These Highlights Do Not Include All The Information Needed To Use Voriconazole For Injection Safely And Effectively. See Full Prescribing Information For Voriconazole For Injection.
25eb8d91-5abd-4fdb-a331-dacc960a95d9
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions, Photosensitivity ( 5.6 ) 10/2022
Indications and Usage
Voriconazole for injection is an azole antifungal indicated for the treatment of adults and pediatric patients 2 years of age and older with: Invasive aspergillosis ( 1.1 ) Candidemia in non-neutropenics and other deep tissue Candida infections ( 1.2 ) Esophageal candidiasis ( 1.3 ) Serious fungal infections caused by Scedosporium apiospermum and Fusarium species including Fusarium solani , in patients intolerant of, or refractory to, other therapy ( 1.4 )
Dosage and Administration
Dosage in Adults ( 2.3 ) Infection Loading dose Maintenance Dose Intravenous infusion Intravenous infusion Oral Invasive Aspergillosis 6 mg/kg every 12 hours for the first 24 hours 4 mg/kg every 12 hours 200 mg every 12 hours Candidemia in nonneutropenics and other deep tissue Candida infections 3 to 4 mg/kg every 12 hours 200 mg every 12 hours Scedosporiosis and Fusariosis 4 mg/kg every 12 hours 200 mg every 12 hours Esophageal Candidiasis Not Evaluated Not Evaluated 200 mg every 12 hours Adult patients weighing less than 40 kg: oral maintenance dose 100 mg or 150 mg every 12 hours Hepatic Impairment: Use half the maintenance dose in adult patients with mild to moderate hepatic impairment (Child-Pugh Class A and B) ( 2.5 ) Renal Impairment: Avoid intravenous administration in adult patients with moderate to severe renal impairment (creatinine clearance <50 mL/min) ( 2.6 ) Dosage in Pediatric Patients 2 years of age and older ( 2.4 ) For pediatric patients 2 to less than 12 years of age and 12 to 14 years of age weighing less than 50 kg see Table below. Infection Loading Dose Maintenance Dose Intravenous infusion Intravenous infusion Oral Invasive Aspergillosis 9 mg/kg every12 hours for the first 24 hours 8 mg/kg every 12 hours after the first 24 hours 9 mg/kg every 12 hours (maximum dose of 350 mg every 12 hours) Candidemia in nonneutropenics and other deep tissue Candida infections Scedosporiosis and Fusariosis Esophageal Candidiasis Not Evaluated 4 mg/kg every 12 hours 9 mg/kg every 12 hours (maximum dose of 350 mg every 12 hours) For pediatric patients aged 12 to 14 years weighing greater than or equal to 50 kg and those aged 15 years and older regardless of body weight use adult dosage. ( 2.4 ) Dosage adjustment of voriconazole for injection in pediatric patients with renal or hepatic impairment has not been established ( 2.5 , 2.6 ) See full prescribing information for instructions on reconstitution of voriconazole for injection lyophilized powder for intravenous use and important administration instructions ( 2.1 , 2.6 , 2.7 )
Warnings and Precautions
Hepatic Toxicity : Serious hepatic reactions reported. Evaluate liver function tests at start of and during voriconazole therapy ( 5.1 ) Arrhythmias and QT Prolongation : Correct potassium, magnesium and calcium prior to use; caution patients with proarrhythmic conditions ( 5.2 ) Infusion Related Reactions (including anaphylaxis) : Stop the infusion ( 5.3 ) Visual Disturbances (including optic neuritis and papilledema): Monitor visual function if treatment continues beyond 28 days ( 5.4 ) Severe Cutaneous Adverse Reactions : Discontinue for exfoliative cutaneous reactions ( 5.5 ) Photosensitivity : Avoid sunlight due to risk of photosensitivity ( 5.6 ) Adrenal Dysfunction : Carefully monitor patients receiving voriconazole and corticosteroids (via all routes of administration) for adrenal dysfunction both during and after voriconazole treatment. Instruct patients to seek immediate medical care if they develop signs and symptoms of Cushing's syndrome or adrenal insufficiency ( 5.8 ) Embryo-Fetal Toxicity : Voriconazole can cause fetal harm when administered to a pregnant woman. Inform pregnant patients of the potential hazard to the fetus. Advise females of reproductive potential to use effective contraception during treatment with voriconazole ( 5.9 , 8.1 , 8.3 ) Skeletal Adverse Reactions : Fluorosis and periostitis with long-term voriconazole therapy. Discontinue if these adverse reactions occur ( 5.12 ) Clinically Significant Drug Interactions : Review patient's concomitant medications ( 5.13 , 7 )
Contraindications
Voriconazole is contraindicated in patients with known hypersensitivity to voriconazole or its excipients. There is no information regarding cross-sensitivity between voriconazole and other azole antifungal agents. Caution should be used when prescribing voriconazole to patients with hypersensitivity to other azoles. Coadministration of pimozide, quinidine or ivabradine with voriconazole is contraindicated because increased plasma concentrations of these drugs can lead to QT prolongation and rare occurrences of torsade de pointes [see Drug Interactions ( 7 )] . Coadministration of voriconazole with sirolimus is contraindicated because voriconazole for injection significantly increases sirolimus concentrations [see Drug Interactions ( 7 ) and Clinical Pharmacology ( 12.3 )] . Coadministration of voriconazole with rifampin, carbamazepine, long-acting barbiturates, and St John's Wort is contraindicated because these drugs are likely to decrease plasma voriconazole concentrations significantly [see Drug Interactions ( 7 ) and Clinical Pharmacology ( 12.3 )] . Coadministration of standard doses of voriconazole with efavirenz doses of 400 mg every 24 hours or higher is contraindicated, because efavirenz significantly decreases plasma voriconazole concentrations in healthy subjects at these doses. Voriconazole also significantly increases efavirenz plasma concentrations [see Drug Interactions ( 7 ) and Clinical Pharmacology ( 12.3 )] . Coadministration of voriconazole with high-dose ritonavir (400 mg every 12 hours) is contraindicated because ritonavir (400 mg every 12 hours) significantly decreases plasma voriconazole concentrations. Coadministration of voriconazole and low-dose ritonavir (100 mg every 12 hours) should be avoided, unless an assessment of the benefit/risk to the patient justifies the use of voriconazole [see Drug Interactions ( 7 ) and Clinical Pharmacology ( 12.3 )] . Coadministration of voriconazole with rifabutin is contraindicated since voriconazole significantly increases rifabutin plasma concentrations and rifabutin also significantly decreases voriconazole plasma concentrations [see Drug Interactions ( 7 ) and Clinical Pharmacology ( 12.3 )] . Coadministration of voriconazole with ergot alkaloids (ergotamine and dihydroergotamine) is contraindicated because voriconazole may increase the plasma concentration of ergot alkaloids, which may lead to ergotism [see Drug Interactions ( 7 )] . Coadministration of voriconazole with naloxegol is contraindicated because voriconazole may increase plasma concentrations of naloxegol which may precipitate opioid withdrawal symptoms [see Drug Interactions ( 7 )] . Coadministration of voriconazole with tolvaptan is contraindicated because voriconazole may increase tolvaptan plasma concentrations and increase risk of adverse reactions [see Drug Interactions ( 7 )] . Coadministration of voriconazole with venetoclax at initiation and during the ramp-up phase is contraindicated in patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) due to the potential for increased risk of tumor lysis syndrome [see Drug Interactions ( 7 )] . Coadministration of voriconazole with lurasidone is contraindicated since it may result in significant increases in lurasidone exposure and the potential for serious adverse reactions [see Drug Interactions ( 7 )] .
Adverse Reactions
Severe cutaneous adverse reactions (SCARs), such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS), which can be life-threatening or fatal, have been reported during treatment with voriconazole. If a patient develops a severe cutaneous adverse reaction, voriconazole should be discontinued [see Adverse Reactions ( 6.1 , 6.2 )].
Drug Interactions
See Table 10 for a listing of drugs that may significantly alter voriconazole concentrations. Also, see Table 11 for a listing of drugs that may interact with voriconazole resulting in altered pharmacokinetics or pharmacodynamics of the other drug [see Contraindications ( 4 ) and Drug Interactions ( 7 )] .
Medication Information
Warnings and Precautions
Hepatic Toxicity : Serious hepatic reactions reported. Evaluate liver function tests at start of and during voriconazole therapy ( 5.1 ) Arrhythmias and QT Prolongation : Correct potassium, magnesium and calcium prior to use; caution patients with proarrhythmic conditions ( 5.2 ) Infusion Related Reactions (including anaphylaxis) : Stop the infusion ( 5.3 ) Visual Disturbances (including optic neuritis and papilledema): Monitor visual function if treatment continues beyond 28 days ( 5.4 ) Severe Cutaneous Adverse Reactions : Discontinue for exfoliative cutaneous reactions ( 5.5 ) Photosensitivity : Avoid sunlight due to risk of photosensitivity ( 5.6 ) Adrenal Dysfunction : Carefully monitor patients receiving voriconazole and corticosteroids (via all routes of administration) for adrenal dysfunction both during and after voriconazole treatment. Instruct patients to seek immediate medical care if they develop signs and symptoms of Cushing's syndrome or adrenal insufficiency ( 5.8 ) Embryo-Fetal Toxicity : Voriconazole can cause fetal harm when administered to a pregnant woman. Inform pregnant patients of the potential hazard to the fetus. Advise females of reproductive potential to use effective contraception during treatment with voriconazole ( 5.9 , 8.1 , 8.3 ) Skeletal Adverse Reactions : Fluorosis and periostitis with long-term voriconazole therapy. Discontinue if these adverse reactions occur ( 5.12 ) Clinically Significant Drug Interactions : Review patient's concomitant medications ( 5.13 , 7 )
Indications and Usage
Voriconazole for injection is an azole antifungal indicated for the treatment of adults and pediatric patients 2 years of age and older with: Invasive aspergillosis ( 1.1 ) Candidemia in non-neutropenics and other deep tissue Candida infections ( 1.2 ) Esophageal candidiasis ( 1.3 ) Serious fungal infections caused by Scedosporium apiospermum and Fusarium species including Fusarium solani , in patients intolerant of, or refractory to, other therapy ( 1.4 )
Dosage and Administration
Dosage in Adults ( 2.3 ) Infection Loading dose Maintenance Dose Intravenous infusion Intravenous infusion Oral Invasive Aspergillosis 6 mg/kg every 12 hours for the first 24 hours 4 mg/kg every 12 hours 200 mg every 12 hours Candidemia in nonneutropenics and other deep tissue Candida infections 3 to 4 mg/kg every 12 hours 200 mg every 12 hours Scedosporiosis and Fusariosis 4 mg/kg every 12 hours 200 mg every 12 hours Esophageal Candidiasis Not Evaluated Not Evaluated 200 mg every 12 hours Adult patients weighing less than 40 kg: oral maintenance dose 100 mg or 150 mg every 12 hours Hepatic Impairment: Use half the maintenance dose in adult patients with mild to moderate hepatic impairment (Child-Pugh Class A and B) ( 2.5 ) Renal Impairment: Avoid intravenous administration in adult patients with moderate to severe renal impairment (creatinine clearance <50 mL/min) ( 2.6 ) Dosage in Pediatric Patients 2 years of age and older ( 2.4 ) For pediatric patients 2 to less than 12 years of age and 12 to 14 years of age weighing less than 50 kg see Table below. Infection Loading Dose Maintenance Dose Intravenous infusion Intravenous infusion Oral Invasive Aspergillosis 9 mg/kg every12 hours for the first 24 hours 8 mg/kg every 12 hours after the first 24 hours 9 mg/kg every 12 hours (maximum dose of 350 mg every 12 hours) Candidemia in nonneutropenics and other deep tissue Candida infections Scedosporiosis and Fusariosis Esophageal Candidiasis Not Evaluated 4 mg/kg every 12 hours 9 mg/kg every 12 hours (maximum dose of 350 mg every 12 hours) For pediatric patients aged 12 to 14 years weighing greater than or equal to 50 kg and those aged 15 years and older regardless of body weight use adult dosage. ( 2.4 ) Dosage adjustment of voriconazole for injection in pediatric patients with renal or hepatic impairment has not been established ( 2.5 , 2.6 ) See full prescribing information for instructions on reconstitution of voriconazole for injection lyophilized powder for intravenous use and important administration instructions ( 2.1 , 2.6 , 2.7 )
Contraindications
Voriconazole is contraindicated in patients with known hypersensitivity to voriconazole or its excipients. There is no information regarding cross-sensitivity between voriconazole and other azole antifungal agents. Caution should be used when prescribing voriconazole to patients with hypersensitivity to other azoles. Coadministration of pimozide, quinidine or ivabradine with voriconazole is contraindicated because increased plasma concentrations of these drugs can lead to QT prolongation and rare occurrences of torsade de pointes [see Drug Interactions ( 7 )] . Coadministration of voriconazole with sirolimus is contraindicated because voriconazole for injection significantly increases sirolimus concentrations [see Drug Interactions ( 7 ) and Clinical Pharmacology ( 12.3 )] . Coadministration of voriconazole with rifampin, carbamazepine, long-acting barbiturates, and St John's Wort is contraindicated because these drugs are likely to decrease plasma voriconazole concentrations significantly [see Drug Interactions ( 7 ) and Clinical Pharmacology ( 12.3 )] . Coadministration of standard doses of voriconazole with efavirenz doses of 400 mg every 24 hours or higher is contraindicated, because efavirenz significantly decreases plasma voriconazole concentrations in healthy subjects at these doses. Voriconazole also significantly increases efavirenz plasma concentrations [see Drug Interactions ( 7 ) and Clinical Pharmacology ( 12.3 )] . Coadministration of voriconazole with high-dose ritonavir (400 mg every 12 hours) is contraindicated because ritonavir (400 mg every 12 hours) significantly decreases plasma voriconazole concentrations. Coadministration of voriconazole and low-dose ritonavir (100 mg every 12 hours) should be avoided, unless an assessment of the benefit/risk to the patient justifies the use of voriconazole [see Drug Interactions ( 7 ) and Clinical Pharmacology ( 12.3 )] . Coadministration of voriconazole with rifabutin is contraindicated since voriconazole significantly increases rifabutin plasma concentrations and rifabutin also significantly decreases voriconazole plasma concentrations [see Drug Interactions ( 7 ) and Clinical Pharmacology ( 12.3 )] . Coadministration of voriconazole with ergot alkaloids (ergotamine and dihydroergotamine) is contraindicated because voriconazole may increase the plasma concentration of ergot alkaloids, which may lead to ergotism [see Drug Interactions ( 7 )] . Coadministration of voriconazole with naloxegol is contraindicated because voriconazole may increase plasma concentrations of naloxegol which may precipitate opioid withdrawal symptoms [see Drug Interactions ( 7 )] . Coadministration of voriconazole with tolvaptan is contraindicated because voriconazole may increase tolvaptan plasma concentrations and increase risk of adverse reactions [see Drug Interactions ( 7 )] . Coadministration of voriconazole with venetoclax at initiation and during the ramp-up phase is contraindicated in patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) due to the potential for increased risk of tumor lysis syndrome [see Drug Interactions ( 7 )] . Coadministration of voriconazole with lurasidone is contraindicated since it may result in significant increases in lurasidone exposure and the potential for serious adverse reactions [see Drug Interactions ( 7 )] .
Adverse Reactions
Severe cutaneous adverse reactions (SCARs), such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS), which can be life-threatening or fatal, have been reported during treatment with voriconazole. If a patient develops a severe cutaneous adverse reaction, voriconazole should be discontinued [see Adverse Reactions ( 6.1 , 6.2 )].
Drug Interactions
See Table 10 for a listing of drugs that may significantly alter voriconazole concentrations. Also, see Table 11 for a listing of drugs that may interact with voriconazole resulting in altered pharmacokinetics or pharmacodynamics of the other drug [see Contraindications ( 4 ) and Drug Interactions ( 7 )] .
Description
Warnings and Precautions, Photosensitivity ( 5.6 ) 10/2022
Section 42229-5
Blood products and concentrated electrolytes
Voriconazole for injection must not be infused concomitantly with any blood product or short-term infusion of concentrated electrolytes, even if the two infusions are running in separate intravenous lines (or cannulas). Electrolyte disturbances such as hypokalemia, hypomagnesemia and hypocalcemia should be corrected prior to initiation of and during voriconazole for injection therapy [see Warnings and Precautions (5.10)].
Section 42230-3
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: 8/2023 |
|||||||||||||||||||||||||||||
|
PATIENT INFORMATION
Voriconazole for Injection, for intravenous use |
||||||||||||||||||||||||||||||
| Read the Patient Information that comes with voriconazole for injection before you start taking it and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your condition or treatment. | ||||||||||||||||||||||||||||||
|
What is voriconazole for injection?
Voriconazole for injection is a prescription medicine used to treat certain serious fungal infections in your blood and body. These infections are called “aspergillosis,” “esophageal candidiasis,” “Scedosporium,” “Fusarium,” and “candidemia”. It is not known if voriconazole for injection is safe and effective in children younger than 2 years old. |
||||||||||||||||||||||||||||||
Do not take voriconazole for injection if you:
|
||||||||||||||||||||||||||||||
| Ask your healthcare provider or pharmacist if you are not sure if you are taking any of the medicines listed above. Do not start taking a new medicine without talking to your healthcare provider or pharmacist. |
||||||||||||||||||||||||||||||
Before you take voriconazole for injection, tell your healthcare provider about all of your medical conditions, including if you:
|
||||||||||||||||||||||||||||||
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements. Voriconazole for injection may affect the way other medicines work, and other medicines may affect how voriconazole for injection works. Know what medicines you take. Keep a list of them to show your healthcare provider or pharmacist when you get a new medicine. |
||||||||||||||||||||||||||||||
How should I take voriconazole for injection?
|
||||||||||||||||||||||||||||||
What should I avoid while taking voriconazole for injection?
|
||||||||||||||||||||||||||||||
|
What are possible side effects of voriconazole for injection?
Voriconazole for injection may cause serious side effects including:
|
||||||||||||||||||||||||||||||
| Call your healthcare provider or go to the nearest hospital emergency room right away if you have any of the symptoms listed above. | ||||||||||||||||||||||||||||||
These are not all the possible side effects of voriconazole for injection. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||||||||||||||||||||||||||||
How should I store voriconazole for injection?
|
||||||||||||||||||||||||||||||
|
General information about the safe and effective use of voriconazole for injection.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use voriconazole for injection for a condition for which it was not prescribed. Do not give voriconazole for injection to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about voriconazole for injection that is written for health professionals. |
||||||||||||||||||||||||||||||
|
What are the ingredients in voriconazole for injection?
Lake Zurich, IL 60047 www.fresenius-kabi.com/us 451665C Made in India |
Section 43683-2
| Warnings and Precautions, Photosensitivity (5.6) | 10/2022 |
Section 51945-4
PACKAGE LABEL - PRINCIPAL DISPLAY – Voriconazole for Injection 200mg* per vial - Carton
NDC 65219-190-30
1 Vial
Voriconazole
for Injection
200 mg* per vial
Not made with natural rubber latex
No Preservatives
For Intravenous Infusion Only
Sterile
Single-Dose Vial
Discard Unused Portion
Rx only
1.5 Usage
Specimens for fungal culture and other relevant laboratory studies (including histopathology) should be obtained prior to therapy to isolate and identify causative organism(s). Therapy may be instituted before the results of the cultures and other laboratory studies are known. However, once these results become available, antifungal therapy should be adjusted accordingly.
16.2 Storage
Voriconazole for injection unreconstituted vials should be stored at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Voriconazole for injection is a single dose unpreserved sterile lyophile. From a microbiological point of view, following reconstitution of the lyophile with Water for Injection, the reconstituted solution should be used immediately. If not used immediately, in-use storage times and conditions prior to use are the responsibility of the user and should not be longer than 24 hours at 2°C to 8°C (36°F to 46°F). Chemical and physical in-use stability has been demonstrated for 24 hours at 2°C to 8°C (36°F to 46°F). This medicinal product is for single use only and any unused solution should be discarded. Only clear solutions without particles should be used [see Dosage and Administration (2.1)].
10 Overdosage
In clinical trials, there were three cases of accidental overdose. All occurred in pediatric patients who received up to five times the recommended intravenous dose of voriconazole. A single adverse reaction of photophobia of 10 minutes duration was reported.
There is no known antidote to voriconazole.
Voriconazole is hemodialyzed with clearance of 121 mL/min. The intravenous vehicle, SBECD, is hemodialyzed with clearance of 55 mL/min. In an overdose, hemodialysis may assist in the removal of voriconazole and SBECD from the body.
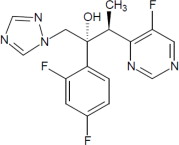
11 Description
Voriconazole, an azole antifungal agent is available as a lyophilized powder for solution for intravenous infusion. The structural formula is:
Voriconazole is designated chemically as (2R,3S)-2-(2, 4-difluorophenyl)-3-(5-fluoro-4-pyrimidinyl)-1-(1H-1,2,4-triazol-1-yl)-2-butanol with an empirical formula of C16H14F3N5O and a molecular weight of 349.3.
Voriconazole drug substance is a white to light-colored powder.
Voriconazole for injection is a white lyophilized powder containing nominally 200 mg voriconazole and 3200 mg sulfobutyl ether beta-cyclodextrin sodium in a 30 mL Type I clear glass vial.
Voriconazole for injection is intended for administration by intravenous infusion. It is a single-dose, unpreserved product. Vials containing 200 mg lyophilized voriconazole are intended for reconstitution with Water for Injection to produce a solution containing 10 mg/mL voriconazole and 160 mg/mL of sulfobutyl ether beta-cyclodextrin sodium. The resultant solution is further diluted prior to administration as an intravenous infusion [see Dosage and Administration (2)].
5.11 Pancreatitis
Pancreatitis has been observed in patients undergoing treatment with voriconazole [see Adverse Reactions (6.1, 6.2)] Patients with risk factors for acute pancreatitis (e.g., recent chemotherapy, hematopoietic stem cell transplantation [HSCT]) should be monitored for the development of pancreatitis during voriconazole treatment.
8.4 Pediatric Use
The safety and effectiveness of voriconazole have been established in pediatric patients 2 years of age and older based on evidence from adequate and well-controlled studies in adult and pediatric patients and additional pediatric pharmacokinetic and safety data. A total of 105 pediatric patients aged 2 to less than 12 [N=26] and aged 12 to less than 18 [N=79] from two, non-comparative Phase 3 pediatric studies and eight adult therapeutic trials provided safety information for voriconazole use in the pediatric population [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
Safety and effectiveness in pediatric patients below the age of 2 years has not been established. Therefore, voriconazole is not recommended for pediatric patients less than 2 years of age.
A higher frequency of liver enzyme elevations was observed in the pediatric patients [see Dosage and Administration (2.5), Warnings and Precautions (5.1), and Adverse Reactions (6.1)].
The frequency of phototoxicity reactions is higher in the pediatric population. Squamous cell carcinoma has been reported in patients who experience photosensitivity reactions. Stringent measures for photoprotection are warranted. Sun avoidance and dermatologic follow-up are recommended in pediatric patients experiencing photoaging injuries, such as lentigines or ephelides, even after treatment discontinuation [see Warnings and Precautions (5.6)].
Voriconazole has not been studied in pediatric patients with hepatic or renal impairment [see Dosage and Administration (2.5, 2.6)]. Hepatic function and serum creatinine levels should be closely monitored in pediatric patients [see Dosage and Administration (2.6) and Warnings and Precautions (5.1, 5.10)].
8.5 Geriatric Use
In multiple dose therapeutic trials of voriconazole, 9.2% of patients were ≥65 years of age and 1.8% of patients were ≥75 years of age. In a study in healthy subjects, the systemic exposure (AUC) and peak plasma concentrations (Cmax) were increased in elderly males compared to young males. Pharmacokinetic data obtained from 552 patients from 10 voriconazole therapeutic trials showed that voriconazole plasma concentrations in the elderly patients were approximately 80% to 90% higher than those in younger patients after either IV or oral administration. However, the overall safety profile of the elderly patients was similar to that of the young so no dosage adjustment is recommended [see Clinical Pharmacology (12.3)].
5.7 Renal Toxicity
Acute renal failure has been observed in patients undergoing treatment with voriconazole. Patients being treated with voriconazole are likely to be treated concomitantly with nephrotoxic medications and may have concurrent conditions that may result in decreased renal function.
Patients should be monitored for the development of abnormal renal function. This should include laboratory evaluation of serum creatinine [see Clinical Pharmacology (12.3) and Dosage and Administration (2.6)].
14 Clinical Studies
Voriconazole, administered orally or parenterally, has been evaluated as primary or salvage therapy in 520 patients aged 12 years and older with infections caused by Aspergillus spp., Fusarium spp., and Scedosporium spp.
4 Contraindications
- Voriconazole is contraindicated in patients with known hypersensitivity to voriconazole or its excipients. There is no information regarding cross-sensitivity between voriconazole and other azole antifungal agents. Caution should be used when prescribing voriconazole to patients with hypersensitivity to other azoles.
- Coadministration of pimozide, quinidine or ivabradine with voriconazole is contraindicated because increased plasma concentrations of these drugs can lead to QT prolongation and rare occurrences of torsade de pointes [see Drug Interactions (7)].
- Coadministration of voriconazole with sirolimus is contraindicated because voriconazole for injection significantly increases sirolimus concentrations [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
- Coadministration of voriconazole with rifampin, carbamazepine, long-acting barbiturates, and St John's Wort is contraindicated because these drugs are likely to decrease plasma voriconazole concentrations significantly [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
- Coadministration of standard doses of voriconazole with efavirenz doses of 400 mg every 24 hours or higher is contraindicated, because efavirenz significantly decreases plasma voriconazole concentrations in healthy subjects at these doses. Voriconazole also significantly increases efavirenz plasma concentrations [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
- Coadministration of voriconazole with high-dose ritonavir (400 mg every 12 hours) is contraindicated because ritonavir (400 mg every 12 hours) significantly decreases plasma voriconazole concentrations. Coadministration of voriconazole and low-dose ritonavir (100 mg every 12 hours) should be avoided, unless an assessment of the benefit/risk to the patient justifies the use of voriconazole [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
- Coadministration of voriconazole with rifabutin is contraindicated since voriconazole significantly increases rifabutin plasma concentrations and rifabutin also significantly decreases voriconazole plasma concentrations [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
- Coadministration of voriconazole with ergot alkaloids (ergotamine and dihydroergotamine) is contraindicated because voriconazole may increase the plasma concentration of ergot alkaloids, which may lead to ergotism [see Drug Interactions (7)].
- Coadministration of voriconazole with naloxegol is contraindicated because voriconazole may increase plasma concentrations of naloxegol which may precipitate opioid withdrawal symptoms [see Drug Interactions (7)].
- Coadministration of voriconazole with tolvaptan is contraindicated because voriconazole may increase tolvaptan plasma concentrations and increase risk of adverse reactions [see Drug Interactions (7)].
- Coadministration of voriconazole with venetoclax at initiation and during the ramp-up phase is contraindicated in patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) due to the potential for increased risk of tumor lysis syndrome [see Drug Interactions (7)].
- Coadministration of voriconazole with lurasidone is contraindicated since it may result in significant increases in lurasidone exposure and the potential for serious adverse reactions [see Drug Interactions (7)].
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling:
Hepatic Toxicity [see Warnings and Precautions (5.1)]
Arrhythmias and QT Prolongation [see Warnings and Precautions (5.2)]
Infusion Related Reactions [see Warnings and Precautions (5.3)]
Visual Disturbances [see Warnings and Precautions (5.4)]
Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.5)]
Photosensitivity [see Warnings and Precautions (5.6)]
Renal Toxicity [see Warnings and Precautions (5.7)]
7 Drug Interactions
Voriconazole is metabolized by cytochrome P450 isoenzymes, CYP2C19, CYP2C9, and CYP3A4. Therefore, inhibitors or inducers of these isoenzymes may increase or decrease voriconazole plasma concentrations, respectively. Voriconazole is a strong inhibitor of CYP3A4, and also inhibits CYP2C19 and CYP2C9. Therefore, voriconazole may increase the plasma concentrations of substances metabolized by these CYP450 isoenzymes.
Tables 10 and 11 provide the clinically significant interactions between voriconazole and other medical products.
|
* Results based on in vivo clinical studies generally following repeat oral dosing with 200 mg every 12 hours voriconazole to healthy subjects |
||
|
** Results based on in vivo clinical study following repeat oral dosing with 400 mg every 12 hours for 1 day, then 200 mg every 12 hours for at least 2 days voriconazole to healthy subjects |
||
|
*** Non-Nucleoside Reverse Transcriptase Inhibitors |
||
|
Drug/Drug Class
(Mechanism of Interaction by the Drug) |
Voriconazole Plasma Exposure (C max and AUC τ after 200 mg every 12 hours) |
Recommendations for Voriconazole Dosage Adjustment/Comments |
| Rifampin* and Rifabutin* (CYP450 Induction) |
Significantly Reduced | Contraindicated |
| Efavirenz (400 mg every 24 hours)** (CYP450 Induction) Efavirenz (300 mg every 24 hours)** (CYP450 Induction) |
Significantly Reduced Slight Decrease in AUCτ |
Contraindicated
When voriconazole is coadministered with efavirenz, voriconazole oral maintenance dose should be increased to 400 mg every 12 hours and efavirenz should be decreased to 300 mg every 24 hours. |
| High-dose Ritonavir (400 mg every 12 hours)** (CYP450 Induction) Low-dose Ritonavir (100 mg every 12 hours)** (CYP450 Induction) |
Significantly Reduced Reduced |
Contraindicated
Coadministration of voriconazole and low-dose ritonavir (100 mg every 12 hours) should be avoided, unless an assessment of the benefit/risk to the patient justifies the use of voriconazole. |
| Carbamazepine (CYP450 Induction) |
Not Studied In Vivo or In Vitro, but Likely to Result in Significant Reduction | Contraindicated |
| Long Acting Barbiturates (e.g., phenobarbital, mephobarbital) (CYP450 Induction) |
Not Studied In Vivo or In Vitro, but Likely to Result in Significant Reduction | Contraindicated |
| Phenytoin* (CYP450 Induction) |
Significantly Reduced | Increase voriconazole maintenance dose from 4 mg/kg to 5 mg/kg IV every 12 hours or from 200 mg to 400 mg orally every 12 hours (100 mg to 200 mg orally every 12 hours in patients weighing less than 40 kg). |
| Letermovir (CYP2C9/2C19 Induction) |
Reduced |
If concomitant administration of voriconazole with letermovir cannot be avoided, monitor for reduced effectiveness of voriconazole. |
| St. John's Wort (CYP450 inducer; P-gp inducer) |
Significantly Reduced | Contraindicated |
| Oral Contraceptives** containing ethinyl estradiol and norethindrone (CYP2C19 Inhibition) |
Increased | Monitoring for adverse reactions and toxicity related to voriconazole is recommended when coadministered with oral contraceptives. |
| Fluconazole** (CYP2C9, CYP2C19 and CYP3A4 Inhibition) | Significantly Increased | Avoid concomitant administration of voriconazole and fluconazole. Monitoring for adverse reactions and toxicity related to voriconazole is started within 24 hours after the last dose of fluconazole. |
| Other HIV Protease Inhibitors (CYP3A4 Inhibition) |
In Vivo Studies Showed No Significant Effects of Indinavir on Voriconazole Exposure In Vitro Studies Demonstrated Potential for Inhibition of Voriconazole Metabolism (Increased Plasma Exposure) |
No dosage adjustment in the voriconazole dosage needed when coadministered with indinavir. Frequent monitoring for adverse reactions and toxicity related to voriconazole when coadministered with other HIV protease inhibitors. |
| Other NNRTIs*** (CYP3A4 Inhibition or CYP450 Induction) |
In Vitro Studies Demonstrated Potential for Inhibition of Voriconazole Metabolism by Delavirdine and Other NNRTIs (Increased Plasma Exposure) A Voriconazole-Efavirenz Drug Interaction Study Demonstrated the Potential for the Metabolism of Voriconazole to be Induced by Efavirenz and Other NNRTIs (Decreased Plasma Exposure) |
Frequent monitoring for adverse reactions and toxicity related to voriconazole. Careful assessment of voriconazole effectiveness. |
|
* Results based on in vivo clinical studies generally following repeat oral dosing with 200 mg BID voriconazole to healthy subjects |
||
|
** Results based on in vivo clinical study following repeat oral dosing with 400 mg every 12 hours for 1 day, then 200 mg every 12 hours for at least 2 days voriconazole to healthy subjects |
||
|
*** Results based on in vivo clinical study following repeat oral dosing with 400 mg every 12 hours for 1 day, then 200 mg every 12 hours for 4 days voriconazole to subjects receiving a methadone maintenance dose (30 to 100 mg every 24 hours) |
||
|
**** Non-Steroidal Anti-Inflammatory Drug |
||
|
***** Non-Nucleoside Reverse Transcriptase Inhibitors |
||
|
Drug/Drug Class
(Mechanism of Interaction by Voriconazole) |
Drug Plasma Exposure
(C max and AUC τ ) |
Recommendations for Drug Dosage Adjustment/
Comments |
| Sirolimus* (CYP3A4 Inhibition) |
Significantly Increased | Contraindicated |
| Rifabutin* (CYP3A4 Inhibition) |
Significantly Increased | Contraindicated |
| Efavirenz (400 mg every 24 hours)** (CYP3A4 Inhibition) Efavirenz (300 mg every 24 hours)** (CYP3A4 Inhibition) |
Significantly Increased Slight Increase in AUCτ |
Contraindicated
When voriconazole is coadministered with efavirenz, voriconazole oral maintenance dose should be increased to 400 mg every 12 hours and efavirenz should be decreased to 300 mg every 24 hours. |
| High-dose Ritonavir (400 mg every 12 hours)** (CYP3A4 Inhibition) Low-dose Ritonavir (100 mg every 12 hours)** |
No Significant Effect of Voriconazole on Ritonavir Cmax or AUCτ Slight Decrease in Ritonavir Cmax and AUCτ |
Contraindicated because of significant reduction of voriconazole Cmax and AUCτ. Coadministration of voriconazole and low-dose ritonavir (100 mg every 12 hours) should be avoided (due to the reduction in voriconazole Cmax and AUCτ) unless an assessment of the benefit/risk to the patient justifies the use of voriconazole. |
| Pimozide, Quinidine, Ivabradine (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased |
Contraindicated because of potential for QT prolongation and rare occurrence of torsade de pointes. |
| Ergot Alkaloids (CYP450 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased |
Contraindicated |
| Naloxegol (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased which may Increase the Risk of Adverse Reactions |
Contraindicated
|
| Tolvaptan (CYP3A4 Inhibition) |
Although Not Studied Clinically, Voriconazole is Likely to Significantly Increase the Plasma Concentrations of Tolvaptan |
Contraindicated
|
| Venetoclax (CYP3A4 Inhibition) |
Not studied In Vivo or In Vitro, but Venetoclax Plasma Exposure Likely to be Significantly Increased |
Coadministration of voriconazole is contraindicated at initiation and during the ramp-up phase in patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL). Refer to the venetoclax labeling for safety monitoring and dose reduction in the steady daily dosing phase in CLL/SLL patients. For patients with acute myeloid leukemia (AML), dose reduction and safety monitoring are recommended across all dosing phases when coadministering voriconazole for injection with venetoclax. Refer to the venetoclax prescribing information for dosing instructions. |
| Lemborexant (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased | Avoid concomitant use of voriconazole with lemborexant. |
| Glasdegib (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased | Consider alternative therapies. If concomitant use cannot be avoided, monitor patients for increased risk of adverse reactions including QTc interval prolongation. |
| Tyrosine kinase inhibitors (including but not limited to axitinib, bosutinib, cabozantinib, ceritinib, cobimetinib, dabrafenib, dasatinib, nilotinib, sunitinib, ibrutinib, ribociclib) (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased | Avoid concomitant use of voriconazole. If concomitant use cannot be avoided, dose reduction of the tyrosine kinase inhibitor is recommended. Refer to the prescribing information for the relevant product. |
| Lurasidone (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Voriconazole is Likely to Significantly Increase the Plasma Concentrations of Lurasidone |
Contraindicated
|
| Cyclosporine* (CYP3A4 Inhibition) |
AUCτ Significantly Increased; No Significant Effect on Cmax |
When initiating therapy with voriconazole in patients already receiving cyclosporine, reduce the cyclosporine dose to one-half of the starting dose and follow with frequent monitoring of cyclosporine blood levels. Increased cyclosporine levels have been associated with nephrotoxicity. When voriconazole is discontinued, cyclosporine concentrations must be frequently monitored and the dose increased as necessary. |
| Methadone*** (CYP3A4 Inhibition) | Increased | Increased plasma concentrations of methadone have been associated with toxicity including QT prolongation. Frequent monitoring for adverse reactions and toxicity related to methadone is recommended during coadministration. Dose reduction of methadone may be needed. |
| Fentanyl (CYP3A4 Inhibition) | Increased | Reduction in the dose of fentanyl and other long-acting opiates metabolized by CYP3A4 should be considered when coadministered with voriconazole. Extended and frequent monitoring for opiate- associated adverse reactions may be necessary. |
| Alfentanil (CYP3A4 Inhibition) | Significantly Increased | An increase in the incidence of delayed and persistent alfentanil-associated nausea and vomiting were observed when coadministered with voriconazole for injection. Reduction in the dose of alfentanil and other opiates metabolized by CYP3A4 (e.g., sufentanil) should be considered when coadministered with voriconazole. A longer period for monitoring respiratory and other opiate-associated adverse reactions may be necessary. |
| Oxycodone (CYP3A4 Inhibition) | Significantly Increased | Increased visual effects (heterophoria and miosis) of oxycodone were observed when coadministered with voriconazole. Reduction in the dose of oxycodone and other long-acting opiates metabolized by CYP3A4 should be considered when coadministered with voriconazole. Extended and frequent monitoring for opiate-associated adverse reactions may be necessary. |
| NSAIDs**** including. ibuprofen and diclofenac (CYP2C9 Inhibition) |
Increased | Frequent monitoring for adverse reactions and toxicity related to NSAIDs. Dose reduction of NSAIDs may be needed. |
| Tacrolimus* (CYP3A4 Inhibition) |
Significantly Increased | When initiating therapy with voriconazole in patients already receiving tacrolimus, reduce the tacrolimus dose to one-third of the starting dose and follow with frequent monitoring of tacrolimus blood levels. Increased tacrolimus levels have been associated with nephrotoxicity. When voriconazole is discontinued, tacrolimus concentrations must be frequently monitored and the dose increased as necessary. |
| Phenytoin* (CYP2C9 Inhibition) |
Significantly Increased | Frequent monitoring of phenytoin plasma concentrations and frequent monitoring of adverse effects related to phenytoin. |
| Oral Contraceptives containing ethinyl estradiol and norethindrone (CYP3A4 Inhibition)** |
Increased | Monitoring for adverse reactions related to oral contraceptives is recommended during coadministration. |
| Prednisolone and other corticosteroids (CYP3A4 Inhibition) |
In Vivo Studies Showed No Significant Effects of Voriconazole on Prednisolone Exposure Not Studied In vitro or In vivo for Other Corticosteroids, but Drug Exposure Likely to be Increased |
No dosage adjustment for prednisolone when coadministered with voriconazole [see Clinical Pharmacology (12.3)].
Monitor for potential adrenal dysfunction when voriconazole is administered with other corticosteroids [See Warnings and Precautions (5.8)]. |
| Warfarin* (CYP2C9 Inhibition) Other Oral Coumarin Anticoagulants (CYP2C9/3A4 Inhibition) |
Prothrombin Time Significantly Increased Not Studied In Vivo or In Vitro for other Oral Coumarin Anticoagulants, but Drug Plasma Exposure Likely to be Increased |
If patients receiving coumarin preparations are treated simultaneously with voriconazole, the prothrombin time or other suitable anticoagulation tests should be monitored at close intervals and the dosage of anticoagulants adjusted accordingly. |
| Ivacaftor (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased which may Increase the Risk of Adverse Reactions |
Dose reduction of ivacaftor is recommended. Refer to the prescribing information for ivacaftor |
| Eszopiclone (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased which may Increase the Sedative Effect of Eszopiclone | Dose reduction of eszopiclone is recommended. Refer to the prescribing information for eszopiclone. |
| Omeprazole* (CYP2C19/3A4 Inhibition) |
Significantly Increased | When initiating therapy with voriconazole in patients already receiving omeprazole doses of 40 mg or greater, reduce the omeprazole dose by one-half. The metabolism of other proton pump inhibitors that are CYP2C19 substrates may also be inhibited by voriconazole and may result in increased plasma concentrations of other proton pump inhibitors. |
| Other HIV Protease Inhibitors (CYP3A4 Inhibition) |
In Vivo Studies Showed No Significant Effects on Indinavir Exposure In Vitro Studies Demonstrated Potential for Voriconazole to Inhibit Metabolism (Increased Plasma Exposure) |
No dosage adjustment for indinavir when coadministered with voriconazole. Frequent monitoring for adverse reactions and toxicity related to other HIV protease inhibitors. |
| Other NNRTIs***** (CYP3A4 Inhibition) |
A Voriconazole-Efavirenz Drug Interaction Study Demonstrated the Potential for Voriconazole to Inhibit Metabolism of Other NNRTIs (Increased Plasma Exposure) |
Frequent monitoring for adverse reactions and toxicity related to NNRTI. |
| Tretinoin (CYP3A4 Inhibition) |
Although Not Studied, Voriconazole may Increase Tretinoin Concentrations and Increase the Risk of Adverse Reactions | Frequent monitoring for signs and symptoms of pseudotumor cerebri or hypercalcemia. |
| Midazolam (CYP3A4 Inhibition) Other benzodiazepines including triazolam and alprazolam (CYP3A4 Inhibition) |
Significantly Increased In Vitro Studies Demonstrated Potential for Voriconazole to Inhibit Metabolism (Increased Plasma Exposure) |
Increased plasma exposures may increase the risk of adverse reactions and toxicities related to benzodiazepines. Refer to drug-specific labeling for details. |
| HMG-CoA Reductase Inhibitors (Statins) (CYP3A4 Inhibition) |
In Vitro Studies Demonstrated Potential for Voriconazole to Inhibit Metabolism (Increased Plasma Exposure) |
Frequent monitoring for adverse reactions and toxicity related to statins. Increased statin concentrations in plasma have been associated with rhabdomyolysis. Adjustment of the statin dosage may be needed. |
| Dihydropyridine Calcium Channel Blockers (CYP3A4 Inhibition) |
In Vitro Studies Demonstrated Potential for Voriconazole to Inhibit Metabolism (Increased Plasma Exposure) |
Frequent monitoring for adverse reactions and toxicity related to calcium channel blockers. Adjustment of calcium channel blocker dosage may be needed. |
| Sulfonylurea Oral Hypoglycemics (CYP2C9 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased | Frequent monitoring of blood glucose and for signs and symptoms of hypoglycemia. Adjustment of oral hypoglycemic drug dosage may be needed. |
| Vinca Alkaloids (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased | Frequent monitoring for adverse reactions and toxicity (i.e., neurotoxicity) related to vinca alkaloids. Reserve azole antifungals, including voriconazole, for patients receiving a vinca alkaloid who have no alternative antifungal treatment options. |
| Everolimus (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased | Concomitant administration of voriconazole and everolimus is not recommended. |
5.1 Hepatic Toxicity
In clinical trials, there have been uncommon cases of serious hepatic reactions during treatment with voriconazole (including clinical hepatitis, cholestasis and fulminant hepatic failure, including fatalities). Instances of hepatic reactions were noted to occur primarily in patients with serious underlying medical conditions (predominantly hematological malignancy). Hepatic reactions, including hepatitis and jaundice, have occurred among patients with no other identifiable risk factors. Liver dysfunction has usually been reversible on discontinuation of therapy [see Adverse Reactions (6.1)].
A higher frequency of liver enzyme elevations was observed in the pediatric population [see Adverse Reactions (6.1)]. Hepatic function should be monitored in both adult and pediatric patients.
Measure serum transaminase levels and bilirubin at the initiation of voriconazole therapy and monitor at least weekly for the first month of treatment. Monitoring frequency can be reduced to monthly during continued use if no clinically significant changes are noted. If liver function tests become markedly elevated compared to baseline, voriconazole should be discontinued unless the medical judgment of the benefit/risk of the treatment for the patient justifies continued use [see Dosage and Administration (2.5) and Adverse Reactions (6.1)].
5.6 Photosensitivity
Voriconazole has been associated with photosensitivity skin reaction. Patients, including pediatric patients, should avoid exposure to direct sunlight during voriconazole treatment and should use measures such as protective clothing and sunscreen with high sun protection factor (SPF). If phototoxic reactions occur, the patient should be referred to a dermatologist and voriconazole discontinuation should be considered. If voriconazole is continued despite the occurrence of phototoxicity-related lesions, dermatologic evaluation should be performed on a systematic and regular basis to allow early detection and management of premalignant lesions. Squamous cell carcinoma of the skin (including cutaneous SCC in situ, or Bowen's disease) and melanoma have been reported during long-term voriconazole therapy in patients with photosensitivity skin reactions. If a patient develops a skin lesion consistent with premalignant skin lesions, squamous cell carcinoma or melanoma, voriconazole should be discontinued. In addition, voriconazole has been associated with photosensitivity related skin reactions such as pseudoporphyria, cheilitis, and cutaneous lupus erythematosus, as well as increased risk of skin toxicity with concomitant use of methotrexate, a drug associated with ultraviolet (UV) reactivation. There is the potential for this risk to be observed with other drugs associated with UV reactivation. Patients should avoid strong, direct sunlight during voriconazole therapy.
The frequency of phototoxicity reactions is higher in the pediatric population. Because squamous cell carcinoma has been reported in patients who experience photosensitivity reactions, stringent measures for photoprotection are warranted in children. In children experiencing photoaging injuries such as lentigines or ephelides, sun avoidance and dermatologic follow-up are recommended even after treatment discontinuation.
12.3 Pharmacokinetics
The pharmacokinetics of voriconazole have been characterized in healthy subjects, special populations and patients.
The pharmacokinetics of voriconazole are non-linear due to saturation of its metabolism. The interindividual variability of voriconazole pharmacokinetics is high. Greater than proportional increase in exposure is observed with increasing dose. It is estimated that, on average, increasing the oral dose from 200 mg every 12 hours to 300 mg every 12 hours leads to an approximately 2.5-fold increase in exposure (AUCτ); similarly, increasing the intravenous dose from 3 mg/kg every 12 hours to 4 mg/kg every 12 hours produces an approximately 2.5-fold increase in exposure (Table 12).
|
Note: Parameters were estimated based on non-compartmental analysis from 5 pharmacokinetic studies. |
||||||
|
AUC12 = area under the curve over 12 hour dosing interval, Cmax = maximum plasma concentration, Cmin = minimum plasma concentration. CV = coefficient of variation |
||||||
|
6 mg/kg IV
(loading dose) |
3 mg/kg IV every 12 hours |
4 mg/kg
IV every 12 hours |
400 mg Oral
(loading dose) |
200 mg
Oral every 12 hours |
300 mg
Oral every 12 hours |
|
| N | 35 | 23 | 40 | 17 | 48 | 16 |
| AUC12 (mcg∙h/mL) | 13.9 (32) | 13.7 (53) | 33.9 (54) | 9.31 (38) | 12.4 (78) | 34.0 (53) |
| Cmax (mcg/mL) | 3.13 (20) | 3.03 (25) | 4.77 (36) | 2.30 (19) | 2.31 (48) | 4.74 (35) |
| Cmin (mcg/mL) | -- | 0.46 (97) | 1.73 (74) | -- | 0.46 (120) | 1.63 (79) |
When the recommended intravenous loading dose regimen is administered to healthy subjects, plasma concentrations close to steady state are achieved within the first 24 hours of dosing (e.g., 6 mg/kg IV every 12 hours on day 1 followed by 3 mg/kg IV every 12 hours). Without the loading dose, accumulation occurs during twice daily multiple dosing with steady state plasma voriconazole concentrations being achieved by day 6 in the majority of subjects.
12.5 Pharmacogenomics
CYP2C19, significantly involved in the metabolism of voriconazole, exhibits genetic polymorphism. Approximately 15 to 20% of Asian populations may be expected to be poor metabolizers. For Caucasians and Blacks, the prevalence of poor metabolizers is 3 to 5%. Studies conducted in Caucasian and Japanese healthy subjects have shown that poor metabolizers have, on average, 4-fold higher voriconazole exposure (AUCτ) than their homozygous extensive metabolizer counterparts. Subjects who are heterozygous extensive metabolizers have, on average, 2-fold higher voriconazole exposure than their homozygous extensive metabolizer counterparts [see Clinical Pharmacology (12.3)].
5.10 Laboratory Tests
Electrolyte disturbances such as hypokalemia, hypomagnesemia and hypocalcemia should be corrected prior to initiation of and during voriconazole therapy.
Patient management should include laboratory evaluation of renal (particularly serum creatinine) and hepatic function (particularly liver function tests and bilirubin).
14.5 Pediatric Studies
A total of 22 patients aged 12 to 18 years with IA were included in the adult therapeutic studies. Twelve out of 22 (55%) patients had successful response after treatment with a maintenance dose of voriconazole 4 mg/kg every 12 hours.
Fifty-three pediatric patients aged 2 to less than 18 years old were treated with voriconazole in two prospective, open-label, non-comparative, multicenter clinical studies.
One study was designed to enroll pediatric patients with IA or infections with rare molds (such as Scedosporium or Fusarium). Patients aged 2 to less than 12 years and 12 to 14 years with body weight less than 50 kg received an intravenous voriconazole loading dose of 9 mg/kg every 12 hours for the first 24-hours followed by an 8 mg/kg intravenous maintenance dose every 12 hours. After completing 7 days of intravenous therapy patients had an option to switch to oral voriconazole. The oral maintenance dose was 9 mg/kg every 12 hours (maximum dose of 350 mg). All other pediatric patients aged 12 to less than 18 years received the adult voriconazole dosage regimen. Patients received voriconazole for at least 6 weeks and up to a maximum of 12 weeks.
The study enrolled 31 patients with possible, proven, or probable IA. Fourteen of 31 patients, 5 of whom were 2 to less than 12 years old and 9 of whom were 12 to less than 18 years old, had proven or probable IA and were included in the modified intent-to-treat (MITT) efficacy analyses. No patients with rare mold were enrolled. A successful global response was defined as resolution or improvement in clinical signs and symptoms and at least 50% resolution of radiological lesions attributed to IA. The overall rate of successful global response at 6 weeks in the MITT population is presented in Table 18 below.
|
a Global response rate was defined as the number of subjects with a successful response (complete or partial) as a percentage of all subjects (including subjects with an indeterminate or missing response) at 6 weeks in the MITT population. |
|||
|
b The Modified Intent-to-Treat (MITT) population was defined as all subjects who received at least 1 dose of study drug and who were diagnosed with proven or probable IA as defined by the modified EORTC/MSG criteria. |
|||
| Parameter | Global Response at Week 6 | ||
|
Ages 2-<12 years
N=5 |
Ages 12-<18 years
N=9 |
Overall
N=14 |
|
| Number of successes, n (%) | 2 (40%) | 7 (78%) | 9 (64%) |
The second study enrolled 22 patients with invasive candidiasis including candidemia (ICC) and EC requiring either primary or salvage therapy. Patients with ICC aged 2 to less than 12 years and 12 to 14 years with body weight less than 50 kg received an intravenous voriconazole loading dose of 9 mg/kg every 12 hours for the first 24 hours followed by an 8 mg/kg intravenous maintenance dose every 12-hours. After completing 5 days of intravenous therapy patients had an option to switch to oral voriconazole. The oral maintenance dose was 9 mg/kg every 12 hours (maximum dose of 350 mg). All other pediatric patients aged 12 to less than 18 years received the adult voriconazole dosage regimen. Voriconazole was administered for at least 14 days after the last positive culture. A maximum of 42 days of treatment was permitted.
Patients with primary or salvage EC aged 2 to less than 12 years and 12 to 14 years with body weight less than 50 kg received an intravenous voriconazole dose of 4 mg/kg every 12 hours followed by an oral voriconazole dose of 9 mg/kg every 12 hours (maximum dose of 350 mg) when criteria for oral switch were met. All other pediatric patients aged 12 to less than 18 years received the adult voriconazole dosage regimen. Voriconazole was administered for at least 7 days after the resolution of clinical signs and symptoms. A maximum of 42 days of treatment was permitted.
For EC, study treatment was initiated without a loading dose of intravenous voriconazole. Seventeen of these patients had confirmed Candida infection and were included in the MITT efficacy analyses. Of the 17 patients included in the MITT analyses, 9 were 2 to less than 12 years old (7 with ICC and 2 with EC) and 8 were 12 to less than 18 years old (all with EC). For ICC and EC, a successful global response was defined as clinical cure or improvement with microbiological eradication or presumed eradication. The overall rate of successful global response at EOT in the MITT population is presented in Table 19 below.
|
a Global response was determined based on the investigator's assessment of clinical and microbiological response in the Modified Intent-to-Treat (MITT) analysis population at end of treatment. Subjects with missing data or whose response was deemed indeterminate were considered failures. |
||||
|
b The MITT population was defined as all subjects who received at least 1 dose of study drug and who had microbiologically confirmed invasive candidiasis with candidemia (ICC) and EC, or subjects with EC who had at least confirmation of oropharyngeal candidiasis without confirmation on esophagoscopy. |
||||
|
c All subjects with ICC were aged 2 to less than 12. |
||||
|
Parameter
|
Global Response at End of Treatment | |||
|
EC
N=10 |
ICC
c
N=7 |
|||
|
Ages 2-<12
N=2 |
Ages 12-<18
N=8 |
Overall
N=10 |
Overall
N=7 |
|
| Number of successes, n (%) | 2 (100%) | 5 (63%) | 7 (70%) | 6 (86%) |
1 Indications and Usage
Voriconazole for injection is an azole antifungal indicated for the treatment of adults and pediatric patients 2 years of age and older with:
- Invasive aspergillosis (1.1)
- Candidemia in non-neutropenics and other deep tissue Candida infections (1.2)
- Esophageal candidiasis (1.3)
- Serious fungal infections caused by Scedosporium apiospermum and Fusarium species including Fusarium solani, in patients intolerant of, or refractory to, other therapy (1.4)
5.4 Visual Disturbances
The effect of voriconazole on visual function is not known if treatment continues beyond 28 days. There have been postmarketing reports of prolonged visual adverse reactions, including optic neuritis and papilledema. If treatment continues beyond 28 days, visual function including visual acuity, visual field, and color perception should be monitored [see Adverse Reactions (6.2)].
5.8 Adrenal Dysfunction
Reversible cases of azole-induced adrenal insufficiency have been reported in patients receiving azoles, including voriconazole. Adrenal insufficiency has been reported in patients receiving azoles with or without concomitant corticosteroids. In patients receiving azoles without corticosteroids adrenal insufficiency is related to direct inhibition of steroidogenesis by azoles. In patients taking corticosteroids, voriconazole associated CYP3A4 inhibition of their metabolism may lead to corticosteroid excess and adrenal suppression [see Drug Interactions (7) and Clinical Pharmacology (12.3)]. Cushing's syndrome with and without subsequent adrenal insufficiency has also been reported in patients receiving voriconazole concomitantly with corticosteroids.
Patients receiving voriconazole and corticosteroids (via all routes of administration) should be carefully monitored for adrenal dysfunction both during and after voriconazole treatment. Patients should be instructed to seek immediate medical care if they develop signs and symptoms of Cushing's syndrome or adrenal insufficiency.
12.1 Mechanism of Action
Voriconazole is an antifungal drug [see Microbiology (12.4)].
5.9 Embryo Fetal Toxicity
Voriconazole can cause fetal harm when administered to a pregnant woman.
In animals, voriconazole administration was associated with fetal malformations, embryotoxicity, increased gestational length, dystocia and embryomortality [see Use in Specific Populations (8.1)].
If voriconazole is used during pregnancy, or if the patient becomes pregnant while taking voriconazole, inform the patient of the potential hazard to the fetus. Advise females of reproductive potential to use effective contraception during treatment with voriconazole [see Use in Specific Populations (8.3)].
1.1 Invasive Aspergillosis
Voriconazole for injection is indicated in adults and pediatric patients (2 years of age and older) for the treatment of invasive aspergillosis (IA). In clinical trials, the majority of isolates recovered were Aspergillus fumigatus. There was a small number of cases of culture-proven disease due to species of Aspergillus other than A. fumigatus [see Clinical Studies (14.1, 14.5) and Microbiology (12.4)].
1.3 Esophageal Candidiasis
5 Warnings and Precautions
- Hepatic Toxicity: Serious hepatic reactions reported. Evaluate liver function tests at start of and during voriconazole therapy (5.1)
- Arrhythmias and QT Prolongation: Correct potassium, magnesium and calcium prior to use; caution patients with proarrhythmic conditions (5.2)
- Infusion Related Reactions (including anaphylaxis): Stop the infusion (5.3)
- Visual Disturbances (including optic neuritis and papilledema): Monitor visual function if treatment continues beyond 28 days (5.4)
- Severe Cutaneous Adverse Reactions: Discontinue for exfoliative cutaneous reactions (5.5)
- Photosensitivity: Avoid sunlight due to risk of photosensitivity (5.6)
- Adrenal Dysfunction: Carefully monitor patients receiving voriconazole and corticosteroids (via all routes of administration) for adrenal dysfunction both during and after voriconazole treatment. Instruct patients to seek immediate medical care if they develop signs and symptoms of Cushing's syndrome or adrenal insufficiency (5.8)
- Embryo-Fetal Toxicity: Voriconazole can cause fetal harm when administered to a pregnant woman. Inform pregnant patients of the potential hazard to the fetus. Advise females of reproductive potential to use effective contraception during treatment with voriconazole (5.9, 8.1, 8.3)
- Skeletal Adverse Reactions: Fluorosis and periostitis with long-term voriconazole therapy. Discontinue if these adverse reactions occur (5.12)
- Clinically Significant Drug Interactions: Review patient's concomitant medications (5.13, 7)
2 Dosage and Administration
- Dosage in Adults (2.3)
|
Infection |
Loading dose | Maintenance Dose | |
| Intravenous infusion | Intravenous infusion | Oral | |
| Invasive Aspergillosis | 6 mg/kg every 12 hours for the first 24 hours |
4 mg/kg every 12 hours | 200 mg every 12 hours |
|
Candidemia in
nonneutropenics and other deep tissue Candida infections |
3 to 4 mg/kg every 12 hours | 200 mg every 12 hours | |
| Scedosporiosis and Fusariosis | 4 mg/kg every 12 hours | 200 mg every 12 hours | |
| Esophageal Candidiasis | Not Evaluated | Not Evaluated | 200 mg every 12 hours |
- Adult patients weighing less than 40 kg: oral maintenance dose 100 mg or 150 mg every 12 hours
- Hepatic Impairment: Use half the maintenance dose in adult patients with mild to moderate hepatic impairment (Child-Pugh Class A and B) (2.5)
- Renal Impairment: Avoid intravenous administration in adult patients with moderate to severe renal impairment (creatinine clearance <50 mL/min) (2.6)
- Dosage in Pediatric Patients 2 years of age and older (2.4)
- For pediatric patients 2 to less than 12 years of age and 12 to 14 years of age weighing less than 50 kg see Table below.
|
Infection |
Loading Dose | Maintenance Dose | |
| Intravenous infusion | Intravenous infusion |
Oral
|
|
| Invasive Aspergillosis | 9 mg/kg every12 hours for the first 24 hours |
8 mg/kg every 12 hours after the first 24 hours |
9 mg/kg every 12 hours (maximum dose of 350 mg every 12 hours) |
| Candidemia in nonneutropenics and other deep tissue Candida infections | |||
| Scedosporiosis and Fusariosis | |||
| Esophageal Candidiasis | Not Evaluated | 4 mg/kg every 12 hours | 9 mg/kg every 12 hours (maximum dose of 350 mg every 12 hours) |
- For pediatric patients aged 12 to 14 years weighing greater than or equal to 50 kg and those aged 15 years and older regardless of body weight use adult dosage. (2.4)
- Dosage adjustment of voriconazole for injection in pediatric patients with renal or hepatic impairment has not been established (2.5, 2.6)
3 Dosage Forms and Strengths
- For Injection: Lyophilized powder containing 200 mg of voriconazole and 3,200 mg of sulfobutyl ether beta-cyclodextrin sodium (SBECD); after reconstitution 10 mg/mL of voriconazole and 160 mg/mL of SBECD (3)
8 Use in Specific Populations
- Pediatrics: Safety and effectiveness in patients younger than 2 years has not been established (8.4)
5.3 Infusion Related Reactions
During infusion of the intravenous formulation of voriconazole in healthy subjects, anaphylactoid-type reactions, including flushing, fever, sweating, tachycardia, chest tightness, dyspnea, faintness, nausea, pruritus and rash, have occurred uncommonly. Symptoms appeared immediately upon initiating the infusion. Consideration should be given to stopping the infusion should these reactions occur.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.12 Skeletal Adverse Reactions
Fluorosis and periostitis have been reported during long-term voriconazole therapy. If a patient develops skeletal pain and radiologic findings compatible with fluorosis or periostitis, voriconazole should be discontinued [see Adverse Reactions (6.2)].
14.1 Invasive Aspergillosis (ia)
Voriconazole was studied in patients for primary therapy of IA (randomized, controlled study 307/602), for primary and salvage therapy of aspergillosis (non-comparative study 304) and for treatment of patients with IA who were refractory to, or intolerant of, other antifungal therapy (non-comparative study 309/604).
14.3 Esophageal Candidiasis (ec)
The efficacy of oral voriconazole 200 mg twice daily compared to oral fluconazole 200 mg once daily in the primary treatment of EC was demonstrated in Study 150 to 305, a double-blind, double-dummy study in immunocompromised patients with endoscopically-proven EC. Patients were treated for a median of 15 days (range 1 to 49 days). Outcome was assessed by repeat endoscopy at end of treatment (EOT). A successful response was defined as a normal endoscopy at EOT or at least a 1 grade improvement over baseline endoscopic score. For patients in the Intent-to-Treat (ITT) population with only a baseline endoscopy, a successful response was defined as symptomatic cure or improvement at EOT compared to baseline. Voriconazole and fluconazole (200 mg once daily) showed comparable efficacy rates against EC, as presented in Table 16.
|
a Confidence Interval for the difference (Voriconazole - Fluconazole) in success rates. |
|||
|
b PP (Per Protocol) patients had confirmation of Candida esophagitis by endoscopy, received at least 12 days of treatment, and had a repeat endoscopy at EOT (end of treatment). |
|||
|
c ITT (Intent to Treat) patients without endoscopy or clinical assessment at EOT were treated as failures. |
|||
| Population | Voriconazole | Fluconazole |
Difference %
(95% CI) a |
| PPb | 113/115 (98.2%) | 134/141 (95.0%) | 3.2 (-1.1, 7.5) |
| ITTc | 175/200 (87.5%) | 171/191 (89.5%) | -2.0 (-8.3, 4.3) |
Microbiologic success rates by Candida species are presented in Table 17.
|
a Some patients had more than one species isolated at baseline. |
||||
|
b Patients with endoscopic and/or mycological assessment at end of therapy. |
||||
| Pathogen a | Voriconazole | Fluconazole | ||
| Favorable endoscopic response b | Mycological eradication b | Favorable endoscopic response b | Mycological eradication b | |
| Success/Total (%) | Eradication/Total (%) | Success/Total (%) | Eradication/Total (%) | |
| C. albicans | 134/140 (96%) | 90/107 (84%) | 147/156 (94%) | 91/115 (79%) |
| C. glabrata | 8/8 (100%) | 4/7 (57%) | 4/4 (100%) | 1/4 (25%) |
| C. krusei | 1/1 | 1/1 | 2/2 (100%) | 0/0 |
1.4 Scedosporiosis and Fusariosis
Voriconazole for injection is indicated for the treatment of serious fungal infections caused by Scedosporium apiospermum (asexual form of Pseudallescheria boydii) and Fusarium spp. including Fusarium solani, in adults and pediatric patients (2 years of age and older) intolerant of, or refractory to, other therapy [see Clinical Studies (14.4) and Microbiology (12.4)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
14.4 Other Serious Fungal Pathogens
In pooled analyses of patients, voriconazole was shown to be effective against the following additional fungal pathogens:
Scedosporium apiospermum - Successful response to voriconazole therapy was seen in 15 of 24 patients (63%). Three of these patients relapsed within 4 weeks, including 1 patient with pulmonary, skin and eye infections, 1 patient with cerebral disease, and 1 patient with skin infection. Ten patients had evidence of cerebral disease and 6 of these had a successful outcome (1 relapse). In addition, a successful response was seen in 1 of 3 patients with mixed organism infections.
Fusarium spp. - Nine of 21 (43%) patients were successfully treated with voriconazole. Of these 9 patients, 3 had eye infections, 1 had an eye and blood infection, 1 had a skin infection, 1 had a blood infection alone, 2 had sinus infections, and 1 had disseminated infection (pulmonary, skin, hepatosplenic). Three of these patients (1 with disseminated disease, 1 with an eye infection and 1 with a blood infection) had Fusarium solani and were complete successes. Two of these patients relapsed, 1 with a sinus infection and profound neutropenia and 1 post surgical patient with blood and eye infections.
5.2 Arrhythmias and Qt Prolongation
Some azoles, including voriconazole, have been associated with prolongation of the QT interval on the electrocardiogram. During clinical development and postmarketing surveillance, there have been rare cases of arrhythmias, (including ventricular arrhythmias such as torsade de pointes), cardiac arrests and sudden deaths in patients taking voriconazole. These cases usually involved seriously ill patients with multiple confounding risk factors, such as history of cardiotoxic chemotherapy, cardiomyopathy, hypokalemia and concomitant medications that may have been contributory.
Voriconazole should be administered with caution to patients with potentially proarrhythmic conditions, such as:
- Congenital or acquired QT prolongation
- Cardiomyopathy, in particular when heart failure is present
- Sinus bradycardia
- Existing symptomatic arrhythmias
- Concomitant medicinal product that is known to prolong QT interval [see Contraindications (4), Drug Interactions (7), and Clinical Pharmacology (12.3)]
Rigorous attempts to correct potassium, magnesium and calcium should be made before starting and during voriconazole therapy [see Clinical Pharmacology (12.3)].
5.5 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCARs), such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS), which can be life-threatening or fatal, have been reported during treatment with voriconazole. If a patient develops a severe cutaneous adverse reaction, voriconazole should be discontinued [see Adverse Reactions (6.1, 6.2)].
5.13 Clinically Significant Drug Interactions
See Table 10 for a listing of drugs that may significantly alter voriconazole concentrations. Also, see Table 11 for a listing of drugs that may interact with voriconazole resulting in altered pharmacokinetics or pharmacodynamics of the other drug [see Contraindications (4) and Drug Interactions (7)].
2.4 Recommended Dosing Regimen in Pediatric Patients
The recommended dosing regimen for pediatric patients 2 to less than 12 years of age and 12 to 14 years of age with body weight less than 50 kg is shown in Table 2. For pediatric patients 12 to 14 years of age with a body weight greater than or equal to 50 kg and those 15 years of age and above regardless of body weight, administer the adult dosing regimen of voriconazole [see Dosage and Administration (2.3)].
|
^ Based on a population pharmacokinetic analysis in 112 immunocompromised pediatric patients aged 2 to less than 12 years of age and 26 immunocompromised pediatric patients aged 12 to less than 17 years of age. |
|||
|
* In the Phase 3 clinical trials, patients with IA received intravenous (IV) treatment for at least 6 weeks and up to a maximum of 12 weeks. Patients received IV treatment for at least the first 7 days of therapy and then could be switched to oral voriconazole therapy. |
|||
|
† Study treatment for primary or salvage invasive candidiasis and candidemia (ICC) or EC consisted of intravenous voriconazole, with an option to switch to oral therapy after at least 5 days of IV therapy, based on subjects meeting switch criteria. For subjects with primary or salvage ICC, voriconazole was administered for at least 14 days after the last positive culture. A maximum of 42 days of treatment was permitted. Patients with primary or salvage EC were treated for at least 7 days after the resolution of clinical signs and symptoms. A maximum of 42 days of treatment was permitted. |
|||
| Loading Dose | Maintenance Dose | ||
| Intravenous infusion | Intravenous infusion | Oral | |
| Invasive Aspergillosis* | 9 mg/kg every 12 hours for the first 24 hours | 8 mg/kg every 12 hours after the first 24 hours | 9 mg/kg every 12 hours (maximum dose of 350 mg every 12 hours) |
| Candidemia in nonneutropenics and other deep tissue Candida infections † | |||
| Scedosporiosis and Fusariosis | |||
| Esophageal Candidiasis † | Not Evaluated | 4 mg/kg every 12 hours | 9 mg/kg every 12 hours (maximum dose of 350 mg every 12 hours) |
Initiate therapy with an intravenous infusion regimen. Consider an oral regimen only after there is a significant clinical improvement. Note that an 8 mg/kg intravenous dose will provide voriconazole exposure approximately 2-fold higher than a 9 mg/kg oral dose.
The oral dose recommendation for children is based on studies in which voriconazole was administered as the powder for oral suspension formulation. Bioequivalence between the voriconazole powder for oral suspension and voriconazole tablets has not been investigated in a pediatric population.
Oral bioavailability may be limited in pediatric patients 2 to 12 years with malabsorption and very low body weight for age. In that case, intravenous voriconazole administration is recommended.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year carcinogenicity studies were conducted in rats and mice. Rats were given oral doses of 6, 18 or 50 mg/kg voriconazole, or 0.2, 0.6, or 1.6 times the RMD on a body surface area basis. Hepatocellular adenomas were detected in females at 50 mg/kg and hepatocellular carcinomas were found in males at 6 and 50 mg/kg. Mice were given oral doses of 10, 30 or 100 mg/kg voriconazole, or 0.1, 0.4, or 1.4 times the RMD on a body surface area basis. In mice, hepatocellular adenomas were detected in males and females and hepatocellular carcinomas were detected in males at 1.4 times the RMD of voriconazole.
Voriconazole demonstrated clastogenic activity (mostly chromosome breaks) in human lymphocyte cultures in vitro. Voriconazole was not genotoxic in the Ames assay, CHO HGPRT assay, the mouse micronucleus assay or the in vivo DNA repair test (Unscheduled DNA Synthesis assay).
Voriconazole administration induced no impairment of male or female fertility in rats dosed at 50 mg/kg, or 1.6 times the RMD.
6.2 Postmarketing Experience in Adult and Pediatric Patients
The following adverse reactions have been identified during post-approval use of voriconazole. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
2.1 Important Administration Instructions for Use in All Patients
Voriconazole for injection requires reconstitution to 10 mg/mL and subsequent dilution to 5 mg/mL or less prior to administration as an infusion, at a maximum rate of 3 mg/kg per hour over 1 to 3 hours.
Administer diluted voriconazole for injection by intravenous infusion over 1 to 3 hours only. Do not administer as an IV bolus injection.
2.7 Dosage Adjustment When Co Administered With Phenytoin Or Efavirenz
1.2 Candidemia in Non Neutropenic Patients and Other Deep Tissue Candida
Voriconazole for injection is indicated in adults and pediatric patients (2 years of age and older) for the treatment of candidemia in non-neutropenic patients and the following Candida infections: disseminated infections in skin and infections in abdomen, kidney, bladder wall, and wounds [see Clinical Studies (14.2, 14.5) and Microbiology (12.4)].
14.2 Candidemia in Non Neutropenic Patients and Other Deep Tissue Candida
Voriconazole was compared to the regimen of amphotericin B followed by fluconazole in Study 608, an open-label, comparative study in nonneutropenic patients with candidemia associated with clinical signs of infection. Patients were randomized in 2:1 ratio to receive either voriconazole (n=283) or the regimen of amphotericin B followed by fluconazole (n=139). Patients were treated with randomized study drug for a median of 15 days. Most of the candidemia in patients evaluated for efficacy was caused by C. albicans (46%), followed by C. tropicalis (19%), C. parapsilosis (17%), C. glabrata (15%), and C. krusei (1%).
An independent Data Review Committee (DRC), blinded to study treatment, reviewed the clinical and mycological data from this study, and generated one assessment of response for each patient. A successful response required all of the following: resolution or improvement in all clinical signs and symptoms of infection, blood cultures negative for Candida, infected deep tissue sites negative for Candida or resolution of all local signs of infection, and no systemic antifungal therapy other than study drug. The primary analysis, which counted DRC-assessed successes at the fixed time point (12 weeks after End of Therapy [EOT]), demonstrated that voriconazole was comparable to the regimen of amphotericin B followed by fluconazole (response rates of 41% and 41%, respectively) in the treatment of candidemia. Patients who did not have a 12-week assessment for any reason were considered a treatment failure.
The overall clinical and mycological success rates by Candida species in Study 150 to 608 are presented in Table 15.
|
a A few patients had more than one pathogen at baseline. |
||
|
b Patients who did not have a 12-week assessment for any reason were considered a treatment failure. |
||
| Baseline Pathogen | Clinical and Mycological Success (%) | |
| Voriconazole | Amphotericin B --> Fluconazole | |
| C. albicans | 46/107 (43%) | 30/63 (48%) |
| C. tropicalis | 17/53 (32%) | 1/16 (6%) |
| C. parapsilosis | 24/45 (53%) | 10/19 (53%) |
| C. glabrata | 12/36 (33%) | 7/21 (33%) |
| C. krusei | 1/4 | 0/1 |
In a secondary analysis, which counted DRC-assessed successes at any time point (EOT, or 2, 6, or 12 weeks after EOT), the response rates were 65% for voriconazole and 71% for the regimen of amphotericin B followed by fluconazole.
In Studies 608 and 309/604 (non-comparative study in patients with invasive fungal infections who were refractory to, or intolerant of, other antifungal agents), voriconazole was evaluated in 35 patients with deep tissue Candida infections. A favorable response was seen in 4 of 7 patients with intra-abdominal infections, 5 of 6 patients with kidney and bladder wall infections, 3 of 3 patients with deep tissue abscess or wound infection, 1 of 2 patients with pneumonia/pleural space infections, 2 of 4 patients with skin lesions, 1 of 1 patients with mixed intra-abdominal and pulmonary infection, 1 of 2 patients with suppurative phlebitis, 1 of 3 patients with hepatosplenic infection, 1 of 5 patients with osteomyelitis, 0 of 1 with liver infection, and 0 of 1 with cervical lymph node infection.
Structured Label Content
Section 42229-5 (42229-5)
Blood products and concentrated electrolytes
Voriconazole for injection must not be infused concomitantly with any blood product or short-term infusion of concentrated electrolytes, even if the two infusions are running in separate intravenous lines (or cannulas). Electrolyte disturbances such as hypokalemia, hypomagnesemia and hypocalcemia should be corrected prior to initiation of and during voriconazole for injection therapy [see Warnings and Precautions (5.10)].
Section 42230-3 (42230-3)
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: 8/2023 |
|||||||||||||||||||||||||||||
|
PATIENT INFORMATION
Voriconazole for Injection, for intravenous use |
||||||||||||||||||||||||||||||
| Read the Patient Information that comes with voriconazole for injection before you start taking it and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your condition or treatment. | ||||||||||||||||||||||||||||||
|
What is voriconazole for injection?
Voriconazole for injection is a prescription medicine used to treat certain serious fungal infections in your blood and body. These infections are called “aspergillosis,” “esophageal candidiasis,” “Scedosporium,” “Fusarium,” and “candidemia”. It is not known if voriconazole for injection is safe and effective in children younger than 2 years old. |
||||||||||||||||||||||||||||||
Do not take voriconazole for injection if you:
|
||||||||||||||||||||||||||||||
| Ask your healthcare provider or pharmacist if you are not sure if you are taking any of the medicines listed above. Do not start taking a new medicine without talking to your healthcare provider or pharmacist. |
||||||||||||||||||||||||||||||
Before you take voriconazole for injection, tell your healthcare provider about all of your medical conditions, including if you:
|
||||||||||||||||||||||||||||||
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements. Voriconazole for injection may affect the way other medicines work, and other medicines may affect how voriconazole for injection works. Know what medicines you take. Keep a list of them to show your healthcare provider or pharmacist when you get a new medicine. |
||||||||||||||||||||||||||||||
How should I take voriconazole for injection?
|
||||||||||||||||||||||||||||||
What should I avoid while taking voriconazole for injection?
|
||||||||||||||||||||||||||||||
|
What are possible side effects of voriconazole for injection?
Voriconazole for injection may cause serious side effects including:
|
||||||||||||||||||||||||||||||
| Call your healthcare provider or go to the nearest hospital emergency room right away if you have any of the symptoms listed above. | ||||||||||||||||||||||||||||||
These are not all the possible side effects of voriconazole for injection. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||||||||||||||||||||||||||||
How should I store voriconazole for injection?
|
||||||||||||||||||||||||||||||
|
General information about the safe and effective use of voriconazole for injection.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use voriconazole for injection for a condition for which it was not prescribed. Do not give voriconazole for injection to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about voriconazole for injection that is written for health professionals. |
||||||||||||||||||||||||||||||
|
What are the ingredients in voriconazole for injection?
Lake Zurich, IL 60047 www.fresenius-kabi.com/us 451665C Made in India |
Section 43683-2 (43683-2)
| Warnings and Precautions, Photosensitivity (5.6) | 10/2022 |
Section 51945-4 (51945-4)
PACKAGE LABEL - PRINCIPAL DISPLAY – Voriconazole for Injection 200mg* per vial - Carton
NDC 65219-190-30
1 Vial
Voriconazole
for Injection
200 mg* per vial
Not made with natural rubber latex
No Preservatives
For Intravenous Infusion Only
Sterile
Single-Dose Vial
Discard Unused Portion
Rx only
1.5 Usage
Specimens for fungal culture and other relevant laboratory studies (including histopathology) should be obtained prior to therapy to isolate and identify causative organism(s). Therapy may be instituted before the results of the cultures and other laboratory studies are known. However, once these results become available, antifungal therapy should be adjusted accordingly.
16.2 Storage
Voriconazole for injection unreconstituted vials should be stored at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Voriconazole for injection is a single dose unpreserved sterile lyophile. From a microbiological point of view, following reconstitution of the lyophile with Water for Injection, the reconstituted solution should be used immediately. If not used immediately, in-use storage times and conditions prior to use are the responsibility of the user and should not be longer than 24 hours at 2°C to 8°C (36°F to 46°F). Chemical and physical in-use stability has been demonstrated for 24 hours at 2°C to 8°C (36°F to 46°F). This medicinal product is for single use only and any unused solution should be discarded. Only clear solutions without particles should be used [see Dosage and Administration (2.1)].
10 Overdosage (10 OVERDOSAGE)
In clinical trials, there were three cases of accidental overdose. All occurred in pediatric patients who received up to five times the recommended intravenous dose of voriconazole. A single adverse reaction of photophobia of 10 minutes duration was reported.
There is no known antidote to voriconazole.
Voriconazole is hemodialyzed with clearance of 121 mL/min. The intravenous vehicle, SBECD, is hemodialyzed with clearance of 55 mL/min. In an overdose, hemodialysis may assist in the removal of voriconazole and SBECD from the body.
11 Description (11 DESCRIPTION)
Voriconazole, an azole antifungal agent is available as a lyophilized powder for solution for intravenous infusion. The structural formula is:
Voriconazole is designated chemically as (2R,3S)-2-(2, 4-difluorophenyl)-3-(5-fluoro-4-pyrimidinyl)-1-(1H-1,2,4-triazol-1-yl)-2-butanol with an empirical formula of C16H14F3N5O and a molecular weight of 349.3.
Voriconazole drug substance is a white to light-colored powder.
Voriconazole for injection is a white lyophilized powder containing nominally 200 mg voriconazole and 3200 mg sulfobutyl ether beta-cyclodextrin sodium in a 30 mL Type I clear glass vial.
Voriconazole for injection is intended for administration by intravenous infusion. It is a single-dose, unpreserved product. Vials containing 200 mg lyophilized voriconazole are intended for reconstitution with Water for Injection to produce a solution containing 10 mg/mL voriconazole and 160 mg/mL of sulfobutyl ether beta-cyclodextrin sodium. The resultant solution is further diluted prior to administration as an intravenous infusion [see Dosage and Administration (2)].
5.11 Pancreatitis
Pancreatitis has been observed in patients undergoing treatment with voriconazole [see Adverse Reactions (6.1, 6.2)] Patients with risk factors for acute pancreatitis (e.g., recent chemotherapy, hematopoietic stem cell transplantation [HSCT]) should be monitored for the development of pancreatitis during voriconazole treatment.
8.4 Pediatric Use
The safety and effectiveness of voriconazole have been established in pediatric patients 2 years of age and older based on evidence from adequate and well-controlled studies in adult and pediatric patients and additional pediatric pharmacokinetic and safety data. A total of 105 pediatric patients aged 2 to less than 12 [N=26] and aged 12 to less than 18 [N=79] from two, non-comparative Phase 3 pediatric studies and eight adult therapeutic trials provided safety information for voriconazole use in the pediatric population [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
Safety and effectiveness in pediatric patients below the age of 2 years has not been established. Therefore, voriconazole is not recommended for pediatric patients less than 2 years of age.
A higher frequency of liver enzyme elevations was observed in the pediatric patients [see Dosage and Administration (2.5), Warnings and Precautions (5.1), and Adverse Reactions (6.1)].
The frequency of phototoxicity reactions is higher in the pediatric population. Squamous cell carcinoma has been reported in patients who experience photosensitivity reactions. Stringent measures for photoprotection are warranted. Sun avoidance and dermatologic follow-up are recommended in pediatric patients experiencing photoaging injuries, such as lentigines or ephelides, even after treatment discontinuation [see Warnings and Precautions (5.6)].
Voriconazole has not been studied in pediatric patients with hepatic or renal impairment [see Dosage and Administration (2.5, 2.6)]. Hepatic function and serum creatinine levels should be closely monitored in pediatric patients [see Dosage and Administration (2.6) and Warnings and Precautions (5.1, 5.10)].
8.5 Geriatric Use
In multiple dose therapeutic trials of voriconazole, 9.2% of patients were ≥65 years of age and 1.8% of patients were ≥75 years of age. In a study in healthy subjects, the systemic exposure (AUC) and peak plasma concentrations (Cmax) were increased in elderly males compared to young males. Pharmacokinetic data obtained from 552 patients from 10 voriconazole therapeutic trials showed that voriconazole plasma concentrations in the elderly patients were approximately 80% to 90% higher than those in younger patients after either IV or oral administration. However, the overall safety profile of the elderly patients was similar to that of the young so no dosage adjustment is recommended [see Clinical Pharmacology (12.3)].
5.7 Renal Toxicity
Acute renal failure has been observed in patients undergoing treatment with voriconazole. Patients being treated with voriconazole are likely to be treated concomitantly with nephrotoxic medications and may have concurrent conditions that may result in decreased renal function.
Patients should be monitored for the development of abnormal renal function. This should include laboratory evaluation of serum creatinine [see Clinical Pharmacology (12.3) and Dosage and Administration (2.6)].
14 Clinical Studies (14 CLINICAL STUDIES)
Voriconazole, administered orally or parenterally, has been evaluated as primary or salvage therapy in 520 patients aged 12 years and older with infections caused by Aspergillus spp., Fusarium spp., and Scedosporium spp.
4 Contraindications (4 CONTRAINDICATIONS)
- Voriconazole is contraindicated in patients with known hypersensitivity to voriconazole or its excipients. There is no information regarding cross-sensitivity between voriconazole and other azole antifungal agents. Caution should be used when prescribing voriconazole to patients with hypersensitivity to other azoles.
- Coadministration of pimozide, quinidine or ivabradine with voriconazole is contraindicated because increased plasma concentrations of these drugs can lead to QT prolongation and rare occurrences of torsade de pointes [see Drug Interactions (7)].
- Coadministration of voriconazole with sirolimus is contraindicated because voriconazole for injection significantly increases sirolimus concentrations [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
- Coadministration of voriconazole with rifampin, carbamazepine, long-acting barbiturates, and St John's Wort is contraindicated because these drugs are likely to decrease plasma voriconazole concentrations significantly [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
- Coadministration of standard doses of voriconazole with efavirenz doses of 400 mg every 24 hours or higher is contraindicated, because efavirenz significantly decreases plasma voriconazole concentrations in healthy subjects at these doses. Voriconazole also significantly increases efavirenz plasma concentrations [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
- Coadministration of voriconazole with high-dose ritonavir (400 mg every 12 hours) is contraindicated because ritonavir (400 mg every 12 hours) significantly decreases plasma voriconazole concentrations. Coadministration of voriconazole and low-dose ritonavir (100 mg every 12 hours) should be avoided, unless an assessment of the benefit/risk to the patient justifies the use of voriconazole [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
- Coadministration of voriconazole with rifabutin is contraindicated since voriconazole significantly increases rifabutin plasma concentrations and rifabutin also significantly decreases voriconazole plasma concentrations [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
- Coadministration of voriconazole with ergot alkaloids (ergotamine and dihydroergotamine) is contraindicated because voriconazole may increase the plasma concentration of ergot alkaloids, which may lead to ergotism [see Drug Interactions (7)].
- Coadministration of voriconazole with naloxegol is contraindicated because voriconazole may increase plasma concentrations of naloxegol which may precipitate opioid withdrawal symptoms [see Drug Interactions (7)].
- Coadministration of voriconazole with tolvaptan is contraindicated because voriconazole may increase tolvaptan plasma concentrations and increase risk of adverse reactions [see Drug Interactions (7)].
- Coadministration of voriconazole with venetoclax at initiation and during the ramp-up phase is contraindicated in patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) due to the potential for increased risk of tumor lysis syndrome [see Drug Interactions (7)].
- Coadministration of voriconazole with lurasidone is contraindicated since it may result in significant increases in lurasidone exposure and the potential for serious adverse reactions [see Drug Interactions (7)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in the labeling:
Hepatic Toxicity [see Warnings and Precautions (5.1)]
Arrhythmias and QT Prolongation [see Warnings and Precautions (5.2)]
Infusion Related Reactions [see Warnings and Precautions (5.3)]
Visual Disturbances [see Warnings and Precautions (5.4)]
Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.5)]
Photosensitivity [see Warnings and Precautions (5.6)]
Renal Toxicity [see Warnings and Precautions (5.7)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Voriconazole is metabolized by cytochrome P450 isoenzymes, CYP2C19, CYP2C9, and CYP3A4. Therefore, inhibitors or inducers of these isoenzymes may increase or decrease voriconazole plasma concentrations, respectively. Voriconazole is a strong inhibitor of CYP3A4, and also inhibits CYP2C19 and CYP2C9. Therefore, voriconazole may increase the plasma concentrations of substances metabolized by these CYP450 isoenzymes.
Tables 10 and 11 provide the clinically significant interactions between voriconazole and other medical products.
|
* Results based on in vivo clinical studies generally following repeat oral dosing with 200 mg every 12 hours voriconazole to healthy subjects |
||
|
** Results based on in vivo clinical study following repeat oral dosing with 400 mg every 12 hours for 1 day, then 200 mg every 12 hours for at least 2 days voriconazole to healthy subjects |
||
|
*** Non-Nucleoside Reverse Transcriptase Inhibitors |
||
|
Drug/Drug Class
(Mechanism of Interaction by the Drug) |
Voriconazole Plasma Exposure (C max and AUC τ after 200 mg every 12 hours) |
Recommendations for Voriconazole Dosage Adjustment/Comments |
| Rifampin* and Rifabutin* (CYP450 Induction) |
Significantly Reduced | Contraindicated |
| Efavirenz (400 mg every 24 hours)** (CYP450 Induction) Efavirenz (300 mg every 24 hours)** (CYP450 Induction) |
Significantly Reduced Slight Decrease in AUCτ |
Contraindicated
When voriconazole is coadministered with efavirenz, voriconazole oral maintenance dose should be increased to 400 mg every 12 hours and efavirenz should be decreased to 300 mg every 24 hours. |
| High-dose Ritonavir (400 mg every 12 hours)** (CYP450 Induction) Low-dose Ritonavir (100 mg every 12 hours)** (CYP450 Induction) |
Significantly Reduced Reduced |
Contraindicated
Coadministration of voriconazole and low-dose ritonavir (100 mg every 12 hours) should be avoided, unless an assessment of the benefit/risk to the patient justifies the use of voriconazole. |
| Carbamazepine (CYP450 Induction) |
Not Studied In Vivo or In Vitro, but Likely to Result in Significant Reduction | Contraindicated |
| Long Acting Barbiturates (e.g., phenobarbital, mephobarbital) (CYP450 Induction) |
Not Studied In Vivo or In Vitro, but Likely to Result in Significant Reduction | Contraindicated |
| Phenytoin* (CYP450 Induction) |
Significantly Reduced | Increase voriconazole maintenance dose from 4 mg/kg to 5 mg/kg IV every 12 hours or from 200 mg to 400 mg orally every 12 hours (100 mg to 200 mg orally every 12 hours in patients weighing less than 40 kg). |
| Letermovir (CYP2C9/2C19 Induction) |
Reduced |
If concomitant administration of voriconazole with letermovir cannot be avoided, monitor for reduced effectiveness of voriconazole. |
| St. John's Wort (CYP450 inducer; P-gp inducer) |
Significantly Reduced | Contraindicated |
| Oral Contraceptives** containing ethinyl estradiol and norethindrone (CYP2C19 Inhibition) |
Increased | Monitoring for adverse reactions and toxicity related to voriconazole is recommended when coadministered with oral contraceptives. |
| Fluconazole** (CYP2C9, CYP2C19 and CYP3A4 Inhibition) | Significantly Increased | Avoid concomitant administration of voriconazole and fluconazole. Monitoring for adverse reactions and toxicity related to voriconazole is started within 24 hours after the last dose of fluconazole. |
| Other HIV Protease Inhibitors (CYP3A4 Inhibition) |
In Vivo Studies Showed No Significant Effects of Indinavir on Voriconazole Exposure In Vitro Studies Demonstrated Potential for Inhibition of Voriconazole Metabolism (Increased Plasma Exposure) |
No dosage adjustment in the voriconazole dosage needed when coadministered with indinavir. Frequent monitoring for adverse reactions and toxicity related to voriconazole when coadministered with other HIV protease inhibitors. |
| Other NNRTIs*** (CYP3A4 Inhibition or CYP450 Induction) |
In Vitro Studies Demonstrated Potential for Inhibition of Voriconazole Metabolism by Delavirdine and Other NNRTIs (Increased Plasma Exposure) A Voriconazole-Efavirenz Drug Interaction Study Demonstrated the Potential for the Metabolism of Voriconazole to be Induced by Efavirenz and Other NNRTIs (Decreased Plasma Exposure) |
Frequent monitoring for adverse reactions and toxicity related to voriconazole. Careful assessment of voriconazole effectiveness. |
|
* Results based on in vivo clinical studies generally following repeat oral dosing with 200 mg BID voriconazole to healthy subjects |
||
|
** Results based on in vivo clinical study following repeat oral dosing with 400 mg every 12 hours for 1 day, then 200 mg every 12 hours for at least 2 days voriconazole to healthy subjects |
||
|
*** Results based on in vivo clinical study following repeat oral dosing with 400 mg every 12 hours for 1 day, then 200 mg every 12 hours for 4 days voriconazole to subjects receiving a methadone maintenance dose (30 to 100 mg every 24 hours) |
||
|
**** Non-Steroidal Anti-Inflammatory Drug |
||
|
***** Non-Nucleoside Reverse Transcriptase Inhibitors |
||
|
Drug/Drug Class
(Mechanism of Interaction by Voriconazole) |
Drug Plasma Exposure
(C max and AUC τ ) |
Recommendations for Drug Dosage Adjustment/
Comments |
| Sirolimus* (CYP3A4 Inhibition) |
Significantly Increased | Contraindicated |
| Rifabutin* (CYP3A4 Inhibition) |
Significantly Increased | Contraindicated |
| Efavirenz (400 mg every 24 hours)** (CYP3A4 Inhibition) Efavirenz (300 mg every 24 hours)** (CYP3A4 Inhibition) |
Significantly Increased Slight Increase in AUCτ |
Contraindicated
When voriconazole is coadministered with efavirenz, voriconazole oral maintenance dose should be increased to 400 mg every 12 hours and efavirenz should be decreased to 300 mg every 24 hours. |
| High-dose Ritonavir (400 mg every 12 hours)** (CYP3A4 Inhibition) Low-dose Ritonavir (100 mg every 12 hours)** |
No Significant Effect of Voriconazole on Ritonavir Cmax or AUCτ Slight Decrease in Ritonavir Cmax and AUCτ |
Contraindicated because of significant reduction of voriconazole Cmax and AUCτ. Coadministration of voriconazole and low-dose ritonavir (100 mg every 12 hours) should be avoided (due to the reduction in voriconazole Cmax and AUCτ) unless an assessment of the benefit/risk to the patient justifies the use of voriconazole. |
| Pimozide, Quinidine, Ivabradine (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased |
Contraindicated because of potential for QT prolongation and rare occurrence of torsade de pointes. |
| Ergot Alkaloids (CYP450 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased |
Contraindicated |
| Naloxegol (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased which may Increase the Risk of Adverse Reactions |
Contraindicated
|
| Tolvaptan (CYP3A4 Inhibition) |
Although Not Studied Clinically, Voriconazole is Likely to Significantly Increase the Plasma Concentrations of Tolvaptan |
Contraindicated
|
| Venetoclax (CYP3A4 Inhibition) |
Not studied In Vivo or In Vitro, but Venetoclax Plasma Exposure Likely to be Significantly Increased |
Coadministration of voriconazole is contraindicated at initiation and during the ramp-up phase in patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL). Refer to the venetoclax labeling for safety monitoring and dose reduction in the steady daily dosing phase in CLL/SLL patients. For patients with acute myeloid leukemia (AML), dose reduction and safety monitoring are recommended across all dosing phases when coadministering voriconazole for injection with venetoclax. Refer to the venetoclax prescribing information for dosing instructions. |
| Lemborexant (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased | Avoid concomitant use of voriconazole with lemborexant. |
| Glasdegib (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased | Consider alternative therapies. If concomitant use cannot be avoided, monitor patients for increased risk of adverse reactions including QTc interval prolongation. |
| Tyrosine kinase inhibitors (including but not limited to axitinib, bosutinib, cabozantinib, ceritinib, cobimetinib, dabrafenib, dasatinib, nilotinib, sunitinib, ibrutinib, ribociclib) (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased | Avoid concomitant use of voriconazole. If concomitant use cannot be avoided, dose reduction of the tyrosine kinase inhibitor is recommended. Refer to the prescribing information for the relevant product. |
| Lurasidone (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Voriconazole is Likely to Significantly Increase the Plasma Concentrations of Lurasidone |
Contraindicated
|
| Cyclosporine* (CYP3A4 Inhibition) |
AUCτ Significantly Increased; No Significant Effect on Cmax |
When initiating therapy with voriconazole in patients already receiving cyclosporine, reduce the cyclosporine dose to one-half of the starting dose and follow with frequent monitoring of cyclosporine blood levels. Increased cyclosporine levels have been associated with nephrotoxicity. When voriconazole is discontinued, cyclosporine concentrations must be frequently monitored and the dose increased as necessary. |
| Methadone*** (CYP3A4 Inhibition) | Increased | Increased plasma concentrations of methadone have been associated with toxicity including QT prolongation. Frequent monitoring for adverse reactions and toxicity related to methadone is recommended during coadministration. Dose reduction of methadone may be needed. |
| Fentanyl (CYP3A4 Inhibition) | Increased | Reduction in the dose of fentanyl and other long-acting opiates metabolized by CYP3A4 should be considered when coadministered with voriconazole. Extended and frequent monitoring for opiate- associated adverse reactions may be necessary. |
| Alfentanil (CYP3A4 Inhibition) | Significantly Increased | An increase in the incidence of delayed and persistent alfentanil-associated nausea and vomiting were observed when coadministered with voriconazole for injection. Reduction in the dose of alfentanil and other opiates metabolized by CYP3A4 (e.g., sufentanil) should be considered when coadministered with voriconazole. A longer period for monitoring respiratory and other opiate-associated adverse reactions may be necessary. |
| Oxycodone (CYP3A4 Inhibition) | Significantly Increased | Increased visual effects (heterophoria and miosis) of oxycodone were observed when coadministered with voriconazole. Reduction in the dose of oxycodone and other long-acting opiates metabolized by CYP3A4 should be considered when coadministered with voriconazole. Extended and frequent monitoring for opiate-associated adverse reactions may be necessary. |
| NSAIDs**** including. ibuprofen and diclofenac (CYP2C9 Inhibition) |
Increased | Frequent monitoring for adverse reactions and toxicity related to NSAIDs. Dose reduction of NSAIDs may be needed. |
| Tacrolimus* (CYP3A4 Inhibition) |
Significantly Increased | When initiating therapy with voriconazole in patients already receiving tacrolimus, reduce the tacrolimus dose to one-third of the starting dose and follow with frequent monitoring of tacrolimus blood levels. Increased tacrolimus levels have been associated with nephrotoxicity. When voriconazole is discontinued, tacrolimus concentrations must be frequently monitored and the dose increased as necessary. |
| Phenytoin* (CYP2C9 Inhibition) |
Significantly Increased | Frequent monitoring of phenytoin plasma concentrations and frequent monitoring of adverse effects related to phenytoin. |
| Oral Contraceptives containing ethinyl estradiol and norethindrone (CYP3A4 Inhibition)** |
Increased | Monitoring for adverse reactions related to oral contraceptives is recommended during coadministration. |
| Prednisolone and other corticosteroids (CYP3A4 Inhibition) |
In Vivo Studies Showed No Significant Effects of Voriconazole on Prednisolone Exposure Not Studied In vitro or In vivo for Other Corticosteroids, but Drug Exposure Likely to be Increased |
No dosage adjustment for prednisolone when coadministered with voriconazole [see Clinical Pharmacology (12.3)].
Monitor for potential adrenal dysfunction when voriconazole is administered with other corticosteroids [See Warnings and Precautions (5.8)]. |
| Warfarin* (CYP2C9 Inhibition) Other Oral Coumarin Anticoagulants (CYP2C9/3A4 Inhibition) |
Prothrombin Time Significantly Increased Not Studied In Vivo or In Vitro for other Oral Coumarin Anticoagulants, but Drug Plasma Exposure Likely to be Increased |
If patients receiving coumarin preparations are treated simultaneously with voriconazole, the prothrombin time or other suitable anticoagulation tests should be monitored at close intervals and the dosage of anticoagulants adjusted accordingly. |
| Ivacaftor (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased which may Increase the Risk of Adverse Reactions |
Dose reduction of ivacaftor is recommended. Refer to the prescribing information for ivacaftor |
| Eszopiclone (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased which may Increase the Sedative Effect of Eszopiclone | Dose reduction of eszopiclone is recommended. Refer to the prescribing information for eszopiclone. |
| Omeprazole* (CYP2C19/3A4 Inhibition) |
Significantly Increased | When initiating therapy with voriconazole in patients already receiving omeprazole doses of 40 mg or greater, reduce the omeprazole dose by one-half. The metabolism of other proton pump inhibitors that are CYP2C19 substrates may also be inhibited by voriconazole and may result in increased plasma concentrations of other proton pump inhibitors. |
| Other HIV Protease Inhibitors (CYP3A4 Inhibition) |
In Vivo Studies Showed No Significant Effects on Indinavir Exposure In Vitro Studies Demonstrated Potential for Voriconazole to Inhibit Metabolism (Increased Plasma Exposure) |
No dosage adjustment for indinavir when coadministered with voriconazole. Frequent monitoring for adverse reactions and toxicity related to other HIV protease inhibitors. |
| Other NNRTIs***** (CYP3A4 Inhibition) |
A Voriconazole-Efavirenz Drug Interaction Study Demonstrated the Potential for Voriconazole to Inhibit Metabolism of Other NNRTIs (Increased Plasma Exposure) |
Frequent monitoring for adverse reactions and toxicity related to NNRTI. |
| Tretinoin (CYP3A4 Inhibition) |
Although Not Studied, Voriconazole may Increase Tretinoin Concentrations and Increase the Risk of Adverse Reactions | Frequent monitoring for signs and symptoms of pseudotumor cerebri or hypercalcemia. |
| Midazolam (CYP3A4 Inhibition) Other benzodiazepines including triazolam and alprazolam (CYP3A4 Inhibition) |
Significantly Increased In Vitro Studies Demonstrated Potential for Voriconazole to Inhibit Metabolism (Increased Plasma Exposure) |
Increased plasma exposures may increase the risk of adverse reactions and toxicities related to benzodiazepines. Refer to drug-specific labeling for details. |
| HMG-CoA Reductase Inhibitors (Statins) (CYP3A4 Inhibition) |
In Vitro Studies Demonstrated Potential for Voriconazole to Inhibit Metabolism (Increased Plasma Exposure) |
Frequent monitoring for adverse reactions and toxicity related to statins. Increased statin concentrations in plasma have been associated with rhabdomyolysis. Adjustment of the statin dosage may be needed. |
| Dihydropyridine Calcium Channel Blockers (CYP3A4 Inhibition) |
In Vitro Studies Demonstrated Potential for Voriconazole to Inhibit Metabolism (Increased Plasma Exposure) |
Frequent monitoring for adverse reactions and toxicity related to calcium channel blockers. Adjustment of calcium channel blocker dosage may be needed. |
| Sulfonylurea Oral Hypoglycemics (CYP2C9 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased | Frequent monitoring of blood glucose and for signs and symptoms of hypoglycemia. Adjustment of oral hypoglycemic drug dosage may be needed. |
| Vinca Alkaloids (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased | Frequent monitoring for adverse reactions and toxicity (i.e., neurotoxicity) related to vinca alkaloids. Reserve azole antifungals, including voriconazole, for patients receiving a vinca alkaloid who have no alternative antifungal treatment options. |
| Everolimus (CYP3A4 Inhibition) |
Not Studied In Vivo or In Vitro, but Drug Plasma Exposure Likely to be Increased | Concomitant administration of voriconazole and everolimus is not recommended. |
5.1 Hepatic Toxicity
In clinical trials, there have been uncommon cases of serious hepatic reactions during treatment with voriconazole (including clinical hepatitis, cholestasis and fulminant hepatic failure, including fatalities). Instances of hepatic reactions were noted to occur primarily in patients with serious underlying medical conditions (predominantly hematological malignancy). Hepatic reactions, including hepatitis and jaundice, have occurred among patients with no other identifiable risk factors. Liver dysfunction has usually been reversible on discontinuation of therapy [see Adverse Reactions (6.1)].
A higher frequency of liver enzyme elevations was observed in the pediatric population [see Adverse Reactions (6.1)]. Hepatic function should be monitored in both adult and pediatric patients.
Measure serum transaminase levels and bilirubin at the initiation of voriconazole therapy and monitor at least weekly for the first month of treatment. Monitoring frequency can be reduced to monthly during continued use if no clinically significant changes are noted. If liver function tests become markedly elevated compared to baseline, voriconazole should be discontinued unless the medical judgment of the benefit/risk of the treatment for the patient justifies continued use [see Dosage and Administration (2.5) and Adverse Reactions (6.1)].
5.6 Photosensitivity
Voriconazole has been associated with photosensitivity skin reaction. Patients, including pediatric patients, should avoid exposure to direct sunlight during voriconazole treatment and should use measures such as protective clothing and sunscreen with high sun protection factor (SPF). If phototoxic reactions occur, the patient should be referred to a dermatologist and voriconazole discontinuation should be considered. If voriconazole is continued despite the occurrence of phototoxicity-related lesions, dermatologic evaluation should be performed on a systematic and regular basis to allow early detection and management of premalignant lesions. Squamous cell carcinoma of the skin (including cutaneous SCC in situ, or Bowen's disease) and melanoma have been reported during long-term voriconazole therapy in patients with photosensitivity skin reactions. If a patient develops a skin lesion consistent with premalignant skin lesions, squamous cell carcinoma or melanoma, voriconazole should be discontinued. In addition, voriconazole has been associated with photosensitivity related skin reactions such as pseudoporphyria, cheilitis, and cutaneous lupus erythematosus, as well as increased risk of skin toxicity with concomitant use of methotrexate, a drug associated with ultraviolet (UV) reactivation. There is the potential for this risk to be observed with other drugs associated with UV reactivation. Patients should avoid strong, direct sunlight during voriconazole therapy.
The frequency of phototoxicity reactions is higher in the pediatric population. Because squamous cell carcinoma has been reported in patients who experience photosensitivity reactions, stringent measures for photoprotection are warranted in children. In children experiencing photoaging injuries such as lentigines or ephelides, sun avoidance and dermatologic follow-up are recommended even after treatment discontinuation.
12.3 Pharmacokinetics
The pharmacokinetics of voriconazole have been characterized in healthy subjects, special populations and patients.
The pharmacokinetics of voriconazole are non-linear due to saturation of its metabolism. The interindividual variability of voriconazole pharmacokinetics is high. Greater than proportional increase in exposure is observed with increasing dose. It is estimated that, on average, increasing the oral dose from 200 mg every 12 hours to 300 mg every 12 hours leads to an approximately 2.5-fold increase in exposure (AUCτ); similarly, increasing the intravenous dose from 3 mg/kg every 12 hours to 4 mg/kg every 12 hours produces an approximately 2.5-fold increase in exposure (Table 12).
|
Note: Parameters were estimated based on non-compartmental analysis from 5 pharmacokinetic studies. |
||||||
|
AUC12 = area under the curve over 12 hour dosing interval, Cmax = maximum plasma concentration, Cmin = minimum plasma concentration. CV = coefficient of variation |
||||||
|
6 mg/kg IV
(loading dose) |
3 mg/kg IV every 12 hours |
4 mg/kg
IV every 12 hours |
400 mg Oral
(loading dose) |
200 mg
Oral every 12 hours |
300 mg
Oral every 12 hours |
|
| N | 35 | 23 | 40 | 17 | 48 | 16 |
| AUC12 (mcg∙h/mL) | 13.9 (32) | 13.7 (53) | 33.9 (54) | 9.31 (38) | 12.4 (78) | 34.0 (53) |
| Cmax (mcg/mL) | 3.13 (20) | 3.03 (25) | 4.77 (36) | 2.30 (19) | 2.31 (48) | 4.74 (35) |
| Cmin (mcg/mL) | -- | 0.46 (97) | 1.73 (74) | -- | 0.46 (120) | 1.63 (79) |
When the recommended intravenous loading dose regimen is administered to healthy subjects, plasma concentrations close to steady state are achieved within the first 24 hours of dosing (e.g., 6 mg/kg IV every 12 hours on day 1 followed by 3 mg/kg IV every 12 hours). Without the loading dose, accumulation occurs during twice daily multiple dosing with steady state plasma voriconazole concentrations being achieved by day 6 in the majority of subjects.
12.5 Pharmacogenomics
CYP2C19, significantly involved in the metabolism of voriconazole, exhibits genetic polymorphism. Approximately 15 to 20% of Asian populations may be expected to be poor metabolizers. For Caucasians and Blacks, the prevalence of poor metabolizers is 3 to 5%. Studies conducted in Caucasian and Japanese healthy subjects have shown that poor metabolizers have, on average, 4-fold higher voriconazole exposure (AUCτ) than their homozygous extensive metabolizer counterparts. Subjects who are heterozygous extensive metabolizers have, on average, 2-fold higher voriconazole exposure than their homozygous extensive metabolizer counterparts [see Clinical Pharmacology (12.3)].
5.10 Laboratory Tests
Electrolyte disturbances such as hypokalemia, hypomagnesemia and hypocalcemia should be corrected prior to initiation of and during voriconazole therapy.
Patient management should include laboratory evaluation of renal (particularly serum creatinine) and hepatic function (particularly liver function tests and bilirubin).
14.5 Pediatric Studies
A total of 22 patients aged 12 to 18 years with IA were included in the adult therapeutic studies. Twelve out of 22 (55%) patients had successful response after treatment with a maintenance dose of voriconazole 4 mg/kg every 12 hours.
Fifty-three pediatric patients aged 2 to less than 18 years old were treated with voriconazole in two prospective, open-label, non-comparative, multicenter clinical studies.
One study was designed to enroll pediatric patients with IA or infections with rare molds (such as Scedosporium or Fusarium). Patients aged 2 to less than 12 years and 12 to 14 years with body weight less than 50 kg received an intravenous voriconazole loading dose of 9 mg/kg every 12 hours for the first 24-hours followed by an 8 mg/kg intravenous maintenance dose every 12 hours. After completing 7 days of intravenous therapy patients had an option to switch to oral voriconazole. The oral maintenance dose was 9 mg/kg every 12 hours (maximum dose of 350 mg). All other pediatric patients aged 12 to less than 18 years received the adult voriconazole dosage regimen. Patients received voriconazole for at least 6 weeks and up to a maximum of 12 weeks.
The study enrolled 31 patients with possible, proven, or probable IA. Fourteen of 31 patients, 5 of whom were 2 to less than 12 years old and 9 of whom were 12 to less than 18 years old, had proven or probable IA and were included in the modified intent-to-treat (MITT) efficacy analyses. No patients with rare mold were enrolled. A successful global response was defined as resolution or improvement in clinical signs and symptoms and at least 50% resolution of radiological lesions attributed to IA. The overall rate of successful global response at 6 weeks in the MITT population is presented in Table 18 below.
|
a Global response rate was defined as the number of subjects with a successful response (complete or partial) as a percentage of all subjects (including subjects with an indeterminate or missing response) at 6 weeks in the MITT population. |
|||
|
b The Modified Intent-to-Treat (MITT) population was defined as all subjects who received at least 1 dose of study drug and who were diagnosed with proven or probable IA as defined by the modified EORTC/MSG criteria. |
|||
| Parameter | Global Response at Week 6 | ||
|
Ages 2-<12 years
N=5 |
Ages 12-<18 years
N=9 |
Overall
N=14 |
|
| Number of successes, n (%) | 2 (40%) | 7 (78%) | 9 (64%) |
The second study enrolled 22 patients with invasive candidiasis including candidemia (ICC) and EC requiring either primary or salvage therapy. Patients with ICC aged 2 to less than 12 years and 12 to 14 years with body weight less than 50 kg received an intravenous voriconazole loading dose of 9 mg/kg every 12 hours for the first 24 hours followed by an 8 mg/kg intravenous maintenance dose every 12-hours. After completing 5 days of intravenous therapy patients had an option to switch to oral voriconazole. The oral maintenance dose was 9 mg/kg every 12 hours (maximum dose of 350 mg). All other pediatric patients aged 12 to less than 18 years received the adult voriconazole dosage regimen. Voriconazole was administered for at least 14 days after the last positive culture. A maximum of 42 days of treatment was permitted.
Patients with primary or salvage EC aged 2 to less than 12 years and 12 to 14 years with body weight less than 50 kg received an intravenous voriconazole dose of 4 mg/kg every 12 hours followed by an oral voriconazole dose of 9 mg/kg every 12 hours (maximum dose of 350 mg) when criteria for oral switch were met. All other pediatric patients aged 12 to less than 18 years received the adult voriconazole dosage regimen. Voriconazole was administered for at least 7 days after the resolution of clinical signs and symptoms. A maximum of 42 days of treatment was permitted.
For EC, study treatment was initiated without a loading dose of intravenous voriconazole. Seventeen of these patients had confirmed Candida infection and were included in the MITT efficacy analyses. Of the 17 patients included in the MITT analyses, 9 were 2 to less than 12 years old (7 with ICC and 2 with EC) and 8 were 12 to less than 18 years old (all with EC). For ICC and EC, a successful global response was defined as clinical cure or improvement with microbiological eradication or presumed eradication. The overall rate of successful global response at EOT in the MITT population is presented in Table 19 below.
|
a Global response was determined based on the investigator's assessment of clinical and microbiological response in the Modified Intent-to-Treat (MITT) analysis population at end of treatment. Subjects with missing data or whose response was deemed indeterminate were considered failures. |
||||
|
b The MITT population was defined as all subjects who received at least 1 dose of study drug and who had microbiologically confirmed invasive candidiasis with candidemia (ICC) and EC, or subjects with EC who had at least confirmation of oropharyngeal candidiasis without confirmation on esophagoscopy. |
||||
|
c All subjects with ICC were aged 2 to less than 12. |
||||
|
Parameter
|
Global Response at End of Treatment | |||
|
EC
N=10 |
ICC
c
N=7 |
|||
|
Ages 2-<12
N=2 |
Ages 12-<18
N=8 |
Overall
N=10 |
Overall
N=7 |
|
| Number of successes, n (%) | 2 (100%) | 5 (63%) | 7 (70%) | 6 (86%) |
1 Indications and Usage (1 INDICATIONS AND USAGE)
Voriconazole for injection is an azole antifungal indicated for the treatment of adults and pediatric patients 2 years of age and older with:
- Invasive aspergillosis (1.1)
- Candidemia in non-neutropenics and other deep tissue Candida infections (1.2)
- Esophageal candidiasis (1.3)
- Serious fungal infections caused by Scedosporium apiospermum and Fusarium species including Fusarium solani, in patients intolerant of, or refractory to, other therapy (1.4)
5.4 Visual Disturbances
The effect of voriconazole on visual function is not known if treatment continues beyond 28 days. There have been postmarketing reports of prolonged visual adverse reactions, including optic neuritis and papilledema. If treatment continues beyond 28 days, visual function including visual acuity, visual field, and color perception should be monitored [see Adverse Reactions (6.2)].
5.8 Adrenal Dysfunction
Reversible cases of azole-induced adrenal insufficiency have been reported in patients receiving azoles, including voriconazole. Adrenal insufficiency has been reported in patients receiving azoles with or without concomitant corticosteroids. In patients receiving azoles without corticosteroids adrenal insufficiency is related to direct inhibition of steroidogenesis by azoles. In patients taking corticosteroids, voriconazole associated CYP3A4 inhibition of their metabolism may lead to corticosteroid excess and adrenal suppression [see Drug Interactions (7) and Clinical Pharmacology (12.3)]. Cushing's syndrome with and without subsequent adrenal insufficiency has also been reported in patients receiving voriconazole concomitantly with corticosteroids.
Patients receiving voriconazole and corticosteroids (via all routes of administration) should be carefully monitored for adrenal dysfunction both during and after voriconazole treatment. Patients should be instructed to seek immediate medical care if they develop signs and symptoms of Cushing's syndrome or adrenal insufficiency.
12.1 Mechanism of Action
Voriconazole is an antifungal drug [see Microbiology (12.4)].
5.9 Embryo Fetal Toxicity (5.9 Embryo-Fetal Toxicity)
Voriconazole can cause fetal harm when administered to a pregnant woman.
In animals, voriconazole administration was associated with fetal malformations, embryotoxicity, increased gestational length, dystocia and embryomortality [see Use in Specific Populations (8.1)].
If voriconazole is used during pregnancy, or if the patient becomes pregnant while taking voriconazole, inform the patient of the potential hazard to the fetus. Advise females of reproductive potential to use effective contraception during treatment with voriconazole [see Use in Specific Populations (8.3)].
1.1 Invasive Aspergillosis
Voriconazole for injection is indicated in adults and pediatric patients (2 years of age and older) for the treatment of invasive aspergillosis (IA). In clinical trials, the majority of isolates recovered were Aspergillus fumigatus. There was a small number of cases of culture-proven disease due to species of Aspergillus other than A. fumigatus [see Clinical Studies (14.1, 14.5) and Microbiology (12.4)].
1.3 Esophageal Candidiasis
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hepatic Toxicity: Serious hepatic reactions reported. Evaluate liver function tests at start of and during voriconazole therapy (5.1)
- Arrhythmias and QT Prolongation: Correct potassium, magnesium and calcium prior to use; caution patients with proarrhythmic conditions (5.2)
- Infusion Related Reactions (including anaphylaxis): Stop the infusion (5.3)
- Visual Disturbances (including optic neuritis and papilledema): Monitor visual function if treatment continues beyond 28 days (5.4)
- Severe Cutaneous Adverse Reactions: Discontinue for exfoliative cutaneous reactions (5.5)
- Photosensitivity: Avoid sunlight due to risk of photosensitivity (5.6)
- Adrenal Dysfunction: Carefully monitor patients receiving voriconazole and corticosteroids (via all routes of administration) for adrenal dysfunction both during and after voriconazole treatment. Instruct patients to seek immediate medical care if they develop signs and symptoms of Cushing's syndrome or adrenal insufficiency (5.8)
- Embryo-Fetal Toxicity: Voriconazole can cause fetal harm when administered to a pregnant woman. Inform pregnant patients of the potential hazard to the fetus. Advise females of reproductive potential to use effective contraception during treatment with voriconazole (5.9, 8.1, 8.3)
- Skeletal Adverse Reactions: Fluorosis and periostitis with long-term voriconazole therapy. Discontinue if these adverse reactions occur (5.12)
- Clinically Significant Drug Interactions: Review patient's concomitant medications (5.13, 7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Dosage in Adults (2.3)
|
Infection |
Loading dose | Maintenance Dose | |
| Intravenous infusion | Intravenous infusion | Oral | |
| Invasive Aspergillosis | 6 mg/kg every 12 hours for the first 24 hours |
4 mg/kg every 12 hours | 200 mg every 12 hours |
|
Candidemia in
nonneutropenics and other deep tissue Candida infections |
3 to 4 mg/kg every 12 hours | 200 mg every 12 hours | |
| Scedosporiosis and Fusariosis | 4 mg/kg every 12 hours | 200 mg every 12 hours | |
| Esophageal Candidiasis | Not Evaluated | Not Evaluated | 200 mg every 12 hours |
- Adult patients weighing less than 40 kg: oral maintenance dose 100 mg or 150 mg every 12 hours
- Hepatic Impairment: Use half the maintenance dose in adult patients with mild to moderate hepatic impairment (Child-Pugh Class A and B) (2.5)
- Renal Impairment: Avoid intravenous administration in adult patients with moderate to severe renal impairment (creatinine clearance <50 mL/min) (2.6)
- Dosage in Pediatric Patients 2 years of age and older (2.4)
- For pediatric patients 2 to less than 12 years of age and 12 to 14 years of age weighing less than 50 kg see Table below.
|
Infection |
Loading Dose | Maintenance Dose | |
| Intravenous infusion | Intravenous infusion |
Oral
|
|
| Invasive Aspergillosis | 9 mg/kg every12 hours for the first 24 hours |
8 mg/kg every 12 hours after the first 24 hours |
9 mg/kg every 12 hours (maximum dose of 350 mg every 12 hours) |
| Candidemia in nonneutropenics and other deep tissue Candida infections | |||
| Scedosporiosis and Fusariosis | |||
| Esophageal Candidiasis | Not Evaluated | 4 mg/kg every 12 hours | 9 mg/kg every 12 hours (maximum dose of 350 mg every 12 hours) |
- For pediatric patients aged 12 to 14 years weighing greater than or equal to 50 kg and those aged 15 years and older regardless of body weight use adult dosage. (2.4)
- Dosage adjustment of voriconazole for injection in pediatric patients with renal or hepatic impairment has not been established (2.5, 2.6)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- For Injection: Lyophilized powder containing 200 mg of voriconazole and 3,200 mg of sulfobutyl ether beta-cyclodextrin sodium (SBECD); after reconstitution 10 mg/mL of voriconazole and 160 mg/mL of SBECD (3)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pediatrics: Safety and effectiveness in patients younger than 2 years has not been established (8.4)
5.3 Infusion Related Reactions
During infusion of the intravenous formulation of voriconazole in healthy subjects, anaphylactoid-type reactions, including flushing, fever, sweating, tachycardia, chest tightness, dyspnea, faintness, nausea, pruritus and rash, have occurred uncommonly. Symptoms appeared immediately upon initiating the infusion. Consideration should be given to stopping the infusion should these reactions occur.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.12 Skeletal Adverse Reactions
Fluorosis and periostitis have been reported during long-term voriconazole therapy. If a patient develops skeletal pain and radiologic findings compatible with fluorosis or periostitis, voriconazole should be discontinued [see Adverse Reactions (6.2)].
14.1 Invasive Aspergillosis (ia) (14.1 Invasive Aspergillosis (IA))
Voriconazole was studied in patients for primary therapy of IA (randomized, controlled study 307/602), for primary and salvage therapy of aspergillosis (non-comparative study 304) and for treatment of patients with IA who were refractory to, or intolerant of, other antifungal therapy (non-comparative study 309/604).
14.3 Esophageal Candidiasis (ec) (14.3 Esophageal Candidiasis (EC))
The efficacy of oral voriconazole 200 mg twice daily compared to oral fluconazole 200 mg once daily in the primary treatment of EC was demonstrated in Study 150 to 305, a double-blind, double-dummy study in immunocompromised patients with endoscopically-proven EC. Patients were treated for a median of 15 days (range 1 to 49 days). Outcome was assessed by repeat endoscopy at end of treatment (EOT). A successful response was defined as a normal endoscopy at EOT or at least a 1 grade improvement over baseline endoscopic score. For patients in the Intent-to-Treat (ITT) population with only a baseline endoscopy, a successful response was defined as symptomatic cure or improvement at EOT compared to baseline. Voriconazole and fluconazole (200 mg once daily) showed comparable efficacy rates against EC, as presented in Table 16.
|
a Confidence Interval for the difference (Voriconazole - Fluconazole) in success rates. |
|||
|
b PP (Per Protocol) patients had confirmation of Candida esophagitis by endoscopy, received at least 12 days of treatment, and had a repeat endoscopy at EOT (end of treatment). |
|||
|
c ITT (Intent to Treat) patients without endoscopy or clinical assessment at EOT were treated as failures. |
|||
| Population | Voriconazole | Fluconazole |
Difference %
(95% CI) a |
| PPb | 113/115 (98.2%) | 134/141 (95.0%) | 3.2 (-1.1, 7.5) |
| ITTc | 175/200 (87.5%) | 171/191 (89.5%) | -2.0 (-8.3, 4.3) |
Microbiologic success rates by Candida species are presented in Table 17.
|
a Some patients had more than one species isolated at baseline. |
||||
|
b Patients with endoscopic and/or mycological assessment at end of therapy. |
||||
| Pathogen a | Voriconazole | Fluconazole | ||
| Favorable endoscopic response b | Mycological eradication b | Favorable endoscopic response b | Mycological eradication b | |
| Success/Total (%) | Eradication/Total (%) | Success/Total (%) | Eradication/Total (%) | |
| C. albicans | 134/140 (96%) | 90/107 (84%) | 147/156 (94%) | 91/115 (79%) |
| C. glabrata | 8/8 (100%) | 4/7 (57%) | 4/4 (100%) | 1/4 (25%) |
| C. krusei | 1/1 | 1/1 | 2/2 (100%) | 0/0 |
1.4 Scedosporiosis and Fusariosis
Voriconazole for injection is indicated for the treatment of serious fungal infections caused by Scedosporium apiospermum (asexual form of Pseudallescheria boydii) and Fusarium spp. including Fusarium solani, in adults and pediatric patients (2 years of age and older) intolerant of, or refractory to, other therapy [see Clinical Studies (14.4) and Microbiology (12.4)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
14.4 Other Serious Fungal Pathogens
In pooled analyses of patients, voriconazole was shown to be effective against the following additional fungal pathogens:
Scedosporium apiospermum - Successful response to voriconazole therapy was seen in 15 of 24 patients (63%). Three of these patients relapsed within 4 weeks, including 1 patient with pulmonary, skin and eye infections, 1 patient with cerebral disease, and 1 patient with skin infection. Ten patients had evidence of cerebral disease and 6 of these had a successful outcome (1 relapse). In addition, a successful response was seen in 1 of 3 patients with mixed organism infections.
Fusarium spp. - Nine of 21 (43%) patients were successfully treated with voriconazole. Of these 9 patients, 3 had eye infections, 1 had an eye and blood infection, 1 had a skin infection, 1 had a blood infection alone, 2 had sinus infections, and 1 had disseminated infection (pulmonary, skin, hepatosplenic). Three of these patients (1 with disseminated disease, 1 with an eye infection and 1 with a blood infection) had Fusarium solani and were complete successes. Two of these patients relapsed, 1 with a sinus infection and profound neutropenia and 1 post surgical patient with blood and eye infections.
5.2 Arrhythmias and Qt Prolongation (5.2 Arrhythmias and QT Prolongation)
Some azoles, including voriconazole, have been associated with prolongation of the QT interval on the electrocardiogram. During clinical development and postmarketing surveillance, there have been rare cases of arrhythmias, (including ventricular arrhythmias such as torsade de pointes), cardiac arrests and sudden deaths in patients taking voriconazole. These cases usually involved seriously ill patients with multiple confounding risk factors, such as history of cardiotoxic chemotherapy, cardiomyopathy, hypokalemia and concomitant medications that may have been contributory.
Voriconazole should be administered with caution to patients with potentially proarrhythmic conditions, such as:
- Congenital or acquired QT prolongation
- Cardiomyopathy, in particular when heart failure is present
- Sinus bradycardia
- Existing symptomatic arrhythmias
- Concomitant medicinal product that is known to prolong QT interval [see Contraindications (4), Drug Interactions (7), and Clinical Pharmacology (12.3)]
Rigorous attempts to correct potassium, magnesium and calcium should be made before starting and during voriconazole therapy [see Clinical Pharmacology (12.3)].
5.5 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCARs), such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS), which can be life-threatening or fatal, have been reported during treatment with voriconazole. If a patient develops a severe cutaneous adverse reaction, voriconazole should be discontinued [see Adverse Reactions (6.1, 6.2)].
5.13 Clinically Significant Drug Interactions
See Table 10 for a listing of drugs that may significantly alter voriconazole concentrations. Also, see Table 11 for a listing of drugs that may interact with voriconazole resulting in altered pharmacokinetics or pharmacodynamics of the other drug [see Contraindications (4) and Drug Interactions (7)].
2.4 Recommended Dosing Regimen in Pediatric Patients
The recommended dosing regimen for pediatric patients 2 to less than 12 years of age and 12 to 14 years of age with body weight less than 50 kg is shown in Table 2. For pediatric patients 12 to 14 years of age with a body weight greater than or equal to 50 kg and those 15 years of age and above regardless of body weight, administer the adult dosing regimen of voriconazole [see Dosage and Administration (2.3)].
|
^ Based on a population pharmacokinetic analysis in 112 immunocompromised pediatric patients aged 2 to less than 12 years of age and 26 immunocompromised pediatric patients aged 12 to less than 17 years of age. |
|||
|
* In the Phase 3 clinical trials, patients with IA received intravenous (IV) treatment for at least 6 weeks and up to a maximum of 12 weeks. Patients received IV treatment for at least the first 7 days of therapy and then could be switched to oral voriconazole therapy. |
|||
|
† Study treatment for primary or salvage invasive candidiasis and candidemia (ICC) or EC consisted of intravenous voriconazole, with an option to switch to oral therapy after at least 5 days of IV therapy, based on subjects meeting switch criteria. For subjects with primary or salvage ICC, voriconazole was administered for at least 14 days after the last positive culture. A maximum of 42 days of treatment was permitted. Patients with primary or salvage EC were treated for at least 7 days after the resolution of clinical signs and symptoms. A maximum of 42 days of treatment was permitted. |
|||
| Loading Dose | Maintenance Dose | ||
| Intravenous infusion | Intravenous infusion | Oral | |
| Invasive Aspergillosis* | 9 mg/kg every 12 hours for the first 24 hours | 8 mg/kg every 12 hours after the first 24 hours | 9 mg/kg every 12 hours (maximum dose of 350 mg every 12 hours) |
| Candidemia in nonneutropenics and other deep tissue Candida infections † | |||
| Scedosporiosis and Fusariosis | |||
| Esophageal Candidiasis † | Not Evaluated | 4 mg/kg every 12 hours | 9 mg/kg every 12 hours (maximum dose of 350 mg every 12 hours) |
Initiate therapy with an intravenous infusion regimen. Consider an oral regimen only after there is a significant clinical improvement. Note that an 8 mg/kg intravenous dose will provide voriconazole exposure approximately 2-fold higher than a 9 mg/kg oral dose.
The oral dose recommendation for children is based on studies in which voriconazole was administered as the powder for oral suspension formulation. Bioequivalence between the voriconazole powder for oral suspension and voriconazole tablets has not been investigated in a pediatric population.
Oral bioavailability may be limited in pediatric patients 2 to 12 years with malabsorption and very low body weight for age. In that case, intravenous voriconazole administration is recommended.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year carcinogenicity studies were conducted in rats and mice. Rats were given oral doses of 6, 18 or 50 mg/kg voriconazole, or 0.2, 0.6, or 1.6 times the RMD on a body surface area basis. Hepatocellular adenomas were detected in females at 50 mg/kg and hepatocellular carcinomas were found in males at 6 and 50 mg/kg. Mice were given oral doses of 10, 30 or 100 mg/kg voriconazole, or 0.1, 0.4, or 1.4 times the RMD on a body surface area basis. In mice, hepatocellular adenomas were detected in males and females and hepatocellular carcinomas were detected in males at 1.4 times the RMD of voriconazole.
Voriconazole demonstrated clastogenic activity (mostly chromosome breaks) in human lymphocyte cultures in vitro. Voriconazole was not genotoxic in the Ames assay, CHO HGPRT assay, the mouse micronucleus assay or the in vivo DNA repair test (Unscheduled DNA Synthesis assay).
Voriconazole administration induced no impairment of male or female fertility in rats dosed at 50 mg/kg, or 1.6 times the RMD.
6.2 Postmarketing Experience in Adult and Pediatric Patients
The following adverse reactions have been identified during post-approval use of voriconazole. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
2.1 Important Administration Instructions for Use in All Patients
Voriconazole for injection requires reconstitution to 10 mg/mL and subsequent dilution to 5 mg/mL or less prior to administration as an infusion, at a maximum rate of 3 mg/kg per hour over 1 to 3 hours.
Administer diluted voriconazole for injection by intravenous infusion over 1 to 3 hours only. Do not administer as an IV bolus injection.
2.7 Dosage Adjustment When Co Administered With Phenytoin Or Efavirenz (2.7 Dosage Adjustment When Co-Administered With Phenytoin or Efavirenz)
1.2 Candidemia in Non Neutropenic Patients and Other Deep Tissue Candida (1.2 Candidemia in Non-neutropenic Patients and Other Deep Tissue Candida)
Voriconazole for injection is indicated in adults and pediatric patients (2 years of age and older) for the treatment of candidemia in non-neutropenic patients and the following Candida infections: disseminated infections in skin and infections in abdomen, kidney, bladder wall, and wounds [see Clinical Studies (14.2, 14.5) and Microbiology (12.4)].
14.2 Candidemia in Non Neutropenic Patients and Other Deep Tissue Candida (14.2 Candidemia in Non-neutropenic Patients and Other Deep Tissue Candida)
Voriconazole was compared to the regimen of amphotericin B followed by fluconazole in Study 608, an open-label, comparative study in nonneutropenic patients with candidemia associated with clinical signs of infection. Patients were randomized in 2:1 ratio to receive either voriconazole (n=283) or the regimen of amphotericin B followed by fluconazole (n=139). Patients were treated with randomized study drug for a median of 15 days. Most of the candidemia in patients evaluated for efficacy was caused by C. albicans (46%), followed by C. tropicalis (19%), C. parapsilosis (17%), C. glabrata (15%), and C. krusei (1%).
An independent Data Review Committee (DRC), blinded to study treatment, reviewed the clinical and mycological data from this study, and generated one assessment of response for each patient. A successful response required all of the following: resolution or improvement in all clinical signs and symptoms of infection, blood cultures negative for Candida, infected deep tissue sites negative for Candida or resolution of all local signs of infection, and no systemic antifungal therapy other than study drug. The primary analysis, which counted DRC-assessed successes at the fixed time point (12 weeks after End of Therapy [EOT]), demonstrated that voriconazole was comparable to the regimen of amphotericin B followed by fluconazole (response rates of 41% and 41%, respectively) in the treatment of candidemia. Patients who did not have a 12-week assessment for any reason were considered a treatment failure.
The overall clinical and mycological success rates by Candida species in Study 150 to 608 are presented in Table 15.
|
a A few patients had more than one pathogen at baseline. |
||
|
b Patients who did not have a 12-week assessment for any reason were considered a treatment failure. |
||
| Baseline Pathogen | Clinical and Mycological Success (%) | |
| Voriconazole | Amphotericin B --> Fluconazole | |
| C. albicans | 46/107 (43%) | 30/63 (48%) |
| C. tropicalis | 17/53 (32%) | 1/16 (6%) |
| C. parapsilosis | 24/45 (53%) | 10/19 (53%) |
| C. glabrata | 12/36 (33%) | 7/21 (33%) |
| C. krusei | 1/4 | 0/1 |
In a secondary analysis, which counted DRC-assessed successes at any time point (EOT, or 2, 6, or 12 weeks after EOT), the response rates were 65% for voriconazole and 71% for the regimen of amphotericin B followed by fluconazole.
In Studies 608 and 309/604 (non-comparative study in patients with invasive fungal infections who were refractory to, or intolerant of, other antifungal agents), voriconazole was evaluated in 35 patients with deep tissue Candida infections. A favorable response was seen in 4 of 7 patients with intra-abdominal infections, 5 of 6 patients with kidney and bladder wall infections, 3 of 3 patients with deep tissue abscess or wound infection, 1 of 2 patients with pneumonia/pleural space infections, 2 of 4 patients with skin lesions, 1 of 1 patients with mixed intra-abdominal and pulmonary infection, 1 of 2 patients with suppurative phlebitis, 1 of 3 patients with hepatosplenic infection, 1 of 5 patients with osteomyelitis, 0 of 1 with liver infection, and 0 of 1 with cervical lymph node infection.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:40:17.478517 · Updated: 2026-03-14T22:01:30.956876