These Highlights Do Not Include All The Information Needed To Use Methylene Blue Injection Safely And Effectively. See Full Prescribing Information For Methylene Blue Injection.
2566ce72-9d19-42e5-80f9-8fc4612b1eff
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Methylene blue injection may cause serious or fatal serotonergic syndrome when used in combination with serotonergic drugs and opioids. Avoid concomitant use of methylene blue injection with selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors (MAOIs) and opioids [see Warnings and Precautions (5.1) and Drug Interactions (7.1) ] .
Indications and Usage
Methylene Blue Injection is indicated for the treatment of pediatric and adult patients with acquired methemoglobinemia.
Dosage and Administration
Administer 1 mg/kg intravenously over 5-30 minutes. ( 2.1 ) If methemoglobin level remains above 30% or if clinical symptoms persist, give a repeat dose of up to 1 mg/kg one hour after the first dose. ( 2.1 ) Administer a single dose of 1 mg/kg in patients with moderate or severe renal impairment. ( 2.2 )
Warnings and Precautions
Hypersensitivity: If severe or life threatening allergic reaction occurs, discontinue methylene blue injection, treat the allergic reaction, and monitor until signs and symptoms resolve. ( 5.2 ) Lack of Effectiveness: Consider alternative treatments if there is no resolution of methemoglobinemia after 2 doses. ( 2.1 , 5.3 ) Hemolytic Anemia: Discontinue methylene blue injection and transfuse. ( 5.4 ) Interference with In Vivo Monitoring Devices: Use methods other than pulse oximetry to assess oxygen saturation. ( 5.5 ) Effects on Ability to Drive and Operate Machinery: Advise patients to refrain from these activities until neurologic and visual symptoms have resolved. ( 5.6 )
Contraindications
Methylene blue injection is contraindicated in the following conditions: Severe hypersensitivity reactions to methylene blue or any other thiazine dye [see Warnings and Precautions (5.2) ] . Patients with glucose-6-phosphate dehydrogenase deficiency (G6PD) due to the risk of hemolytic anemia [see Warnings and Precautions (5.3 , 5.4 )] .
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: Serotonin Syndrome with Concomitant Use of Serotonergic Drugs [see Warnings and Precautions (5.1) ] Anaphylaxis [see Warnings and Precautions (5.2) ] Lack of Effectiveness [see Warnings and Precautions (5.3) ] Hemolytic Anemia [see Warnings and Precautions (5.4) ] Interference with In Vivo Monitoring Devices [see Warnings and Precautions (5.5) ] Effects on Ability to Drive and Operate Machinery [see Warnings and Precautions (5.6) ] Interference with Laboratory Tests [see Warnings and Precautions (5.7) ]
Storage and Handling
Methylene Blue Injection, USP is supplied in 10 mL single-dose vials. Each 10 mL vial contains 50 mg of methylene blue as a clear dark blue solution. It is supplied as follows: NDC Strength and Package Type Package Size 71288- 811 -11 50 mg per 10 mL (5 mg per mL) (0.5%) Single-Dose Vial 5 vials per carton
How Supplied
Methylene Blue Injection, USP is supplied in 10 mL single-dose vials. Each 10 mL vial contains 50 mg of methylene blue as a clear dark blue solution. It is supplied as follows: NDC Strength and Package Type Package Size 71288- 811 -11 50 mg per 10 mL (5 mg per mL) (0.5%) Single-Dose Vial 5 vials per carton
Medication Information
Warnings and Precautions
Hypersensitivity: If severe or life threatening allergic reaction occurs, discontinue methylene blue injection, treat the allergic reaction, and monitor until signs and symptoms resolve. ( 5.2 ) Lack of Effectiveness: Consider alternative treatments if there is no resolution of methemoglobinemia after 2 doses. ( 2.1 , 5.3 ) Hemolytic Anemia: Discontinue methylene blue injection and transfuse. ( 5.4 ) Interference with In Vivo Monitoring Devices: Use methods other than pulse oximetry to assess oxygen saturation. ( 5.5 ) Effects on Ability to Drive and Operate Machinery: Advise patients to refrain from these activities until neurologic and visual symptoms have resolved. ( 5.6 )
Indications and Usage
Methylene Blue Injection is indicated for the treatment of pediatric and adult patients with acquired methemoglobinemia.
Dosage and Administration
Administer 1 mg/kg intravenously over 5-30 minutes. ( 2.1 ) If methemoglobin level remains above 30% or if clinical symptoms persist, give a repeat dose of up to 1 mg/kg one hour after the first dose. ( 2.1 ) Administer a single dose of 1 mg/kg in patients with moderate or severe renal impairment. ( 2.2 )
Contraindications
Methylene blue injection is contraindicated in the following conditions: Severe hypersensitivity reactions to methylene blue or any other thiazine dye [see Warnings and Precautions (5.2) ] . Patients with glucose-6-phosphate dehydrogenase deficiency (G6PD) due to the risk of hemolytic anemia [see Warnings and Precautions (5.3 , 5.4 )] .
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: Serotonin Syndrome with Concomitant Use of Serotonergic Drugs [see Warnings and Precautions (5.1) ] Anaphylaxis [see Warnings and Precautions (5.2) ] Lack of Effectiveness [see Warnings and Precautions (5.3) ] Hemolytic Anemia [see Warnings and Precautions (5.4) ] Interference with In Vivo Monitoring Devices [see Warnings and Precautions (5.5) ] Effects on Ability to Drive and Operate Machinery [see Warnings and Precautions (5.6) ] Interference with Laboratory Tests [see Warnings and Precautions (5.7) ]
Storage and Handling
Methylene Blue Injection, USP is supplied in 10 mL single-dose vials. Each 10 mL vial contains 50 mg of methylene blue as a clear dark blue solution. It is supplied as follows: NDC Strength and Package Type Package Size 71288- 811 -11 50 mg per 10 mL (5 mg per mL) (0.5%) Single-Dose Vial 5 vials per carton
How Supplied
Methylene Blue Injection, USP is supplied in 10 mL single-dose vials. Each 10 mL vial contains 50 mg of methylene blue as a clear dark blue solution. It is supplied as follows: NDC Strength and Package Type Package Size 71288- 811 -11 50 mg per 10 mL (5 mg per mL) (0.5%) Single-Dose Vial 5 vials per carton
Description
Methylene blue injection may cause serious or fatal serotonergic syndrome when used in combination with serotonergic drugs and opioids. Avoid concomitant use of methylene blue injection with selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors (MAOIs) and opioids [see Warnings and Precautions (5.1) and Drug Interactions (7.1) ] .
Section 42229-5
Other adverse reactions reported to occur following the administration of methylene blue class products include the following:
Blood and lymphatic system disorders: hemolytic anemia, hemolysis, hyperbilirubinemia
Cardiac disorders: palpitations, tachycardia
Eye disorders: eye pruritus, ocular hyperemia, vision blurred
Gastrointestinal disorders: abdominal pain lower, dry mouth, flatulence, glossodynia, tongue eruption
General disorders and administration site conditions: death, infusion site extravasation, infusion site induration, infusion site pruritus, infusion site swelling, infusion site urticaria, peripheral swelling, thirst
Investigations: elevated liver enzymes
Musculoskeletal and connective tissue disorders: myalgia
Renal and urinary disorders: dysuria
Respiratory, thoracic and mediastinal disorders: nasal congestion, oropharyngeal pain, rhinorrhea, sneezing
Skin and subcutaneous tissue disorders: necrotic ulcer, papule, phototoxicity
Vascular disorders: hypertension
Section 43683-2
Section 44425-7
Storage
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.]
Any unused product or waste material should be disposed of in accordance with local practice.
Do not refrigerate or freeze.
Keep the vial in the original package to protect from light.
The container closure is not made with natural rubber latex.
10 Overdosage
Hypotension, wheezing and reduced oxygenation have been reported in patients who received methylene blue class products in single doses of 3 mg/kg or more.
Administration of large intravenous doses (cumulative dose ≥ 7 mg/kg) of a methylene blue class product caused nausea, vomiting, precordial pain, dyspnea, tachypnea, chest tightness, tachycardia, apprehension, tremor, mydriasis, blue staining of the urine, the skin and mucous membranes, abdominal pain, dizziness, paresthesia, headache, confusion, mild methemoglobinemia (up to 7%) and electrocardiogram changes (T-wave flattening or inversion). These effects lasted 2-12 hours following administration.
A severe overdosage (single dose of 20 mg/kg or more) of a methylene blue class product caused severe intravascular hemolysis, hyperbilirubinemia and death.
In case of overdose of methylene blue injection, maintain the patient under observation until signs and symptoms have resolved, monitor for cardiopulmonary, hematologic and neurologic toxicities, and institute supportive measures as necessary.
11 Description
Methylene blue is an oxidation-reduction agent.
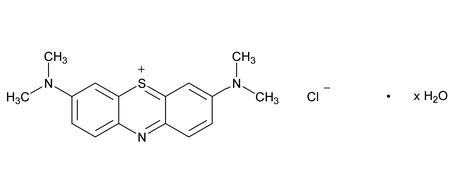
Its chemical name is 3,7-bis(dimethylamino)phenothiazin-5-ium, chloride hydrate. The molecular formula of methylene blue is C16H18ClN3S•xH2O and its molecular weight of 319.85 g/mol for the anhydrous form. The structural formula of methylene blue is:
Methylene Blue Injection, USP is a sterile solution intended for intravenous administration. Each mL of solution contains 5 mg methylene blue, USP and water for injection, USP. Methylene Blue Injection, USP is a clear dark blue solution with a pH value between 3.0 and 4.5. The osmolality is between 10 and 15 mOsm/kg. Methylene Blue Injection, USP strength is expressed in terms of trihydrate.
2.3 preparation
Methylene blue injection is hypotonic and may be diluted before use in a solution of 50 mL 5% Dextrose Injection in order to avoid local pain, particularly in the pediatric population. Use the diluted solution immediately after preparation.
Avoid diluting with sodium chloride solutions, because it has been demonstrated that chloride reduces the solubility of methylene blue.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Discard unused portion.
8.4 Pediatric Use
The safety and effectiveness of methylene blue injection for the treatment of acquired methemoglobinemia have been established in pediatric patients. Use of methylene blue injection is supported by two retrospective case series that included 2 pediatric patients treated with methylene blue injection and 12 treated with another methylene blue class product. The case series included pediatric patients in the following age groups: 3 neonates (less than 1 month), 4 infants (1 month up to less than 2 years), 4 children (2 years up to less than 12 years), and 3 adolescents (12 years to less than 17 years). The efficacy outcomes were consistent across pediatric and adult patients in both case series.
8.5 Geriatric Use
Clinical studies of methylene blue injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. Methylene blue injection is known to be substantially excreted by the kidney, so the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, treatment of methemoglobinemia in these patients should use the lowest number of doses needed to achieve a response [see Dosage and Administration (2)].
14 Clinical Studies
4 Contraindications
Methylene blue injection is contraindicated in the following conditions:
- Severe hypersensitivity reactions to methylene blue or any other thiazine dye [see Warnings and Precautions (5.2)].
- Patients with glucose-6-phosphate dehydrogenase deficiency (G6PD) due to the risk of hemolytic anemia [see Warnings and Precautions (5.3, 5.4)].
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Serotonin Syndrome with Concomitant Use of Serotonergic Drugs [see Warnings and Precautions (5.1)]
- Anaphylaxis [see Warnings and Precautions (5.2)]
- Lack of Effectiveness [see Warnings and Precautions (5.3)]
- Hemolytic Anemia [see Warnings and Precautions (5.4)]
- Interference with In Vivo Monitoring Devices [see Warnings and Precautions (5.5)]
- Effects on Ability to Drive and Operate Machinery [see Warnings and Precautions (5.6)]
- Interference with Laboratory Tests [see Warnings and Precautions (5.7)]
5.2 Hypersensitivity
Anaphylactic reactions to methylene blue class products have been reported. Patients treated with methylene blue injection should be monitored for anaphylaxis. If anaphylaxis or other severe hypersensitivity reactions (e.g., angioedema, urticaria, bronchospasm) should occur, discontinue use of methylene blue injection and initiate supportive treatment. Methylene blue injection is contraindicated in patients who have experienced anaphylaxis or other severe hypersensitivity reactions to a methylene blue class product in the past.
8.6 Renal Impairment
Methylene blue concentrations increased in subjects with renal impairment (eGFR 15 to 89 mL/min/1.73 m2) significantly [see Clinical Pharmacology (12.3)]. Adjust methylene blue injection dosage in patients with moderate or severe renal impairment (eGFR 15 to 59 mL/min/1.73 m2) [see Dosage and Administration (2.2)]. No dose adjustment is recommended in patients with mild renal impairment (eGFR 60 – 89 mL/min/1.73 m2).
12.2 Pharmacodynamics
Low concentrations of methylene blue speeds up the in vivo conversion of methemoglobin to hemoglobin. Methylene blue has been observed to stain tissues selectively. The exposure-response or –safety relationship for methylene is unknown.
Cardiac Electrophysiology
The results of a thorough QT study demonstrated methylene blue injection at an intravenous dose of 2 mg/kg as a 5-minute intravenous infusion had no effect on the QT, PR or QRS intervals.
5.4 hemolytic Anemia
Hemolysis can occur during treatment of methemoglobinemia with methylene blue injection. Laboratory testing may show Heinz bodies, elevated indirect bilirubin and low haptoglobin, but the Coombs test is negative. The onset of anemia may be delayed 1 or more days after treatment with methylene blue injection. The anemia may require red blood cell transfusions [see Adverse Reactions (6.1)]. Use the lowest effective number of doses of methylene blue injection to treat methemoglobinemia. Discontinue methylene blue injection and consider alternative treatments of methemoglobinemia if severe hemolysis occurs.
Treatment of patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency with methylene blue injection may result in severe hemolysis and severe anemia. Methylene blue injection is contraindicated for use in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency [see Contraindications (4)].
8.7 Hepatic Impairment
Methylene blue is extensively metabolized in the liver. Monitor patients with any hepatic impairment for toxicities and potential drug interactions for an extended period of time following treatment with methylene blue injection.
1 Indications and Usage
Methylene Blue Injection is indicated for the treatment of pediatric and adult patients with acquired methemoglobinemia.
12.1 Mechanism of Action
Methylene blue is a water soluble thiazine dye that promotes a non-enzymatic redox conversion of metHb to hemoglobin. In situ, methylene blue is first converted to leucomethylene blue (LMB) via NADPH reductase. It is the LMB molecule which then reduces the ferric iron of metHb to the ferrous state of normal hemoglobin.
5 Warnings and Precautions
- Hypersensitivity: If severe or life threatening allergic reaction occurs, discontinue methylene blue injection, treat the allergic reaction, and monitor until signs and symptoms resolve. (5.2)
- Lack of Effectiveness: Consider alternative treatments if there is no resolution of methemoglobinemia after 2 doses. (2.1, 5.3)
- Hemolytic Anemia: Discontinue methylene blue injection and transfuse. (5.4)
- Interference with In Vivo Monitoring Devices: Use methods other than pulse oximetry to assess oxygen saturation. (5.5)
- Effects on Ability to Drive and Operate Machinery: Advise patients to refrain from these activities until neurologic and visual symptoms have resolved. (5.6)
5.3 lack of Effectiveness
Methemoglobinemia may not resolve or may rebound after response to treatment with methylene blue injection in patients with methemoglobinemia due to aryl amines such as aniline or sulfa drugs such as dapsone. Monitor response to therapy with methylene blue injection through resolution of methemoglobinemia. If methemoglobinemia does not respond to 2 doses of methylene blue injection or if methemoglobinemia rebounds after a response, consider additional treatment options [see Dosage and Administration (2.2)].
Patients with glucose-6-phosphate dehydrogenase deficiency may not reduce methylene blue injection to its active form in vivo. Methylene blue injection may not be effective in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency.
2 Dosage and Administration
- Administer 1 mg/kg intravenously over 5-30 minutes. (2.1)
- If methemoglobin level remains above 30% or if clinical symptoms persist, give a repeat dose of up to 1 mg/kg one hour after the first dose. (2.1)
- Administer a single dose of 1 mg/kg in patients with moderate or severe renal impairment. (2.2)
3 Dosage Forms and Strengths
Methylene Blue Injection, USP: 50 mg per 10 mL (5 mg per mL) (0.5%) clear dark blue solution in single-dose vials.
5.5 Interference With in Vivo
- Inaccurate Pulse Oximeter Readings
The presence of methylene blue in the blood may result in an underestimation of the oxygen saturation reading by pulse oximetry. If a measure of oxygen saturation is required during or shortly after infusion of methylene blue injection, it is advisable to obtain an arterial blood sample for testing by an alternative method.
- Bispectral index monitor
A fall in the Bispectral Index (BIS) has been reported following administration of methylene blue class products. If methylene blue injection is administered during surgery, alternative methods for assessing the depth of anesthesia should be employed.
8 Use in Specific Populations
2.1 dosage and Administration
- Ensure patent venous access prior to administration of methylene blue injection. Do not administer methylene blue injection subcutaneously.
- Administer methylene blue injection 1 mg/kg intravenously over 5-30 minutes.
- If the methemoglobin level remains greater than 30% or if clinical signs and symptoms persist, a repeat dose of methylene blue injection 1 mg/kg may be given one hour after the first dose.
- If methemoglobinemia does not resolve after 2 doses of methylene blue injection, consider initiating alternative interventions for treatment of methemoglobinemia.
6.1 clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of methylene blue injection in adults with acquired methemoglobinemia was assessed in 24 patients who received at least 1 dose of methylene blue injection [see Clinical Studies (14)]. Most doses administered were 1 mg/kg (88.5%), but doses from 1 mg/kg to 2 mg/kg were administered. All patients received at least one dose of methylene blue injection; two received two doses. Serious adverse reactions occurred in 4.2% of patients who received methylene blue injection. A serious adverse reaction of seizure-like phenomenon was reported in one patient. Adverse reactions (≥2%) included headache, hypokalemia, diarrhea, hypomagnesemia, myoclonus, nausea, and seizure-like phenomena.
The safety of methylene blue injection in pediatric patients with acquired methemoglobinemia was assessed in two retrospective case series that included two pediatric patients treated with methylene blue injection and 12 treated with another methylene blue product. The case series included patients in the following age groups: 3 neonates (<1 month), 4 infants (1 month to <2 years), 4 children (2 years to <12 years), and 3 adolescents (12 years to <17 years). The safety profile in pediatric patients was similar to that in adult patients.
16 How Supplied/storage and Handling
Methylene Blue Injection, USP is supplied in 10 mL single-dose vials. Each 10 mL vial contains 50 mg of methylene blue as a clear dark blue solution. It is supplied as follows:
| NDC | Strength and Package Type | Package Size |
| 71288-811-11 | 50 mg per 10 mL (5 mg per mL) (0.5%) Single-Dose Vial | 5 vials per carton |
5.7 interference With Laboratory Tests
Methylene blue injection is a blue dye which passes freely into the urine and may interfere with the interpretation of any urine test which relies on a blue indicator, such as the dipstick test for leucocyte esterase.
14.1 Treatment of Acquired Methemoglobinemia
The efficacy of methylene blue injection in the treatment of patients with methemoglobinemia was evaluated in 24 adult patients with acquired methemoglobinemia: in study NCT03542760, a prospective, multicenter, observational registry. Of the 24 subjects enrolled 92% were white, 8% were black, 67% were female, and 33% were male. Hispanic or Latino was 12.5%; non-Hispanic or Latino was 87.5%, and ethnicity data were missing for 0%. The mean age was 46.0 years, and the ages ranged from 19 to 72 years. Each individual received at least 1 intravenous dose of methylene blue injection; two received 2 doses. Most doses administered were 1 mg/kg (88.5%), but doses from 1 mg/kg to 2 mg/kg were administered. The recommended methylene blue injection dose is 1 mg/kg; lower or greater doses are not recommended. The maximum recommended number of doses is two [see Dosage and Administration (2.1)].
In total, 22 of the 24 (91.7%) subjects had post treatment methemoglobin (metHb) assessment; 22 of the 22 subjects had baseline metHb with a mean concentration of 12.3% and a range of 4.1% to 30.0%. 21 of 22 (95.5%) subjects who had baseline metHb had at least a 50% reduction in metHB from baseline in their first assessment post baseline. This first post dosing assessment occurred from 0.7 to 27.3 hours from the end of first methylene blue injection infusion with a median time of 2.9 hours. There were 9 subjects that had baseline metHb and had metHb assessed within 2 hours of the end of the first methylene blue injection treatment; 6 of the 9 (67%; 95% CI (30.9%, 91.0%)) had at least a 50% reduction in metHb at 1 hour postdosing.
Available vital sign data including blood pressure, heart rate and respiratory rate were reviewed at baseline and compared to data collected within 2 hours post methylene blue injection infusion. Prior to treatment with methylene blue injection, 12 of the 18 (67%) of patients had a respiratory rate exceeding the upper limit of normal (≥ 20 bpm). Of these, 8 of the 12 (67%) experienced a normalization of respiratory rate within 2 hours post methylene blue injection infusion. There was minimal impact on other vital signs.
At baseline, the most common prespecified signs and symptoms of methemoglobinemia (reported by ≥2 subjects [8.3%] overall) were fatigue (33.3%), dyspnea (29.2%), cyanosis (12.5%), depressed CNS (12.5%), dizziness (8.3%), headache (8.3%), and weakness (8.3%). Following treatment with methylene blue injection, signs and symptoms of methemoglobinemia improved.
The efficacy of methylene blue injection in the treatment of methemoglobinemia in pediatric patients was assessed in 14 patients in two retrospective case series (2 patients received methylene blue injection and 12 who received another methylene blue product). The ages ranged from 6 days to 16 years. The efficacy outcomes were consistent across the pediatric and adult populations.
2.2 recommended Dosage for Renal Impairment
- The recommended dosage of methylene blue injection in patients with moderate or severe renal impairment (eGFR 15-59 mL/min/1.73 m2) is a single dose of 1 mg/kg.
- If the methemoglobin level remains greater than 30% or if the clinical symptoms persist 1 hour after dosing, consider initiating alternative interventions for the treatment of methemoglobinemia.
5.6 effects On Ability to Drive and Operate Machinery
Treatment with methylene blue injection may cause confusion, dizziness and disturbances in vision [see Adverse Reactions (6)]. Advise patients to refrain from driving or engaging in hazardous occupations or activities such as operating heavy or potentially dangerous machinery until such adverse reactions to methylene blue injection have resolved.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a two-year carcinogenicity study, rats were administered oral doses of methylene blue at 5, 25, or 50 mg/kg. Methylene blue caused pancreatic islet adenomas or carcinomas (combined) in male rats. In a two-year carcinogenicity study, mice were administered oral doses of methylene blue at 2.5, 12.5, or 25 mg/kg. There were no drug-related neoplastic findings in mice.
Methylene blue was genotoxic in gene mutation assays in bacteria (Ames test), and in an in vitro sister chromatid exchange test and an in vitro chromosomal aberration test in Chinese hamster ovary (CHO) cells. Methylene blue was negative for micronucleus induction in bone marrow or peripheral blood collected from mice treated with methylene blue.
Fertility studies with methylene blue have not been conducted. In vitro, methylene blue reduced motility of human sperm in a concentration dependent manner.
Principal Display Panel – Methylene Blue Injection, Usp 10 Ml Carton
NDC 71288-811-11
Rx Only
Methylene Blue Injection, USP
50 mg per 10 mL
(5 mg per mL) (0.5%)
Intravenous Use Only
For Slow Intravenous Injection
Use 5% Dextrose Injection When Diluting
5 x 10 mL Single-Dose Vials
Discard unused portion
Principal Display Panel – Methylene Blue Injection, Usp 10 Ml Vial Label
NDC 71288-811-10
Rx Only
Methylene Blue Injection, USP
50 mg per 10 mL
(5 mg per mL) (0.5%)
Intravenous Use Only
Use 5% Dextrose Injection When Diluting
10 mL Single-Dose Vial
5.1 serotonin Syndrome With Concomitant Use of Serotonergic Drugs and Opioids
The development of serotonin syndrome has been reported with the use of methylene blue class products. Most reports have been associated with concomitant use of serotonergic drugs (e.g., selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors (MAOIs)). Opioids and dextromethorphan may increase the risk of developing serotonin syndrome. Some of the reported cases were fatal. Symptoms associated with serotonin syndrome may include the following combination of signs and symptoms: mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, and hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, and incoordination), seizures, and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Avoid concomitant use of methylene blue injection with serotonergic drugs and opioids.
Patients treated with methylene blue injection should be monitored for the emergence of serotonin syndrome. If symptoms of serotonin syndrome occur, discontinue use of methylene blue injection, and initiate supportive treatment. Inform patients of the increased risk of serotonin syndrome and advise them to not to take serotonergic drugs within 72 hours after the last dose of methylene blue injection [see Drug Interactions (7), Patient Counseling Information (17)].
Warning: Serotonin Syndrome With Concomitant Use of Serotonergic Drugs and Opioids
Methylene blue injection may cause serious or fatal serotonergic syndrome when used in combination with serotonergic drugs and opioids. Avoid concomitant use of methylene blue injection with selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors (MAOIs) and opioids [see Warnings and Precautions (5.1) and Drug Interactions (7.1)].
Structured Label Content
Section 42229-5 (42229-5)
Other adverse reactions reported to occur following the administration of methylene blue class products include the following:
Blood and lymphatic system disorders: hemolytic anemia, hemolysis, hyperbilirubinemia
Cardiac disorders: palpitations, tachycardia
Eye disorders: eye pruritus, ocular hyperemia, vision blurred
Gastrointestinal disorders: abdominal pain lower, dry mouth, flatulence, glossodynia, tongue eruption
General disorders and administration site conditions: death, infusion site extravasation, infusion site induration, infusion site pruritus, infusion site swelling, infusion site urticaria, peripheral swelling, thirst
Investigations: elevated liver enzymes
Musculoskeletal and connective tissue disorders: myalgia
Renal and urinary disorders: dysuria
Respiratory, thoracic and mediastinal disorders: nasal congestion, oropharyngeal pain, rhinorrhea, sneezing
Skin and subcutaneous tissue disorders: necrotic ulcer, papule, phototoxicity
Vascular disorders: hypertension
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.]
Any unused product or waste material should be disposed of in accordance with local practice.
Do not refrigerate or freeze.
Keep the vial in the original package to protect from light.
The container closure is not made with natural rubber latex.
10 Overdosage (10 OVERDOSAGE)
Hypotension, wheezing and reduced oxygenation have been reported in patients who received methylene blue class products in single doses of 3 mg/kg or more.
Administration of large intravenous doses (cumulative dose ≥ 7 mg/kg) of a methylene blue class product caused nausea, vomiting, precordial pain, dyspnea, tachypnea, chest tightness, tachycardia, apprehension, tremor, mydriasis, blue staining of the urine, the skin and mucous membranes, abdominal pain, dizziness, paresthesia, headache, confusion, mild methemoglobinemia (up to 7%) and electrocardiogram changes (T-wave flattening or inversion). These effects lasted 2-12 hours following administration.
A severe overdosage (single dose of 20 mg/kg or more) of a methylene blue class product caused severe intravascular hemolysis, hyperbilirubinemia and death.
In case of overdose of methylene blue injection, maintain the patient under observation until signs and symptoms have resolved, monitor for cardiopulmonary, hematologic and neurologic toxicities, and institute supportive measures as necessary.
11 Description (11 DESCRIPTION)
Methylene blue is an oxidation-reduction agent.
Its chemical name is 3,7-bis(dimethylamino)phenothiazin-5-ium, chloride hydrate. The molecular formula of methylene blue is C16H18ClN3S•xH2O and its molecular weight of 319.85 g/mol for the anhydrous form. The structural formula of methylene blue is:
Methylene Blue Injection, USP is a sterile solution intended for intravenous administration. Each mL of solution contains 5 mg methylene blue, USP and water for injection, USP. Methylene Blue Injection, USP is a clear dark blue solution with a pH value between 3.0 and 4.5. The osmolality is between 10 and 15 mOsm/kg. Methylene Blue Injection, USP strength is expressed in terms of trihydrate.
2.3 preparation (2.3 Preparation)
Methylene blue injection is hypotonic and may be diluted before use in a solution of 50 mL 5% Dextrose Injection in order to avoid local pain, particularly in the pediatric population. Use the diluted solution immediately after preparation.
Avoid diluting with sodium chloride solutions, because it has been demonstrated that chloride reduces the solubility of methylene blue.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Discard unused portion.
8.4 Pediatric Use
The safety and effectiveness of methylene blue injection for the treatment of acquired methemoglobinemia have been established in pediatric patients. Use of methylene blue injection is supported by two retrospective case series that included 2 pediatric patients treated with methylene blue injection and 12 treated with another methylene blue class product. The case series included pediatric patients in the following age groups: 3 neonates (less than 1 month), 4 infants (1 month up to less than 2 years), 4 children (2 years up to less than 12 years), and 3 adolescents (12 years to less than 17 years). The efficacy outcomes were consistent across pediatric and adult patients in both case series.
8.5 Geriatric Use
Clinical studies of methylene blue injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. Methylene blue injection is known to be substantially excreted by the kidney, so the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, treatment of methemoglobinemia in these patients should use the lowest number of doses needed to achieve a response [see Dosage and Administration (2)].
14 Clinical Studies (14 CLINICAL STUDIES)
4 Contraindications (4 CONTRAINDICATIONS)
Methylene blue injection is contraindicated in the following conditions:
- Severe hypersensitivity reactions to methylene blue or any other thiazine dye [see Warnings and Precautions (5.2)].
- Patients with glucose-6-phosphate dehydrogenase deficiency (G6PD) due to the risk of hemolytic anemia [see Warnings and Precautions (5.3, 5.4)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Serotonin Syndrome with Concomitant Use of Serotonergic Drugs [see Warnings and Precautions (5.1)]
- Anaphylaxis [see Warnings and Precautions (5.2)]
- Lack of Effectiveness [see Warnings and Precautions (5.3)]
- Hemolytic Anemia [see Warnings and Precautions (5.4)]
- Interference with In Vivo Monitoring Devices [see Warnings and Precautions (5.5)]
- Effects on Ability to Drive and Operate Machinery [see Warnings and Precautions (5.6)]
- Interference with Laboratory Tests [see Warnings and Precautions (5.7)]
5.2 Hypersensitivity
Anaphylactic reactions to methylene blue class products have been reported. Patients treated with methylene blue injection should be monitored for anaphylaxis. If anaphylaxis or other severe hypersensitivity reactions (e.g., angioedema, urticaria, bronchospasm) should occur, discontinue use of methylene blue injection and initiate supportive treatment. Methylene blue injection is contraindicated in patients who have experienced anaphylaxis or other severe hypersensitivity reactions to a methylene blue class product in the past.
8.6 Renal Impairment
Methylene blue concentrations increased in subjects with renal impairment (eGFR 15 to 89 mL/min/1.73 m2) significantly [see Clinical Pharmacology (12.3)]. Adjust methylene blue injection dosage in patients with moderate or severe renal impairment (eGFR 15 to 59 mL/min/1.73 m2) [see Dosage and Administration (2.2)]. No dose adjustment is recommended in patients with mild renal impairment (eGFR 60 – 89 mL/min/1.73 m2).
12.2 Pharmacodynamics
Low concentrations of methylene blue speeds up the in vivo conversion of methemoglobin to hemoglobin. Methylene blue has been observed to stain tissues selectively. The exposure-response or –safety relationship for methylene is unknown.
Cardiac Electrophysiology
The results of a thorough QT study demonstrated methylene blue injection at an intravenous dose of 2 mg/kg as a 5-minute intravenous infusion had no effect on the QT, PR or QRS intervals.
5.4 hemolytic Anemia (5.4 Hemolytic Anemia)
Hemolysis can occur during treatment of methemoglobinemia with methylene blue injection. Laboratory testing may show Heinz bodies, elevated indirect bilirubin and low haptoglobin, but the Coombs test is negative. The onset of anemia may be delayed 1 or more days after treatment with methylene blue injection. The anemia may require red blood cell transfusions [see Adverse Reactions (6.1)]. Use the lowest effective number of doses of methylene blue injection to treat methemoglobinemia. Discontinue methylene blue injection and consider alternative treatments of methemoglobinemia if severe hemolysis occurs.
Treatment of patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency with methylene blue injection may result in severe hemolysis and severe anemia. Methylene blue injection is contraindicated for use in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency [see Contraindications (4)].
8.7 Hepatic Impairment
Methylene blue is extensively metabolized in the liver. Monitor patients with any hepatic impairment for toxicities and potential drug interactions for an extended period of time following treatment with methylene blue injection.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Methylene Blue Injection is indicated for the treatment of pediatric and adult patients with acquired methemoglobinemia.
12.1 Mechanism of Action
Methylene blue is a water soluble thiazine dye that promotes a non-enzymatic redox conversion of metHb to hemoglobin. In situ, methylene blue is first converted to leucomethylene blue (LMB) via NADPH reductase. It is the LMB molecule which then reduces the ferric iron of metHb to the ferrous state of normal hemoglobin.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity: If severe or life threatening allergic reaction occurs, discontinue methylene blue injection, treat the allergic reaction, and monitor until signs and symptoms resolve. (5.2)
- Lack of Effectiveness: Consider alternative treatments if there is no resolution of methemoglobinemia after 2 doses. (2.1, 5.3)
- Hemolytic Anemia: Discontinue methylene blue injection and transfuse. (5.4)
- Interference with In Vivo Monitoring Devices: Use methods other than pulse oximetry to assess oxygen saturation. (5.5)
- Effects on Ability to Drive and Operate Machinery: Advise patients to refrain from these activities until neurologic and visual symptoms have resolved. (5.6)
5.3 lack of Effectiveness (5.3 Lack of Effectiveness)
Methemoglobinemia may not resolve or may rebound after response to treatment with methylene blue injection in patients with methemoglobinemia due to aryl amines such as aniline or sulfa drugs such as dapsone. Monitor response to therapy with methylene blue injection through resolution of methemoglobinemia. If methemoglobinemia does not respond to 2 doses of methylene blue injection or if methemoglobinemia rebounds after a response, consider additional treatment options [see Dosage and Administration (2.2)].
Patients with glucose-6-phosphate dehydrogenase deficiency may not reduce methylene blue injection to its active form in vivo. Methylene blue injection may not be effective in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Administer 1 mg/kg intravenously over 5-30 minutes. (2.1)
- If methemoglobin level remains above 30% or if clinical symptoms persist, give a repeat dose of up to 1 mg/kg one hour after the first dose. (2.1)
- Administer a single dose of 1 mg/kg in patients with moderate or severe renal impairment. (2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Methylene Blue Injection, USP: 50 mg per 10 mL (5 mg per mL) (0.5%) clear dark blue solution in single-dose vials.
5.5 Interference With in Vivo (5.5 Interference with In Vivo)
- Inaccurate Pulse Oximeter Readings
The presence of methylene blue in the blood may result in an underestimation of the oxygen saturation reading by pulse oximetry. If a measure of oxygen saturation is required during or shortly after infusion of methylene blue injection, it is advisable to obtain an arterial blood sample for testing by an alternative method.
- Bispectral index monitor
A fall in the Bispectral Index (BIS) has been reported following administration of methylene blue class products. If methylene blue injection is administered during surgery, alternative methods for assessing the depth of anesthesia should be employed.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
2.1 dosage and Administration (2.1 Dosage and Administration)
- Ensure patent venous access prior to administration of methylene blue injection. Do not administer methylene blue injection subcutaneously.
- Administer methylene blue injection 1 mg/kg intravenously over 5-30 minutes.
- If the methemoglobin level remains greater than 30% or if clinical signs and symptoms persist, a repeat dose of methylene blue injection 1 mg/kg may be given one hour after the first dose.
- If methemoglobinemia does not resolve after 2 doses of methylene blue injection, consider initiating alternative interventions for treatment of methemoglobinemia.
6.1 clinical Trials Experience (6.1 Clinical Trials Experience)
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of methylene blue injection in adults with acquired methemoglobinemia was assessed in 24 patients who received at least 1 dose of methylene blue injection [see Clinical Studies (14)]. Most doses administered were 1 mg/kg (88.5%), but doses from 1 mg/kg to 2 mg/kg were administered. All patients received at least one dose of methylene blue injection; two received two doses. Serious adverse reactions occurred in 4.2% of patients who received methylene blue injection. A serious adverse reaction of seizure-like phenomenon was reported in one patient. Adverse reactions (≥2%) included headache, hypokalemia, diarrhea, hypomagnesemia, myoclonus, nausea, and seizure-like phenomena.
The safety of methylene blue injection in pediatric patients with acquired methemoglobinemia was assessed in two retrospective case series that included two pediatric patients treated with methylene blue injection and 12 treated with another methylene blue product. The case series included patients in the following age groups: 3 neonates (<1 month), 4 infants (1 month to <2 years), 4 children (2 years to <12 years), and 3 adolescents (12 years to <17 years). The safety profile in pediatric patients was similar to that in adult patients.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Methylene Blue Injection, USP is supplied in 10 mL single-dose vials. Each 10 mL vial contains 50 mg of methylene blue as a clear dark blue solution. It is supplied as follows:
| NDC | Strength and Package Type | Package Size |
| 71288-811-11 | 50 mg per 10 mL (5 mg per mL) (0.5%) Single-Dose Vial | 5 vials per carton |
5.7 interference With Laboratory Tests (5.7 Interference with Laboratory Tests)
Methylene blue injection is a blue dye which passes freely into the urine and may interfere with the interpretation of any urine test which relies on a blue indicator, such as the dipstick test for leucocyte esterase.
14.1 Treatment of Acquired Methemoglobinemia
The efficacy of methylene blue injection in the treatment of patients with methemoglobinemia was evaluated in 24 adult patients with acquired methemoglobinemia: in study NCT03542760, a prospective, multicenter, observational registry. Of the 24 subjects enrolled 92% were white, 8% were black, 67% were female, and 33% were male. Hispanic or Latino was 12.5%; non-Hispanic or Latino was 87.5%, and ethnicity data were missing for 0%. The mean age was 46.0 years, and the ages ranged from 19 to 72 years. Each individual received at least 1 intravenous dose of methylene blue injection; two received 2 doses. Most doses administered were 1 mg/kg (88.5%), but doses from 1 mg/kg to 2 mg/kg were administered. The recommended methylene blue injection dose is 1 mg/kg; lower or greater doses are not recommended. The maximum recommended number of doses is two [see Dosage and Administration (2.1)].
In total, 22 of the 24 (91.7%) subjects had post treatment methemoglobin (metHb) assessment; 22 of the 22 subjects had baseline metHb with a mean concentration of 12.3% and a range of 4.1% to 30.0%. 21 of 22 (95.5%) subjects who had baseline metHb had at least a 50% reduction in metHB from baseline in their first assessment post baseline. This first post dosing assessment occurred from 0.7 to 27.3 hours from the end of first methylene blue injection infusion with a median time of 2.9 hours. There were 9 subjects that had baseline metHb and had metHb assessed within 2 hours of the end of the first methylene blue injection treatment; 6 of the 9 (67%; 95% CI (30.9%, 91.0%)) had at least a 50% reduction in metHb at 1 hour postdosing.
Available vital sign data including blood pressure, heart rate and respiratory rate were reviewed at baseline and compared to data collected within 2 hours post methylene blue injection infusion. Prior to treatment with methylene blue injection, 12 of the 18 (67%) of patients had a respiratory rate exceeding the upper limit of normal (≥ 20 bpm). Of these, 8 of the 12 (67%) experienced a normalization of respiratory rate within 2 hours post methylene blue injection infusion. There was minimal impact on other vital signs.
At baseline, the most common prespecified signs and symptoms of methemoglobinemia (reported by ≥2 subjects [8.3%] overall) were fatigue (33.3%), dyspnea (29.2%), cyanosis (12.5%), depressed CNS (12.5%), dizziness (8.3%), headache (8.3%), and weakness (8.3%). Following treatment with methylene blue injection, signs and symptoms of methemoglobinemia improved.
The efficacy of methylene blue injection in the treatment of methemoglobinemia in pediatric patients was assessed in 14 patients in two retrospective case series (2 patients received methylene blue injection and 12 who received another methylene blue product). The ages ranged from 6 days to 16 years. The efficacy outcomes were consistent across the pediatric and adult populations.
2.2 recommended Dosage for Renal Impairment (2.2 Recommended Dosage for Renal Impairment)
- The recommended dosage of methylene blue injection in patients with moderate or severe renal impairment (eGFR 15-59 mL/min/1.73 m2) is a single dose of 1 mg/kg.
- If the methemoglobin level remains greater than 30% or if the clinical symptoms persist 1 hour after dosing, consider initiating alternative interventions for the treatment of methemoglobinemia.
5.6 effects On Ability to Drive and Operate Machinery (5.6 Effects on Ability to Drive and Operate Machinery)
Treatment with methylene blue injection may cause confusion, dizziness and disturbances in vision [see Adverse Reactions (6)]. Advise patients to refrain from driving or engaging in hazardous occupations or activities such as operating heavy or potentially dangerous machinery until such adverse reactions to methylene blue injection have resolved.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a two-year carcinogenicity study, rats were administered oral doses of methylene blue at 5, 25, or 50 mg/kg. Methylene blue caused pancreatic islet adenomas or carcinomas (combined) in male rats. In a two-year carcinogenicity study, mice were administered oral doses of methylene blue at 2.5, 12.5, or 25 mg/kg. There were no drug-related neoplastic findings in mice.
Methylene blue was genotoxic in gene mutation assays in bacteria (Ames test), and in an in vitro sister chromatid exchange test and an in vitro chromosomal aberration test in Chinese hamster ovary (CHO) cells. Methylene blue was negative for micronucleus induction in bone marrow or peripheral blood collected from mice treated with methylene blue.
Fertility studies with methylene blue have not been conducted. In vitro, methylene blue reduced motility of human sperm in a concentration dependent manner.
Principal Display Panel – Methylene Blue Injection, Usp 10 Ml Carton (PRINCIPAL DISPLAY PANEL – Methylene Blue Injection, USP 10 mL Carton)
NDC 71288-811-11
Rx Only
Methylene Blue Injection, USP
50 mg per 10 mL
(5 mg per mL) (0.5%)
Intravenous Use Only
For Slow Intravenous Injection
Use 5% Dextrose Injection When Diluting
5 x 10 mL Single-Dose Vials
Discard unused portion
Principal Display Panel – Methylene Blue Injection, Usp 10 Ml Vial Label (PRINCIPAL DISPLAY PANEL – Methylene Blue Injection, USP 10 mL Vial Label)
NDC 71288-811-10
Rx Only
Methylene Blue Injection, USP
50 mg per 10 mL
(5 mg per mL) (0.5%)
Intravenous Use Only
Use 5% Dextrose Injection When Diluting
10 mL Single-Dose Vial
5.1 serotonin Syndrome With Concomitant Use of Serotonergic Drugs and Opioids (5.1 Serotonin Syndrome with Concomitant Use of Serotonergic Drugs and Opioids)
The development of serotonin syndrome has been reported with the use of methylene blue class products. Most reports have been associated with concomitant use of serotonergic drugs (e.g., selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors (MAOIs)). Opioids and dextromethorphan may increase the risk of developing serotonin syndrome. Some of the reported cases were fatal. Symptoms associated with serotonin syndrome may include the following combination of signs and symptoms: mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, and hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, and incoordination), seizures, and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Avoid concomitant use of methylene blue injection with serotonergic drugs and opioids.
Patients treated with methylene blue injection should be monitored for the emergence of serotonin syndrome. If symptoms of serotonin syndrome occur, discontinue use of methylene blue injection, and initiate supportive treatment. Inform patients of the increased risk of serotonin syndrome and advise them to not to take serotonergic drugs within 72 hours after the last dose of methylene blue injection [see Drug Interactions (7), Patient Counseling Information (17)].
Warning: Serotonin Syndrome With Concomitant Use of Serotonergic Drugs and Opioids (WARNING: SEROTONIN SYNDROME WITH CONCOMITANT USE OF SEROTONERGIC DRUGS AND OPIOIDS)
Methylene blue injection may cause serious or fatal serotonergic syndrome when used in combination with serotonergic drugs and opioids. Avoid concomitant use of methylene blue injection with selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors (MAOIs) and opioids [see Warnings and Precautions (5.1) and Drug Interactions (7.1)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:27.593864 · Updated: 2026-03-14T22:22:33.812720