Atenolol Tablets, Usp
2492d5f6-00b0-42c9-a793-2be83ea81ad1
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
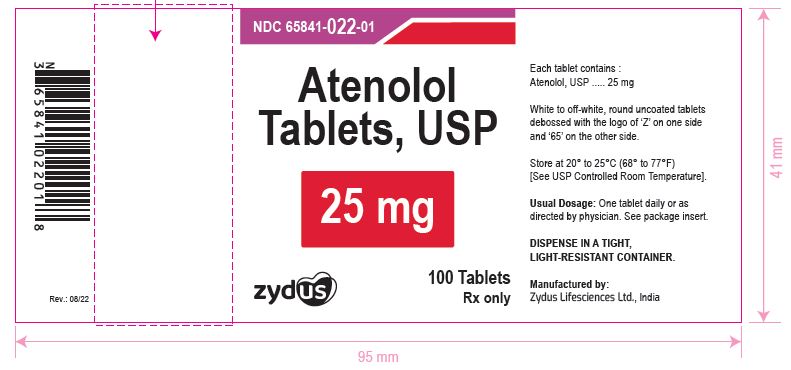
NDC 65841-022-01 in bottles of 100 tablets Atenolol Tablets USP, 25 mg R x only 100 Tablets NDC 65841-023-01 in bottles of 100 tablets Atenolol Tablets USP, 50 mg R x only 100 Tablets NDC 65841-024-01 in bottles of 100 tablets Atenolol Tablets USP, 100 mg R x only 100 Tablets
Medication Information
Description
NDC 65841-022-01 in bottles of 100 tablets Atenolol Tablets USP, 25 mg R x only 100 Tablets NDC 65841-023-01 in bottles of 100 tablets Atenolol Tablets USP, 50 mg R x only 100 Tablets NDC 65841-024-01 in bottles of 100 tablets Atenolol Tablets USP, 100 mg R x only 100 Tablets
Package Label.principal Display Panel
NDC 65841-022-01 in bottles of 100 tablets
Atenolol Tablets USP, 25 mg
Rx only
100 Tablets
NDC 65841-023-01 in bottles of 100 tablets
Atenolol Tablets USP, 50 mg
Rx only
100 Tablets
NDC 65841-024-01 in bottles of 100 tablets
Atenolol Tablets USP, 100 mg
Rx only
100 Tablets
Structured Label Content
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
NDC 65841-022-01 in bottles of 100 tablets
Atenolol Tablets USP, 25 mg
Rx only
100 Tablets
NDC 65841-023-01 in bottles of 100 tablets
Atenolol Tablets USP, 50 mg
Rx only
100 Tablets
NDC 65841-024-01 in bottles of 100 tablets
Atenolol Tablets USP, 100 mg
Rx only
100 Tablets
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:57.604971 · Updated: 2026-03-14T22:18:11.806691