Ivizia Dry Eye Drops 5ml Sample
25787325-c4e9-4cb1-bbc8-6cd3da18af0f
34390-5
HUMAN OTC DRUG LABEL
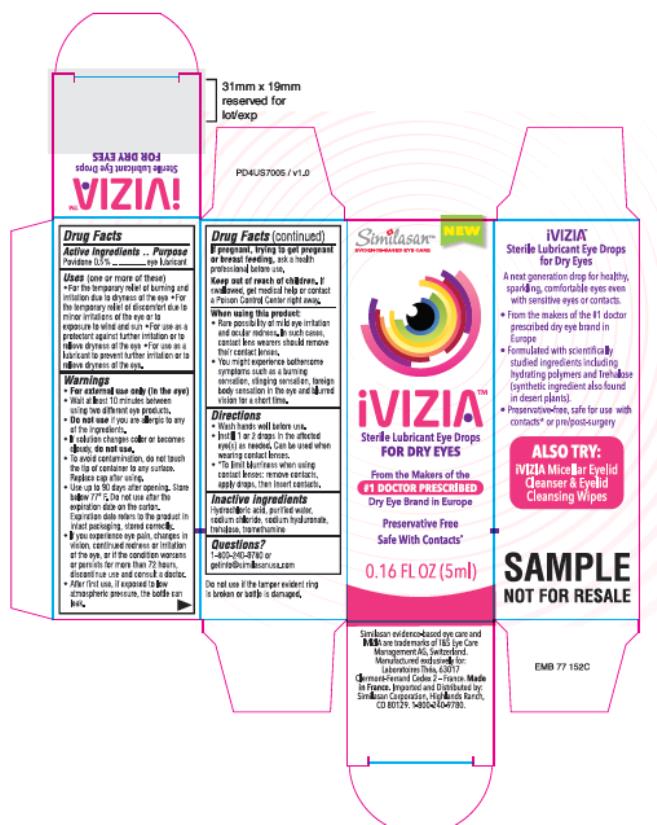
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Povidone 0.5%
Purpose
Eye lubricant
Medication Information
Purpose
Eye lubricant
Description
Povidone 0.5%
Uses
(one or more of these)
- For the temporary relief of burning and irritation due to dryness of the eye
- For the temporary relief of discomfort due to minor irritations of the eye or to exposure to wind and sun
- For use as a protectant against further irritation or to relieve dryness of the eye
- For use as a lubricant to prevent further irritation or to relieve dryness of the eye.
Warnings:
-
For external use only
(in the eye)
- Wait at least 10 minutes between using two different eye products
-
Do not use if you are allergic to any of the ingredients.
- If solution changes color or becomes cloudy, do not use.
- To avoid contamination, do not touch the tip of container to any surface. Replace cap after using.
- Use up to 90 days after opening. Store below 77°F. Do not use after the expiration date on the carton. Expiration date refers to the product in intact packaging, stored correctly.
- If you experience eye pain, changes in vision, continued redness or irritation of the eye, or if the condition worsens or persists for more than 72 hours, discontinue use and consult a doctor.
- After first use, if exposed to low atmospheric pressure, the bottle can leak.
Questions?
1-800-240-9780 or [email protected].
Directions:
- Wash hands well before use.
- Instill 1 or 2 drops in the affected eye(s) as needed. Can be used when wearing contact lenses.
- *To limit blurriness when using contact lenses: remove contacts, apply drops, then insert contacts.
If Pregnant
ask a health professional before use.
Active Ingredient
Povidone 0.5%
Inactive Ingredients
Hydrochloric acid, purified water, sodium chloride, sodium hyaluronate, trehalose, tromethamine
Principle Display Panel
iVizia
Sterile Lubricant Eye Drops
For Dry Eyes
0.16 FL OZ (5ml)
|
|
When Using This Product
- Rare possibility of mild eye irritation and ocular redness. In such cases, contact lens wearers should remove their contact lenses.
- You might experience bothersome symptoms such as a burning sensation, stinging sensation, foreign body sensation in the eye and blurred vision for a short time.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
(one or more of these)
- For the temporary relief of burning and irritation due to dryness of the eye
- For the temporary relief of discomfort due to minor irritations of the eye or to exposure to wind and sun
- For use as a protectant against further irritation or to relieve dryness of the eye
- For use as a lubricant to prevent further irritation or to relieve dryness of the eye.
Purpose
Eye lubricant
Warnings:
-
For external use only
(in the eye)
- Wait at least 10 minutes between using two different eye products
-
Do not use if you are allergic to any of the ingredients.
- If solution changes color or becomes cloudy, do not use.
- To avoid contamination, do not touch the tip of container to any surface. Replace cap after using.
- Use up to 90 days after opening. Store below 77°F. Do not use after the expiration date on the carton. Expiration date refers to the product in intact packaging, stored correctly.
- If you experience eye pain, changes in vision, continued redness or irritation of the eye, or if the condition worsens or persists for more than 72 hours, discontinue use and consult a doctor.
- After first use, if exposed to low atmospheric pressure, the bottle can leak.
Questions?
1-800-240-9780 or [email protected].
Directions:
- Wash hands well before use.
- Instill 1 or 2 drops in the affected eye(s) as needed. Can be used when wearing contact lenses.
- *To limit blurriness when using contact lenses: remove contacts, apply drops, then insert contacts.
If Pregnant (If pregnant)
ask a health professional before use.
Active Ingredient
Povidone 0.5%
Inactive Ingredients
Hydrochloric acid, purified water, sodium chloride, sodium hyaluronate, trehalose, tromethamine
Principle Display Panel
iVizia
Sterile Lubricant Eye Drops
For Dry Eyes
0.16 FL OZ (5ml)
|
|
When Using This Product (When using this product)
- Rare possibility of mild eye irritation and ocular redness. In such cases, contact lens wearers should remove their contact lenses.
- You might experience bothersome symptoms such as a burning sensation, stinging sensation, foreign body sensation in the eye and blurred vision for a short time.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:59:45.030286 · Updated: 2026-03-14T22:59:51.166569