate slowly and monitor vital signs frequently ( 5.3 ). Rebound Hypertension: Abrupt discontinuation of guanfacine extended-release tablets can lead to clinically significant and persistent rebound hypertension. Subsequent hypertensive encephalopathy was also reported. To minimize the risk of rebound hypertension upon discontinuation, the total daily dose of guanfacine extended-release tablets should be tapered in decrements of no more than 1 mg every 3 to 7 days ( 5.4 ).
Contraindications
Guanfacine extended-release tablets are contraindicated in patients with a history of a hypersensitivity reaction to guanfacine extended-release tablets or their inactive ingredients, or other products containing guanfacine. Rash and pruritus have been reported.
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Hypotension, bradycardia, and syncope [see Warnings and Precautions (5.1) ] Sedation and somnolence [see Warnings and Precautions (5.2) ] Cardiac conduction abnormalities [see Warnings and Precautions (5.3) ] Rebound Hypertension [see Warnings and Precautions (5.4) ]
Drug Interactions
Table 14 contains clinically important drug interactions with guanfacine extended-release tablets [see Clinical Pharmacology (12.3) ]. Table 14: Clinically Important Drug Interactions: Effect of other Drugs on Guanfacine Extended-Release Tablets Concomitant Drug Name or Drug Class Clinical Rationale and Magnitude of Drug Interaction Clinical Recommendation Strong and moderate CYP3A4 inhibitors, e.g., ketoconazole, fluconazole Guanfacine is primarily metabolized by CYP3A4 and its plasma concentrations can be significantly affected resulting in an increase in exposure Consider dose reduction [see Dosage and administration (2.7) ] Strong and moderate CYP3A4 inducers, e.g., rifampin, efavirenz Guanfacine is primarily metabolized by CYP3A4 and its plasma concentrations can be significantly affected resulting in a decrease in exposure Consider dose increase [see Dosage and administration (2.7) ]
Storage and Handling
Guanfacine extended-release tablets, USP are supplied in 1 mg, 2 mg, 3 mg, and 4 mg strength extended-release tablets in 100 count bottles. 1 mg 2 mg 3 mg 4 mg Color White/off-white White/off-white Green Green Shape Round Caplet Round Caplet Debossment (top/bottom) F152 / Plain F153 / Plain F154 / Plain F155 / Plain NDC number 72266-255-55 72266-256-55 72266-257-55 72266-258-55
How Supplied
Guanfacine extended-release tablets, USP are supplied in 1 mg, 2 mg, 3 mg, and 4 mg strength extended-release tablets in 100 count bottles. 1 mg 2 mg 3 mg 4 mg Color White/off-white White/off-white Green Green Shape Round Caplet Round Caplet Debossment (top/bottom) F152 / Plain F153 / Plain F154 / Plain F155 / Plain NDC number 72266-255-55 72266-256-55 72266-257-55 72266-258-55
Medication Information
Warnings and Precautions
Hypotension, bradycardia, syncope: Titrate slowly and monitor vital signs frequently in patients at risk for hypotension, heart block, bradycardia, syncope, cardiovascular disease, vascular disease, cerebrovascular disease or chronic renal failure. Measure heart rate and blood pressure prior to initiation of therapy, following dose increases, and periodically while on therapy. Avoid concomitant use of drugs with additive effects unless clinically indicated. Advise patients to avoid becoming dehydrated or overheated ( 5.1 ). Sedation and somnolence: Occur commonly with guanfacine extended-release tablets. Consider the potential for additive sedative effects with CNS depressant drugs. Caution patients against operating heavy equipment or driving until they know how they respond to guanfacine extended-release tablets ( 5.2 ). Cardiac Conduction Abnormalities: May worsen sinus node dysfunction and atrioventricular (AV) block, especially in patients taking other sympatholytic drugs. Titrate slowly and monitor vital signs frequently ( 5.3 ). Rebound Hypertension: Abrupt discontinuation of guanfacine extended-release tablets can lead to clinically significant and persistent rebound hypertension. Subsequent hypertensive encephalopathy was also reported. To minimize the risk of rebound hypertension upon discontinuation, the total daily dose of guanfacine extended-release tablets should be tapered in decrements of no more than 1 mg every 3 to 7 days ( 5.4 ).
Indications and Usage
Guanfacine extended-release tablets are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) as monotherapy and as adjunctive therapy to stimulant medications [see Clinical Studies (14) ] .
Dosage and Administration
Recommended dose: 1 mg to 7 mg (0.05-0.12 mg/kg target weight based dose range) once daily in the morning or evening based on clinical response and tolerability ( 2.2 ). Begin at a dose of 1 mg once daily and adjust in increments of no more than 1 mg/week ( 2.2 ). Do not crush, chew or break tablets before swallowing ( 2.1 ). Do not administer with high-fat meals, because of increased exposure ( 2.1 ). Do not substitute for immediate-release guanfacine tablets on a mg-per-mg basis, because of differing pharmacokinetic profiles ( 2.3 ). If switching from immediate-release guanfacine, discontinue that treatment and titrate with guanfacine extended-release tablets as directed ( 2.3 ). When discontinuing, taper the dose in decrements of no more than 1 mg every 3 to 7 days to avoid rebound hypertension ( 2.5 ).
Contraindications
Guanfacine extended-release tablets are contraindicated in patients with a history of a hypersensitivity reaction to guanfacine extended-release tablets or their inactive ingredients, or other products containing guanfacine. Rash and pruritus have been reported.
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Hypotension, bradycardia, and syncope [see Warnings and Precautions (5.1) ] Sedation and somnolence [see Warnings and Precautions (5.2) ] Cardiac conduction abnormalities [see Warnings and Precautions (5.3) ] Rebound Hypertension [see Warnings and Precautions (5.4) ]
Drug Interactions
Table 14 contains clinically important drug interactions with guanfacine extended-release tablets [see Clinical Pharmacology (12.3) ]. Table 14: Clinically Important Drug Interactions: Effect of other Drugs on Guanfacine Extended-Release Tablets Concomitant Drug Name or Drug Class Clinical Rationale and Magnitude of Drug Interaction Clinical Recommendation Strong and moderate CYP3A4 inhibitors, e.g., ketoconazole, fluconazole Guanfacine is primarily metabolized by CYP3A4 and its plasma concentrations can be significantly affected resulting in an increase in exposure Consider dose reduction [see Dosage and administration (2.7) ] Strong and moderate CYP3A4 inducers, e.g., rifampin, efavirenz Guanfacine is primarily metabolized by CYP3A4 and its plasma concentrations can be significantly affected resulting in a decrease in exposure Consider dose increase [see Dosage and administration (2.7) ]
Storage and Handling
Guanfacine extended-release tablets, USP are supplied in 1 mg, 2 mg, 3 mg, and 4 mg strength extended-release tablets in 100 count bottles. 1 mg 2 mg 3 mg 4 mg Color White/off-white White/off-white Green Green Shape Round Caplet Round Caplet Debossment (top/bottom) F152 / Plain F153 / Plain F154 / Plain F155 / Plain NDC number 72266-255-55 72266-256-55 72266-257-55 72266-258-55
How Supplied
Guanfacine extended-release tablets, USP are supplied in 1 mg, 2 mg, 3 mg, and 4 mg strength extended-release tablets in 100 count bottles. 1 mg 2 mg 3 mg 4 mg Color White/off-white White/off-white Green Green Shape Round Caplet Round Caplet Debossment (top/bottom) F152 / Plain F153 / Plain F154 / Plain F155 / Plain NDC number 72266-255-55 72266-256-55 72266-257-55 72266-258-55
Description
Guanfacine extended-release tablets are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) as monotherapy and as adjunctive therapy to stimulant medications [see Clinical Studies (14) ] .
Section 42229-5
Fixed Dose Trials
| Guanfacine Extended-Release Tablets (mg) | ||||||
|---|---|---|---|---|---|---|
| Adverse Reaction Term | Placebo
(N=149) |
1mg*
(N=61) |
2mg
(N=150) |
3mg
(N=151) |
4mg
(N=151) |
All Doses of Guanfacine Extended-Release Tablets (N=513) |
| *The lowest dose of 1 mg used in Study 2 was not randomized to patients weighing more than 50 kg.
a: The somnolence term includes somnolence, sedation, and hypersomnia. b: The hypotension term includes hypotension, diastolic hypotension, orthostatic hypotension, blood pressure decreased, blood pressure diastolic decreased, blood pressure systolic decreased). |
||||||
| Somnolence a | 11% | 28% | 30% | 38% | 51% | 38% |
| Fatigue | 3% | 10% | 13% | 17% | 15% | 14% |
| Hypotension b | 3% | 8% | 5% | 7% | 8% | 7% |
| Dizziness | 4% | 5% | 3% | 7% | 10% | 6% |
| Lethargy | 3% | 2% | 3% | 8% | 7% | 6% |
| Nausea | 2% | 7% | 5% | 5% | 6% | 6% |
| Dry mouth | 1% | 0% | 1% | 6% | 7% | 4% |
| Guanfacine Extended-Release Tablets (mg) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adverse Reaction Term | Placebo
(N=149) |
1mg*
(N=61) |
2mg
(N=150) |
3mg
(N=151) |
4mg
(N=151) |
All Doses of Guanfacine Extended-Release Tablets (N=513) | ||||||
| n | (%) | n | (%) | n | (%) | n | (%) | n | (%) | n | (%) | |
| Adverse reactions leading to discontinuation in ≥2% in any dose group but did not meet this criteria in all doses combined: hypotension (hypotension, diastolic hypotension, orthostatic hypotension, blood pressure decreased, blood pressure diastolic decreased, blood pressure systolic decreased), headache, and dizziness.
* The lowest dose of 1 mg used in Study 2 was not randomized to patients weighing more than 50 kg. a: The somnolence term includes somnolence, sedation, and hypersomnia. |
||||||||||||
| Total patients | 4 | (3%) | 2 | (3%) | 10 | (7%) | 15 | (10%) | 27 | (18%) | 54 | (11%) |
| Somnolence a | 1 | (1%) | 2 | (3%) | 5 | (3%) | 6 | (4%) | 17 | (11%) | 30 | (6%) |
| Fatigue | 0 | (0%) | 0 | (0%) | 2 | (1%) | 2 | (1%) | 4 | (3%) | 8 | (2%) |
| Guanfacine Extended-Release Tablets (mg) | ||||||
|---|---|---|---|---|---|---|
| Adverse Reaction Term | Placebo
(N=149) |
1mg*
(N=61) |
2mg
(N=150) |
3mg
(N=151) |
4mg
(N=151) |
All Doses of Guanfacine Extended-Release Tablets (N=513) |
| Adverse reactions ≥2% for all doses of guanfacine extend-release tablets and >rate in placebo in any dose group but did not meet this criteria in all doses combined: insomnia (insomnia, initial insomnia, middle insomnia, terminal insomnia, sleep disorder), vomiting, diarrhea, abdominal/stomach discomfort (abdominal discomfort, epigastric discomfort, stomach discomfort), rash (rash, rash generalized, rash papular), dyspepsia, increased weight, bradycardia (bradycardia, sinus bradycardia), asthma (asthma, bronchospasm, wheezing), agitation, anxiety (anxiety, nervousness), sinus arrhythmia, blood pressure increased (blood pressure increased, blood pressure diastolic increased), and first degree atrioventricular block.
* The lowest dose of 1 mg used in Study 2 was not randomized to patients weighing more than 50 kg. a: The abdominal pain term includes abdominal pain, abdominal pain lower, abdominal pain upper, and abdominal tenderness. b: The nightmare term includes abnormal dreams, nightmare, and sleep terror. c: The enuresis term includes enuresis, nocturia, and urinary incontinence. d: The affect lability term includes affect lability and mood swings. |
||||||
| Headache | 19% | 26% | 25% | 16% | 28% | 23% |
| Abdominal Pain a | 9% | 10% | 7% | 11% | 15% | 11% |
| Decreased Appetite | 4% | 5% | 4% | 9% | 6% | 6% |
| Irritability | 4% | 5% | 8% | 3% | 7% | 6% |
| Constipation | 1% | 2% | 2% | 3% | 4% | 3% |
| Nightmare b | 0% | 0% | 0% | 3% | 4% | 2% |
| Enuresis c | 1% | 0% | 1% | 3% | 2% | 2% |
| Affect Lability d | 1% | 2% | 1% | 3% | 1% | 2% |
Section 42230-3
Patient Information
Guanfacine (gwahn-fa-seen)
Extended-Release Tablets
Read the Patient Information that comes with guanfacine extended-release tablets before you start taking it and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your doctor about your medical condition or your treatment.
What is guanfacine extended-release tablets ?
Guanfacine extended-release tablets are prescription medicine used to treat the symptoms of attention deficit hyperactivity disorder (ADHD). Guanfacine extended-release tablets may be used alone or with ADHD stimulant medicines.
Guanfacine extended-release tablets are not central nervous system (CNS) stimulant.
It is not known if guanfacine extended-release tablets are safe and effective in children younger than 6 years of age.
Who should not take guanfacine extended-release tablets ?
Do not take guanfacine extended-release tablets if you are allergic to guanfacine or any of the ingredients in guanfacine extended-release tablets. See the end of this leaflet for a complete list of ingredients in guanfacine extended-release tablets.
What should I tell my doctor before taking guanfacine extended-release tablets ?
Before you take guanfacine extended-release tablets , tell your doctor if you:
- have heart problems or a low heart rate
- have fainted
- have low or high blood pressure
- have liver or kidney problems
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if guanfacine extended-release tablets will harm your unborn baby. Talk to your doctor if you are pregnant or plan to become pregnant.
- are breastfeeding or plan to breastfeed. It is not known if guanfacine passes into your breast milk. Talk to your doctor about the best way to feed your baby while taking guanfacine extended-release tablets.
Tell your doctor about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Guanfacine extended-release tablets may affect the way other medicines work, and other medicines may affect how guanfacine extended-release tablets work.
Especially tell your doctor if you take:
- ketoconazole
- medicines that can affect enzyme metabolism
- high blood pressure medicine
- sedatives
- benzodiazepines
- barbiturates
- antipsychotics
Ask your doctor or pharmacist for a list of these medicines, if you are not sure.
Know the medicines you take. Keep a list of them and show it to your doctor and pharmacist when you get a new medicine.
How should I take guanfacine extended-release tablets ?
- Take guanfacine extended-release tablets exactly as your doctor tells you.
- Your doctor may change your dose. Do not change your dose of guanfacine extended-release tablets without talking to your doctor.
- Do not stop taking guanfacine extended-release tablets without talking to your doctor.
- Try not to miss your dose of guanfacine extended-release tablets. If you miss a dose of guanfacine extended-release tablets, take the next dose at your regular time. If you miss 2 or more doses, talk to your doctor, as you may need to restart guanfacine extended-release tablets with a lower dose.
- Do not take a double dose to make up for a missed dose.
- Guanfacine extended-release tablets should be taken 1 time a day in the morning or in the evening, either alone or in combination with an ADHD stimulant medicine that your doctor may prescribe. Your doctor will tell you when to take guanfacine extended-release tablets and when to take your ADHD stimulant medication.
- Guanfacine extended-release tablets should be swallowed whole with a small amount of water, milk, or other liquid.
- Do not crush, chew, or break guanfacine extended-release tablets. Tell your doctor if you cannot swallow guanfacine extended-release tablets whole.
- Do not take guanfacine extended-release tablets with a high-fat meal.
- Your doctor will check your blood pressure and heart rate while you take guanfacine extended-release tablets.
- If you take too much guanfacine extended-release tablets, call your local Poison Control Center at 1-800-222-1222 or go to the nearest emergency room right away.
What should I avoid while taking guanfacine extended-release tablets ?
- Do not drive, operate heavy machinery, or do other dangerous activities until you know how guanfacine extended-release tablets affects you. Guanfacine extended-release tablets can slow your thinking and motor skills.
- Do not drink alcohol or take other medicines that make you sleepy or dizzy while taking guanfacine extended-release tablets until you talk with your doctor. Guanfacine extended-release tablets taken with alcohol or medicines that cause sleepiness or dizziness may make your sleepiness or dizziness worse.
- Do not become dehydrated or overheated. This may increase your chance of having low blood pressure or fainting while taking guanfacine extended-release tablets.
- Do not suddenly stop guanfacine extended-release tablets. Tell your healthcare provider if you have been vomiting and cannot take guanfacine extended-release tablets, you may be at risk for rebound hypertension.
What are the possible side effects of guanfacine extended-release tablets ?
Guanfacine extended-release tablets may cause serious side effects including:
- low blood pressure
- low heart rate
- fainting
- sleepiness
- increased blood pressure and heart rate after suddenly stopping guanfacine extended-release tablets (rebound hypertension). Suddenly stopping guanfacine extended-release tablets can cause increased blood pressure and heart rate and other withdrawal symptoms such as headache, confusion, nervousness, agitation, and tremors. If these symptoms continue to get worse and are left untreated, it could lead to a very serious condition including very high blood pressure, feeling very sleepy or tired, severe headache, vomiting, vision problems, seizures.
Get medical help right away, if you have any of the symptoms listed above.
The most common side effects of guanfacine extended-release tablets include:
- sleepiness
- tiredness
- trouble sleeping
- low blood pressure
- nausea
- stomach pain
- dizziness
- dry mouth
- irritability
- vomiting
- slow heart rate
Tell the doctor if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of guanfacine extended-release tablets. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store guanfacine extended-release tablets ?
- Store guanfacine extended-release tablets between 68°F to 77°F (20°C to 25°C)
Keep guanfacine extended-release tablets and all medicines out of the reach of children.
General Information about the safe and effective use guanfacine extended-release tablets
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information Leaflet. Do not use guanfacine extended-release tablets for a condition for which it was not prescribed. Do not give guanfacine extended-release tablets to other people, even if they have the same symptoms that you have. It may harm them.
This leaflet summarizes the most important information about guanfacine extended-release tablets. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about guanfacine extended-release tablets that is written for health professionals.
For more information, call 1-866-611-3762.
What are the ingredients in guanfacine extended-release tablets ?
Active ingredient: guanfacine hydrochloride
Inactive ingredients: hypromellose, methacrylic acid and ethyl acrylate copolymer, lactose monohydrate, povidone, colloidal silicon dioxide, microcrystalline cellulose, fumaric acid, and glyceryl dibehenate. In addition, the 3 mg and 4 mg tablets also contain FD&C Blue No. 1 and FD&C Yellow No.5.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Distributed by:
Fosun Pharma USA Inc.
Princeton, NJ 08540
Made in China
Issued: November 2024
41-1133
Section 44425-7
Storage- Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). See USP Controlled Room Temperature.
Section 51945-4
NDC 72266-255-55
Guanfacine Extended-Release Tablets, USP
1 mg
Tablets should not be crushed,
chewed or broken before swallowing.
Rx only
100 Tablets
11 Description
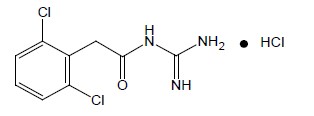
Guanfacine extended-release tablets, USP are once-daily, extended-release formulation of guanfacine hydrochloride (HCl) in a matrix tablet formulation for oral administration only. The chemical designation is N-amidino-2-(2,6-dichlorophenyl) acetamide monohydrochloride. The molecular formula is C 9H 9Cl 2N 3O∙HCl corresponding to a molecular weight of 282.55. The chemical structure is:
Guanfacine HCl is a white to off-white crystalline powder, sparingly soluble in water (approximately 1 mg/mL) and alcohol and slightly soluble in acetone. The only organic solvent in which it has relatively high solubility is methanol (>30 mg/mL). Each tablet contains guanfacine HCl equivalent to 1 mg, 2 mg, 3 mg, or 4 mg of guanfacine base. The tablets also contain hypromellose, methacrylic acid and ethyl acrylate copolymer, lactose monohydrate, povidone, colloidal silicon dioxide, microcrystalline cellulose, fumaric acid, and glyceryl dibehenate. In addition, the 3-mg and 4-mg tablets also contain FD&C Blue No. 1 and FD&C Yellow No. 5.
FDA approved dissolution test specifications differ from USP.
2.6 missed Doses
When reinitiating patients to the previous maintenance dose after two or more missed consecutive doses, consider titration based on patient tolerability.
8.4 Pediatric Use
Safety and efficacy of guanfacine extended-release tablets in pediatric patients less than 6 years of age have not been established. The efficacy of guanfacine extended-release tablets was studied for the treatment of ADHD in five controlled monotherapy clinical trials (up to 15 weeks in duration), one randomized withdrawal study and one controlled adjunctive trial with psychostimulants (8 weeks in duration) in children and adolescents ages 6-17 who met DSM-IV ®criteria for ADHD [see Adverse Reactions (6)and Clinical Studies (14)].
8.5 Geriatric Use
The safety and efficacy of guanfacine extended-release tablets in geriatric patients have not been established.
2.2 Dose Selection
Take guanfacine extended-release tablets orally once daily, either in the morning or evening, at approximately the same time each day. Begin at a dose of 1 mg/day, and adjust in increments of no more than 1 mg/week.
In monotherapy clinical trials, there was dose- and exposure-related clinical improvement as well as risks for several clinically significant adverse reactions (hypotension, bradycardia, sedative events). To balance the exposure-related potential benefits and risks, the recommended target dose range depending on clinical response and tolerability for guanfacine extended-release tablets is 0.05-0.12 mg/kg/day (total daily dose between 1-7 mg) (See Table 1).
| Weight | Target dose range (0.05 - 0.12 mg/kg/day) |
|---|---|
| Doses above 4 mg/day have not been evaluated in children (ages 6-12 years) and doses above 7 mg/day have not been evaluated in adolescents (ages 13-17 years) | |
| 25-33.9 kg | 2-3 mg/day |
| 34-41.4 kg | 2-4 mg/day |
| 41.5-49.4 kg | 3-5 mg/day |
| 49.5-58.4 kg | 3-6 mg/day |
| 58.5-91 kg | 4-7 mg/day |
| >91 kg | 5-7 mg/day |
In the adjunctive trial which evaluated guanfacine extended-release tablets treatment with psychostimulants, the majority of patients reached optimal doses in the 0.05-0.12 mg/kg/day range. Doses above 4 mg/day have not been studied in adjunctive trials.
14 Clinical Studies
Efficacy of guanfacine extended-release tablets in the treatment of ADHD was established in children and adolescents (6 to 17 years) in:
- Five short-term, placebo-controlled monotherapy trials (Studies 1, 2, 4, 5, and 6).
- One short-term, placebo-controlled adjunctive trial with psychostimulants (Study 3).
- One long-term, placebo-controlled monotherapy maintenance trial (Study 7).
4 Contraindications
Guanfacine extended-release tablets are contraindicated in patients with a history of a hypersensitivity reaction to guanfacine extended-release tablets or their inactive ingredients, or other products containing guanfacine. Rash and pruritus have been reported.
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling:
- Hypotension, bradycardia, and syncope [see Warnings and Precautions (5.1)]
- Sedation and somnolence [see Warnings and Precautions (5.2)]
- Cardiac conduction abnormalities [see Warnings and Precautions (5.3)]
- Rebound Hypertension [see Warnings and Precautions (5.4)]
7 Drug Interactions
Table 14 contains clinically important drug interactions with guanfacine extended-release tablets [see Clinical Pharmacology (12.3)].
| Concomitant Drug Name or Drug Class | Clinical Rationale and Magnitude of Drug Interaction | Clinical Recommendation |
|---|---|---|
| Strong and moderate CYP3A4 inhibitors, e.g., ketoconazole, fluconazole | Guanfacine is primarily metabolized by CYP3A4 and its plasma concentrations can be significantly affected resulting in an increase in exposure | Consider dose reduction [see Dosage and administration (2.7)] |
| Strong and moderate CYP3A4 inducers, e.g., rifampin, efavirenz | Guanfacine is primarily metabolized by CYP3A4 and its plasma concentrations can be significantly affected resulting in a decrease in exposure | Consider dose increase [see Dosage and administration (2.7)] |
8.6 Renal Impairment
It may be necessary to reduce the dosage in patients with significant impairment of renal function [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Guanfacine is a selective central alpha 2A-adrenergic receptor agonist in that it has a 15-20 times higher affinity for this receptor subtype than for the alpha 2B or alpha 2C subtypes.
Guanfacine is a known antihypertensive agent. By stimulating central alpha 2A-adrenergic receptors, guanfacine reduces sympathetic nerve impulses from the vasomotor center to the heart and blood vessels. This results in a decrease in peripheral vascular resistance and a reduction in heart rate.
In a thorough QT study, the administration of two dose levels of immediate-release guanfacine (4 mg and 8 mg) produced concentrations approximately 2 to 4 times the concentrations observed with the maximum recommended dose of guanfacine extended-release tablets of 0.12 mg/kg. Guanfacine was not shown to prolong the QTc interval to any clinically relevant extent.
8.7 Hepatic Impairment
It may be necessary to reduce the dosage in patients with significant impairment of hepatic function [see Clinical Pharmacology (12.3)].
1 Indications and Usage
Guanfacine extended-release tablets are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) as monotherapy and as adjunctive therapy to stimulant medications [see Clinical Studies (14)] .
12.1 Mechanism of Action
Guanfacine is a central alpha 2A-adrenergic receptor agonist. Guanfacine is not a central nervous system (CNS) stimulant. The mechanism of action of guanfacine in ADHD is not known.
5.4 Rebound Hypertension
In post marketing experience, abrupt discontinuation of guanfacine extended-release tablets has resulted in clinically significant and persistent rebound hypertension above baseline levels and increases in heart rate. Hypertensive encephalopathy has also been reported in association with rebound hypertension with both guanfacine extended-release tablets and immediate release guanfacine [see Adverse Reactions (6.2)] . In these cases, high-dosage guanfacine was discontinued; concomitant stimulant use was also reported, which may potentially increase hypertensive response upon abrupt discontinuation of guanfacine. Children commonly have gastrointestinal illnesses that lead to vomiting, and a resulting inability to take medications, so they may be especially at risk for rebound hypertension.
To minimize the risk of rebound hypertension upon discontinuation, the total daily dose of guanfacine extended-release tablets should be tapered in decrements of no more than 1 mg every 3 to 7 days [see Dosage and Administration (2.5)] . Blood pressure and heart rate should be monitored when reducing the dose or discontinuing guanfacine extended-release tablets. If abrupt discontinuation occurs (especially with concomitant stimulant use), patients should be closely followed for rebound hypertension.
9.1 Controlled Substance
Guanfacine extended-release tablets are not controlled substance and have no known potential for abuse or dependence.
2.4 Maintenance Treatment
Pharmacological treatment of ADHD may be needed for extended periods. Healthcare providers should periodically re-evaluate the long-term use of guanfacine extended-release tablets, and adjust weight-based dosage as needed. The majority of children and adolescents reach optimal doses in the 0.05-0.12 mg/kg/day range. Doses above 4 mg/day have not been evaluated in children (ages 6-12 years) and above 7 mg/day have not been evaluated in adolescents (ages 13-17 years) [see Clinical Studies (14)] .
5 Warnings and Precautions
- Hypotension, bradycardia, syncope: Titrate slowly and monitor vital signs frequently in patients at risk for hypotension, heart block, bradycardia, syncope, cardiovascular disease, vascular disease, cerebrovascular disease or chronic renal failure. Measure heart rate and blood pressure prior to initiation of therapy, following dose increases, and periodically while on therapy. Avoid concomitant use of drugs with additive effects unless clinically indicated. Advise patients to avoid becoming dehydrated or overheated ( 5.1).
- Sedation and somnolence: Occur commonly with guanfacine extended-release tablets. Consider the potential for additive sedative effects with CNS depressant drugs. Caution patients against operating heavy equipment or driving until they know how they respond to guanfacine extended-release tablets ( 5.2).
- Cardiac Conduction Abnormalities: May worsen sinus node dysfunction and atrioventricular (AV) block, especially in patients taking other sympatholytic drugs. Titrate slowly and monitor vital signs frequently ( 5.3).
- Rebound Hypertension: Abrupt discontinuation of guanfacine extended-release tablets can lead to clinically significant and persistent rebound hypertension. Subsequent hypertensive encephalopathy was also reported. To minimize the risk of rebound hypertension upon discontinuation, the total daily dose of guanfacine extended-release tablets should be tapered in decrements of no more than 1 mg every 3 to 7 days ( 5.4).
2 Dosage and Administration
- Recommended dose: 1 mg to 7 mg (0.05-0.12 mg/kg target weight based dose range) once daily in the morning or evening based on clinical response and tolerability ( 2.2).
- Begin at a dose of 1 mg once daily and adjust in increments of no more than 1 mg/week ( 2.2).
- Do not crush, chew or break tablets before swallowing ( 2.1).
- Do not administer with high-fat meals, because of increased exposure ( 2.1).
- Do not substitute for immediate-release guanfacine tablets on a mg-per-mg basis, because of differing pharmacokinetic profiles ( 2.3).
- If switching from immediate-release guanfacine, discontinue that treatment and titrate with guanfacine extended-release tablets as directed ( 2.3).
- When discontinuing, taper the dose in decrements of no more than 1 mg every 3 to 7 days to avoid rebound hypertension ( 2.5).
5.2 Sedation and Somnolence
Somnolence and sedation were commonly reported adverse reactions in clinical studies [see Adverse Reactions (6.1)] . Before using guanfacine extended-release tablets with other centrally active depressants, consider the potential for additive sedative effects. Caution patients against operating heavy equipment or driving until they know how they respond to treatment with guanfacine extended-release tablets .Advise patients to avoid use with alcohol.
3 Dosage Forms and Strengths
1 mg, 2 mg, 3 mg and 4 mg extended-release tablets
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of guanfacine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Less frequent, possibly guanfacine-related events observed in the post-marketing study and/or reported spontaneously, not included in section 6.1, include:
General: edema, malaise, tremor
Cardiovascular: palpitations, tachycardia, rebound hypertension, hypertensive encephalopathy
Central Nervous System: paresthesias, vertigo
Eye Disorders: blurred vision
Musculo-Skeletal System: arthralgia, leg cramps, leg pain, myalgia
Psychiatric: confusion, hallucinations
Reproductive System, Male: erectile dysfunction
Respiratory System: dyspnea
Skin and Appendages: alopecia, dermatitis, exfoliative dermatitis, pruritus, rash
Special Senses: alterations in taste
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect clinical trial exposure to guanfacine extended-release tablets in 2,825 patients. This includes 2,330 patients from completed studies in children and adolescents, ages 6 to 17 years and 495 patients in completed studies in adult healthy volunteers.
The mean duration of exposure of 446 patients that previously participated in two 2-year, open-label long-term studies was approximately 10 months.
2.1 General Instruction for Use
Swallow tablets whole. Do not crush, chew, or break tablets because this will increase the rate of guanfacine release. Do not administer with high fat meals, due to increased exposure.
2.5 Discontinuation of Treatment
Following discontinuation of guanfacine extended-release tablets, patients may experience increases in blood pressure and heart rate [see Warnings and Precautions (5.4)and Adverse Reactions (6)] . Patients/caregivers should be instructed not to discontinue guanfacine extended-release tablets without consulting their health care provider. Monitor blood pressure and pulse when reducing the dose or discontinuing the drug. Taper the daily dose in decrements of no more than 1 mg every 3 to 7 days to minimize the risk of rebound hypertension.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling ( Patient Information).
16 How Supplied/storage and Handling
Guanfacine extended-release tablets, USP are supplied in 1 mg, 2 mg, 3 mg, and 4 mg strength extended-release tablets in 100 count bottles.
| 1 mg | 2 mg | 3 mg | 4 mg | |
|---|---|---|---|---|
| Color | White/off-white | White/off-white | Green | Green |
| Shape | Round | Caplet | Round | Caplet |
|
Debossment
(top/bottom) |
F152 / Plain | F153 / Plain | F154 / Plain | F155 / Plain |
| NDC number | 72266-255-55 | 72266-256-55 | 72266-257-55 | 72266-258-55 |
5.3 Cardiac Conduction Abnormalities
The sympatholytic action of guanfacine extended-release tablets may worsen sinus node dysfunction and atrioventricular (AV) block, especially in patients taking other sympatholytic drugs. Titrate guanfacine extended-release tablets slowly and monitor vital signs frequently in patients with cardiac conduction abnormalities or patients concomitantly treated with other sympatholytic drugs.
5.1 Hypotension, Bradycardia, and Syncope
Treatment with guanfacine extended-release tablets can cause dose-dependent decreases in blood pressure and heart rate. Decreases were less pronounced over time of treatment. Orthostatic hypotension and syncope have been reported [see Adverse Reactions (6.1)] .
Measure heart rate and blood pressure prior to initiation of therapy, following dose increases, and periodically while on therapy. Titrate guanfacine extended-release tablets slowly in patients with a history of hypotension, and those with underlying conditions that may be worsened by hypotension and bradycardia; e.g., heart block, bradycardia, cardiovascular disease, vascular disease, cerebrovascular disease, or chronic renal failure. In patients who have a history of syncope or may have a condition that predisposes them to syncope, such as hypotension, orthostatic hypotension, bradycardia, or dehydration, advise patients to avoid becoming dehydrated or overheated. Monitor blood pressure and heart rate, and adjust dosages accordingly in patients treated concomitantly with antihypertensives or other drugs that can reduce blood pressure or heart rate or increase the risk of syncope.
5.5 Risk of Allergic Reactions Due to Tartrazine
This product contains FD&C Yellow No. 5 (tartrazine) which may cause allergic-type reactions (including bronchial asthma) in certain susceptible persons. Although the overall incidence of FD&C Yellow No. 5 (tartrazine) sensitivity in the general population is low, it is frequently seen in patients who also have aspirin hypersensitivity.
2.3 Switching From Immediate Release Guanfacine to Guanfacine Extended Release Tablets
If switching from immediate-release guanfacine, discontinue that treatment, and titrate with guanfacine extended-release tablets following above recommended schedule.
Do not substitute for immediate-release guanfacine tablets on a milligram-per-milligram basis, because of differing pharmacokinetic profiles. Guanfacine extended-release tablets have significantly reduced C max (60% lower), bioavailability (43% lower), and a delayed T max (3 hours later) compared to those of the same dose of immediate-release guanfacine [see Clinical Pharmacology (12.3)] .
2.7 Dosage Adjustment With Concomitant Use of Strong and Moderate Cyp3a4 Inhibitors Or Inducers
Dosage adjustments for guanfacine extended-release tablets are recommended with concomitant use of strong and moderate CYP3A4 inhibitors (e.g., ketoconazole), or CYP3A4 inducers (e.g., carbamazepine) (Table 2) [see Drug Interactions (7)] .
| Clinical Scenarios | |||
|---|---|---|---|
| Starting guanfacine extended-release tablets while currently on a CYP3A4 modulator | Continuing guanfacine extended-release tablets while adding a CYP3A4 modulator | Continuing guanfacine extended-release tablets while stopping a CYP3A4 modulator | |
|
CYP3A4
Strong and moderate Inhibitors |
Decrease guanfacine extended-release tablets dosage to half the recommended level.
(see Table 1) |
Decrease guanfacine extended-release tablets dosage to half the recommended level.
(see Table 1) |
Increase guanfacine extended-release tablets dosage to recommended level.
(see Table 1) |
|
CYP3A4
Strong and moderate Inducers |
Consider increasing guanfacine extended-release tablets dosage up to double the recommended level.
(see Table 1) |
Consider increasing guanfacine extended-release tablets dosage up to double the recommended level over 1 to 2 weeks.
(see Table 1) |
Decrease guanfacine extended-release tablets dosage to recommended level over 1 to 2 weeks.
(see Table 1) |
Structured Label Content
Section 42229-5 (42229-5)
Fixed Dose Trials
| Guanfacine Extended-Release Tablets (mg) | ||||||
|---|---|---|---|---|---|---|
| Adverse Reaction Term | Placebo
(N=149) |
1mg*
(N=61) |
2mg
(N=150) |
3mg
(N=151) |
4mg
(N=151) |
All Doses of Guanfacine Extended-Release Tablets (N=513) |
| *The lowest dose of 1 mg used in Study 2 was not randomized to patients weighing more than 50 kg.
a: The somnolence term includes somnolence, sedation, and hypersomnia. b: The hypotension term includes hypotension, diastolic hypotension, orthostatic hypotension, blood pressure decreased, blood pressure diastolic decreased, blood pressure systolic decreased). |
||||||
| Somnolence a | 11% | 28% | 30% | 38% | 51% | 38% |
| Fatigue | 3% | 10% | 13% | 17% | 15% | 14% |
| Hypotension b | 3% | 8% | 5% | 7% | 8% | 7% |
| Dizziness | 4% | 5% | 3% | 7% | 10% | 6% |
| Lethargy | 3% | 2% | 3% | 8% | 7% | 6% |
| Nausea | 2% | 7% | 5% | 5% | 6% | 6% |
| Dry mouth | 1% | 0% | 1% | 6% | 7% | 4% |
| Guanfacine Extended-Release Tablets (mg) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adverse Reaction Term | Placebo
(N=149) |
1mg*
(N=61) |
2mg
(N=150) |
3mg
(N=151) |
4mg
(N=151) |
All Doses of Guanfacine Extended-Release Tablets (N=513) | ||||||
| n | (%) | n | (%) | n | (%) | n | (%) | n | (%) | n | (%) | |
| Adverse reactions leading to discontinuation in ≥2% in any dose group but did not meet this criteria in all doses combined: hypotension (hypotension, diastolic hypotension, orthostatic hypotension, blood pressure decreased, blood pressure diastolic decreased, blood pressure systolic decreased), headache, and dizziness.
* The lowest dose of 1 mg used in Study 2 was not randomized to patients weighing more than 50 kg. a: The somnolence term includes somnolence, sedation, and hypersomnia. |
||||||||||||
| Total patients | 4 | (3%) | 2 | (3%) | 10 | (7%) | 15 | (10%) | 27 | (18%) | 54 | (11%) |
| Somnolence a | 1 | (1%) | 2 | (3%) | 5 | (3%) | 6 | (4%) | 17 | (11%) | 30 | (6%) |
| Fatigue | 0 | (0%) | 0 | (0%) | 2 | (1%) | 2 | (1%) | 4 | (3%) | 8 | (2%) |
| Guanfacine Extended-Release Tablets (mg) | ||||||
|---|---|---|---|---|---|---|
| Adverse Reaction Term | Placebo
(N=149) |
1mg*
(N=61) |
2mg
(N=150) |
3mg
(N=151) |
4mg
(N=151) |
All Doses of Guanfacine Extended-Release Tablets (N=513) |
| Adverse reactions ≥2% for all doses of guanfacine extend-release tablets and >rate in placebo in any dose group but did not meet this criteria in all doses combined: insomnia (insomnia, initial insomnia, middle insomnia, terminal insomnia, sleep disorder), vomiting, diarrhea, abdominal/stomach discomfort (abdominal discomfort, epigastric discomfort, stomach discomfort), rash (rash, rash generalized, rash papular), dyspepsia, increased weight, bradycardia (bradycardia, sinus bradycardia), asthma (asthma, bronchospasm, wheezing), agitation, anxiety (anxiety, nervousness), sinus arrhythmia, blood pressure increased (blood pressure increased, blood pressure diastolic increased), and first degree atrioventricular block.
* The lowest dose of 1 mg used in Study 2 was not randomized to patients weighing more than 50 kg. a: The abdominal pain term includes abdominal pain, abdominal pain lower, abdominal pain upper, and abdominal tenderness. b: The nightmare term includes abnormal dreams, nightmare, and sleep terror. c: The enuresis term includes enuresis, nocturia, and urinary incontinence. d: The affect lability term includes affect lability and mood swings. |
||||||
| Headache | 19% | 26% | 25% | 16% | 28% | 23% |
| Abdominal Pain a | 9% | 10% | 7% | 11% | 15% | 11% |
| Decreased Appetite | 4% | 5% | 4% | 9% | 6% | 6% |
| Irritability | 4% | 5% | 8% | 3% | 7% | 6% |
| Constipation | 1% | 2% | 2% | 3% | 4% | 3% |
| Nightmare b | 0% | 0% | 0% | 3% | 4% | 2% |
| Enuresis c | 1% | 0% | 1% | 3% | 2% | 2% |
| Affect Lability d | 1% | 2% | 1% | 3% | 1% | 2% |
Section 42230-3 (42230-3)
Patient Information
Guanfacine (gwahn-fa-seen)
Extended-Release Tablets
Read the Patient Information that comes with guanfacine extended-release tablets before you start taking it and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your doctor about your medical condition or your treatment.
What is guanfacine extended-release tablets ?
Guanfacine extended-release tablets are prescription medicine used to treat the symptoms of attention deficit hyperactivity disorder (ADHD). Guanfacine extended-release tablets may be used alone or with ADHD stimulant medicines.
Guanfacine extended-release tablets are not central nervous system (CNS) stimulant.
It is not known if guanfacine extended-release tablets are safe and effective in children younger than 6 years of age.
Who should not take guanfacine extended-release tablets ?
Do not take guanfacine extended-release tablets if you are allergic to guanfacine or any of the ingredients in guanfacine extended-release tablets. See the end of this leaflet for a complete list of ingredients in guanfacine extended-release tablets.
What should I tell my doctor before taking guanfacine extended-release tablets ?
Before you take guanfacine extended-release tablets , tell your doctor if you:
- have heart problems or a low heart rate
- have fainted
- have low or high blood pressure
- have liver or kidney problems
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if guanfacine extended-release tablets will harm your unborn baby. Talk to your doctor if you are pregnant or plan to become pregnant.
- are breastfeeding or plan to breastfeed. It is not known if guanfacine passes into your breast milk. Talk to your doctor about the best way to feed your baby while taking guanfacine extended-release tablets.
Tell your doctor about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Guanfacine extended-release tablets may affect the way other medicines work, and other medicines may affect how guanfacine extended-release tablets work.
Especially tell your doctor if you take:
- ketoconazole
- medicines that can affect enzyme metabolism
- high blood pressure medicine
- sedatives
- benzodiazepines
- barbiturates
- antipsychotics
Ask your doctor or pharmacist for a list of these medicines, if you are not sure.
Know the medicines you take. Keep a list of them and show it to your doctor and pharmacist when you get a new medicine.
How should I take guanfacine extended-release tablets ?
- Take guanfacine extended-release tablets exactly as your doctor tells you.
- Your doctor may change your dose. Do not change your dose of guanfacine extended-release tablets without talking to your doctor.
- Do not stop taking guanfacine extended-release tablets without talking to your doctor.
- Try not to miss your dose of guanfacine extended-release tablets. If you miss a dose of guanfacine extended-release tablets, take the next dose at your regular time. If you miss 2 or more doses, talk to your doctor, as you may need to restart guanfacine extended-release tablets with a lower dose.
- Do not take a double dose to make up for a missed dose.
- Guanfacine extended-release tablets should be taken 1 time a day in the morning or in the evening, either alone or in combination with an ADHD stimulant medicine that your doctor may prescribe. Your doctor will tell you when to take guanfacine extended-release tablets and when to take your ADHD stimulant medication.
- Guanfacine extended-release tablets should be swallowed whole with a small amount of water, milk, or other liquid.
- Do not crush, chew, or break guanfacine extended-release tablets. Tell your doctor if you cannot swallow guanfacine extended-release tablets whole.
- Do not take guanfacine extended-release tablets with a high-fat meal.
- Your doctor will check your blood pressure and heart rate while you take guanfacine extended-release tablets.
- If you take too much guanfacine extended-release tablets, call your local Poison Control Center at 1-800-222-1222 or go to the nearest emergency room right away.
What should I avoid while taking guanfacine extended-release tablets ?
- Do not drive, operate heavy machinery, or do other dangerous activities until you know how guanfacine extended-release tablets affects you. Guanfacine extended-release tablets can slow your thinking and motor skills.
- Do not drink alcohol or take other medicines that make you sleepy or dizzy while taking guanfacine extended-release tablets until you talk with your doctor. Guanfacine extended-release tablets taken with alcohol or medicines that cause sleepiness or dizziness may make your sleepiness or dizziness worse.
- Do not become dehydrated or overheated. This may increase your chance of having low blood pressure or fainting while taking guanfacine extended-release tablets.
- Do not suddenly stop guanfacine extended-release tablets. Tell your healthcare provider if you have been vomiting and cannot take guanfacine extended-release tablets, you may be at risk for rebound hypertension.
What are the possible side effects of guanfacine extended-release tablets ?
Guanfacine extended-release tablets may cause serious side effects including:
- low blood pressure
- low heart rate
- fainting
- sleepiness
- increased blood pressure and heart rate after suddenly stopping guanfacine extended-release tablets (rebound hypertension). Suddenly stopping guanfacine extended-release tablets can cause increased blood pressure and heart rate and other withdrawal symptoms such as headache, confusion, nervousness, agitation, and tremors. If these symptoms continue to get worse and are left untreated, it could lead to a very serious condition including very high blood pressure, feeling very sleepy or tired, severe headache, vomiting, vision problems, seizures.
Get medical help right away, if you have any of the symptoms listed above.
The most common side effects of guanfacine extended-release tablets include:
- sleepiness
- tiredness
- trouble sleeping
- low blood pressure
- nausea
- stomach pain
- dizziness
- dry mouth
- irritability
- vomiting
- slow heart rate
Tell the doctor if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of guanfacine extended-release tablets. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store guanfacine extended-release tablets ?
- Store guanfacine extended-release tablets between 68°F to 77°F (20°C to 25°C)
Keep guanfacine extended-release tablets and all medicines out of the reach of children.
General Information about the safe and effective use guanfacine extended-release tablets
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information Leaflet. Do not use guanfacine extended-release tablets for a condition for which it was not prescribed. Do not give guanfacine extended-release tablets to other people, even if they have the same symptoms that you have. It may harm them.
This leaflet summarizes the most important information about guanfacine extended-release tablets. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about guanfacine extended-release tablets that is written for health professionals.
For more information, call 1-866-611-3762.
What are the ingredients in guanfacine extended-release tablets ?
Active ingredient: guanfacine hydrochloride
Inactive ingredients: hypromellose, methacrylic acid and ethyl acrylate copolymer, lactose monohydrate, povidone, colloidal silicon dioxide, microcrystalline cellulose, fumaric acid, and glyceryl dibehenate. In addition, the 3 mg and 4 mg tablets also contain FD&C Blue No. 1 and FD&C Yellow No.5.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Distributed by:
Fosun Pharma USA Inc.
Princeton, NJ 08540
Made in China
Issued: November 2024
41-1133
Section 44425-7 (44425-7)
Storage- Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). See USP Controlled Room Temperature.
Section 51945-4 (51945-4)
NDC 72266-255-55
Guanfacine Extended-Release Tablets, USP
1 mg
Tablets should not be crushed,
chewed or broken before swallowing.
Rx only
100 Tablets
11 Description (11 DESCRIPTION)
Guanfacine extended-release tablets, USP are once-daily, extended-release formulation of guanfacine hydrochloride (HCl) in a matrix tablet formulation for oral administration only. The chemical designation is N-amidino-2-(2,6-dichlorophenyl) acetamide monohydrochloride. The molecular formula is C 9H 9Cl 2N 3O∙HCl corresponding to a molecular weight of 282.55. The chemical structure is:
Guanfacine HCl is a white to off-white crystalline powder, sparingly soluble in water (approximately 1 mg/mL) and alcohol and slightly soluble in acetone. The only organic solvent in which it has relatively high solubility is methanol (>30 mg/mL). Each tablet contains guanfacine HCl equivalent to 1 mg, 2 mg, 3 mg, or 4 mg of guanfacine base. The tablets also contain hypromellose, methacrylic acid and ethyl acrylate copolymer, lactose monohydrate, povidone, colloidal silicon dioxide, microcrystalline cellulose, fumaric acid, and glyceryl dibehenate. In addition, the 3-mg and 4-mg tablets also contain FD&C Blue No. 1 and FD&C Yellow No. 5.
FDA approved dissolution test specifications differ from USP.
2.6 missed Doses (2.6 Missed Doses)
When reinitiating patients to the previous maintenance dose after two or more missed consecutive doses, consider titration based on patient tolerability.
8.4 Pediatric Use
Safety and efficacy of guanfacine extended-release tablets in pediatric patients less than 6 years of age have not been established. The efficacy of guanfacine extended-release tablets was studied for the treatment of ADHD in five controlled monotherapy clinical trials (up to 15 weeks in duration), one randomized withdrawal study and one controlled adjunctive trial with psychostimulants (8 weeks in duration) in children and adolescents ages 6-17 who met DSM-IV ®criteria for ADHD [see Adverse Reactions (6)and Clinical Studies (14)].
8.5 Geriatric Use
The safety and efficacy of guanfacine extended-release tablets in geriatric patients have not been established.
2.2 Dose Selection
Take guanfacine extended-release tablets orally once daily, either in the morning or evening, at approximately the same time each day. Begin at a dose of 1 mg/day, and adjust in increments of no more than 1 mg/week.
In monotherapy clinical trials, there was dose- and exposure-related clinical improvement as well as risks for several clinically significant adverse reactions (hypotension, bradycardia, sedative events). To balance the exposure-related potential benefits and risks, the recommended target dose range depending on clinical response and tolerability for guanfacine extended-release tablets is 0.05-0.12 mg/kg/day (total daily dose between 1-7 mg) (See Table 1).
| Weight | Target dose range (0.05 - 0.12 mg/kg/day) |
|---|---|
| Doses above 4 mg/day have not been evaluated in children (ages 6-12 years) and doses above 7 mg/day have not been evaluated in adolescents (ages 13-17 years) | |
| 25-33.9 kg | 2-3 mg/day |
| 34-41.4 kg | 2-4 mg/day |
| 41.5-49.4 kg | 3-5 mg/day |
| 49.5-58.4 kg | 3-6 mg/day |
| 58.5-91 kg | 4-7 mg/day |
| >91 kg | 5-7 mg/day |
In the adjunctive trial which evaluated guanfacine extended-release tablets treatment with psychostimulants, the majority of patients reached optimal doses in the 0.05-0.12 mg/kg/day range. Doses above 4 mg/day have not been studied in adjunctive trials.
14 Clinical Studies (14 CLINICAL STUDIES)
Efficacy of guanfacine extended-release tablets in the treatment of ADHD was established in children and adolescents (6 to 17 years) in:
- Five short-term, placebo-controlled monotherapy trials (Studies 1, 2, 4, 5, and 6).
- One short-term, placebo-controlled adjunctive trial with psychostimulants (Study 3).
- One long-term, placebo-controlled monotherapy maintenance trial (Study 7).
4 Contraindications (4 CONTRAINDICATIONS)
Guanfacine extended-release tablets are contraindicated in patients with a history of a hypersensitivity reaction to guanfacine extended-release tablets or their inactive ingredients, or other products containing guanfacine. Rash and pruritus have been reported.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in the labeling:
- Hypotension, bradycardia, and syncope [see Warnings and Precautions (5.1)]
- Sedation and somnolence [see Warnings and Precautions (5.2)]
- Cardiac conduction abnormalities [see Warnings and Precautions (5.3)]
- Rebound Hypertension [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Table 14 contains clinically important drug interactions with guanfacine extended-release tablets [see Clinical Pharmacology (12.3)].
| Concomitant Drug Name or Drug Class | Clinical Rationale and Magnitude of Drug Interaction | Clinical Recommendation |
|---|---|---|
| Strong and moderate CYP3A4 inhibitors, e.g., ketoconazole, fluconazole | Guanfacine is primarily metabolized by CYP3A4 and its plasma concentrations can be significantly affected resulting in an increase in exposure | Consider dose reduction [see Dosage and administration (2.7)] |
| Strong and moderate CYP3A4 inducers, e.g., rifampin, efavirenz | Guanfacine is primarily metabolized by CYP3A4 and its plasma concentrations can be significantly affected resulting in a decrease in exposure | Consider dose increase [see Dosage and administration (2.7)] |
8.6 Renal Impairment
It may be necessary to reduce the dosage in patients with significant impairment of renal function [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Guanfacine is a selective central alpha 2A-adrenergic receptor agonist in that it has a 15-20 times higher affinity for this receptor subtype than for the alpha 2B or alpha 2C subtypes.
Guanfacine is a known antihypertensive agent. By stimulating central alpha 2A-adrenergic receptors, guanfacine reduces sympathetic nerve impulses from the vasomotor center to the heart and blood vessels. This results in a decrease in peripheral vascular resistance and a reduction in heart rate.
In a thorough QT study, the administration of two dose levels of immediate-release guanfacine (4 mg and 8 mg) produced concentrations approximately 2 to 4 times the concentrations observed with the maximum recommended dose of guanfacine extended-release tablets of 0.12 mg/kg. Guanfacine was not shown to prolong the QTc interval to any clinically relevant extent.
8.7 Hepatic Impairment
It may be necessary to reduce the dosage in patients with significant impairment of hepatic function [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Guanfacine extended-release tablets are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) as monotherapy and as adjunctive therapy to stimulant medications [see Clinical Studies (14)] .
12.1 Mechanism of Action
Guanfacine is a central alpha 2A-adrenergic receptor agonist. Guanfacine is not a central nervous system (CNS) stimulant. The mechanism of action of guanfacine in ADHD is not known.
5.4 Rebound Hypertension
In post marketing experience, abrupt discontinuation of guanfacine extended-release tablets has resulted in clinically significant and persistent rebound hypertension above baseline levels and increases in heart rate. Hypertensive encephalopathy has also been reported in association with rebound hypertension with both guanfacine extended-release tablets and immediate release guanfacine [see Adverse Reactions (6.2)] . In these cases, high-dosage guanfacine was discontinued; concomitant stimulant use was also reported, which may potentially increase hypertensive response upon abrupt discontinuation of guanfacine. Children commonly have gastrointestinal illnesses that lead to vomiting, and a resulting inability to take medications, so they may be especially at risk for rebound hypertension.
To minimize the risk of rebound hypertension upon discontinuation, the total daily dose of guanfacine extended-release tablets should be tapered in decrements of no more than 1 mg every 3 to 7 days [see Dosage and Administration (2.5)] . Blood pressure and heart rate should be monitored when reducing the dose or discontinuing guanfacine extended-release tablets. If abrupt discontinuation occurs (especially with concomitant stimulant use), patients should be closely followed for rebound hypertension.
9.1 Controlled Substance
Guanfacine extended-release tablets are not controlled substance and have no known potential for abuse or dependence.
2.4 Maintenance Treatment
Pharmacological treatment of ADHD may be needed for extended periods. Healthcare providers should periodically re-evaluate the long-term use of guanfacine extended-release tablets, and adjust weight-based dosage as needed. The majority of children and adolescents reach optimal doses in the 0.05-0.12 mg/kg/day range. Doses above 4 mg/day have not been evaluated in children (ages 6-12 years) and above 7 mg/day have not been evaluated in adolescents (ages 13-17 years) [see Clinical Studies (14)] .
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypotension, bradycardia, syncope: Titrate slowly and monitor vital signs frequently in patients at risk for hypotension, heart block, bradycardia, syncope, cardiovascular disease, vascular disease, cerebrovascular disease or chronic renal failure. Measure heart rate and blood pressure prior to initiation of therapy, following dose increases, and periodically while on therapy. Avoid concomitant use of drugs with additive effects unless clinically indicated. Advise patients to avoid becoming dehydrated or overheated ( 5.1).
- Sedation and somnolence: Occur commonly with guanfacine extended-release tablets. Consider the potential for additive sedative effects with CNS depressant drugs. Caution patients against operating heavy equipment or driving until they know how they respond to guanfacine extended-release tablets ( 5.2).
- Cardiac Conduction Abnormalities: May worsen sinus node dysfunction and atrioventricular (AV) block, especially in patients taking other sympatholytic drugs. Titrate slowly and monitor vital signs frequently ( 5.3).
- Rebound Hypertension: Abrupt discontinuation of guanfacine extended-release tablets can lead to clinically significant and persistent rebound hypertension. Subsequent hypertensive encephalopathy was also reported. To minimize the risk of rebound hypertension upon discontinuation, the total daily dose of guanfacine extended-release tablets should be tapered in decrements of no more than 1 mg every 3 to 7 days ( 5.4).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended dose: 1 mg to 7 mg (0.05-0.12 mg/kg target weight based dose range) once daily in the morning or evening based on clinical response and tolerability ( 2.2).
- Begin at a dose of 1 mg once daily and adjust in increments of no more than 1 mg/week ( 2.2).
- Do not crush, chew or break tablets before swallowing ( 2.1).
- Do not administer with high-fat meals, because of increased exposure ( 2.1).
- Do not substitute for immediate-release guanfacine tablets on a mg-per-mg basis, because of differing pharmacokinetic profiles ( 2.3).
- If switching from immediate-release guanfacine, discontinue that treatment and titrate with guanfacine extended-release tablets as directed ( 2.3).
- When discontinuing, taper the dose in decrements of no more than 1 mg every 3 to 7 days to avoid rebound hypertension ( 2.5).
5.2 Sedation and Somnolence
Somnolence and sedation were commonly reported adverse reactions in clinical studies [see Adverse Reactions (6.1)] . Before using guanfacine extended-release tablets with other centrally active depressants, consider the potential for additive sedative effects. Caution patients against operating heavy equipment or driving until they know how they respond to treatment with guanfacine extended-release tablets .Advise patients to avoid use with alcohol.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
1 mg, 2 mg, 3 mg and 4 mg extended-release tablets
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of guanfacine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Less frequent, possibly guanfacine-related events observed in the post-marketing study and/or reported spontaneously, not included in section 6.1, include:
General: edema, malaise, tremor
Cardiovascular: palpitations, tachycardia, rebound hypertension, hypertensive encephalopathy
Central Nervous System: paresthesias, vertigo
Eye Disorders: blurred vision
Musculo-Skeletal System: arthralgia, leg cramps, leg pain, myalgia
Psychiatric: confusion, hallucinations
Reproductive System, Male: erectile dysfunction
Respiratory System: dyspnea
Skin and Appendages: alopecia, dermatitis, exfoliative dermatitis, pruritus, rash
Special Senses: alterations in taste
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect clinical trial exposure to guanfacine extended-release tablets in 2,825 patients. This includes 2,330 patients from completed studies in children and adolescents, ages 6 to 17 years and 495 patients in completed studies in adult healthy volunteers.
The mean duration of exposure of 446 patients that previously participated in two 2-year, open-label long-term studies was approximately 10 months.
2.1 General Instruction for Use
Swallow tablets whole. Do not crush, chew, or break tablets because this will increase the rate of guanfacine release. Do not administer with high fat meals, due to increased exposure.
2.5 Discontinuation of Treatment
Following discontinuation of guanfacine extended-release tablets, patients may experience increases in blood pressure and heart rate [see Warnings and Precautions (5.4)and Adverse Reactions (6)] . Patients/caregivers should be instructed not to discontinue guanfacine extended-release tablets without consulting their health care provider. Monitor blood pressure and pulse when reducing the dose or discontinuing the drug. Taper the daily dose in decrements of no more than 1 mg every 3 to 7 days to minimize the risk of rebound hypertension.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling ( Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Guanfacine extended-release tablets, USP are supplied in 1 mg, 2 mg, 3 mg, and 4 mg strength extended-release tablets in 100 count bottles.
| 1 mg | 2 mg | 3 mg | 4 mg | |
|---|---|---|---|---|
| Color | White/off-white | White/off-white | Green | Green |
| Shape | Round | Caplet | Round | Caplet |
|
Debossment
(top/bottom) |
F152 / Plain | F153 / Plain | F154 / Plain | F155 / Plain |
| NDC number | 72266-255-55 | 72266-256-55 | 72266-257-55 | 72266-258-55 |
5.3 Cardiac Conduction Abnormalities
The sympatholytic action of guanfacine extended-release tablets may worsen sinus node dysfunction and atrioventricular (AV) block, especially in patients taking other sympatholytic drugs. Titrate guanfacine extended-release tablets slowly and monitor vital signs frequently in patients with cardiac conduction abnormalities or patients concomitantly treated with other sympatholytic drugs.
5.1 Hypotension, Bradycardia, and Syncope
Treatment with guanfacine extended-release tablets can cause dose-dependent decreases in blood pressure and heart rate. Decreases were less pronounced over time of treatment. Orthostatic hypotension and syncope have been reported [see Adverse Reactions (6.1)] .
Measure heart rate and blood pressure prior to initiation of therapy, following dose increases, and periodically while on therapy. Titrate guanfacine extended-release tablets slowly in patients with a history of hypotension, and those with underlying conditions that may be worsened by hypotension and bradycardia; e.g., heart block, bradycardia, cardiovascular disease, vascular disease, cerebrovascular disease, or chronic renal failure. In patients who have a history of syncope or may have a condition that predisposes them to syncope, such as hypotension, orthostatic hypotension, bradycardia, or dehydration, advise patients to avoid becoming dehydrated or overheated. Monitor blood pressure and heart rate, and adjust dosages accordingly in patients treated concomitantly with antihypertensives or other drugs that can reduce blood pressure or heart rate or increase the risk of syncope.
5.5 Risk of Allergic Reactions Due to Tartrazine
This product contains FD&C Yellow No. 5 (tartrazine) which may cause allergic-type reactions (including bronchial asthma) in certain susceptible persons. Although the overall incidence of FD&C Yellow No. 5 (tartrazine) sensitivity in the general population is low, it is frequently seen in patients who also have aspirin hypersensitivity.
2.3 Switching From Immediate Release Guanfacine to Guanfacine Extended Release Tablets (2.3 Switching from Immediate-Release Guanfacine to Guanfacine Extended-Release Tablets)
If switching from immediate-release guanfacine, discontinue that treatment, and titrate with guanfacine extended-release tablets following above recommended schedule.
Do not substitute for immediate-release guanfacine tablets on a milligram-per-milligram basis, because of differing pharmacokinetic profiles. Guanfacine extended-release tablets have significantly reduced C max (60% lower), bioavailability (43% lower), and a delayed T max (3 hours later) compared to those of the same dose of immediate-release guanfacine [see Clinical Pharmacology (12.3)] .
2.7 Dosage Adjustment With Concomitant Use of Strong and Moderate Cyp3a4 Inhibitors Or Inducers (2.7 Dosage Adjustment with Concomitant Use of Strong and Moderate CYP3A4 Inhibitors or Inducers)
Dosage adjustments for guanfacine extended-release tablets are recommended with concomitant use of strong and moderate CYP3A4 inhibitors (e.g., ketoconazole), or CYP3A4 inducers (e.g., carbamazepine) (Table 2) [see Drug Interactions (7)] .
| Clinical Scenarios | |||
|---|---|---|---|
| Starting guanfacine extended-release tablets while currently on a CYP3A4 modulator | Continuing guanfacine extended-release tablets while adding a CYP3A4 modulator | Continuing guanfacine extended-release tablets while stopping a CYP3A4 modulator | |
|
CYP3A4
Strong and moderate Inhibitors |
Decrease guanfacine extended-release tablets dosage to half the recommended level.
(see Table 1) |
Decrease guanfacine extended-release tablets dosage to half the recommended level.
(see Table 1) |
Increase guanfacine extended-release tablets dosage to recommended level.
(see Table 1) |
|
CYP3A4
Strong and moderate Inducers |
Consider increasing guanfacine extended-release tablets dosage up to double the recommended level.
(see Table 1) |
Consider increasing guanfacine extended-release tablets dosage up to double the recommended level over 1 to 2 weeks.
(see Table 1) |
Decrease guanfacine extended-release tablets dosage to recommended level over 1 to 2 weeks.
(see Table 1) |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:03.936802 · Updated: 2026-03-14T22:28:55.050842