25d0bfab-3dd1-c34b-e063-6294a90ab2d1
34390-5
HUMAN OTC DRUG LABEL
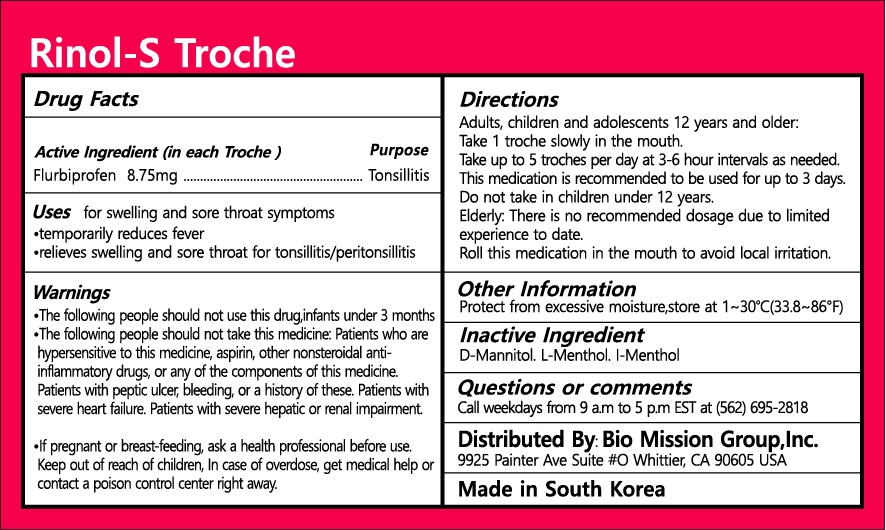
Drug Facts
Composition & Product
Identifiers & Packaging
Description
flurbiprofen
Medication Information
Warnings and Precautions
please see attached Drug Facts
Indications and Usage
please see attached Drug Facts
Dosage and Administration
oral use only
Description
flurbiprofen
Section 50565-1
Keep out of reach of children
Section 51727-6
d-mannitil, l-menthol, L-menthol
Section 51945-4
Section 55105-1
for swelling and sore throat symptoms, temporarily reduces feber, relieves swelling and sore throat for tonsillitis/peritonsillitis
Section 55106-9
flurbiprofen
Structured Label Content
Indications and Usage (34067-9)
please see attached Drug Facts
Dosage and Administration (34068-7)
oral use only
Warnings and Precautions (34071-1)
please see attached Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children
Section 51727-6 (51727-6)
d-mannitil, l-menthol, L-menthol
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
for swelling and sore throat symptoms, temporarily reduces feber, relieves swelling and sore throat for tonsillitis/peritonsillitis
Section 55106-9 (55106-9)
flurbiprofen
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:09.684349 · Updated: 2026-03-14T23:04:39.787858