These Highlights Do Not Include All The Information Needed To Use Gemtesa®
25f21d25-14f8-4fda-91f6-7aa8b68aa1c8
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage, Overactive Bladder in Adult Males with Benign Prostatic Hyperplasia ( 1.2 ) 12/2024 Contraindications ( 4 ) 10/2024 Warnings and Precautions, Angioedema ( 5.2 ) 10/2024
Indications and Usage
GEMTESA is a beta-3 adrenergic agonist indicated for the treatment of: overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency in adults. ( 1.1 ) overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency in adult males on pharmacological therapy for benign prostatic hyperplasia (BPH). ( 1.2 )
Dosage and Administration
The recommended dose is one 75 mg tablet orally once daily. ( 2.1 ) Swallow tablet whole with water. ( 2.1 ) Tablet may be crushed and mixed with applesauce. ( 2.1 )
Warnings and Precautions
Urinary Retention : Monitor for urinary retention, especially in patients with bladder outlet obstruction and also in patients taking muscarinic antagonist medications for OAB, in whom the risk of urinary retention may be greater. If urinary retention develops, discontinue GEMTESA. ( 5.1 ) Angioedema : Angioedema of the face and/or larynx has been reported with GEMTESA. ( 5.2 )
Contraindications
GEMTESA is contraindicated in patients with known hypersensitivity to vibegron or any components of GEMTESA. Hypersensitivity reactions, such as angioedema, have occurred [see Warnings and Precautions ( 5.2 ) and Adverse Reactions ( 6.2 )] .
Adverse Reactions
The following clinically significant adverse reaction is described elsewhere in the labeling: Urinary retention [see Warnings and Precautions ( 5.1 )]
Drug Interactions
Concomitant use of GEMTESA increases digoxin maximal concentrations (C max ) and systemic exposure as assessed by area under the concentration-time curve (AUC) [see Clinical Pharmacology ( 12.3 )] . Serum digoxin concentrations should be monitored before initiating and during therapy with GEMTESA and used for titration of the digoxin dose to obtain the desired clinical effect. Continue monitoring digoxin concentrations upon discontinuation of GEMTESA and adjust digoxin dose as needed.
Storage and Handling
GEMTESA 75 mg tablets are light green, oval, film-coated tablets, debossed with V75 on one side and no debossing on the other side. GEMTESA is marketed in two packaging configurations: Thirty (30) tablets in a HDPE bottle with a child-resistant cap, NDC 73336-075-30 Ninety (90) tablets in a HDPE bottle with a child-resistant cap, NDC 73336-075-90 Store at 20°C to 25°C (68°F to 77°F), excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Keep this and all medications out of sight and reach of children. Dispose unused medication via a take-back option if available; otherwise follow FDA instructions for disposal in the household trash. See www.fda.gov/drugdisposal for more information.
How Supplied
GEMTESA 75 mg tablets are light green, oval, film-coated tablets, debossed with V75 on one side and no debossing on the other side. GEMTESA is marketed in two packaging configurations: Thirty (30) tablets in a HDPE bottle with a child-resistant cap, NDC 73336-075-30 Ninety (90) tablets in a HDPE bottle with a child-resistant cap, NDC 73336-075-90 Store at 20°C to 25°C (68°F to 77°F), excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Keep this and all medications out of sight and reach of children. Dispose unused medication via a take-back option if available; otherwise follow FDA instructions for disposal in the household trash. See www.fda.gov/drugdisposal for more information.
Medication Information
Warnings and Precautions
Urinary Retention : Monitor for urinary retention, especially in patients with bladder outlet obstruction and also in patients taking muscarinic antagonist medications for OAB, in whom the risk of urinary retention may be greater. If urinary retention develops, discontinue GEMTESA. ( 5.1 ) Angioedema : Angioedema of the face and/or larynx has been reported with GEMTESA. ( 5.2 )
Indications and Usage
GEMTESA is a beta-3 adrenergic agonist indicated for the treatment of: overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency in adults. ( 1.1 ) overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency in adult males on pharmacological therapy for benign prostatic hyperplasia (BPH). ( 1.2 )
Dosage and Administration
The recommended dose is one 75 mg tablet orally once daily. ( 2.1 ) Swallow tablet whole with water. ( 2.1 ) Tablet may be crushed and mixed with applesauce. ( 2.1 )
Contraindications
GEMTESA is contraindicated in patients with known hypersensitivity to vibegron or any components of GEMTESA. Hypersensitivity reactions, such as angioedema, have occurred [see Warnings and Precautions ( 5.2 ) and Adverse Reactions ( 6.2 )] .
Adverse Reactions
The following clinically significant adverse reaction is described elsewhere in the labeling: Urinary retention [see Warnings and Precautions ( 5.1 )]
Drug Interactions
Concomitant use of GEMTESA increases digoxin maximal concentrations (C max ) and systemic exposure as assessed by area under the concentration-time curve (AUC) [see Clinical Pharmacology ( 12.3 )] . Serum digoxin concentrations should be monitored before initiating and during therapy with GEMTESA and used for titration of the digoxin dose to obtain the desired clinical effect. Continue monitoring digoxin concentrations upon discontinuation of GEMTESA and adjust digoxin dose as needed.
Storage and Handling
GEMTESA 75 mg tablets are light green, oval, film-coated tablets, debossed with V75 on one side and no debossing on the other side. GEMTESA is marketed in two packaging configurations: Thirty (30) tablets in a HDPE bottle with a child-resistant cap, NDC 73336-075-30 Ninety (90) tablets in a HDPE bottle with a child-resistant cap, NDC 73336-075-90 Store at 20°C to 25°C (68°F to 77°F), excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Keep this and all medications out of sight and reach of children. Dispose unused medication via a take-back option if available; otherwise follow FDA instructions for disposal in the household trash. See www.fda.gov/drugdisposal for more information.
How Supplied
GEMTESA 75 mg tablets are light green, oval, film-coated tablets, debossed with V75 on one side and no debossing on the other side. GEMTESA is marketed in two packaging configurations: Thirty (30) tablets in a HDPE bottle with a child-resistant cap, NDC 73336-075-30 Ninety (90) tablets in a HDPE bottle with a child-resistant cap, NDC 73336-075-90 Store at 20°C to 25°C (68°F to 77°F), excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Keep this and all medications out of sight and reach of children. Dispose unused medication via a take-back option if available; otherwise follow FDA instructions for disposal in the household trash. See www.fda.gov/drugdisposal for more information.
Description
Indications and Usage, Overactive Bladder in Adult Males with Benign Prostatic Hyperplasia ( 1.2 ) 12/2024 Contraindications ( 4 ) 10/2024 Warnings and Precautions, Angioedema ( 5.2 ) 10/2024
Section 42229-5
GEMTESA is indicated for the treatment of overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency in adult males on pharmacological therapy for benign prostatic hyperplasia (BPH).
Section 42230-3
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Approved: 12/2024 |
|
|
PATIENT INFORMATION
GEMTESA [gem tes' ah] (vibegron) tablets, for oral use |
||
|
What is GEMTESA?
GEMTESA is a prescription medicine used to treat the following symptoms due to a condition called overactive bladder in adults, and in adult males taking medicine for benign prostatic hyperplasia (BPH):
|
||
Do not take GEMTESA if you:
|
||
|
Before you take GEMTESA, tell your doctor about all of your medical conditions, including if you:
have liver problems.
|
||
|
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Know the medicines you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine. |
||
How should I take GEMTESA?
|
||
|
What are the possible side effects of GEMTESA?
GEMTESA may cause serious side effects, including:
|
||
| The most common side effects of GEMTESA include: | ||
|
|
|
| These are not all the possible side effects of GEMTESA. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store GEMTESA?
|
||
| Keep GEMTESA and all medicines out of the reach of children. | ||
|
General information about the safe and effective use of GEMTESA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use GEMTESA for a condition for which it was not prescribed. Do not give GEMTESA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your doctor or pharmacist for information about GEMTESA that is written for health professionals. |
||
|
What are the ingredients in GEMTESA?
Active ingredient: vibegron Inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, mannitol, and microcrystalline cellulose. The light green film coating contains FD&C Blue No. 2 - aluminum lake, hypromellose, iron oxide yellow, lactose monohydrate, titanium dioxide, and triacetin. |
||
|
Manufactured for and Distributed by: SUMITOMO PHARMA is a trademark of Sumitomo Pharma Co., Ltd., used under license. SUMITOMO is a registered trademark of Sumitomo Chemical Co., Ltd., used under license. Sumitomo Pharma America, Inc. is a U.S. subsidiary of Sumitomo Pharma Co., Ltd. All other trademarks are the property of their respective owners. For more information, go to www.GEMTESA.com or call 1-833-876-8268. |
Section 43683-2
10 Overdosage
There is no experience with inadvertent GEMTESA overdosage. In case of suspected overdose, treatment should be symptomatic and supportive.
11 Description
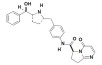
Vibegron is a selective beta-3 adrenergic agonist. The chemical name is (6S)-N-[4-[[(2S,5R)-5-[(R)-hydroxy(phenyl)methyl]pyrrolidin-2-yl]methyl]phenyl]-4-oxo-7,8-dihydro-6H-pyrrolo[1,2-a]pyrimidine-6-carboxamide having a molecular formula of C26H28N4O3 and a molecular weight of 444.538 g/mol. The structural formula of vibegron is:
Vibegron is a crystalline, white to off-white to tan powder.
GEMTESA tablets, for oral administration contain 75 mg of vibegron and the following inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, mannitol, and microcrystalline cellulose. The light green film coating contains FD&C Blue No. 2 - aluminum lake, hypromellose, iron oxide yellow, lactose monohydrate, titanium dioxide, and triacetin.
5.2 Angioedema
Angioedema of the face and/or larynx has been reported with GEMTESA. Angioedema has been reported to occur hours after the first dose or after multiple doses. Angioedema, associated with upper airway swelling, may be life-threatening. If involvement of the tongue, hypopharynx, or larynx occurs, immediately discontinue GEMTESA and provide appropriate therapy and/or measures necessary to ensure a patent airway. GEMTESA is contraindicated in patients with known hypersensitivity to vibegron or any component of GEMTESA [see Contraindications (4) and Adverse Reactions (6.2)].
8.4 Pediatric Use
The safety and effectiveness of GEMTESA in pediatric patients have not been established.
8.5 Geriatric Use
Of 526 patients who received GEMTESA in the clinical studies for OAB with symptoms of urge urinary incontinence, urgency, and urinary frequency, 242 (46%) were 65 years of age or older, and 75 (14%) were 75 years of age or older [see Clinical Studies (14.1)]. No overall differences in safety or effectiveness of GEMTESA have been observed between patients 65 years of age and older and younger adult patients.
Of the total number of GEMTESA-treated patients in clinical studies for OAB with symptoms of urge urinary incontinence, urgency, and urinary frequency in adult males on pharmacological therapy for benign prostatic hyperplasia (BPH), 347 (63%) were 65 years of age and older, while 100 (18%) were 75 years of age and older [see Clinical Studies (14.2)]. No overall differences in safety of GEMTESA have been observed between patients 65 years of age and older and younger adult patients.
4 Contraindications
6 Adverse Reactions
The following clinically significant adverse reaction is described elsewhere in the labeling:
- Urinary retention [see Warnings and Precautions (5.1)]
7 Drug Interactions
Concomitant use of GEMTESA increases digoxin maximal concentrations (Cmax) and systemic exposure as assessed by area under the concentration-time curve (AUC) [see Clinical Pharmacology (12.3)]. Serum digoxin concentrations should be monitored before initiating and during therapy with GEMTESA and used for titration of the digoxin dose to obtain the desired clinical effect. Continue monitoring digoxin concentrations upon discontinuation of GEMTESA and adjust digoxin dose as needed.
8.6 Renal Impairment
No dosage adjustment for GEMTESA is recommended for patients with mild, moderate, or severe renal impairment (eGFR 15 to <90 mL/min/1.73 m2). GEMTESA has not been studied in patients with eGFR <15 mL/min/1.73 m2 (with or without hemodialysis) and is not recommended in these patients [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Vibegron's exposure-response relationship and the time course of pharmacodynamic response are not fully characterized.
12.3 Pharmacokinetics
Mean vibegron Cmax and AUC increased in a greater than dose-proportional manner up to 600 mg (8 times the approved recommended dosage). Steady state concentrations are achieved within 7 days of once daily dosing. The mean accumulation ratio (Rac) was 1.7 for Cmax and 2.4 for AUC0-24hr.
5.1 Urinary Retention
Urinary retention has been reported in patients taking GEMTESA. The risk of urinary retention may be increased in patients with bladder outlet obstruction and also in patients taking muscarinic antagonist medications for the treatment of OAB. Monitor patients for signs and symptoms of urinary retention, particularly in patients with bladder outlet obstruction or patients taking muscarinic antagonist medications for the treatment of OAB. Discontinue GEMTESA in patients who develop urinary retention [see Adverse Reactions (6.1)].
2.1 Recommended Dosage
The recommended dosage of GEMTESA is one 75 mg tablet orally, once daily with or without food. Swallow GEMTESA tablets whole with a glass of water.
In adults, GEMTESA tablets also may be crushed, mixed with a tablespoon (approximately 15 mL) of applesauce and taken immediately with a glass of water [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
No dosage adjustment for GEMTESA is recommended for patients with mild to moderate hepatic impairment (Child-Pugh A and B). GEMTESA has not been studied in patients with severe hepatic impairment (Child-Pugh C) and is not recommended in this patient population [see Clinical Pharmacology (12.3)].
1 Indications and Usage
GEMTESA is a beta-3 adrenergic agonist indicated for the treatment of:
- overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency in adults. (1.1)
- overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency in adult males on pharmacological therapy for benign prostatic hyperplasia (BPH). (1.2)
12.1 Mechanism of Action
Vibegron is a selective human beta-3 adrenergic receptor agonist. Activation of the beta-3 adrenergic receptor increases bladder capacity by relaxing the detrusor smooth muscle during bladder filling.
5 Warnings and Precautions
Urinary Retention: Monitor for urinary retention, especially in patients with bladder outlet obstruction and also in patients taking muscarinic antagonist medications for OAB, in whom the risk of urinary retention may be greater. If urinary retention develops, discontinue GEMTESA. (5.1)
Angioedema: Angioedema of the face and/or larynx has been reported with GEMTESA. (5.2)
2 Dosage and Administration
3 Dosage Forms and Strengths
Tablets: 75 mg, oval, light green, film-coated, debossed with V75 on one side and no debossing on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of vibegron. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following adverse events have been reported in association with vibegron use in worldwide postmarketing experience:
Urologic disorders: urinary retention
Skin and subcutaneous tissue disorders: angioedema of the face and larynx; hypersensitivity reactions, including urticaria, pruritus, rash and drug eruption; eczema
Gastrointestinal disorders: constipation
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
1.1 Overactive Bladder in Adults
GEMTESA® is indicated for the treatment of overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency in adults.
14.1 Overactive Bladder in Adults
The efficacy of GEMTESA was evaluated in a 12-week, double-blind, randomized, placebo-controlled, and active-controlled trial (Study 3003, NCT03492281) in patients with OAB (urge urinary incontinence, urgency, and urinary frequency). Patients were randomized 5:5:4 to receive either GEMTESA 75 mg, placebo, or active control orally, once daily for 12 weeks. For study entry, patients had to have symptoms of OAB for at least 3 months with an average of 8 or more micturitions per day and at least 1 urge urinary incontinence (UUI) per day, or an average of 8 or more micturitions per day and an average of at least 3 urgency episodes per day. Urge urinary incontinence was defined as leakage of urine of any amount because the patient felt an urge or need to urinate immediately. The study population included OAB medication-naïve patients as well as patients who had received prior therapy with OAB medications.
The co-primary endpoints were change from baseline in average daily number of micturitions and average daily number of UUI episodes at week 12. Additional endpoints included change from baseline in average daily number of “need to urinate immediately” (urgency) episodes and average volume voided per micturition.
A total of 1,515 patients received at least one daily dose of placebo (n=540), GEMTESA 75 mg (n=545), or an active control treatment (n=430). The majority of patients were White (78%) and female (85%) with a mean age of 60 (range 18 to 93) years.
Table 3 shows changes from baseline at week 12 for average daily number of micturitions, average daily number of UUI episodes, average daily number of “need to urinate immediately” (urgency) episodes, and average volume voided per micturition.
| Parameter |
GEMTESA 75 mg |
Placebo |
|---|---|---|
|
* Least squares mean adjusted for treatment, baseline, sex, geographical region, study visit, and study visit by treatment interaction term. |
||
| Average Daily Number of Micturitions | ||
| Baseline mean (n) | 11.3 (526) | 11.8 (520) |
| Change from Baseline* (n) | -1.8 (492) | -1.3 (475) |
| Difference from Placebo | -0.5 | |
| 95% Confidence Interval | -0.8, -0.2 | |
| p-value | <0.001 | |
| Average Daily Number of UUI Episodes | ||
| Baseline mean (n) | 3.4 (403) | 3.5 (405) |
| Change from Baseline* (n) | -2.0 (383) | -1.4 (372) |
| Difference from Placebo | -0.6 | |
| 95% Confidence Interval | -0.9, -0.3 | |
| p-value | <0.0001 | |
| Average Daily Number of “Need to Urinate Immediately” (Urgency) Episodes | ||
| Baseline mean (n) | 8.1 (526) | 8.1 (520) |
| Change from Baseline* (n) | -2.7 (492) | -2.0 (475) |
| Difference from Placebo | -0.7 | |
| 95% Confidence Interval | -1.1, -0.2 | |
| p-value | 0.002 | |
| Average Volume Voided (mL) per Micturition | ||
| Baseline mean (n) | 155 (524) | 148 (514) |
| Change from Baseline* (n) | 23 (490) | 2 (478) |
| Difference from Placebo | 21 | |
| 95% Confidence Interval | 14, 28 | |
| p-value | <0.0001 |
Figure 1 and Figure 2 show the mean change from baseline over time in average daily number of micturitions and mean change from baseline over time in average daily number of UUI episodes, respectively.
Figure 1: Mean (SE) Change from Baseline in the Average Daily Number of Micturitions
Figure 2: Mean (SE) Change from Baseline in the Average Daily Number of UUI Episodes in Patients with At Least 1 Average Daily UUI Episode at Baseline
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
GEMTESA 75 mg tablets are light green, oval, film-coated tablets, debossed with V75 on one side and no debossing on the other side.
GEMTESA is marketed in two packaging configurations:
Thirty (30) tablets in a HDPE bottle with a child-resistant cap, NDC 73336-075-30
Ninety (90) tablets in a HDPE bottle with a child-resistant cap, NDC 73336-075-90
Store at 20°C to 25°C (68°F to 77°F), excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Keep this and all medications out of sight and reach of children.
Dispose unused medication via a take-back option if available; otherwise follow FDA instructions for disposal in the household trash. See www.fda.gov/drugdisposal for more information.
14.2 Overactive Bladder in Adult Males With Bph
The safety, tolerability, and efficacy of GEMTESA was evaluated in a multinational 24-week, double-blind, randomized, placebo-controlled trial (Study 3005, NCT03902080) in male patients at least 45 years of age with OAB on pharmacological therapy (i.e., treatment with an alpha blocker, with or without a 5-alpha reductase inhibitor) for BPH. A total of 1105 patients were randomized 1:1 to receive either GEMTESA 75 mg or placebo once daily for 24 weeks. For study entry, patients had symptoms of OAB (an average of 8 or more micturitions per day, 3 or more urgency episodes per day with or without incontinence, and 2 or more nocturia episodes per night) while taking pharmacological therapy for at least 2 months for the treatment of lower urinary tract symptoms due to BPH. Randomization was stratified based on the baseline average number of micturition episodes per day, alpha blocker use with or without 5 alpha reductase inhibitor use, and urinary incontinence.
The co-primary endpoints were change from baseline in the average daily number of micturitions and the average daily number of “need to urinate immediately” (urgency) episodes at week 12. Additional endpoints included change from baseline in the average daily number of urge urinary incontinence (UUI) episodes and the average volume voided per micturition.
A total of 1104 patients received at least one daily dose of placebo (n=551) or GEMTESA 75 mg (n=553). The majority of patients were White (87%) and enrolled in the U.S. (56%). The mean age was 67 years (range 45 to 97) and at least 63% were ≥ 65 years.
Table 4 shows changes from baseline at week 12 for average daily number of micturitions, average daily number of urgency episodes, average daily number of UUI episodes and average volume voided per micturition.
| Parameter |
GEMTESA 75 mg
(N = 538) |
Placebo
(N = 542) |
|---|---|---|
|
* Least squares mean adjusted for study visit, baseline value, geographical region, interaction of visit by treatment, and stratification factors as randomized (baseline average micturitions per day**, alpha blocker use with or without 5-ARI, baseline urinary incontinence**), geographical region, study visit, and study visit by treatment interaction term. |
||
|
** Baseline average micturitions per day stratification factor is not included in the model where the continuous value of baseline average micturitions per day is present; baseline urinary incontinence is not included in the model for UUI; |
||
|
*** Only subjects with baseline incontinence have UUI analyzed. |
||
| Average Daily Number of Micturitions | ||
| Baseline mean | 11.84 | 11.96 |
| Change from Baseline* | -2.04 | -1.30 |
| Difference from Placebo | -0.74 | |
| 95% Confidence Interval | -1.02, -0.46 | |
| Average Daily Number of “Need to Urinate Immediately” (Urgency) Episodes | ||
| Baseline mean | 9.05 | 9.00 |
| Change from Baseline* | -2.88 | -1.93 |
| Difference from Placebo | -0.95 | |
| 95% Confidence Interval | -1.37, -0.54 | |
| Average Daily Number of UUI Episodes | ||
| N *** | 146 | 151 |
| Baseline mean | 3.33 | 3.23 |
| Change from Baseline* | -2.19 | -1.39 |
| Difference from Placebo | -0.80 | |
| 95% Confidence Interval | -1.33, -0.27 | |
| Average Volume Voided (mL) per Micturition | ||
| Baseline mean | 166.37 | 166.43 |
| Change from Baseline* | 25.63 | 10.56 |
| Difference from Placebo | 15.07 | |
| 95% Confidence Interval | 9.13, 21.02 |
Figure 3 and Figure 4 show the mean change from baseline over time in average daily number of micturitions and mean change from baseline over time in average daily number of urgency episodes, respectively.
Figure 3: Mean (SE) Change from Baseline in the Average Daily Number of Micturitions in Male Patients with BPH on Pharmacological Therapy
Figure 4: Mean (SE) Change from Baseline in the Average Daily Number of Urgency Episodes in Male Patients with BPH on Pharmacological Therapy
Principal Display Panel 75 Mg Tablet Bottle Label
30 tablets
NDC 73336-075-30
Rx Only
GEMTESA®
(vibegron) tablets
75 mg
One Tablet • Once Daily
May be swallowed whole or crushed
Structured Label Content
Section 42229-5 (42229-5)
GEMTESA is indicated for the treatment of overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency in adult males on pharmacological therapy for benign prostatic hyperplasia (BPH).
Section 42230-3 (42230-3)
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Approved: 12/2024 |
|
|
PATIENT INFORMATION
GEMTESA [gem tes' ah] (vibegron) tablets, for oral use |
||
|
What is GEMTESA?
GEMTESA is a prescription medicine used to treat the following symptoms due to a condition called overactive bladder in adults, and in adult males taking medicine for benign prostatic hyperplasia (BPH):
|
||
Do not take GEMTESA if you:
|
||
|
Before you take GEMTESA, tell your doctor about all of your medical conditions, including if you:
have liver problems.
|
||
|
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Know the medicines you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine. |
||
How should I take GEMTESA?
|
||
|
What are the possible side effects of GEMTESA?
GEMTESA may cause serious side effects, including:
|
||
| The most common side effects of GEMTESA include: | ||
|
|
|
| These are not all the possible side effects of GEMTESA. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store GEMTESA?
|
||
| Keep GEMTESA and all medicines out of the reach of children. | ||
|
General information about the safe and effective use of GEMTESA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use GEMTESA for a condition for which it was not prescribed. Do not give GEMTESA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your doctor or pharmacist for information about GEMTESA that is written for health professionals. |
||
|
What are the ingredients in GEMTESA?
Active ingredient: vibegron Inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, mannitol, and microcrystalline cellulose. The light green film coating contains FD&C Blue No. 2 - aluminum lake, hypromellose, iron oxide yellow, lactose monohydrate, titanium dioxide, and triacetin. |
||
|
Manufactured for and Distributed by: SUMITOMO PHARMA is a trademark of Sumitomo Pharma Co., Ltd., used under license. SUMITOMO is a registered trademark of Sumitomo Chemical Co., Ltd., used under license. Sumitomo Pharma America, Inc. is a U.S. subsidiary of Sumitomo Pharma Co., Ltd. All other trademarks are the property of their respective owners. For more information, go to www.GEMTESA.com or call 1-833-876-8268. |
Section 43683-2 (43683-2)
10 Overdosage (10 OVERDOSAGE)
There is no experience with inadvertent GEMTESA overdosage. In case of suspected overdose, treatment should be symptomatic and supportive.
11 Description (11 DESCRIPTION)
Vibegron is a selective beta-3 adrenergic agonist. The chemical name is (6S)-N-[4-[[(2S,5R)-5-[(R)-hydroxy(phenyl)methyl]pyrrolidin-2-yl]methyl]phenyl]-4-oxo-7,8-dihydro-6H-pyrrolo[1,2-a]pyrimidine-6-carboxamide having a molecular formula of C26H28N4O3 and a molecular weight of 444.538 g/mol. The structural formula of vibegron is:
Vibegron is a crystalline, white to off-white to tan powder.
GEMTESA tablets, for oral administration contain 75 mg of vibegron and the following inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, mannitol, and microcrystalline cellulose. The light green film coating contains FD&C Blue No. 2 - aluminum lake, hypromellose, iron oxide yellow, lactose monohydrate, titanium dioxide, and triacetin.
5.2 Angioedema
Angioedema of the face and/or larynx has been reported with GEMTESA. Angioedema has been reported to occur hours after the first dose or after multiple doses. Angioedema, associated with upper airway swelling, may be life-threatening. If involvement of the tongue, hypopharynx, or larynx occurs, immediately discontinue GEMTESA and provide appropriate therapy and/or measures necessary to ensure a patent airway. GEMTESA is contraindicated in patients with known hypersensitivity to vibegron or any component of GEMTESA [see Contraindications (4) and Adverse Reactions (6.2)].
8.4 Pediatric Use
The safety and effectiveness of GEMTESA in pediatric patients have not been established.
8.5 Geriatric Use
Of 526 patients who received GEMTESA in the clinical studies for OAB with symptoms of urge urinary incontinence, urgency, and urinary frequency, 242 (46%) were 65 years of age or older, and 75 (14%) were 75 years of age or older [see Clinical Studies (14.1)]. No overall differences in safety or effectiveness of GEMTESA have been observed between patients 65 years of age and older and younger adult patients.
Of the total number of GEMTESA-treated patients in clinical studies for OAB with symptoms of urge urinary incontinence, urgency, and urinary frequency in adult males on pharmacological therapy for benign prostatic hyperplasia (BPH), 347 (63%) were 65 years of age and older, while 100 (18%) were 75 years of age and older [see Clinical Studies (14.2)]. No overall differences in safety of GEMTESA have been observed between patients 65 years of age and older and younger adult patients.
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reaction is described elsewhere in the labeling:
- Urinary retention [see Warnings and Precautions (5.1)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Concomitant use of GEMTESA increases digoxin maximal concentrations (Cmax) and systemic exposure as assessed by area under the concentration-time curve (AUC) [see Clinical Pharmacology (12.3)]. Serum digoxin concentrations should be monitored before initiating and during therapy with GEMTESA and used for titration of the digoxin dose to obtain the desired clinical effect. Continue monitoring digoxin concentrations upon discontinuation of GEMTESA and adjust digoxin dose as needed.
8.6 Renal Impairment
No dosage adjustment for GEMTESA is recommended for patients with mild, moderate, or severe renal impairment (eGFR 15 to <90 mL/min/1.73 m2). GEMTESA has not been studied in patients with eGFR <15 mL/min/1.73 m2 (with or without hemodialysis) and is not recommended in these patients [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Vibegron's exposure-response relationship and the time course of pharmacodynamic response are not fully characterized.
12.3 Pharmacokinetics
Mean vibegron Cmax and AUC increased in a greater than dose-proportional manner up to 600 mg (8 times the approved recommended dosage). Steady state concentrations are achieved within 7 days of once daily dosing. The mean accumulation ratio (Rac) was 1.7 for Cmax and 2.4 for AUC0-24hr.
5.1 Urinary Retention
Urinary retention has been reported in patients taking GEMTESA. The risk of urinary retention may be increased in patients with bladder outlet obstruction and also in patients taking muscarinic antagonist medications for the treatment of OAB. Monitor patients for signs and symptoms of urinary retention, particularly in patients with bladder outlet obstruction or patients taking muscarinic antagonist medications for the treatment of OAB. Discontinue GEMTESA in patients who develop urinary retention [see Adverse Reactions (6.1)].
2.1 Recommended Dosage
The recommended dosage of GEMTESA is one 75 mg tablet orally, once daily with or without food. Swallow GEMTESA tablets whole with a glass of water.
In adults, GEMTESA tablets also may be crushed, mixed with a tablespoon (approximately 15 mL) of applesauce and taken immediately with a glass of water [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
No dosage adjustment for GEMTESA is recommended for patients with mild to moderate hepatic impairment (Child-Pugh A and B). GEMTESA has not been studied in patients with severe hepatic impairment (Child-Pugh C) and is not recommended in this patient population [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
GEMTESA is a beta-3 adrenergic agonist indicated for the treatment of:
- overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency in adults. (1.1)
- overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency in adult males on pharmacological therapy for benign prostatic hyperplasia (BPH). (1.2)
12.1 Mechanism of Action
Vibegron is a selective human beta-3 adrenergic receptor agonist. Activation of the beta-3 adrenergic receptor increases bladder capacity by relaxing the detrusor smooth muscle during bladder filling.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Urinary Retention: Monitor for urinary retention, especially in patients with bladder outlet obstruction and also in patients taking muscarinic antagonist medications for OAB, in whom the risk of urinary retention may be greater. If urinary retention develops, discontinue GEMTESA. (5.1)
Angioedema: Angioedema of the face and/or larynx has been reported with GEMTESA. (5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 75 mg, oval, light green, film-coated, debossed with V75 on one side and no debossing on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of vibegron. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following adverse events have been reported in association with vibegron use in worldwide postmarketing experience:
Urologic disorders: urinary retention
Skin and subcutaneous tissue disorders: angioedema of the face and larynx; hypersensitivity reactions, including urticaria, pruritus, rash and drug eruption; eczema
Gastrointestinal disorders: constipation
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
1.1 Overactive Bladder in Adults
GEMTESA® is indicated for the treatment of overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency in adults.
14.1 Overactive Bladder in Adults
The efficacy of GEMTESA was evaluated in a 12-week, double-blind, randomized, placebo-controlled, and active-controlled trial (Study 3003, NCT03492281) in patients with OAB (urge urinary incontinence, urgency, and urinary frequency). Patients were randomized 5:5:4 to receive either GEMTESA 75 mg, placebo, or active control orally, once daily for 12 weeks. For study entry, patients had to have symptoms of OAB for at least 3 months with an average of 8 or more micturitions per day and at least 1 urge urinary incontinence (UUI) per day, or an average of 8 or more micturitions per day and an average of at least 3 urgency episodes per day. Urge urinary incontinence was defined as leakage of urine of any amount because the patient felt an urge or need to urinate immediately. The study population included OAB medication-naïve patients as well as patients who had received prior therapy with OAB medications.
The co-primary endpoints were change from baseline in average daily number of micturitions and average daily number of UUI episodes at week 12. Additional endpoints included change from baseline in average daily number of “need to urinate immediately” (urgency) episodes and average volume voided per micturition.
A total of 1,515 patients received at least one daily dose of placebo (n=540), GEMTESA 75 mg (n=545), or an active control treatment (n=430). The majority of patients were White (78%) and female (85%) with a mean age of 60 (range 18 to 93) years.
Table 3 shows changes from baseline at week 12 for average daily number of micturitions, average daily number of UUI episodes, average daily number of “need to urinate immediately” (urgency) episodes, and average volume voided per micturition.
| Parameter |
GEMTESA 75 mg |
Placebo |
|---|---|---|
|
* Least squares mean adjusted for treatment, baseline, sex, geographical region, study visit, and study visit by treatment interaction term. |
||
| Average Daily Number of Micturitions | ||
| Baseline mean (n) | 11.3 (526) | 11.8 (520) |
| Change from Baseline* (n) | -1.8 (492) | -1.3 (475) |
| Difference from Placebo | -0.5 | |
| 95% Confidence Interval | -0.8, -0.2 | |
| p-value | <0.001 | |
| Average Daily Number of UUI Episodes | ||
| Baseline mean (n) | 3.4 (403) | 3.5 (405) |
| Change from Baseline* (n) | -2.0 (383) | -1.4 (372) |
| Difference from Placebo | -0.6 | |
| 95% Confidence Interval | -0.9, -0.3 | |
| p-value | <0.0001 | |
| Average Daily Number of “Need to Urinate Immediately” (Urgency) Episodes | ||
| Baseline mean (n) | 8.1 (526) | 8.1 (520) |
| Change from Baseline* (n) | -2.7 (492) | -2.0 (475) |
| Difference from Placebo | -0.7 | |
| 95% Confidence Interval | -1.1, -0.2 | |
| p-value | 0.002 | |
| Average Volume Voided (mL) per Micturition | ||
| Baseline mean (n) | 155 (524) | 148 (514) |
| Change from Baseline* (n) | 23 (490) | 2 (478) |
| Difference from Placebo | 21 | |
| 95% Confidence Interval | 14, 28 | |
| p-value | <0.0001 |
Figure 1 and Figure 2 show the mean change from baseline over time in average daily number of micturitions and mean change from baseline over time in average daily number of UUI episodes, respectively.
Figure 1: Mean (SE) Change from Baseline in the Average Daily Number of Micturitions
Figure 2: Mean (SE) Change from Baseline in the Average Daily Number of UUI Episodes in Patients with At Least 1 Average Daily UUI Episode at Baseline
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
GEMTESA 75 mg tablets are light green, oval, film-coated tablets, debossed with V75 on one side and no debossing on the other side.
GEMTESA is marketed in two packaging configurations:
Thirty (30) tablets in a HDPE bottle with a child-resistant cap, NDC 73336-075-30
Ninety (90) tablets in a HDPE bottle with a child-resistant cap, NDC 73336-075-90
Store at 20°C to 25°C (68°F to 77°F), excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Keep this and all medications out of sight and reach of children.
Dispose unused medication via a take-back option if available; otherwise follow FDA instructions for disposal in the household trash. See www.fda.gov/drugdisposal for more information.
14.2 Overactive Bladder in Adult Males With Bph (14.2 Overactive Bladder in Adult Males with BPH)
The safety, tolerability, and efficacy of GEMTESA was evaluated in a multinational 24-week, double-blind, randomized, placebo-controlled trial (Study 3005, NCT03902080) in male patients at least 45 years of age with OAB on pharmacological therapy (i.e., treatment with an alpha blocker, with or without a 5-alpha reductase inhibitor) for BPH. A total of 1105 patients were randomized 1:1 to receive either GEMTESA 75 mg or placebo once daily for 24 weeks. For study entry, patients had symptoms of OAB (an average of 8 or more micturitions per day, 3 or more urgency episodes per day with or without incontinence, and 2 or more nocturia episodes per night) while taking pharmacological therapy for at least 2 months for the treatment of lower urinary tract symptoms due to BPH. Randomization was stratified based on the baseline average number of micturition episodes per day, alpha blocker use with or without 5 alpha reductase inhibitor use, and urinary incontinence.
The co-primary endpoints were change from baseline in the average daily number of micturitions and the average daily number of “need to urinate immediately” (urgency) episodes at week 12. Additional endpoints included change from baseline in the average daily number of urge urinary incontinence (UUI) episodes and the average volume voided per micturition.
A total of 1104 patients received at least one daily dose of placebo (n=551) or GEMTESA 75 mg (n=553). The majority of patients were White (87%) and enrolled in the U.S. (56%). The mean age was 67 years (range 45 to 97) and at least 63% were ≥ 65 years.
Table 4 shows changes from baseline at week 12 for average daily number of micturitions, average daily number of urgency episodes, average daily number of UUI episodes and average volume voided per micturition.
| Parameter |
GEMTESA 75 mg
(N = 538) |
Placebo
(N = 542) |
|---|---|---|
|
* Least squares mean adjusted for study visit, baseline value, geographical region, interaction of visit by treatment, and stratification factors as randomized (baseline average micturitions per day**, alpha blocker use with or without 5-ARI, baseline urinary incontinence**), geographical region, study visit, and study visit by treatment interaction term. |
||
|
** Baseline average micturitions per day stratification factor is not included in the model where the continuous value of baseline average micturitions per day is present; baseline urinary incontinence is not included in the model for UUI; |
||
|
*** Only subjects with baseline incontinence have UUI analyzed. |
||
| Average Daily Number of Micturitions | ||
| Baseline mean | 11.84 | 11.96 |
| Change from Baseline* | -2.04 | -1.30 |
| Difference from Placebo | -0.74 | |
| 95% Confidence Interval | -1.02, -0.46 | |
| Average Daily Number of “Need to Urinate Immediately” (Urgency) Episodes | ||
| Baseline mean | 9.05 | 9.00 |
| Change from Baseline* | -2.88 | -1.93 |
| Difference from Placebo | -0.95 | |
| 95% Confidence Interval | -1.37, -0.54 | |
| Average Daily Number of UUI Episodes | ||
| N *** | 146 | 151 |
| Baseline mean | 3.33 | 3.23 |
| Change from Baseline* | -2.19 | -1.39 |
| Difference from Placebo | -0.80 | |
| 95% Confidence Interval | -1.33, -0.27 | |
| Average Volume Voided (mL) per Micturition | ||
| Baseline mean | 166.37 | 166.43 |
| Change from Baseline* | 25.63 | 10.56 |
| Difference from Placebo | 15.07 | |
| 95% Confidence Interval | 9.13, 21.02 |
Figure 3 and Figure 4 show the mean change from baseline over time in average daily number of micturitions and mean change from baseline over time in average daily number of urgency episodes, respectively.
Figure 3: Mean (SE) Change from Baseline in the Average Daily Number of Micturitions in Male Patients with BPH on Pharmacological Therapy
Figure 4: Mean (SE) Change from Baseline in the Average Daily Number of Urgency Episodes in Male Patients with BPH on Pharmacological Therapy
Principal Display Panel 75 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 75 mg Tablet Bottle Label)
30 tablets
NDC 73336-075-30
Rx Only
GEMTESA®
(vibegron) tablets
75 mg
One Tablet • Once Daily
May be swallowed whole or crushed
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:31.868668 · Updated: 2026-03-14T22:25:04.498043