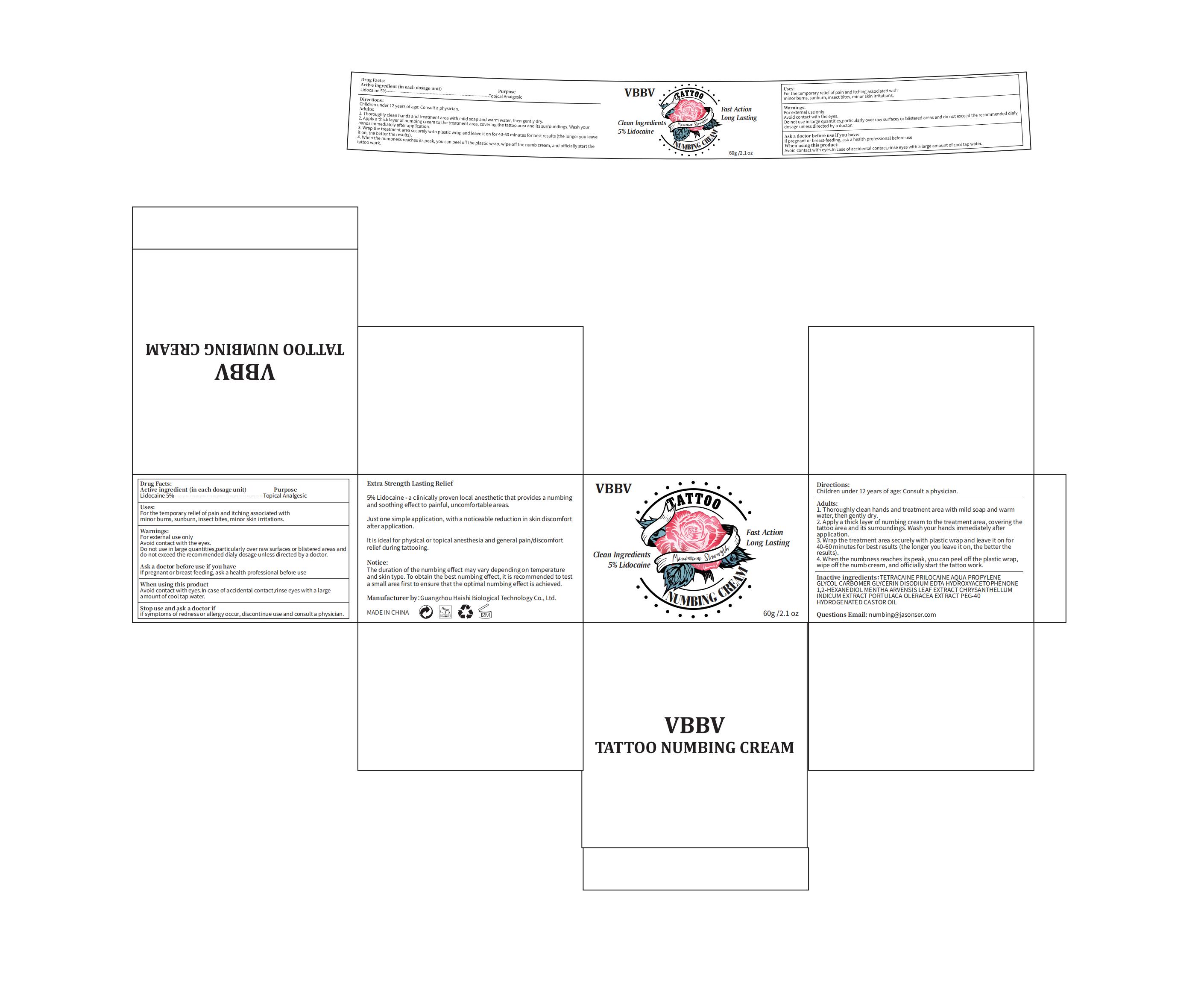
Vbbv Numbing Cream
24a36b2c-5f71-4574-e063-6394a90a3192
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 5%
Purpose
Topical Analgesic
Medication Information
Purpose
Topical Analgesic
Description
Lidocaine 5%
Uses
1. Thoroughly clean hands and treatment area with mild soap and warm water, then gently dry.
2. Apply a thick layer of numbing cream to the treatment area, covering the tattoo area and its surroundings. Wash your hands immediately after
application:
3. Wrap the treatment area securely with plastic wrap and leave it on for 40-60 minutes for best results (the longer you leave it on, the better the results).
4. When the numbness reaches its peak, you can peel off the plastic wrap, wipe off the numb cream, and officially start the tattoo work.
Dosage
Apply a thick layer of numbing cream to the treatment area, covering the tattoo area and its surroundings.
Section 50565-1
Children under 12 years of age: Consult a physician.
Section 51945-4
Stop Use
If symptoms of redness or allergy occur, discontinue use and consult a physician.
Warnings
For external use only
Avoid contact with the eyes.
Do not use in large quantities,particularly over raw surfaces or blistered areas and
do not exceed the recommended dialy dosage unless directed by a doctor.
Do Not Use
on damaged or broken skin.
When Using
Avoid contact with eyes.In case of accidental contact,rinse eyes with a large
amount of cool tap water.
Active Ingredients
Lidocaine 5%
Inactive Ingredients
AQUA 73%
PROPYLENE GLYCOL 4%
CARBOMER 0.3%
GLYCERIN 5%
DISODIUM EDTA 0.05%
HYDROXYACETOPHENONE 0.5%
1,2-HEXANEDIOL 0.4%
MENTHA ARVENSIS LEAF EXTRACT 3.05%
CHRYSANTHELLUM INDICUM EXTRACT 3.5%
PORTULACA OLERACEA EXTRACT 3%
PEG-40 HYDROGENATED CASTOR OIL 0.2%
Tetracaine 1%
Prilocaine 1%
Structured Label Content
Uses
1. Thoroughly clean hands and treatment area with mild soap and warm water, then gently dry.
2. Apply a thick layer of numbing cream to the treatment area, covering the tattoo area and its surroundings. Wash your hands immediately after
application:
3. Wrap the treatment area securely with plastic wrap and leave it on for 40-60 minutes for best results (the longer you leave it on, the better the results).
4. When the numbness reaches its peak, you can peel off the plastic wrap, wipe off the numb cream, and officially start the tattoo work.
Dosage
Apply a thick layer of numbing cream to the treatment area, covering the tattoo area and its surroundings.
Section 50565-1 (50565-1)
Children under 12 years of age: Consult a physician.
Section 51945-4 (51945-4)
Purpose
Topical Analgesic
Stop Use
If symptoms of redness or allergy occur, discontinue use and consult a physician.
Warnings
For external use only
Avoid contact with the eyes.
Do not use in large quantities,particularly over raw surfaces or blistered areas and
do not exceed the recommended dialy dosage unless directed by a doctor.
Do Not Use (Do not use)
on damaged or broken skin.
When Using
Avoid contact with eyes.In case of accidental contact,rinse eyes with a large
amount of cool tap water.
Active Ingredients (Active ingredients)
Lidocaine 5%
Inactive Ingredients (Inactive ingredients)
AQUA 73%
PROPYLENE GLYCOL 4%
CARBOMER 0.3%
GLYCERIN 5%
DISODIUM EDTA 0.05%
HYDROXYACETOPHENONE 0.5%
1,2-HEXANEDIOL 0.4%
MENTHA ARVENSIS LEAF EXTRACT 3.05%
CHRYSANTHELLUM INDICUM EXTRACT 3.5%
PORTULACA OLERACEA EXTRACT 3%
PEG-40 HYDROGENATED CASTOR OIL 0.2%
Tetracaine 1%
Prilocaine 1%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:51.597768 · Updated: 2026-03-14T23:04:09.079304