tadalafil tablets
248a4705-1403-e94a-e063-6294a90a3544
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Tadalafil Tablets are a phosphodiesterase 5 (PDE5) inhibitor indicated for the treatment of: erectile dysfunction (ED) ( 1.1 ) the signs and symptoms of benign prostatic hyperplasia (BPH) ( 1.2 ) ED and the signs and symptoms of BPH (ED/BPH) ( 1.3 ) If Tadalafil Tablets are used with finasteride to initiate BPH treatment, such use is recommended for up to 26 weeks ( 1.4 ).
Dosage and Administration
Do not split Tadalafil Tablets; entire dose should be taken.
Contraindications
Administration of Tadalafil Tablets to patients using any form of organic nitrate is contraindicated. Tadalafil Tablets were shown to potentiate the hypotensive effect of nitrates ( 4.1 ). History of known serious hypersensitivity reaction to Tadalafil Tablets or ADCIRCA®( 4.2 ). Administration with guanylate cyclase (GC) stimulators, such as riociguat ( 4.3 ).
Warnings and Precautions
Evaluation of erectile dysfunction and BPH should include an appropriate medical assessment to identify potential underlying causes, as well as treatment options. Before prescribing Tadalafil Tablets, it is important to note the following:
Adverse Reactions
Most common adverse reactions (≥2%) include headache, dyspepsia, back pain, myalgia, nasal congestion, flushing, and pain in limb ( 6.1 ). To report SUSPECTED ADVERSE REACTIONS, contact TWi Pharmaceuticals, Inc. at 1-844-518-2989 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Drug Interactions
Physicians should be aware that Tadalafil Tablets for once daily use provides continuous plasma tadalafil levels and should consider this when evaluating the potential for interactions with medications (e.g., nitrates, alpha-blockers, anti-hypertensives and potent inhibitors of CYP3A4) and with substantial consumption of alcohol [see Drug Interactions ( 7.1 , 7.2 , 7.3 )] .
How Supplied
Tadalafil Tablets, USP are supplied as follows: Four strengths of round, pale yellow to yellow tablets are available in different sizes, and supplied in the following package sizes: Bottle of 7 Tablets NDC: 80425-0443-01
Description
Warnings and Precautions ( 5.4 ) 05/2017
Medication Information
Warnings and Precautions
Evaluation of erectile dysfunction and BPH should include an appropriate medical assessment to identify potential underlying causes, as well as treatment options. Before prescribing Tadalafil Tablets, it is important to note the following:
Indications and Usage
Tadalafil Tablets are a phosphodiesterase 5 (PDE5) inhibitor indicated for the treatment of: erectile dysfunction (ED) ( 1.1 ) the signs and symptoms of benign prostatic hyperplasia (BPH) ( 1.2 ) ED and the signs and symptoms of BPH (ED/BPH) ( 1.3 ) If Tadalafil Tablets are used with finasteride to initiate BPH treatment, such use is recommended for up to 26 weeks ( 1.4 ).
Dosage and Administration
Do not split Tadalafil Tablets; entire dose should be taken.
Contraindications
Administration of Tadalafil Tablets to patients using any form of organic nitrate is contraindicated. Tadalafil Tablets were shown to potentiate the hypotensive effect of nitrates ( 4.1 ). History of known serious hypersensitivity reaction to Tadalafil Tablets or ADCIRCA®( 4.2 ). Administration with guanylate cyclase (GC) stimulators, such as riociguat ( 4.3 ).
Adverse Reactions
Most common adverse reactions (≥2%) include headache, dyspepsia, back pain, myalgia, nasal congestion, flushing, and pain in limb ( 6.1 ). To report SUSPECTED ADVERSE REACTIONS, contact TWi Pharmaceuticals, Inc. at 1-844-518-2989 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Drug Interactions
Physicians should be aware that Tadalafil Tablets for once daily use provides continuous plasma tadalafil levels and should consider this when evaluating the potential for interactions with medications (e.g., nitrates, alpha-blockers, anti-hypertensives and potent inhibitors of CYP3A4) and with substantial consumption of alcohol [see Drug Interactions ( 7.1 , 7.2 , 7.3 )] .
How Supplied
Tadalafil Tablets, USP are supplied as follows: Four strengths of round, pale yellow to yellow tablets are available in different sizes, and supplied in the following package sizes: Bottle of 7 Tablets NDC: 80425-0443-01
Description
Warnings and Precautions ( 5.4 ) 05/2017
Section 34077-8
Risk Summary
Tadalafil Tablets are not indicated for use in females.
There are no data with the use of Tadalafil Tablets in pregnant women to inform any drug-associated risks for adverse developmental outcomes. In animal reproduction studies, no adverse developmental effects were observed with oral administration of tadalafil to pregnant rats or mice during organogenesis at exposures up to 11 times the maximum recommended human dose (MRHD) of 20 mg/day ( seeData)
Data
Animal Data
Animal reproduction studies showed no evidence of teratogenicity, embryotoxicity, or fetotoxicity when tadalafil was given orally to pregnant rats or mice at exposures up to 11 times the maximum recommended human dose (MRHD) of 20 mg/day during organogenesis. In a prenatal/postnatal developmental study in rats, postnatal pup survival decreased following maternal exposure to tadalafil doses greater than 10 times the MRHD based on AUC. Signs of maternal toxicity occurred at doses greater than 16 times the MRHD based on AUC. Surviving offspring had normal development and reproductive performance.
In another rat prenatal and postnatal development study at doses of 60, 200, and 1000 mg/kg, a reduction in postnatal survival of pups was observed. The no observed effect level (NOEL) for maternal toxicity was 200 mg/kg/day and for developmental toxicity was 30 mg/kg/day. This gives approximately 16 and 10 fold exposure multiples, respectively, of the human AUC for the MRHD of 20 mg.
Tadalafil and/or its metabolites cross the placenta, resulting in fetal exposure in rats.
Section 42229-5
Renal Impairment
Section 42230-3
Patient Information
Tadalafil (ta da’ la fil) Tablets, USP
Read this important information before you start taking Tadalafil Tablets and each time you get a refill. There may be new information. You may also find it helpful to share this information with your partner. This information does not take the place of talking with your healthcare provider. You and your healthcare provider should talk about Tadalafil Tablets when you start taking it and at regular checkups. If you do not understand the information, or have questions, talk with your healthcare provider or pharmacist.
What Is The Most Important Information I Should Know About Tadalafil Tablets?
Tadalafil Tablets can cause your blood pressure to drop suddenly to an unsafe level if it is taken with certain other medicines.You could get dizzy, faint, or have a heart attack or stroke. Never take Tadalafil Tablets with any nitrate or guanylate cyclase stimulator medicines.
Do not take Tadalafil Tablets if you take any medicines called “nitrates.”Nitrates are commonly used to treat angina. Angina is a symptom of heart disease and can cause pain in your chest, jaw, or down your arm.
- Medicines called nitrates include nitroglycerin that is found in tablets, sprays, ointments, pastes, or patches. Nitrates can also be found in other medicines such as isosorbide dinitrate or isosorbide mononitrate. Some recreational drugs called “poppers” also contain nitrates, such as amyl nitrite and butyl nitrite.
Do not take Tadalafil Tablets if you take medicines called guanylate cyclase stimulators which include:
- Riociguat (Adempas ®) a medicine that treats pulmonary arterial hypertension and chronic-thromboembolic pulmonary hypertension.
Ask your healthcare provider or pharmacist if you are not sure if any of your medicines are nitrates or guanylate cyclase stimulators, such as riociguat.
(See “ Who Should Not Take Tadalafil Tablets?” )
Tell all of your healthcare providers that you take Tadalafil Tablets. If you need emergency medical care for a heart problem, it will be important for your healthcare provider to know when you last took Tadalafil Tablets.
After taking a single tablet, some of the active ingredient of Tadalafil Tablets remains in your body for more than 2 days.The active ingredient can remain longer if you have problems with your kidneys or liver, or you are taking certain other medications (see “ Can Other Medicines Affect Tadalafil Tablets?” ).
Stop sexual activity and get medical help right away if you get symptoms such as chest pain, dizziness, or nausea during sex. Sexual activity can put an extra strain on your heart, especially if your heart is already weak from a heart attack or heart disease.
See also “ What Are The Possible Side Effects Of Tadalafil Tablets?”
What Are Tadalafil Tablets?
Tadalafil Tablets are a prescription medicine taken by mouth for the treatment of:
- men with erectile dysfunction (ED)
- men with symptoms of benign prostatic hyperplasia (BPH)
- men with both ED and BPH
Tadalafil Tablets for the Treatment of ED
ED is a condition where the penis does not fill with enough blood to harden and expand when a man is sexually excited, or when he cannot keep an erection. A man who has trouble getting or keeping an erection should see his healthcare provider for help if the condition bothers him. Tadalafil Tablets help increase blood flow to the penis and may help men with ED get and keep an erection satisfactory for sexual activity. Once a man has completed sexual activity, blood flow to his penis decreases, and his erection goes away.
Some form of sexual stimulation is needed for an erection to happen with Tadalafil Tablets.
Tadalafil Tablets do not:
- cure ED
- increase a man's sexual desire
- protect a man or his partner from sexually transmitted diseases, including HIV. Speak to your healthcare provider about ways to guard against sexually transmitted diseases.
- serve as a male form of birth control
Tadalafil Tablets are only for men over the age of 18, including men with diabetes or who have undergone prostatectomy.
Tadalafil Tablets for the Treatment of Symptoms of BPH
BPH is a condition that happens in men, where the prostate gland enlarges which can cause urinary symptoms.
Tadalafil Tablets for the Treatment of ED and Symptoms of BPH
ED and symptoms of BPH may happen in the same person and at the same time. Men who have both ED and symptoms of BPH may take Tadalafil Tablets for the treatment of both conditions.
Tadalafil Tablets are not for women or children.
Tadalafil Tablets must be used only under a healthcare provider's care.
Who Should Not Take Tadalafil Tablets?
Do not take Tadalafil Tablets if you:
- take any medicines called “nitrates”.
- use recreational drugs called “poppers” like amyl nitrite and butyl nitrite. (See “ What Is The Most Important Information I Should Know About Tadalafil Tablets?” )
- take any medicines called guanylate cyclase stimulators, such as riociguat.
-
are allergic to Tadalafil Tablets or ADCIRCA®
, or any of its ingredients.See the end of this leaflet for a complete list of ingredients in Tadalafil Tablets. Symptoms of an allergic reaction may include:
- rash
- hives
- swelling of the lips, tongue, or throat
- difficulty breathing or swallowing
Call your healthcare provider or get help right away if you have any of the symptoms of an allergic reaction listed above.
What Should I Tell My Healthcare Provider Before Taking Tadalafil Tablets?
Tadalafil Tablets are not right for everyone. Only your healthcare provider and you can decide if Tadalafil Tablets are right for you.Before taking Tadalafil Tablets, tell your healthcare provider about all your medical problems, including if you:
- have heart problemssuch as angina, heart failure, irregular heartbeats, or have had a heart attack. Ask your healthcare provider if it is safe for you to have sexual activity. You should not take Tadalafil Tablets if your healthcare provider has told you not to have sexual activity because of your health problems.
- have pulmonary hypertension
- have low blood pressure orhave high blood pressure that is not controlled
- have had a stroke
- have liver problems
- have kidney problems or require dialysis
- have retinitis pigmentosa,a rare genetic (runs in families) eye disease
- have ever had severe vision loss, including a condition called NAION
- have stomach ulcers
- have a bleeding problem
- have a deformed penis shapeor Peyronie's disease
- have had an erection that lasted more than 4 hours
- have blood cell problemssuch as sickle cell anemia, multiple myeloma, or leukemia
Can Other Medicines Affect Tadalafil Tablets?
Tell your healthcare provider about all the medicines you take including prescription and non-prescription medicines, vitamins, and herbal supplements. Tadalafil Tablets and other medicines may affect each other. Always check with your healthcare provider before starting or stopping any medicines. Especially tell your healthcare provider if you take any of the following*:
- medicines called nitrates (see “ What Is The Most Important Information I Should Know About Tadalafil Tablets?” )
- medicines called guanylate cyclase stimulators, such as riociguat (Adempas ®), used to treat pulmonary hypertension
- medicines called alpha blockers. These include Hytrin ® (terazosin HCl), Flomax ® (tamsulosin HCl), Cardura ® (doxazosin mesylate), Minipress ® (prazosin HCl), Uroxatral ® (alfuzosin HCl), Jalyn ®(dutasteride and tamsulosin HCl) or Rapaflo ®(silodosin). Alpha-blockers are sometimes prescribed for prostate problems or high blood pressure. If Tadalafil Tablets are taken with certain alpha blockers, your blood pressure could suddenly drop. You could get dizzy or faint.
- other medicines to treat high blood pressure (hypertension)
- medicines called HIV protease inhibitors, such as ritonavir (Norvir ®, Kaletra ®)
- some types of oral antifungals such as ketoconazole (Nizoral ®), itraconazole (Sporanox ®)
- some types of antibiotics such as clarithromycin (Biaxin ®), telithromycin (Ketek ®), erythromycin (several brand names exist. Please consult your healthcare provider to determine if you are taking this medicine).
- other medicines or treatments for ED.
- Tadalafil Tablets are also marketed as ADCIRCA for the treatment of pulmonary arterial hypertension. Do not take both Tadalafil Tablets and ADCIRCA. Do not take sildenafil citrate (Revatio ®) with Tadalafil Tablets.
How Should I Take Tadalafil Tablets?
- Take Tadalafil Tablets exactly as your healthcare provider prescribes it. Your healthcare provider will prescribe the dose that is right for you.
- Some men can only take a low dose of Tadalafil Tablets or may have to take it less often, because of medical conditions or medicines they take.
- Do not change your dose or the way you take Tadalafil Tablets without talking to your healthcare provider. Your healthcare provider may lower or raise your dose, depending on how your body reacts to Tadalafil Tablets and your health condition.
- Tadalafil Tablets may be taken with or without meals.
- If you take too much Tadalafil Tablets, call your healthcare provider or emergency room right away.
How Should I Take Tadalafil Tablets for Symptoms of BPH?
For symptoms of BPH, Tadalafil Tablets are taken once daily.
- Do not take Tadalafil Tablets more than one time each day.
- Take one Tadalafil Tablet every day at about the same time of day.
- If you miss a dose, you may take it when you remember but do not take more than one dose per day.
How Should I Take Tadalafil Tablets for ED?
For ED, there are two ways to take Tadalafil Tablets - either for use as needed OR for use once daily.
Tadalafil Tablets for use as needed:
- Do not take Tadalafil Tablets more than one time each day.
- Take one Tadalafil Tablet before you expect to have sexual activity. You may be able to have sexual activity at 30 minutes after taking Tadalafil Tablets and up to 36 hours after taking it. You and your healthcare provider should consider this in deciding when you should take Tadalafil Tablets before sexual activity. Some form of sexual stimulation is needed for an erection to happen with Tadalafil Tablets.
- Your healthcare provider may change your dose of Tadalafil Tablets depending on how you respond to the medicine, and on your health condition.
OR
Tadalafil Tablets for once daily use are a lower dose you take every day.
- Do not take Tadalafil Tablets more than one time each day.
- Take one Tadalafil Tablet every day at about the same time of day. You may attempt sexual activity at any time between doses.
- If you miss a dose, you may take it when you remember but do not take more than one dose per day.
- Some form of sexual stimulation is needed for an erection to happen with Tadalafil Tablets.
- Your healthcare provider may change your dose of Tadalafil Tablets depending on how you respond to the medicine, and on your health condition.
How Should I Take Tadalafil Tablets for Both ED and the Symptoms of BPH?
For both ED and the symptoms of BPH, Tadalafil Tablets are taken once daily.
- Do not take Tadalafil Tablets more than one time each day.
- Take one Tadalafil Tablet every day at about the same time of day. You may attempt sexual activity at any time between doses.
- If you miss a dose, you may take it when you remember but do not take more than one dose per day.
- Some form of sexual stimulation is needed for an erection to happen with Tadalafil Tablets.
What Should I Avoid While Taking Tadalafil Tablets?
- Do not use other ED medicines or ED treatments while taking Tadalafil Tablets.
- Do not drink too much alcohol when taking Tadalafil Tablets (for example, 5 glasses of wine or 5 shots of whiskey). Drinking too much alcohol can increase your chances of getting a headache or getting dizzy, increasing your heart rate, or lowering your blood pressure.
What Are The Possible Side Effects Of Tadalafil Tablets?
See “ What Is The Most Important Information I Should Know About Tadalafil Tablets?”
The most common side effects with Tadalafil Tablets are:headache, indigestion, back pain, muscle aches, flushing, and stuffy or runny nose. These side effects usually go away after a few hours. Men who get back pain and muscle aches usually get it 12 to 24 hours after taking Tadalafil Tablets. Back pain and muscle aches usually go away within 2 days.
Call your healthcare provider if you get any side effect that bothers you or one that does not go away.
Uncommon side effects include:
An erection that won't go away (priapism). If you get an erection that lasts more than 4 hours, get medical help right away. Priapism must be treated as soon as possible or lasting damage can happen to your penis, including the inability to have erections.
Color vision changes, such as seeing a blue tinge (shade) to objects or having difficulty telling the difference between the colors blue and green.
In rare instances, men taking PDE5 inhibitors (oral erectile dysfunction medicines, including Tadalafil Tablets) reported a sudden decrease or loss of vision in one or both eyes. It is uncertain whether PDE5 inhibitors directly cause the vision loss. If you experience sudden decrease or loss of vision, stop taking PDE5 inhibitors, including Tadalafil Tablets, and call a healthcare provider right away.
Sudden loss or decrease in hearing, sometimes with ringing in the ears and dizziness, has been rarely reported in people taking PDE5 inhibitors, including Tadalafil Tablets. It is not possible to determine whether these events are related directly to the PDE5 inhibitors, to other diseases or medications, to other factors, or to a combination of factors. If you experience these symptoms, stop taking Tadalafil Tablets and contact a healthcare provider right away.
These are not all the possible side effects of Tadalafil Tablets. For more information, ask your healthcare provider or pharmacist.
How Should I Store Tadalafil Tablets?
Store Tadalafil Tablets at room temperature between 59° and 86°F (15° and 30°C).
Keep Tadalafil Tablets and all medicines out of the reach of children.
General Information About Tadalafil Tablets:
Medicines are sometimes prescribed for conditions other than those described in patient information leaflets. Do not use Tadalafil Tablets for a condition for which it was not prescribed. Do not give Tadalafil Tablets to other people, even if they have the same symptoms that you have. It may harm them.
This is a summary of the most important information about Tadalafil Tablets. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about Tadalafil Tablets that is written for health providers.
What Are The Ingredients In Tadalafil Tablets?
Active Ingredient: tadalafil, USP
Inactive Ingredients: croscarmellose sodium, hydroxypropyl cellulose, hypromellose, iron oxide, lactose monohydrate, magnesium stearate, microcrystalline cellulose, sodium lauryl sulfate, talc, titanium dioxide, and triacetin.
This Patient Information has been approved by the U.S. Food and Drug Administration
Rx only
*Trademarks are the property of their respective owners.
34120062611A Revised: January 2024
Manufactured for:
TWi Pharmaceuticals USA, Inc.
Paramus, NJ 07652
Section 43683-2
Warnings and Precautions ( 5.4) 05/2017
5.9 Alcohol
Patients should be made aware that both alcohol and Tadalafil Tablets, a PDE5 inhibitor, act as mild vasodilators. When mild vasodilators are taken in combination, blood-pressure-lowering effects of each individual compound may be increased. Therefore, physicians should inform patients that substantial consumption of alcohol (e.g., 5 units or greater) in combination with Tadalafil Tablets can increase the potential for orthostatic signs and symptoms, including increase in heart rate, decrease in standing blood pressure, dizziness, and headache [see Clinical Pharmacology ( 12.2)] .
16.2 Storage
Store at 25°C (77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Keep out of reach of children.
17.9 Alcohol
Patients should be made aware that both alcohol and Tadalafil Tablets, a PDE5 inhibitor, act as mild vasodilators. When mild vasodilators are taken in combination, blood-pressure-lowering effects of each individual compound may be increased. Therefore, physicians should inform patients that substantial consumption of alcohol (e.g., 5 units or greater) in combination with Tadalafil Tablets can increase the potential for orthostatic signs and symptoms, including increase in heart rate, decrease in standing blood pressure, dizziness, and headache [see Warnings and Precautions ( 5.9), Drug Interactions ( 7.1), and Clinical Pharmacology ( 12.2)] .
4.1 Nitrates
Administration of Tadalafil Tablets to patients who are using any form of organic nitrate, either regularly and/or intermittently, is contraindicated. In clinical pharmacology studies, Tadalafil Tablets were shown to potentiate the hypotensive effect of nitrates [see Clinical Pharmacology ( 12.2)] .
10 Overdosage
Single doses up to 500 mg have been given to healthy subjects, and multiple daily doses up to 100 mg have been given to patients. Adverse events were similar to those seen at lower doses. In cases of overdose, standard supportive measures should be adopted as required. Hemodialysis contributes negligibly to tadalafil elimination.
17.1 Nitrates
Physicians should discuss with patients the contraindication of Tadalafil Tablets with regular and/or intermittent use of organic nitrates. Patients should be counseled that concomitant use of Tadalafil Tablets with nitrates could cause blood pressure to suddenly drop to an unsafe level, resulting in dizziness, syncope, or even heart attack or stroke.
Physicians should discuss with patients the appropriate action in the event that they experience anginal chest pain requiring nitroglycerin following intake of Tadalafil Tablets. In such a patient, who has taken Tadalafil Tablets, where nitrate administration is deemed medically necessary for a life-threatening situation, at least 48 hours should have elapsed after the last dose of Tadalafil Tablets before nitrate administration is considered. In such circumstances, nitrates should still only be administered under close medical supervision with appropriate hemodynamic monitoring. Therefore, patients who experience anginal chest pain after taking Tadalafil Tablets should seek immediate medical attention [see Contraindications ( 4.1) and Warnings and Precautions ( 5.1)] .
17.6 Priapism
There have been rare reports of prolonged erections greater than 4 hours and priapism (painful erections greater than 6 hours in duration) for this class of compounds. Priapism, if not treated promptly, can result in irreversible damage to the erectile tissue. Physicians should advise patients who have an erection lasting greater than 4 hours, whether painful or not, to seek emergency medical attention.
8.2 Lactation
Risk Summary
Tadalafil Tablets are not indicated for use in females.
There is no information on the presence of tadalafil and/or metabolites in human milk, the effects on the breastfed child, or the effects on milk production. Tadalafil and/or its metabolites are present in the milk of lactating rats at concentrations approximately 2.4-fold greater than found in the plasma.
11 Description
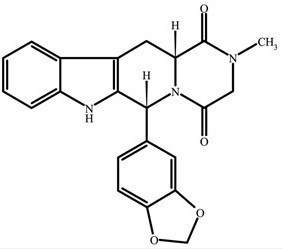
Tadalafil is a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type 5 (PDE5). Tadalafil has the empirical formula C 22H 19N 3O 4representing a molecular weight of 389.41. The structural formula is:
The chemical designation is pyrazino[1´,2´:1,6]pyrido[3,4-b]indole-1,4-dione, 6-(1,3-benzodioxol-5-yl)-2,3,6,7,12,12a-hexahydro-2-methyl-, (6R,12aR)-. It is a crystalline solid that is practically insoluble in water and very slightly soluble in ethanol.
Tadalafil Tablets, USP are available as round, pale yellow to yellow tablets for oral administration. Each tablet contains 2.5, 5, 10, or 20 mg of tadalafil and the following inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, hypromellose, iron oxide, lactose monohydrate, magnesium stearate, microcrystalline cellulose, sodium lauryl sulfate, talc, titanium dioxide, and triacetin.
16.1 How Supplied
Tadalafil Tablets, USP are supplied as follows:
Four strengths of round, pale yellow to yellow tablets are available in different sizes, and supplied in the following package sizes:
Bottle of 7 Tablets NDC: 80425-0443-01
2.5 Use With Food
Tadalafil Tablets may be taken without regard to food.
8.4 Pediatric Use
Tadalafil Tablets are not indicated for use in pediatric patients. Safety and efficacy in patients below the age of 18 years have not been established.
Juvenile Animal Study
No adverse effects were observed in a study in which tadalafil was administered orally at doses of 60, 200, and 1000 mg/kg/day to juvenile rats on postnatal days 14 to 90. The highest plasma tadalafil exposures (AUC) achieved were approximately 10-fold that observed at the MRHD.
Additional information describing a clinical study in which efficacy was not demonstrated is approved for Eli Lilly and Company’s CIALIS (tadalafil) tablets. However, due to Eli Lilly and Company’s marketing exclusivity rights, this drug product is not labeled with that pediatric information.
8.5 Geriatric Use
Of the total number of subjects in ED clinical studies of tadalafil, approximately 19 percent were 65 and over, while approximately 2 percent were 75 and over. Of the total number of subjects in BPH clinical studies of tadalafil (including the ED/BPH study), approximately 40 percent were over 65, while approximately 10 percent were 75 and over. In these clinical trials, no overall differences in efficacy or safety were observed between older (>65 and ≥75 years of age) and younger subjects (≤65 years of age). However, in placebo-controlled studies with Tadalafil Tablets for use as needed for ED, diarrhea was reported more frequently in patients 65 years of age and older who were treated with Tadalafil Tablets (2.5% of patients) [see Adverse Reactions ( 6.1)] . No dose adjustment is warranted based on age alone. However, a greater sensitivity to medications in some older individuals should be considered. [see Clinical Pharmacology ( 12.3)] .
5.1 Cardiovascular
Physicians should consider the cardiovascular status of their patients, since there is a degree of cardiac risk associated with sexual activity. Therefore, treatments for erectile dysfunction, including Tadalafil Tablets, should not be used in men for whom sexual activity is inadvisable as a result of their underlying cardiovascular status. Patients who experience symptoms upon initiation of sexual activity should be advised to refrain from further sexual activity and seek immediate medical attention.
Physicians should discuss with patients the appropriate action in the event that they experience anginal chest pain requiring nitroglycerin following intake of Tadalafil Tablets. In such a patient, who has taken Tadalafil Tablets, where nitrate administration is deemed medically necessary for a life-threatening situation, at least 48 hours should have elapsed after the last dose of Tadalafil Tablets before nitrate administration is considered. In such circumstances, nitrates should still only be administered under close medical supervision with appropriate hemodynamic monitoring. Therefore, patients who experience anginal chest pain after taking Tadalafil Tablets should seek immediate medical attention. [see Contraindications ( 4.1) and Patient Counseling Information ( 17.1)] .
Patients with left ventricular outflow obstruction, (e.g., aortic stenosis and idiopathic hypertrophic subaortic stenosis) can be sensitive to the action of vasodilators, including PDE5 inhibitors.
The following groups of patients with cardiovascular disease were not included in clinical safety and efficacy trials for Tadalafil Tablets, and therefore until further information is available, Tadalafil Tablets are not recommended for the following groups of patients:
- myocardial infarction within the last 90 days
- unstable angina or angina occurring during sexual intercourse
- New York Heart Association Class 2 or greater heart failure in the last 6 months
- uncontrolled arrhythmias, hypotension (<90/50 mm Hg), or uncontrolled hypertension
- stroke within the last 6 months.
As with other PDE5 inhibitors, tadalafil has mild systemic vasodilatory properties that may result in transient decreases in blood pressure. In a clinical pharmacology study, tadalafil 20 mg resulted in a mean maximal decrease in supine blood pressure, relative to placebo, of 1.6/0.8 mm Hg in healthy subjects [see Clinical Pharmacology ( 12.2)] . While this effect should not be of consequence in most patients, prior to prescribing Tadalafil Tablets, physicians should carefully consider whether their patients with underlying cardiovascular disease could be affected adversely by such vasodilatory effects. Patients with severely impaired autonomic control of blood pressure may be particularly sensitive to the actions of vasodilators, including PDE5 inhibitors.
4 Contraindications
- Administration of Tadalafil Tablets to patients using any form of organic nitrate is contraindicated. Tadalafil Tablets were shown to potentiate the hypotensive effect of nitrates ( 4.1).
- History of known serious hypersensitivity reaction to Tadalafil Tablets or ADCIRCA®( 4.2).
- Administration with guanylate cyclase (GC) stimulators, such as riociguat ( 4.3).
6 Adverse Reactions
Most common adverse reactions (≥2%) include headache, dyspepsia, back pain, myalgia, nasal congestion, flushing, and pain in limb ( 6.1).
To report SUSPECTED ADVERSE REACTIONS, contact TWi Pharmaceuticals, Inc. at 1-844-518-2989 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
7 Drug Interactions
- Tadalafil Tablets can potentiate the hypotensive effects of nitrates, alpha-blockers, antihypertensives or alcohol ( 7.1).
- CYP3A4 inhibitors (e.g. ketoconazole, ritonavir) increase Tadalafil Tablets exposure (
2.7,
5.10,
7.2) requiring dose adjustment:
- Tadalafil Tablets for use as needed: no more than 10 mg every 72 hours
- Tadalafil Tablets for once daily use: dose not to exceed 2.5 mg
- CYP3A4 inducers (e.g. rifampin) decrease Tadalafil Tablets exposure ( 7.2).
8.7 Renal Impairment
In clinical pharmacology studies using single-dose tadalafil (5 to 10 mg), tadalafil exposure (AUC) doubled in subjects with creatinine clearance 30 to 80 mL/min. In subjects with end-stage renal disease on hemodialysis, there was a two-fold increase in C maxand 2.7- to 4.8-fold increase in AUC following single-dose administration of 10 or 20 mg tadalafil. Exposure to total methylcatechol (unconjugated plus glucuronide) was 2- to 4-fold higher in subjects with renal impairment, compared to those with normal renal function. Hemodialysis (performed between 24 and 30 hours post-dose) contributed negligibly to tadalafil or metabolite elimination. In a clinical pharmacology study (N=28) at a dose of 10 mg, back pain was reported as a limiting adverse event in male patients with creatinine clearance 30 to 50 mL/min. At a dose of 5 mg, the incidence and severity of back pain was not significantly different than in the general population. In patients on hemodialysis taking 10- or 20-mg tadalafil, there were no reported cases of back pain. [see Dosage and Administration ( 2.6) and Warnings and Precautions ( 5.7)] .
1.4 Limitation of Use
If Tadalafil Tablets are used with finasteride to initiate BPH treatment, such use is recommended for up to 26 weeks because the incremental benefit of Tadalafil Tablets decreases from 4 weeks until 26 weeks, and the incremental benefit of Tadalafil Tablets beyond 26 weeks is unknown [see Clinical Studies ( 14.3)] .
12.3 Pharmacokinetics
Over a dose range of 2.5 to 20 mg, tadalafil exposure (AUC) increases proportionally with dose in healthy subjects. Steady-state plasma concentrations are attained within 5 days of once per day dosing and exposure is approximately 1.6-fold greater than after a single dose. Mean tadalafil concentrations measured after the administration of a single oral dose of 20 mg and single and once daily multiple doses of 5 mg, from a separate study, ( see Figure 4) to healthy male subjects are depicted in Figure 4.
Figure 4: Plasma tadalafil concentrations (mean ± SD) following a single 20-mg tadalafil dose and single and once daily multiple doses of 5 mg
5.3 Prolonged Erection
There have been rare reports of prolonged erections greater than 4 hours and priapism (painful erections greater than 6 hours in duration) for this class of compounds. Priapism, if not treated promptly, can result in irreversible damage to the erectile tissue. Patients who have an erection lasting greater than 4 hours, whether painful or not, should seek emergency medical attention.
Tadalafil Tablets should be used with caution in patients who have conditions that might predispose them to priapism (such as sickle cell anemia, multiple myeloma, or leukemia), or in patients with anatomical deformation of the penis (such as angulation, cavernosal fibrosis, or Peyronie's disease).
5.4 Effects On the Eye
Physicians should advise patients to stop use of all phosphodiesterase type 5 (PDE5) inhibitors, including Tadalafil Tablets, and seek medical attention in the event of a sudden loss of vision in one or both eyes. Such an event may be a sign of non-arteritic anterior ischemic optic neuropathy (NAION), a rare condition and a cause of decreased vision, including permanent loss of vision, that has been reported rarely postmarketing in temporal association with the use of all PDE5 inhibitors. Based on published literature, the annual incidence of NAION is 2.5-11.8 cases per 100,000 in males aged ≥50.Physicians should advise patients to stop use of all phosphodiesterase type 5 (PDE5) inhibitors, including Tadalafil Tablets, and seek medical attention in the event of a sudden loss of vision in one or both eyes. Such an event may be a sign of non-arteritic anterior ischemic optic neuropathy (NAION), a rare condition and a cause of decreased vision, including permanent loss of vision, that has been reported rarely postmarketing in temporal association with the use of all PDE5 inhibitors. Based on published literature, the annual incidence of NAION is 2.5-11.8 cases per 100,000 in males aged ≥50.
An observational case-crossover study evaluated the risk of NAION when PDE5 inhibitor use, as a class, occurred immediately before NAION onset (within 5 half-lives), compared to PDE5 inhibitor use in a prior time period. The results suggest an approximate 2-fold increase in the risk of NAION, with a risk estimate of 2.15 (95% CI 1.06, 4.34). A similar study reported a consistent result, with a risk estimate of 2.27 (95% CI 0.99, 5.20). Other risk factors for NAION, such as the presence of "crowded" optic disc, may have contributed to the occurrence of NAION in these studies.
Neither the rare postmarketing reports, nor the association of PDE5 inhibitor use and NAION in the observational studies, substantiate a causal relationship between PDE5 inhibitor use and NAION [see Adverse Reactions ( 6.2)].
Physicians should consider whether their patients with underlying NAION risk factors could be adversely affected by use of PDE5 inhibitors. Individuals who have already experienced NAION are at increased risk of NAION recurrence. Therefore, PDE5 inhibitors, including Tadalafil Tablets, should be used with caution in these patients and only when the anticipated benefits outweigh the risks. Individuals with "crowded" optic disc are also considered at greater risk for NAION compared to the general population; however, evidence is insufficient to support screening of prospective users of PDE5 inhibitors, including Tadalafil Tablets, for this uncommon condition.Physicians should consider whether their patients with underlying NAION risk factors could be adversely affected by use of PDE5 inhibitors. Individuals who have already experienced NAION are at increased risk of NAION recurrence. Therefore, PDE5 inhibitors, including Tadalafil Tablets, should be used with caution in these patients and only when the anticipated benefits outweigh the risks. Individuals with "crowded" optic disc are also considered at greater risk for NAION compared to the general population; however, evidence is insufficient to support screening of prospective users of PDE5 inhibitors, including Tadalafil Tablets, for this uncommon condition.
Patients with known hereditary degenerative retinal disorders, including retinitis pigmentosa, were not included in the clinical trials, and use in these patients is not recommended.
8.6 Hepatic Impairment
In clinical pharmacology studies, tadalafil exposure (AUC) in subjects with mild or moderate hepatic impairment (Child-Pugh Class A or B) was comparable to exposure in healthy subjects when a dose of 10 mg was administered. There are no available data for doses higher than 10 mg of tadalafil in patients with hepatic impairment. Insufficient data are available for subjects with severe hepatic impairment (Child-Pugh Class C). [see Dosage and Administration ( 2.6) and Warnings and Precautions ( 5.8)] .
1 Indications and Usage
Tadalafil Tablets are a phosphodiesterase 5 (PDE5) inhibitor indicated for the treatment of:
- erectile dysfunction (ED) ( 1.1)
- the signs and symptoms of benign prostatic hyperplasia (BPH) ( 1.2)
- ED and the signs and symptoms of BPH (ED/BPH) ( 1.3)
If Tadalafil Tablets are used with finasteride to initiate BPH treatment, such use is recommended for up to 26 weeks ( 1.4).
5.5 Sudden Hearing Loss
Physicians should advise patients to stop taking PDE5 inhibitors, including Tadalafil Tablets, and seek prompt medical attention in the event of sudden decrease or loss of hearing. These events, which may be accompanied by tinnitus and dizziness, have been reported in temporal association to the intake of PDE5 inhibitors, including Tadalafil Tablets. It is not possible to determine whether these events are related directly to the use of PDE5 inhibitors or to other factors [see Adverse Reactions ( 6.1, 6.2)] .
1.1 Erectile Dysfunction
Tadalafil Tablets are indicated for the treatment of erectile dysfunction (ED).
12.1 Mechanism of Action
Penile erection during sexual stimulation is caused by increased penile blood flow resulting from the relaxation of penile arteries and corpus cavernosal smooth muscle. This response is mediated by the release of nitric oxide (NO) from nerve terminals and endothelial cells, which stimulates the synthesis of cGMP in smooth muscle cells. Cyclic GMP causes smooth muscle relaxation and increased blood flow into the corpus cavernosum. The inhibition of phosphodiesterase type 5 (PDE5) enhances erectile function by increasing the amount of cGMP. Tadalafil inhibits PDE5. Because sexual stimulation is required to initiate the local release of nitric oxide, the inhibition of PDE5 by tadalafil has no effect in the absence of sexual stimulation.
The effect of PDE5 inhibition on cGMP concentration in the corpus cavernosum and pulmonary arteries is also observed in the smooth muscle of the prostate, the bladder and their vascular supply. The mechanism for reducing BPH symptoms has not been established.
Studies in vitrohave demonstrated that tadalafil is a selective inhibitor of PDE5. PDE5 is found in the smooth muscle of the corpus cavernosum, prostate, and bladder as well as in vascular and visceral smooth muscle, skeletal muscle, urethra, platelets, kidney, lung, cerebellum, heart, liver, testis, seminal vesicle, and pancreas.
In vitrostudies have shown that the effect of tadalafil is more potent on PDE5 than on other phosphodiesterases. These studies have shown that tadalafil is >10,000-fold more potent for PDE5 than for PDE1, PDE2, PDE4, and PDE7 enzymes, which are found in the heart, brain, blood vessels, liver, leukocytes, skeletal muscle, and other organs. Tadalafil is >10,000-fold more potent for PDE5 than for PDE3, an enzyme found in the heart and blood vessels. Additionally, tadalafil is 700-fold more potent for PDE5 than for PDE6, which is found in the retina and is responsible for phototransduction. Tadalafil is >9,000-fold more potent for PDE5 than for PDE8, PDE9, and PDE10. Tadalafil is 14-fold more potent for PDE5 than for PDE11A1 and 40-fold more potent for PDE5 than for PDE11A4, two of the four known forms of PDE11. PDE11 is an enzyme found in human prostate, testes, skeletal muscle and in other tissues (e.g., adrenal cortex). In vitro, tadalafil inhibits human recombinant PDE11A1 and, to a lesser degree, PDE11A4 activities at concentrations within the therapeutic range. The physiological role and clinical consequence of PDE11 inhibition in humans have not been defined.
17.8 Sudden Hearing Loss
Physicians should advise patients to stop taking PDE5 inhibitors, including Tadalafil Tablets, and seek prompt medical attention in the event of sudden decrease or loss of hearing. These events, which may be accompanied by tinnitus and dizziness, have been reported in temporal association to the intake of PDE5 inhibitors, including Tadalafil Tablets. It is not possible to determine whether these events are related directly to the use of PDE5 inhibitors or to other factors [see Adverse Reactions ( 6.1, 6.2)] .
5.12 Effects On Bleeding
Studies in vitrohave demonstrated that tadalafil is a selective inhibitor of PDE5. PDE5 is found in platelets. When administered in combination with aspirin, tadalafil 20 mg did not prolong bleeding time, relative to aspirin alone. Tadalafil Tablets have not been administered to patients with bleeding disorders or significant active peptic ulceration. Although Tadalafil Tablets have not been shown to increase bleeding times in healthy subjects, use in patients with bleeding disorders or significant active peptic ulceration should be based upon a careful risk-benefit assessment and caution.
17.7 Sudden Loss of Vision
Physicians should advise patients to stop use of all PDE5 inhibitors, including Tadalafil Tablets, and seek medical attention in the event of a sudden loss of vision in one or both eyes. Such an event may be a sign of non-arteritic anterior ischemic optic neuropathy (NAION), a cause of decreased vision, including possible permanent loss of vision, that has been reported rarely postmarketing in temporal association with the use of all PDE5 inhibitors. Physicians should discuss with patients the increased risk of NAION among the general population in patients with a "crowded" optic disc, although evidence is insufficient to support screening of prospective users of PDE5 inhibitors, including Tadalafil Tablets, for this uncommon condition [see Warnings and Precautions ( 5.4) and Adverse Reactions ( 6.2)] .
5 Warnings and Precautions
Evaluation of erectile dysfunction and BPH should include an appropriate medical assessment to identify potential underlying causes, as well as treatment options.
Before prescribing Tadalafil Tablets, it is important to note the following:
2 Dosage and Administration
Do not split Tadalafil Tablets; entire dose should be taken.
3 Dosage Forms and Strengths
Four strengths of round, pale yellow to yellow tablets are available in different sizes:
2.5 mg tablets debossed with “43”
5 mg tablets debossed with “44”
10 mg tablets debossed with “45”
20 mg tablets debossed with “47”
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of Tadalafil Tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These events have been chosen for inclusion either due to their seriousness, reporting frequency, lack of clear alternative causation, or a combination of these factors.
Cardiovascular and Cerebrovascular — Serious cardiovascular events, including myocardial infarction, sudden cardiac death, stroke, chest pain, palpitations, and tachycardia, have been reported postmarketing in temporal association with the use of tadalafil. Most, but not all, of these patients had preexisting cardiovascular risk factors. Many of these events were reported to occur during or shortly after sexual activity, and a few were reported to occur shortly after the use of Tadalafil Tablets without sexual activity. Others were reported to have occurred hours to days after the use of Tadalafil Tablets and sexual activity. It is not possible to determine whether these events are related directly to Tadalafil Tablets, to sexual activity, to the patient's underlying cardiovascular disease, to a combination of these factors, or to other factors [see Warnings and Precautions ( 5.1)] .
Body as a Whole — hypersensitivity reactions including urticaria, Stevens-Johnson syndrome, and exfoliative dermatitis
Nervous — migraine, seizure and seizure recurrence, transient global amnesia
Ophthalmologic — visual field defect, retinal vein occlusion, retinal artery occlusion
Non-arteritic anterior ischemic optic neuropathy (NAION), a cause of decreased vision including permanent loss of vision, has been reported rarely postmarketing in temporal association with the use of PDE5 inhibitors, including Tadalafil Tablets. Most, but not all, of these patients had underlying anatomic or vascular risk factors for development of NAION, including but not necessarily limited to: low cup to disc ratio (“crowded disc”), age over 50, diabetes, hypertension, coronary artery disease, hyperlipidemia, and smoking. [see Warnings and Precautions ( 5.4)] .
Otologic — Cases of sudden decrease or loss of hearing have been reported postmarketing in temporal association with the use of PDE5 inhibitors, including Tadalafil Tablets. In some of the cases, medical conditions and other factors were reported that may have also played a role in the otologic adverse events. In many cases, medical follow-up information was limited. It is not possible to determine whether these reported events are related directly to the use of Tadalafil Tablets, to the patient's underlying risk factors for hearing loss, a combination of these factors, or to other factors [see Warnings and Precautions ( 5.5)] .
Urogenital — priapism [see Warnings and Precautions ( 5.3)] .
8 Use in Specific Populations
Hepatic Impairment ( 2.6, 5.8, 8.6):
- Mild or Moderate: Dosage adjustment may be needed.
- Severe: Use is not recommended.
Renal Impairment ( 2.6, 5.7, 8.7):
- Patients with creatinine clearance 30 to 50 mL/min: Dosage adjustment may be needed.
- Patients with creatinine clearance less than 30 mL/min or on hemodialysis: For use as needed: Dose should not exceed 5 mg every 72 hours. Once daily use is not recommended.
4.2 Hypersensitivity Reactions
Tadalafil Tablets are contraindicated in patients with a known serious hypersensitivity to tadalafil or ADCIRCA®. Hypersensitivity reactions have been reported, including Stevens-Johnson syndrome and exfoliative dermatitis [see Adverse Reactions ( 6.2)] .
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Tadalafil was administered to over 9000 men during clinical trials worldwide. In trials of Tadalafil Tablets for once daily use, a total of 1434, 905, and 115 were treated for at least 6 months, 1 year, and 2 years, respectively. For Tadalafil Tablets for use as needed, over 1300 and 1000 subjects were treated for at least 6 months and 1 year, respectively.
1.2 Benign Prostatic Hyperplasia
Tadalafil Tablets are indicated for the treatment of the signs and symptoms of benign prostatic hyperplasia (BPH).
17.11 Recommended Administration
Physicians should instruct patients on the appropriate administration of Tadalafil Tablets to allow optimal use.
For Tadalafil Tablets for use as needed in men with ED, patients should be instructed to take one tablet at least 30 minutes before anticipated sexual activity. In most patients, the ability to have sexual intercourse is improved for up to 36 hours.
For Tadalafil Tablets for once daily use in men with ED or ED/BPH, patients should be instructed to take one tablet at approximately the same time every day without regard for the timing of sexual activity. Tadalafil Tablets are effective at improving erectile function over the course of therapy.
For Tadalafil Tablets for once daily use in men with BPH, patients should be instructed to take one tablet at approximately the same time every day.
17 Patient Counseling Information
“See FDA-approved patient labeling ( Patient Information)”
17.10 Sexually Transmitted Disease
The use of Tadalafil Tablets offers no protection against sexually transmitted diseases. Counseling of patients about the protective measures necessary to guard against sexually transmitted diseases, including Human Immunodeficiency Virus (HIV) should be considered.
17.3 Cardiovascular Considerations
Physicians should consider the potential cardiac risk of sexual activity in patients with preexisting cardiovascular disease. Physicians should advise patients who experience symptoms upon initiation of sexual activity to refrain from further sexual activity and seek immediate medical attention [see Warnings and Precautions ( 5.1)] .
Package Label. Principal Display Panel
17.2 Guanylate Cyclase (gc) Stimulators
Physicians should discuss with patients the contraindication of Tadalafil Tablets with any use of a GC stimulator, such as riociguat, for pulmonary arterial hypertension. Patients should be counseled that the concomitant use of Tadalafil Tablets with GC stimulators may cause blood pressure to drop to an unsafe level.
5.6 Alpha Blockers and Antihypertensives
Physicians should discuss with patients the potential for Tadalafil Tablets to augment the blood-pressure-lowering effect of alpha-blockers and antihypertensive medications [see Drug Interactions ( 7.1) and Clinical Pharmacology ( 12.2)] .
Caution is advised when PDE5 inhibitors are coadministered with alpha-blockers. PDE5 inhibitors, including Tadalafil Tablets, and alpha-adrenergic blocking agents are both vasodilators with blood-pressure-lowering effects. When vasodilators are used in combination, an additive effect on blood pressure may be anticipated. In some patients, concomitant use of these two drug classes can lower blood pressure significantly [see Drug Interactions ( 7.1) and Clinical Pharmacology ( 12.2)] , which may lead to symptomatic hypotension (e.g., fainting). Consideration should be given to the following:
13.2 Animal Toxicology And/or Pharmacology
Animal studies showed vascular inflammation in tadalafil-treated mice, rats, and dogs. In mice and rats, lymphoid necrosis and hemorrhage were seen in the spleen, thymus, and mesenteric lymph nodes at unbound tadalafil exposure of 2- to 33-fold above the human exposure (AUCs) at the MRHD of 20 mg. In dogs, an increased incidence of disseminated arteritis was observed in 1- and 6-month studies at unbound tadalafil exposure of 1- to 54-fold above the human exposure (AUC) at the MRHD of 20 mg. In a 12-month dog study, no disseminated arteritis was observed, but 2 dogs exhibited marked decreases in white blood cells (neutrophils) and moderate decreases in platelets with inflammatory signs at unbound tadalafil exposures of approximately 14- to 18-fold the human exposure at the MRHD of 20 mg. The abnormal blood-cell findings were reversible within 2 weeks after stopping treatment.
14.1 Tadalafil Tablets for Use As Needed for Ed
The efficacy and safety of tadalafil in the treatment of erectile dysfunction has been evaluated in 22 clinical trials of up to 24-weeks duration, involving over 4000 patients. Tadalafil Tablets, when taken as needed up to once per day, were shown to be effective in improving erectile function in men with erectile dysfunction (ED).
Tadalafil Tablets were studied in the general ED population in 7 randomized, multicenter, double-blinded, placebo-controlled, parallel-arm design, primary efficacy and safety studies of 12-weeks duration. Two of these studies were conducted in the United States and 5 were conducted in centers outside the US. Additional efficacy and safety studies were performed in ED patients with diabetes mellitus and in patients who developed ED status post bilateral nerve-sparing radical prostatectomy.
In these 7 trials, Tadalafil Tablets were taken as needed, at doses ranging from 2.5 to 20 mg, up to once per day. Patients were free to choose the time interval between dose administration and the time of sexual attempts. Food and alcohol intake were not restricted.
Several assessment tools were used to evaluate the effect of Tadalafil Tablets on erectile function. The 3 primary outcome measures were the Erectile Function (EF) domain of the International Index of Erectile Function (IIEF) and Questions 2 and 3 from Sexual Encounter Profile (SEP). The IIEF is a 4-week recall questionnaire that was administered at the end of a treatment-free baseline period and subsequently at follow-up visits after randomization. The IIEF EF domain has a 30-point total score, where higher scores reflect better erectile function. SEP is a diary in which patients recorded each sexual attempt made throughout the study. SEP Question 2 asks, “Were you able to insert your penis into the partner's vagina?” SEP Question 3 asks, “Did your erection last long enough for you to have successful intercourse?” The overall percentage of successful attempts to insert the penis into the vagina (SEP2) and to maintain the erection for successful intercourse (SEP3) is derived for each patient.
8.3 Females and Males of Reproductive Potential
Infertility
Based on the data from 3 studies in adult males, tadalafil decreased sperm concentrations in the study of 10 mg tadalafil for 6 months and the study of 20 mg tadalafil for 9 months. This effect was not seen in the study of 20 mg tadalafil taken for 6 months. There was no adverse effect of tadalafil 10 mg or 20 mg on mean concentrations of testosterone, luteinizing hormone or follicle stimulating hormone. The clinical significance of the decreased sperm concentrations in the two studies is unknown. There have been no studies evaluating the effect of tadalafil on fertility in men [see Clinical Pharmacology ( 12.2)] .
Based on studies in animals, a decrease in spermatogenesis was observed in dogs, but not in rats [see Nonclinical Toxicology ( 13.1)] .
14.2 Tadalafil Tablets for Once Daily Use for Ed
The efficacy and safety of Tadalafil Tablets for once daily use in the treatment of erectile dysfunction has been evaluated in 2 clinical trials of 12-weeks duration and 1 clinical trial of 24-weeks duration, involving a total of 853 patients. Tadalafil Tablets, when taken once daily, were shown to be effective in improving erectile function in men with erectile dysfunction (ED).
Tadalafil Tablets were studied in the general ED population in 2 randomized, multicenter, double-blinded, placebo-controlled, parallel-arm design, primary efficacy and safety studies of 12- and 24-weeks duration, respectively. One of these studies was conducted in the United States and one was conducted in centers outside the US. An additional efficacy and safety study was performed in ED patients with diabetes mellitus. Tadalafil Tablets were taken once daily at doses ranging from 2.5 to 10 mg. Food and alcohol intake were not restricted. Timing of sexual activity was not restricted relative to when patients took Tadalafil Tablets.
4.3 Concomitant Guanylate Cyclase (gc) Stimulators
Do not use Tadalafil Tablets in patients who are using a GC stimulator, such as riociguat. PDE5 inhibitors, including Tadalafil Tablets, may potentiate the hypotensive effects of GC stimulators.
1.3 Erectile Dysfunction and Benign Prostatic Hyperplasia
Tadalafil Tablets are indicated for the treatment of ED and the signs and symptoms of BPH (ED/BPH).
7.2 Potential for Other Drugs to Affect Tadalafil Tablets
17.4 Concomitant Use With Drugs Which Lower Blood Pressure
5.13 Counseling Patients About Sexually Transmitted Diseases
The use of Tadalafil Tablets offers no protection against sexually transmitted diseases. Counseling patients about the protective measures necessary to guard against sexually transmitted diseases, including Human Immunodeficiency Virus (HIV) should be considered.
14.4 Tadalafil Tablets 5 mg for Once Daily Use for Ed and Bph
The efficacy and safety of Tadalafil Tablets for once daily use for the treatment of ED, and the signs and symptoms of BPH, in patients with both conditions was evaluated in one placebo-controlled, multinational, double-blind, parallel-arm study which randomized 606 patients to receive either Tadalafil Tablets 2.5 mg, 5 mg, for once daily use or placebo. ED severity ranged from mild to severe and BPH severity ranged from moderate to severe. The full study population had a mean age of 63 years (range 45 to 83) and was 93% White, 4% Black, 3% other races; 16% were of Hispanic ethnicity. Patients with multiple co-morbid conditions such as diabetes mellitus, hypertension, and other cardiovascular disease were included.
In this study, the co-primary endpoints were total IPSS and the Erectile Function (EF) domain score of the International Index of Erectile Function (IIEF). One of the key secondary endpoints in this study was Question 3 of the Sexual Encounter Profile diary (SEP3). Timing of sexual activity was not restricted relative to when patients took Tadalafil Tablets.
The efficacy results for patients with both ED and BPH, who received either Tadalafil Tablets 5 mg for once daily use or placebo (N=408) are shown in Tables 21and 22and Figure 8.
Tadalafil Tablets 5 mg for once daily use resulted in statistically significant improvements in the total IPSS and in the EF domain of the IIEF questionnaire. Tadalafil Tablets 5 mg for once daily use also resulted in statistically significant improvement in SEP3. Tadalafil Tablets 2.5 mg did not result in statistically significant improvement in the total IPSS.
| Placebo | Tadalafil Tablets 5 mg | p-value | |
| Total Symptom Score (IPSS) | |||
| (N=193) | (N=206) | ||
| Baseline | 18.2 | 18.5 | |
| Change from Baseline to Week 12 | -3.8 | -6.1 | <.001 |
| EF Domain Score (IIEF EF) | |||
| (N=188) | (N=202) | ||
| Baseline | 15.6 | 16.5 | |
| Endpoint | 17.6 | 22.9 | |
| Change from Baseline to Week 12 | 1.9 | 6.5 | <.001 |
| Placebo | Tadalafil Tablets 5 mg | ||
| (N=187) | (N=199) | p-value | |
| Maintenance of Erection (SEP3) | |||
| Baseline | 36% | 43% | |
| Endpoint | 48% | 72% | |
| Change from Baseline to Week 12 | 12% | 32% | <.001 |
Tadalafil Tablets for once daily use resulted in improvement in the IPSS total score at the first scheduled observation (week 2) and throughout the 12 weeks of treatment ( see Figure 8).
Figure 8: Mean IPSS Changes in ED/BPH Patients by Visit in Study L
In this study, the effect of Tadalafil Tablets 5 mg once daily on Q maxwas evaluated as a safety endpoint. Mean Q maxincreased from baseline in both the treatment and placebo groups (Tadalafil Tablets 5 mg: 1.6 mL/sec, placebo: 1.2 mL/sec); however, these changes were not significantly different between groups.
2.1 Tadalafil Tablets for Use As Needed for Erectile Dysfunction
- The recommended starting dose of Tadalafil Tablets for use as needed in most patients is 10 mg, taken prior to anticipated sexual activity.
- The dose may be increased to 20 mg or decreased to 5 mg, based on individual efficacy and tolerability. The maximum recommended dosing frequency is once per day in most patients.
- Tadalafil Tablets for use as needed was shown to improve erectile function compared to placebo up to 36 hours following dosing. Therefore, when advising patients on optimal use of Tadalafil Tablets, this should be taken into consideration.
2.2 Tadalafil Tablets for Once Daily Use for Erectile Dysfunction
- The recommended starting dose of Tadalafil Tablets for once daily use is 2.5 mg, taken at approximately the same time every day, without regard to timing of sexual activity.
- The Tadalafil Tablets dose for once daily use may be increased to 5 mg, based on individual efficacy and tolerability.
2.3 Tadalafil Tablets for Once Daily Use for Benign Prostatic Hyperplasia
- The recommended dose of Tadalafil Tablets for once daily use is 5 mg, taken at approximately the same time every day.
- When therapy for BPH is initiated with Tadalafil Tablets and finasteride, the recommended dose of Tadalafil Tablets for once daily use is 5 mg, taken at approximately the same time every day for up to 26 weeks.
5.10 Concomitant Use of Potent Inhibitors of Cytochrome P450 3a4 (cyp3a4)
Tadalafil Tablets are metabolized predominantly by CYP3A4 in the liver. The dose of Tadalafil Tablets for use as needed should be limited to 10 mg no more than once every 72 hours in patients taking potent inhibitors of CYP3A4 such as ritonavir, ketoconazole, and itraconazole [see Drug Interactions ( 7.2)] . In patients taking potent inhibitors of CYP3A4 and Tadalafil Tablets for once daily use, the maximum recommended dose is 2.5 mg [see Dosage and Administration ( 2.7)] .
5.11 Combination With Other Pde5 Inhibitors Or Erectile Dysfunction Therapies
The safety and efficacy of combinations of Tadalafil Tablets and other PDE5 inhibitors or treatments for erectile dysfunction have not been studied. Inform patients not to take Tadalafil Tablets with other PDE5 inhibitors, including ADCIRCA.
5.2 Potential for Drug Interactions When Taking Tadalafil Tablets for Once Daily Use
Physicians should be aware that Tadalafil Tablets for once daily use provides continuous plasma tadalafil levels and should consider this when evaluating the potential for interactions with medications (e.g., nitrates, alpha-blockers, anti-hypertensives and potent inhibitors of CYP3A4) and with substantial consumption of alcohol [see Drug Interactions ( 7.1, 7.2, 7.3)] .
17.5 Potential for Drug Interactions When Taking Tadalafil Tablets for Once Daily Use
Physicians should discuss with patients the clinical implications of continuous exposure to tadalafil when prescribing Tadalafil Tablets for once daily use, especially the potential for interactions with medications (e.g., nitrates, alpha-blockers, antihypertensives and potent inhibitors of cytochrome P450 3A4) and with substantial consumption of alcohol. [see Dosage and Administration ( 2.7), Warnings and Precautions ( 5.6), Drug Interactions ( 7.1, 7.2), Clinical Pharmacology ( 12.2), and Clinical Studies ( 14.2)] .
14.3 Tadalafil Tablets 5 mg for Once Daily Use for Benign Prostatic Hyperplasia (bph)
The efficacy and safety of Tadalafil Tablets for once daily use for the treatment of the signs and symptoms of BPH was evaluated in 3 randomized, multinational, double-blinded, placebo-controlled, parallel-design, efficacy and safety studies of 12 weeks duration. Two of these studies were in men with BPH and one study was specific to men with both ED and BPH [see Clinical Studies ( 14.4)] . The first study (Study J) randomized 1058 patients to receive either Tadalafil Tablets 2.5 mg, 5 mg, 10 mg or 20 mg for once daily use or placebo. The second study (Study K) randomized 325 patients to receive either Tadalafil Tablets 5 mg for once daily use or placebo. The full study population was 87% White, 2% Black, 11% other races; 15% was of Hispanic ethnicity. Patients with multiple co-morbid conditions such as diabetes mellitus, hypertension, and other cardiovascular disease were included.
The primary efficacy endpoint in the two studies that evaluated the effect of Tadalafil Tablets for the signs and symptoms of BPH was the International Prostate Symptom Score (IPSS), a four week recall questionnaire that was administered at the beginning and end of a placebo run-in period and subsequently at follow-up visits after randomization. The IPSS assesses the severity of irritative (frequency, urgency, nocturia) and obstructive symptoms (incomplete emptying, stopping and starting, weak stream, and pushing or straining), with scores ranging from 0 to 35; higher numeric scores representing greater severity. Maximum urinary flow rate (Q max), an objective measure of urine flow, was assessed as a secondary efficacy endpoint in Study J and as a safety endpoint in Study K.
The results for BPH patients with moderate to severe symptoms and a mean age of 63.2 years (range 44 to 87) who received either Tadalafil Tablets 5 mg for once daily use or placebo (N=748) in Studies J and K are shown in Table 19and Figures 5and 6, respectively.
In each of these 2 trials, Tadalafil Tablets 5 mg for once daily use resulted in statistically significant improvement in the total IPSS compared to placebo. Mean total IPSS showed a decrease starting at the first scheduled observation (4 weeks) in Study K and remained decreased through 12 weeks.
| Study J | Study K | |||||
| Placebo |
Tadalafil Tablets
5 mg |
Placebo |
Tadalafil Tablets
5 mg |
|||
| (N=205) | (N=205) | p-value | (N=164) | (N=160) | p-value | |
| Total Symptom Score (IPSS) | ||||||
| Baseline | 17.1 | 17.3 | 16.6 | 17.1 | ||
| Change from Baseline to Week 12 | -2.2 | -4.8 | <.001 | -3.6 | -5.6 | .004 |
Figure 5: Mean IPSS Changes in BPH Patients by Visit in Study J
Figure 6: Mean IPSS Changes in BPH Patients by Visit in Study K
In Study J, the effect of Tadalafil Tablets 5 mg once daily on maximum urinary flow rate (Q max) was evaluated as a secondary efficacy endpoint. Mean Q maxincreased from baseline in both the treatment and placebo groups (Tadalafil Tablets 5 mg: 1.6 mL/sec, placebo: 1.2 mL/sec); however, these changes were not significantly different between groups.
In Study K, the effect of Tadalafil Tablets 5 mg once daily on Q maxwas evaluated as a safety endpoint. Mean Q maxincreased from baseline in both the treatment and placebo groups (Tadalafil Tablets 5 mg: 1.6 mL/sec, placebo: 1.1 mL/sec); however, these changes were not significantly different between groups.
Efficacy Results in Patients with BPH initiating Tadalafil Tablets and Finasteride– Tadalafil Tablets for once daily use initiated together with finasteride was shown to be effective in treating the signs and symptoms of BPH in men with an enlarged prostate (>30 cc) for up to 26 weeks. This additional double-blinded, parallel-design study of 26 weeks duration randomized 696 men to initiate either Tadalafil Tablets 5 mg with finasteride 5 mg or placebo with finasteride 5 mg. The study population had a mean age of 64 years (range 46-86). Patients with multiple co-morbid conditions such as erectile dysfunction, diabetes mellitus, hypertension, and other cardiovascular disease were included.
Tadalafil Tablets with finasteride demonstrated statistically significant improvement in the signs and symptoms of BPH compared to placebo with finasteride, as measured by the total IPSS at 12 weeks, the primary study endpoint ( see Table 20). Key secondary endpoints demonstrated improvement in total IPSS starting at the first scheduled observation at week 4 (Tadalafil Tablets -4.0, placebo -2.3: p<.001) and the score remained decreased through 26 weeks (Tadalafil Tablets -5.5, placebo -4.5; p=.022). However, the magnitude of the treatment difference between placebo/finasteride and Tadalafil Tablets/finasteride decreased from 1.7 points at Week 4 to 1.0 point at Week 26, as shown in Table 20and in Figure 7. The incremental benefit of Tadalafil Tablets beyond 26 weeks is unknown.
|
aOverall ITT population. |
||||||
|
bMixed model for repeated measurements. |
||||||
|
cUnadjusted mean. |
||||||
| Placebo and finasteride 5 mg | Tadalafil Tablets 5mg and finasteride 5 mg | Treatment difference | ||||
| n | (N=350) a | n | (N=345) a | p-value b | ||
| Total Symptom Score (IPSS) | ||||||
| Baseline c | 349 | 17.4 | 344 | 17.1 | ||
| Change from Baseline to Week 4 b | 340 | -2.3 | 330 | -4.0 | -1.7 | <.001 |
| Change from Baseline to Week 12 b | 318 | -3.8 | 317 | -5.2 | -1.4 | .001 |
| Change from Baseline to Week 26 b | 295 | -4.5 | 308 | -5.5 | -1.0 | .022 |
Figure 7: Mean Total IPSS Changes By Visit in BPH Patients Taking Tadalafil Tablets for Once Daily Use Together With Finasteride
In the 404 patients who had both ED and BPH at baseline, changes in erectile function were assessed as key secondary endpoints using the EF domain of the IIEF questionnaire. Tadalafil Tablets with finasteride (N=203) were compared to placebo with finasteride (N=201). A statistically significant improvement from baseline (Tadalafil Tablets/finasteride 13.7, placebo/finasteride 15.1) was observed at week 4 (Tadalafil Tablets/finasteride 3.7, placebo/finasteride -1.1; p<.001), week 12 (Tadalafil Tablets/finasteride 4.7, placebo/finasteride 0.6; p<.001), and week 26 (Tadalafil Tablets/finasteride 4.7, placebo/finasteride 0.0; p<.001).
5.14 Consideration of Other Urological Conditions Prior to Initiating Treatment for Bph
Prior to initiating treatment with Tadalafil Tablets for BPH, consideration should be given to other urological conditions that may cause similar symptoms. In addition, prostate cancer and BPH may coexist.
2.4 Tadalafil Tablets for Once Daily Use for Erectile Dysfunction and Benign Prostatic Hyperplasia
The recommended dose of Tadalafil Tablets for once daily use is 5 mg, taken at approximately the same time every day, without regard to timing of sexual activity.
Structured Label Content
Section 34077-8 (34077-8)
Risk Summary
Tadalafil Tablets are not indicated for use in females.
There are no data with the use of Tadalafil Tablets in pregnant women to inform any drug-associated risks for adverse developmental outcomes. In animal reproduction studies, no adverse developmental effects were observed with oral administration of tadalafil to pregnant rats or mice during organogenesis at exposures up to 11 times the maximum recommended human dose (MRHD) of 20 mg/day ( seeData)
Data
Animal Data
Animal reproduction studies showed no evidence of teratogenicity, embryotoxicity, or fetotoxicity when tadalafil was given orally to pregnant rats or mice at exposures up to 11 times the maximum recommended human dose (MRHD) of 20 mg/day during organogenesis. In a prenatal/postnatal developmental study in rats, postnatal pup survival decreased following maternal exposure to tadalafil doses greater than 10 times the MRHD based on AUC. Signs of maternal toxicity occurred at doses greater than 16 times the MRHD based on AUC. Surviving offspring had normal development and reproductive performance.
In another rat prenatal and postnatal development study at doses of 60, 200, and 1000 mg/kg, a reduction in postnatal survival of pups was observed. The no observed effect level (NOEL) for maternal toxicity was 200 mg/kg/day and for developmental toxicity was 30 mg/kg/day. This gives approximately 16 and 10 fold exposure multiples, respectively, of the human AUC for the MRHD of 20 mg.
Tadalafil and/or its metabolites cross the placenta, resulting in fetal exposure in rats.
Section 42229-5 (42229-5)
Renal Impairment
Section 42230-3 (42230-3)
Patient Information
Tadalafil (ta da’ la fil) Tablets, USP
Read this important information before you start taking Tadalafil Tablets and each time you get a refill. There may be new information. You may also find it helpful to share this information with your partner. This information does not take the place of talking with your healthcare provider. You and your healthcare provider should talk about Tadalafil Tablets when you start taking it and at regular checkups. If you do not understand the information, or have questions, talk with your healthcare provider or pharmacist.
What Is The Most Important Information I Should Know About Tadalafil Tablets?
Tadalafil Tablets can cause your blood pressure to drop suddenly to an unsafe level if it is taken with certain other medicines.You could get dizzy, faint, or have a heart attack or stroke. Never take Tadalafil Tablets with any nitrate or guanylate cyclase stimulator medicines.
Do not take Tadalafil Tablets if you take any medicines called “nitrates.”Nitrates are commonly used to treat angina. Angina is a symptom of heart disease and can cause pain in your chest, jaw, or down your arm.
- Medicines called nitrates include nitroglycerin that is found in tablets, sprays, ointments, pastes, or patches. Nitrates can also be found in other medicines such as isosorbide dinitrate or isosorbide mononitrate. Some recreational drugs called “poppers” also contain nitrates, such as amyl nitrite and butyl nitrite.
Do not take Tadalafil Tablets if you take medicines called guanylate cyclase stimulators which include:
- Riociguat (Adempas ®) a medicine that treats pulmonary arterial hypertension and chronic-thromboembolic pulmonary hypertension.
Ask your healthcare provider or pharmacist if you are not sure if any of your medicines are nitrates or guanylate cyclase stimulators, such as riociguat.
(See “ Who Should Not Take Tadalafil Tablets?” )
Tell all of your healthcare providers that you take Tadalafil Tablets. If you need emergency medical care for a heart problem, it will be important for your healthcare provider to know when you last took Tadalafil Tablets.
After taking a single tablet, some of the active ingredient of Tadalafil Tablets remains in your body for more than 2 days.The active ingredient can remain longer if you have problems with your kidneys or liver, or you are taking certain other medications (see “ Can Other Medicines Affect Tadalafil Tablets?” ).
Stop sexual activity and get medical help right away if you get symptoms such as chest pain, dizziness, or nausea during sex. Sexual activity can put an extra strain on your heart, especially if your heart is already weak from a heart attack or heart disease.
See also “ What Are The Possible Side Effects Of Tadalafil Tablets?”
What Are Tadalafil Tablets?
Tadalafil Tablets are a prescription medicine taken by mouth for the treatment of:
- men with erectile dysfunction (ED)
- men with symptoms of benign prostatic hyperplasia (BPH)
- men with both ED and BPH
Tadalafil Tablets for the Treatment of ED
ED is a condition where the penis does not fill with enough blood to harden and expand when a man is sexually excited, or when he cannot keep an erection. A man who has trouble getting or keeping an erection should see his healthcare provider for help if the condition bothers him. Tadalafil Tablets help increase blood flow to the penis and may help men with ED get and keep an erection satisfactory for sexual activity. Once a man has completed sexual activity, blood flow to his penis decreases, and his erection goes away.
Some form of sexual stimulation is needed for an erection to happen with Tadalafil Tablets.
Tadalafil Tablets do not:
- cure ED
- increase a man's sexual desire
- protect a man or his partner from sexually transmitted diseases, including HIV. Speak to your healthcare provider about ways to guard against sexually transmitted diseases.
- serve as a male form of birth control
Tadalafil Tablets are only for men over the age of 18, including men with diabetes or who have undergone prostatectomy.
Tadalafil Tablets for the Treatment of Symptoms of BPH
BPH is a condition that happens in men, where the prostate gland enlarges which can cause urinary symptoms.
Tadalafil Tablets for the Treatment of ED and Symptoms of BPH
ED and symptoms of BPH may happen in the same person and at the same time. Men who have both ED and symptoms of BPH may take Tadalafil Tablets for the treatment of both conditions.
Tadalafil Tablets are not for women or children.
Tadalafil Tablets must be used only under a healthcare provider's care.
Who Should Not Take Tadalafil Tablets?
Do not take Tadalafil Tablets if you:
- take any medicines called “nitrates”.
- use recreational drugs called “poppers” like amyl nitrite and butyl nitrite. (See “ What Is The Most Important Information I Should Know About Tadalafil Tablets?” )
- take any medicines called guanylate cyclase stimulators, such as riociguat.
-
are allergic to Tadalafil Tablets or ADCIRCA®
, or any of its ingredients.See the end of this leaflet for a complete list of ingredients in Tadalafil Tablets. Symptoms of an allergic reaction may include:
- rash
- hives
- swelling of the lips, tongue, or throat
- difficulty breathing or swallowing
Call your healthcare provider or get help right away if you have any of the symptoms of an allergic reaction listed above.
What Should I Tell My Healthcare Provider Before Taking Tadalafil Tablets?
Tadalafil Tablets are not right for everyone. Only your healthcare provider and you can decide if Tadalafil Tablets are right for you.Before taking Tadalafil Tablets, tell your healthcare provider about all your medical problems, including if you:
- have heart problemssuch as angina, heart failure, irregular heartbeats, or have had a heart attack. Ask your healthcare provider if it is safe for you to have sexual activity. You should not take Tadalafil Tablets if your healthcare provider has told you not to have sexual activity because of your health problems.
- have pulmonary hypertension
- have low blood pressure orhave high blood pressure that is not controlled
- have had a stroke
- have liver problems
- have kidney problems or require dialysis
- have retinitis pigmentosa,a rare genetic (runs in families) eye disease
- have ever had severe vision loss, including a condition called NAION
- have stomach ulcers
- have a bleeding problem
- have a deformed penis shapeor Peyronie's disease
- have had an erection that lasted more than 4 hours
- have blood cell problemssuch as sickle cell anemia, multiple myeloma, or leukemia
Can Other Medicines Affect Tadalafil Tablets?
Tell your healthcare provider about all the medicines you take including prescription and non-prescription medicines, vitamins, and herbal supplements. Tadalafil Tablets and other medicines may affect each other. Always check with your healthcare provider before starting or stopping any medicines. Especially tell your healthcare provider if you take any of the following*:
- medicines called nitrates (see “ What Is The Most Important Information I Should Know About Tadalafil Tablets?” )
- medicines called guanylate cyclase stimulators, such as riociguat (Adempas ®), used to treat pulmonary hypertension
- medicines called alpha blockers. These include Hytrin ® (terazosin HCl), Flomax ® (tamsulosin HCl), Cardura ® (doxazosin mesylate), Minipress ® (prazosin HCl), Uroxatral ® (alfuzosin HCl), Jalyn ®(dutasteride and tamsulosin HCl) or Rapaflo ®(silodosin). Alpha-blockers are sometimes prescribed for prostate problems or high blood pressure. If Tadalafil Tablets are taken with certain alpha blockers, your blood pressure could suddenly drop. You could get dizzy or faint.
- other medicines to treat high blood pressure (hypertension)
- medicines called HIV protease inhibitors, such as ritonavir (Norvir ®, Kaletra ®)
- some types of oral antifungals such as ketoconazole (Nizoral ®), itraconazole (Sporanox ®)
- some types of antibiotics such as clarithromycin (Biaxin ®), telithromycin (Ketek ®), erythromycin (several brand names exist. Please consult your healthcare provider to determine if you are taking this medicine).
- other medicines or treatments for ED.
- Tadalafil Tablets are also marketed as ADCIRCA for the treatment of pulmonary arterial hypertension. Do not take both Tadalafil Tablets and ADCIRCA. Do not take sildenafil citrate (Revatio ®) with Tadalafil Tablets.
How Should I Take Tadalafil Tablets?
- Take Tadalafil Tablets exactly as your healthcare provider prescribes it. Your healthcare provider will prescribe the dose that is right for you.
- Some men can only take a low dose of Tadalafil Tablets or may have to take it less often, because of medical conditions or medicines they take.
- Do not change your dose or the way you take Tadalafil Tablets without talking to your healthcare provider. Your healthcare provider may lower or raise your dose, depending on how your body reacts to Tadalafil Tablets and your health condition.
- Tadalafil Tablets may be taken with or without meals.
- If you take too much Tadalafil Tablets, call your healthcare provider or emergency room right away.
How Should I Take Tadalafil Tablets for Symptoms of BPH?
For symptoms of BPH, Tadalafil Tablets are taken once daily.
- Do not take Tadalafil Tablets more than one time each day.
- Take one Tadalafil Tablet every day at about the same time of day.
- If you miss a dose, you may take it when you remember but do not take more than one dose per day.
How Should I Take Tadalafil Tablets for ED?
For ED, there are two ways to take Tadalafil Tablets - either for use as needed OR for use once daily.
Tadalafil Tablets for use as needed:
- Do not take Tadalafil Tablets more than one time each day.
- Take one Tadalafil Tablet before you expect to have sexual activity. You may be able to have sexual activity at 30 minutes after taking Tadalafil Tablets and up to 36 hours after taking it. You and your healthcare provider should consider this in deciding when you should take Tadalafil Tablets before sexual activity. Some form of sexual stimulation is needed for an erection to happen with Tadalafil Tablets.
- Your healthcare provider may change your dose of Tadalafil Tablets depending on how you respond to the medicine, and on your health condition.
OR
Tadalafil Tablets for once daily use are a lower dose you take every day.
- Do not take Tadalafil Tablets more than one time each day.
- Take one Tadalafil Tablet every day at about the same time of day. You may attempt sexual activity at any time between doses.
- If you miss a dose, you may take it when you remember but do not take more than one dose per day.
- Some form of sexual stimulation is needed for an erection to happen with Tadalafil Tablets.
- Your healthcare provider may change your dose of Tadalafil Tablets depending on how you respond to the medicine, and on your health condition.
How Should I Take Tadalafil Tablets for Both ED and the Symptoms of BPH?
For both ED and the symptoms of BPH, Tadalafil Tablets are taken once daily.
- Do not take Tadalafil Tablets more than one time each day.
- Take one Tadalafil Tablet every day at about the same time of day. You may attempt sexual activity at any time between doses.
- If you miss a dose, you may take it when you remember but do not take more than one dose per day.
- Some form of sexual stimulation is needed for an erection to happen with Tadalafil Tablets.
What Should I Avoid While Taking Tadalafil Tablets?
- Do not use other ED medicines or ED treatments while taking Tadalafil Tablets.
- Do not drink too much alcohol when taking Tadalafil Tablets (for example, 5 glasses of wine or 5 shots of whiskey). Drinking too much alcohol can increase your chances of getting a headache or getting dizzy, increasing your heart rate, or lowering your blood pressure.
What Are The Possible Side Effects Of Tadalafil Tablets?
See “ What Is The Most Important Information I Should Know About Tadalafil Tablets?”
The most common side effects with Tadalafil Tablets are:headache, indigestion, back pain, muscle aches, flushing, and stuffy or runny nose. These side effects usually go away after a few hours. Men who get back pain and muscle aches usually get it 12 to 24 hours after taking Tadalafil Tablets. Back pain and muscle aches usually go away within 2 days.
Call your healthcare provider if you get any side effect that bothers you or one that does not go away.
Uncommon side effects include:
An erection that won't go away (priapism). If you get an erection that lasts more than 4 hours, get medical help right away. Priapism must be treated as soon as possible or lasting damage can happen to your penis, including the inability to have erections.
Color vision changes, such as seeing a blue tinge (shade) to objects or having difficulty telling the difference between the colors blue and green.
In rare instances, men taking PDE5 inhibitors (oral erectile dysfunction medicines, including Tadalafil Tablets) reported a sudden decrease or loss of vision in one or both eyes. It is uncertain whether PDE5 inhibitors directly cause the vision loss. If you experience sudden decrease or loss of vision, stop taking PDE5 inhibitors, including Tadalafil Tablets, and call a healthcare provider right away.
Sudden loss or decrease in hearing, sometimes with ringing in the ears and dizziness, has been rarely reported in people taking PDE5 inhibitors, including Tadalafil Tablets. It is not possible to determine whether these events are related directly to the PDE5 inhibitors, to other diseases or medications, to other factors, or to a combination of factors. If you experience these symptoms, stop taking Tadalafil Tablets and contact a healthcare provider right away.
These are not all the possible side effects of Tadalafil Tablets. For more information, ask your healthcare provider or pharmacist.
How Should I Store Tadalafil Tablets?
Store Tadalafil Tablets at room temperature between 59° and 86°F (15° and 30°C).
Keep Tadalafil Tablets and all medicines out of the reach of children.
General Information About Tadalafil Tablets:
Medicines are sometimes prescribed for conditions other than those described in patient information leaflets. Do not use Tadalafil Tablets for a condition for which it was not prescribed. Do not give Tadalafil Tablets to other people, even if they have the same symptoms that you have. It may harm them.
This is a summary of the most important information about Tadalafil Tablets. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about Tadalafil Tablets that is written for health providers.
What Are The Ingredients In Tadalafil Tablets?
Active Ingredient: tadalafil, USP
Inactive Ingredients: croscarmellose sodium, hydroxypropyl cellulose, hypromellose, iron oxide, lactose monohydrate, magnesium stearate, microcrystalline cellulose, sodium lauryl sulfate, talc, titanium dioxide, and triacetin.
This Patient Information has been approved by the U.S. Food and Drug Administration
Rx only
*Trademarks are the property of their respective owners.
34120062611A Revised: January 2024
Manufactured for:
TWi Pharmaceuticals USA, Inc.
Paramus, NJ 07652
Section 43683-2 (43683-2)
Warnings and Precautions ( 5.4) 05/2017
5.9 Alcohol
Patients should be made aware that both alcohol and Tadalafil Tablets, a PDE5 inhibitor, act as mild vasodilators. When mild vasodilators are taken in combination, blood-pressure-lowering effects of each individual compound may be increased. Therefore, physicians should inform patients that substantial consumption of alcohol (e.g., 5 units or greater) in combination with Tadalafil Tablets can increase the potential for orthostatic signs and symptoms, including increase in heart rate, decrease in standing blood pressure, dizziness, and headache [see Clinical Pharmacology ( 12.2)] .
16.2 Storage
Store at 25°C (77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Keep out of reach of children.
17.9 Alcohol
Patients should be made aware that both alcohol and Tadalafil Tablets, a PDE5 inhibitor, act as mild vasodilators. When mild vasodilators are taken in combination, blood-pressure-lowering effects of each individual compound may be increased. Therefore, physicians should inform patients that substantial consumption of alcohol (e.g., 5 units or greater) in combination with Tadalafil Tablets can increase the potential for orthostatic signs and symptoms, including increase in heart rate, decrease in standing blood pressure, dizziness, and headache [see Warnings and Precautions ( 5.9), Drug Interactions ( 7.1), and Clinical Pharmacology ( 12.2)] .
4.1 Nitrates
Administration of Tadalafil Tablets to patients who are using any form of organic nitrate, either regularly and/or intermittently, is contraindicated. In clinical pharmacology studies, Tadalafil Tablets were shown to potentiate the hypotensive effect of nitrates [see Clinical Pharmacology ( 12.2)] .
10 Overdosage (10 OVERDOSAGE)
Single doses up to 500 mg have been given to healthy subjects, and multiple daily doses up to 100 mg have been given to patients. Adverse events were similar to those seen at lower doses. In cases of overdose, standard supportive measures should be adopted as required. Hemodialysis contributes negligibly to tadalafil elimination.
17.1 Nitrates
Physicians should discuss with patients the contraindication of Tadalafil Tablets with regular and/or intermittent use of organic nitrates. Patients should be counseled that concomitant use of Tadalafil Tablets with nitrates could cause blood pressure to suddenly drop to an unsafe level, resulting in dizziness, syncope, or even heart attack or stroke.
Physicians should discuss with patients the appropriate action in the event that they experience anginal chest pain requiring nitroglycerin following intake of Tadalafil Tablets. In such a patient, who has taken Tadalafil Tablets, where nitrate administration is deemed medically necessary for a life-threatening situation, at least 48 hours should have elapsed after the last dose of Tadalafil Tablets before nitrate administration is considered. In such circumstances, nitrates should still only be administered under close medical supervision with appropriate hemodynamic monitoring. Therefore, patients who experience anginal chest pain after taking Tadalafil Tablets should seek immediate medical attention [see Contraindications ( 4.1) and Warnings and Precautions ( 5.1)] .
17.6 Priapism
There have been rare reports of prolonged erections greater than 4 hours and priapism (painful erections greater than 6 hours in duration) for this class of compounds. Priapism, if not treated promptly, can result in irreversible damage to the erectile tissue. Physicians should advise patients who have an erection lasting greater than 4 hours, whether painful or not, to seek emergency medical attention.
8.2 Lactation
Risk Summary
Tadalafil Tablets are not indicated for use in females.
There is no information on the presence of tadalafil and/or metabolites in human milk, the effects on the breastfed child, or the effects on milk production. Tadalafil and/or its metabolites are present in the milk of lactating rats at concentrations approximately 2.4-fold greater than found in the plasma.
11 Description (11 DESCRIPTION)
Tadalafil is a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type 5 (PDE5). Tadalafil has the empirical formula C 22H 19N 3O 4representing a molecular weight of 389.41. The structural formula is:
The chemical designation is pyrazino[1´,2´:1,6]pyrido[3,4-b]indole-1,4-dione, 6-(1,3-benzodioxol-5-yl)-2,3,6,7,12,12a-hexahydro-2-methyl-, (6R,12aR)-. It is a crystalline solid that is practically insoluble in water and very slightly soluble in ethanol.
Tadalafil Tablets, USP are available as round, pale yellow to yellow tablets for oral administration. Each tablet contains 2.5, 5, 10, or 20 mg of tadalafil and the following inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, hypromellose, iron oxide, lactose monohydrate, magnesium stearate, microcrystalline cellulose, sodium lauryl sulfate, talc, titanium dioxide, and triacetin.
16.1 How Supplied
Tadalafil Tablets, USP are supplied as follows:
Four strengths of round, pale yellow to yellow tablets are available in different sizes, and supplied in the following package sizes:
Bottle of 7 Tablets NDC: 80425-0443-01
2.5 Use With Food (2.5 Use with Food)
Tadalafil Tablets may be taken without regard to food.
8.4 Pediatric Use
Tadalafil Tablets are not indicated for use in pediatric patients. Safety and efficacy in patients below the age of 18 years have not been established.
Juvenile Animal Study
No adverse effects were observed in a study in which tadalafil was administered orally at doses of 60, 200, and 1000 mg/kg/day to juvenile rats on postnatal days 14 to 90. The highest plasma tadalafil exposures (AUC) achieved were approximately 10-fold that observed at the MRHD.
Additional information describing a clinical study in which efficacy was not demonstrated is approved for Eli Lilly and Company’s CIALIS (tadalafil) tablets. However, due to Eli Lilly and Company’s marketing exclusivity rights, this drug product is not labeled with that pediatric information.
8.5 Geriatric Use
Of the total number of subjects in ED clinical studies of tadalafil, approximately 19 percent were 65 and over, while approximately 2 percent were 75 and over. Of the total number of subjects in BPH clinical studies of tadalafil (including the ED/BPH study), approximately 40 percent were over 65, while approximately 10 percent were 75 and over. In these clinical trials, no overall differences in efficacy or safety were observed between older (>65 and ≥75 years of age) and younger subjects (≤65 years of age). However, in placebo-controlled studies with Tadalafil Tablets for use as needed for ED, diarrhea was reported more frequently in patients 65 years of age and older who were treated with Tadalafil Tablets (2.5% of patients) [see Adverse Reactions ( 6.1)] . No dose adjustment is warranted based on age alone. However, a greater sensitivity to medications in some older individuals should be considered. [see Clinical Pharmacology ( 12.3)] .
5.1 Cardiovascular
Physicians should consider the cardiovascular status of their patients, since there is a degree of cardiac risk associated with sexual activity. Therefore, treatments for erectile dysfunction, including Tadalafil Tablets, should not be used in men for whom sexual activity is inadvisable as a result of their underlying cardiovascular status. Patients who experience symptoms upon initiation of sexual activity should be advised to refrain from further sexual activity and seek immediate medical attention.
Physicians should discuss with patients the appropriate action in the event that they experience anginal chest pain requiring nitroglycerin following intake of Tadalafil Tablets. In such a patient, who has taken Tadalafil Tablets, where nitrate administration is deemed medically necessary for a life-threatening situation, at least 48 hours should have elapsed after the last dose of Tadalafil Tablets before nitrate administration is considered. In such circumstances, nitrates should still only be administered under close medical supervision with appropriate hemodynamic monitoring. Therefore, patients who experience anginal chest pain after taking Tadalafil Tablets should seek immediate medical attention. [see Contraindications ( 4.1) and Patient Counseling Information ( 17.1)] .
Patients with left ventricular outflow obstruction, (e.g., aortic stenosis and idiopathic hypertrophic subaortic stenosis) can be sensitive to the action of vasodilators, including PDE5 inhibitors.
The following groups of patients with cardiovascular disease were not included in clinical safety and efficacy trials for Tadalafil Tablets, and therefore until further information is available, Tadalafil Tablets are not recommended for the following groups of patients:
- myocardial infarction within the last 90 days
- unstable angina or angina occurring during sexual intercourse
- New York Heart Association Class 2 or greater heart failure in the last 6 months
- uncontrolled arrhythmias, hypotension (<90/50 mm Hg), or uncontrolled hypertension
- stroke within the last 6 months.
As with other PDE5 inhibitors, tadalafil has mild systemic vasodilatory properties that may result in transient decreases in blood pressure. In a clinical pharmacology study, tadalafil 20 mg resulted in a mean maximal decrease in supine blood pressure, relative to placebo, of 1.6/0.8 mm Hg in healthy subjects [see Clinical Pharmacology ( 12.2)] . While this effect should not be of consequence in most patients, prior to prescribing Tadalafil Tablets, physicians should carefully consider whether their patients with underlying cardiovascular disease could be affected adversely by such vasodilatory effects. Patients with severely impaired autonomic control of blood pressure may be particularly sensitive to the actions of vasodilators, including PDE5 inhibitors.
4 Contraindications (4 CONTRAINDICATIONS)
- Administration of Tadalafil Tablets to patients using any form of organic nitrate is contraindicated. Tadalafil Tablets were shown to potentiate the hypotensive effect of nitrates ( 4.1).
- History of known serious hypersensitivity reaction to Tadalafil Tablets or ADCIRCA®( 4.2).
- Administration with guanylate cyclase (GC) stimulators, such as riociguat ( 4.3).
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions (≥2%) include headache, dyspepsia, back pain, myalgia, nasal congestion, flushing, and pain in limb ( 6.1).
To report SUSPECTED ADVERSE REACTIONS, contact TWi Pharmaceuticals, Inc. at 1-844-518-2989 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
7 Drug Interactions (7 DRUG INTERACTIONS)
- Tadalafil Tablets can potentiate the hypotensive effects of nitrates, alpha-blockers, antihypertensives or alcohol ( 7.1).
- CYP3A4 inhibitors (e.g. ketoconazole, ritonavir) increase Tadalafil Tablets exposure (
2.7,
5.10,
7.2) requiring dose adjustment:
- Tadalafil Tablets for use as needed: no more than 10 mg every 72 hours
- Tadalafil Tablets for once daily use: dose not to exceed 2.5 mg
- CYP3A4 inducers (e.g. rifampin) decrease Tadalafil Tablets exposure ( 7.2).
8.7 Renal Impairment
In clinical pharmacology studies using single-dose tadalafil (5 to 10 mg), tadalafil exposure (AUC) doubled in subjects with creatinine clearance 30 to 80 mL/min. In subjects with end-stage renal disease on hemodialysis, there was a two-fold increase in C maxand 2.7- to 4.8-fold increase in AUC following single-dose administration of 10 or 20 mg tadalafil. Exposure to total methylcatechol (unconjugated plus glucuronide) was 2- to 4-fold higher in subjects with renal impairment, compared to those with normal renal function. Hemodialysis (performed between 24 and 30 hours post-dose) contributed negligibly to tadalafil or metabolite elimination. In a clinical pharmacology study (N=28) at a dose of 10 mg, back pain was reported as a limiting adverse event in male patients with creatinine clearance 30 to 50 mL/min. At a dose of 5 mg, the incidence and severity of back pain was not significantly different than in the general population. In patients on hemodialysis taking 10- or 20-mg tadalafil, there were no reported cases of back pain. [see Dosage and Administration ( 2.6) and Warnings and Precautions ( 5.7)] .
1.4 Limitation of Use
If Tadalafil Tablets are used with finasteride to initiate BPH treatment, such use is recommended for up to 26 weeks because the incremental benefit of Tadalafil Tablets decreases from 4 weeks until 26 weeks, and the incremental benefit of Tadalafil Tablets beyond 26 weeks is unknown [see Clinical Studies ( 14.3)] .
12.3 Pharmacokinetics
Over a dose range of 2.5 to 20 mg, tadalafil exposure (AUC) increases proportionally with dose in healthy subjects. Steady-state plasma concentrations are attained within 5 days of once per day dosing and exposure is approximately 1.6-fold greater than after a single dose. Mean tadalafil concentrations measured after the administration of a single oral dose of 20 mg and single and once daily multiple doses of 5 mg, from a separate study, ( see Figure 4) to healthy male subjects are depicted in Figure 4.
Figure 4: Plasma tadalafil concentrations (mean ± SD) following a single 20-mg tadalafil dose and single and once daily multiple doses of 5 mg
5.3 Prolonged Erection
There have been rare reports of prolonged erections greater than 4 hours and priapism (painful erections greater than 6 hours in duration) for this class of compounds. Priapism, if not treated promptly, can result in irreversible damage to the erectile tissue. Patients who have an erection lasting greater than 4 hours, whether painful or not, should seek emergency medical attention.
Tadalafil Tablets should be used with caution in patients who have conditions that might predispose them to priapism (such as sickle cell anemia, multiple myeloma, or leukemia), or in patients with anatomical deformation of the penis (such as angulation, cavernosal fibrosis, or Peyronie's disease).
5.4 Effects On the Eye (5.4 Effects on the Eye)
Physicians should advise patients to stop use of all phosphodiesterase type 5 (PDE5) inhibitors, including Tadalafil Tablets, and seek medical attention in the event of a sudden loss of vision in one or both eyes. Such an event may be a sign of non-arteritic anterior ischemic optic neuropathy (NAION), a rare condition and a cause of decreased vision, including permanent loss of vision, that has been reported rarely postmarketing in temporal association with the use of all PDE5 inhibitors. Based on published literature, the annual incidence of NAION is 2.5-11.8 cases per 100,000 in males aged ≥50.Physicians should advise patients to stop use of all phosphodiesterase type 5 (PDE5) inhibitors, including Tadalafil Tablets, and seek medical attention in the event of a sudden loss of vision in one or both eyes. Such an event may be a sign of non-arteritic anterior ischemic optic neuropathy (NAION), a rare condition and a cause of decreased vision, including permanent loss of vision, that has been reported rarely postmarketing in temporal association with the use of all PDE5 inhibitors. Based on published literature, the annual incidence of NAION is 2.5-11.8 cases per 100,000 in males aged ≥50.
An observational case-crossover study evaluated the risk of NAION when PDE5 inhibitor use, as a class, occurred immediately before NAION onset (within 5 half-lives), compared to PDE5 inhibitor use in a prior time period. The results suggest an approximate 2-fold increase in the risk of NAION, with a risk estimate of 2.15 (95% CI 1.06, 4.34). A similar study reported a consistent result, with a risk estimate of 2.27 (95% CI 0.99, 5.20). Other risk factors for NAION, such as the presence of "crowded" optic disc, may have contributed to the occurrence of NAION in these studies.
Neither the rare postmarketing reports, nor the association of PDE5 inhibitor use and NAION in the observational studies, substantiate a causal relationship between PDE5 inhibitor use and NAION [see Adverse Reactions ( 6.2)].
Physicians should consider whether their patients with underlying NAION risk factors could be adversely affected by use of PDE5 inhibitors. Individuals who have already experienced NAION are at increased risk of NAION recurrence. Therefore, PDE5 inhibitors, including Tadalafil Tablets, should be used with caution in these patients and only when the anticipated benefits outweigh the risks. Individuals with "crowded" optic disc are also considered at greater risk for NAION compared to the general population; however, evidence is insufficient to support screening of prospective users of PDE5 inhibitors, including Tadalafil Tablets, for this uncommon condition.Physicians should consider whether their patients with underlying NAION risk factors could be adversely affected by use of PDE5 inhibitors. Individuals who have already experienced NAION are at increased risk of NAION recurrence. Therefore, PDE5 inhibitors, including Tadalafil Tablets, should be used with caution in these patients and only when the anticipated benefits outweigh the risks. Individuals with "crowded" optic disc are also considered at greater risk for NAION compared to the general population; however, evidence is insufficient to support screening of prospective users of PDE5 inhibitors, including Tadalafil Tablets, for this uncommon condition.
Patients with known hereditary degenerative retinal disorders, including retinitis pigmentosa, were not included in the clinical trials, and use in these patients is not recommended.
8.6 Hepatic Impairment
In clinical pharmacology studies, tadalafil exposure (AUC) in subjects with mild or moderate hepatic impairment (Child-Pugh Class A or B) was comparable to exposure in healthy subjects when a dose of 10 mg was administered. There are no available data for doses higher than 10 mg of tadalafil in patients with hepatic impairment. Insufficient data are available for subjects with severe hepatic impairment (Child-Pugh Class C). [see Dosage and Administration ( 2.6) and Warnings and Precautions ( 5.8)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
Tadalafil Tablets are a phosphodiesterase 5 (PDE5) inhibitor indicated for the treatment of:
- erectile dysfunction (ED) ( 1.1)
- the signs and symptoms of benign prostatic hyperplasia (BPH) ( 1.2)
- ED and the signs and symptoms of BPH (ED/BPH) ( 1.3)
If Tadalafil Tablets are used with finasteride to initiate BPH treatment, such use is recommended for up to 26 weeks ( 1.4).
5.5 Sudden Hearing Loss
Physicians should advise patients to stop taking PDE5 inhibitors, including Tadalafil Tablets, and seek prompt medical attention in the event of sudden decrease or loss of hearing. These events, which may be accompanied by tinnitus and dizziness, have been reported in temporal association to the intake of PDE5 inhibitors, including Tadalafil Tablets. It is not possible to determine whether these events are related directly to the use of PDE5 inhibitors or to other factors [see Adverse Reactions ( 6.1, 6.2)] .
1.1 Erectile Dysfunction
Tadalafil Tablets are indicated for the treatment of erectile dysfunction (ED).
12.1 Mechanism of Action
Penile erection during sexual stimulation is caused by increased penile blood flow resulting from the relaxation of penile arteries and corpus cavernosal smooth muscle. This response is mediated by the release of nitric oxide (NO) from nerve terminals and endothelial cells, which stimulates the synthesis of cGMP in smooth muscle cells. Cyclic GMP causes smooth muscle relaxation and increased blood flow into the corpus cavernosum. The inhibition of phosphodiesterase type 5 (PDE5) enhances erectile function by increasing the amount of cGMP. Tadalafil inhibits PDE5. Because sexual stimulation is required to initiate the local release of nitric oxide, the inhibition of PDE5 by tadalafil has no effect in the absence of sexual stimulation.
The effect of PDE5 inhibition on cGMP concentration in the corpus cavernosum and pulmonary arteries is also observed in the smooth muscle of the prostate, the bladder and their vascular supply. The mechanism for reducing BPH symptoms has not been established.
Studies in vitrohave demonstrated that tadalafil is a selective inhibitor of PDE5. PDE5 is found in the smooth muscle of the corpus cavernosum, prostate, and bladder as well as in vascular and visceral smooth muscle, skeletal muscle, urethra, platelets, kidney, lung, cerebellum, heart, liver, testis, seminal vesicle, and pancreas.
In vitrostudies have shown that the effect of tadalafil is more potent on PDE5 than on other phosphodiesterases. These studies have shown that tadalafil is >10,000-fold more potent for PDE5 than for PDE1, PDE2, PDE4, and PDE7 enzymes, which are found in the heart, brain, blood vessels, liver, leukocytes, skeletal muscle, and other organs. Tadalafil is >10,000-fold more potent for PDE5 than for PDE3, an enzyme found in the heart and blood vessels. Additionally, tadalafil is 700-fold more potent for PDE5 than for PDE6, which is found in the retina and is responsible for phototransduction. Tadalafil is >9,000-fold more potent for PDE5 than for PDE8, PDE9, and PDE10. Tadalafil is 14-fold more potent for PDE5 than for PDE11A1 and 40-fold more potent for PDE5 than for PDE11A4, two of the four known forms of PDE11. PDE11 is an enzyme found in human prostate, testes, skeletal muscle and in other tissues (e.g., adrenal cortex). In vitro, tadalafil inhibits human recombinant PDE11A1 and, to a lesser degree, PDE11A4 activities at concentrations within the therapeutic range. The physiological role and clinical consequence of PDE11 inhibition in humans have not been defined.
17.8 Sudden Hearing Loss
Physicians should advise patients to stop taking PDE5 inhibitors, including Tadalafil Tablets, and seek prompt medical attention in the event of sudden decrease or loss of hearing. These events, which may be accompanied by tinnitus and dizziness, have been reported in temporal association to the intake of PDE5 inhibitors, including Tadalafil Tablets. It is not possible to determine whether these events are related directly to the use of PDE5 inhibitors or to other factors [see Adverse Reactions ( 6.1, 6.2)] .
5.12 Effects On Bleeding (5.12 Effects on Bleeding)
Studies in vitrohave demonstrated that tadalafil is a selective inhibitor of PDE5. PDE5 is found in platelets. When administered in combination with aspirin, tadalafil 20 mg did not prolong bleeding time, relative to aspirin alone. Tadalafil Tablets have not been administered to patients with bleeding disorders or significant active peptic ulceration. Although Tadalafil Tablets have not been shown to increase bleeding times in healthy subjects, use in patients with bleeding disorders or significant active peptic ulceration should be based upon a careful risk-benefit assessment and caution.
17.7 Sudden Loss of Vision
Physicians should advise patients to stop use of all PDE5 inhibitors, including Tadalafil Tablets, and seek medical attention in the event of a sudden loss of vision in one or both eyes. Such an event may be a sign of non-arteritic anterior ischemic optic neuropathy (NAION), a cause of decreased vision, including possible permanent loss of vision, that has been reported rarely postmarketing in temporal association with the use of all PDE5 inhibitors. Physicians should discuss with patients the increased risk of NAION among the general population in patients with a "crowded" optic disc, although evidence is insufficient to support screening of prospective users of PDE5 inhibitors, including Tadalafil Tablets, for this uncommon condition [see Warnings and Precautions ( 5.4) and Adverse Reactions ( 6.2)] .
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Evaluation of erectile dysfunction and BPH should include an appropriate medical assessment to identify potential underlying causes, as well as treatment options.
Before prescribing Tadalafil Tablets, it is important to note the following:
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Do not split Tadalafil Tablets; entire dose should be taken.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Four strengths of round, pale yellow to yellow tablets are available in different sizes:
2.5 mg tablets debossed with “43”
5 mg tablets debossed with “44”
10 mg tablets debossed with “45”
20 mg tablets debossed with “47”
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of Tadalafil Tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These events have been chosen for inclusion either due to their seriousness, reporting frequency, lack of clear alternative causation, or a combination of these factors.
Cardiovascular and Cerebrovascular — Serious cardiovascular events, including myocardial infarction, sudden cardiac death, stroke, chest pain, palpitations, and tachycardia, have been reported postmarketing in temporal association with the use of tadalafil. Most, but not all, of these patients had preexisting cardiovascular risk factors. Many of these events were reported to occur during or shortly after sexual activity, and a few were reported to occur shortly after the use of Tadalafil Tablets without sexual activity. Others were reported to have occurred hours to days after the use of Tadalafil Tablets and sexual activity. It is not possible to determine whether these events are related directly to Tadalafil Tablets, to sexual activity, to the patient's underlying cardiovascular disease, to a combination of these factors, or to other factors [see Warnings and Precautions ( 5.1)] .
Body as a Whole — hypersensitivity reactions including urticaria, Stevens-Johnson syndrome, and exfoliative dermatitis
Nervous — migraine, seizure and seizure recurrence, transient global amnesia
Ophthalmologic — visual field defect, retinal vein occlusion, retinal artery occlusion
Non-arteritic anterior ischemic optic neuropathy (NAION), a cause of decreased vision including permanent loss of vision, has been reported rarely postmarketing in temporal association with the use of PDE5 inhibitors, including Tadalafil Tablets. Most, but not all, of these patients had underlying anatomic or vascular risk factors for development of NAION, including but not necessarily limited to: low cup to disc ratio (“crowded disc”), age over 50, diabetes, hypertension, coronary artery disease, hyperlipidemia, and smoking. [see Warnings and Precautions ( 5.4)] .
Otologic — Cases of sudden decrease or loss of hearing have been reported postmarketing in temporal association with the use of PDE5 inhibitors, including Tadalafil Tablets. In some of the cases, medical conditions and other factors were reported that may have also played a role in the otologic adverse events. In many cases, medical follow-up information was limited. It is not possible to determine whether these reported events are related directly to the use of Tadalafil Tablets, to the patient's underlying risk factors for hearing loss, a combination of these factors, or to other factors [see Warnings and Precautions ( 5.5)] .
Urogenital — priapism [see Warnings and Precautions ( 5.3)] .
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Hepatic Impairment ( 2.6, 5.8, 8.6):
- Mild or Moderate: Dosage adjustment may be needed.
- Severe: Use is not recommended.
Renal Impairment ( 2.6, 5.7, 8.7):
- Patients with creatinine clearance 30 to 50 mL/min: Dosage adjustment may be needed.
- Patients with creatinine clearance less than 30 mL/min or on hemodialysis: For use as needed: Dose should not exceed 5 mg every 72 hours. Once daily use is not recommended.
4.2 Hypersensitivity Reactions
Tadalafil Tablets are contraindicated in patients with a known serious hypersensitivity to tadalafil or ADCIRCA®. Hypersensitivity reactions have been reported, including Stevens-Johnson syndrome and exfoliative dermatitis [see Adverse Reactions ( 6.2)] .
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Tadalafil was administered to over 9000 men during clinical trials worldwide. In trials of Tadalafil Tablets for once daily use, a total of 1434, 905, and 115 were treated for at least 6 months, 1 year, and 2 years, respectively. For Tadalafil Tablets for use as needed, over 1300 and 1000 subjects were treated for at least 6 months and 1 year, respectively.
1.2 Benign Prostatic Hyperplasia
Tadalafil Tablets are indicated for the treatment of the signs and symptoms of benign prostatic hyperplasia (BPH).
17.11 Recommended Administration
Physicians should instruct patients on the appropriate administration of Tadalafil Tablets to allow optimal use.
For Tadalafil Tablets for use as needed in men with ED, patients should be instructed to take one tablet at least 30 minutes before anticipated sexual activity. In most patients, the ability to have sexual intercourse is improved for up to 36 hours.
For Tadalafil Tablets for once daily use in men with ED or ED/BPH, patients should be instructed to take one tablet at approximately the same time every day without regard for the timing of sexual activity. Tadalafil Tablets are effective at improving erectile function over the course of therapy.
For Tadalafil Tablets for once daily use in men with BPH, patients should be instructed to take one tablet at approximately the same time every day.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
“See FDA-approved patient labeling ( Patient Information)”
17.10 Sexually Transmitted Disease
The use of Tadalafil Tablets offers no protection against sexually transmitted diseases. Counseling of patients about the protective measures necessary to guard against sexually transmitted diseases, including Human Immunodeficiency Virus (HIV) should be considered.
17.3 Cardiovascular Considerations
Physicians should consider the potential cardiac risk of sexual activity in patients with preexisting cardiovascular disease. Physicians should advise patients who experience symptoms upon initiation of sexual activity to refrain from further sexual activity and seek immediate medical attention [see Warnings and Precautions ( 5.1)] .
Package Label. Principal Display Panel (PACKAGE LABEL. PRINCIPAL DISPLAY PANEL)
17.2 Guanylate Cyclase (gc) Stimulators (17.2 Guanylate Cyclase (GC) Stimulators)
Physicians should discuss with patients the contraindication of Tadalafil Tablets with any use of a GC stimulator, such as riociguat, for pulmonary arterial hypertension. Patients should be counseled that the concomitant use of Tadalafil Tablets with GC stimulators may cause blood pressure to drop to an unsafe level.
5.6 Alpha Blockers and Antihypertensives (5.6 Alpha-blockers and Antihypertensives)
Physicians should discuss with patients the potential for Tadalafil Tablets to augment the blood-pressure-lowering effect of alpha-blockers and antihypertensive medications [see Drug Interactions ( 7.1) and Clinical Pharmacology ( 12.2)] .
Caution is advised when PDE5 inhibitors are coadministered with alpha-blockers. PDE5 inhibitors, including Tadalafil Tablets, and alpha-adrenergic blocking agents are both vasodilators with blood-pressure-lowering effects. When vasodilators are used in combination, an additive effect on blood pressure may be anticipated. In some patients, concomitant use of these two drug classes can lower blood pressure significantly [see Drug Interactions ( 7.1) and Clinical Pharmacology ( 12.2)] , which may lead to symptomatic hypotension (e.g., fainting). Consideration should be given to the following:
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Animal studies showed vascular inflammation in tadalafil-treated mice, rats, and dogs. In mice and rats, lymphoid necrosis and hemorrhage were seen in the spleen, thymus, and mesenteric lymph nodes at unbound tadalafil exposure of 2- to 33-fold above the human exposure (AUCs) at the MRHD of 20 mg. In dogs, an increased incidence of disseminated arteritis was observed in 1- and 6-month studies at unbound tadalafil exposure of 1- to 54-fold above the human exposure (AUC) at the MRHD of 20 mg. In a 12-month dog study, no disseminated arteritis was observed, but 2 dogs exhibited marked decreases in white blood cells (neutrophils) and moderate decreases in platelets with inflammatory signs at unbound tadalafil exposures of approximately 14- to 18-fold the human exposure at the MRHD of 20 mg. The abnormal blood-cell findings were reversible within 2 weeks after stopping treatment.
14.1 Tadalafil Tablets for Use As Needed for Ed (14.1 Tadalafil Tablets for Use as Needed for ED)
The efficacy and safety of tadalafil in the treatment of erectile dysfunction has been evaluated in 22 clinical trials of up to 24-weeks duration, involving over 4000 patients. Tadalafil Tablets, when taken as needed up to once per day, were shown to be effective in improving erectile function in men with erectile dysfunction (ED).
Tadalafil Tablets were studied in the general ED population in 7 randomized, multicenter, double-blinded, placebo-controlled, parallel-arm design, primary efficacy and safety studies of 12-weeks duration. Two of these studies were conducted in the United States and 5 were conducted in centers outside the US. Additional efficacy and safety studies were performed in ED patients with diabetes mellitus and in patients who developed ED status post bilateral nerve-sparing radical prostatectomy.
In these 7 trials, Tadalafil Tablets were taken as needed, at doses ranging from 2.5 to 20 mg, up to once per day. Patients were free to choose the time interval between dose administration and the time of sexual attempts. Food and alcohol intake were not restricted.
Several assessment tools were used to evaluate the effect of Tadalafil Tablets on erectile function. The 3 primary outcome measures were the Erectile Function (EF) domain of the International Index of Erectile Function (IIEF) and Questions 2 and 3 from Sexual Encounter Profile (SEP). The IIEF is a 4-week recall questionnaire that was administered at the end of a treatment-free baseline period and subsequently at follow-up visits after randomization. The IIEF EF domain has a 30-point total score, where higher scores reflect better erectile function. SEP is a diary in which patients recorded each sexual attempt made throughout the study. SEP Question 2 asks, “Were you able to insert your penis into the partner's vagina?” SEP Question 3 asks, “Did your erection last long enough for you to have successful intercourse?” The overall percentage of successful attempts to insert the penis into the vagina (SEP2) and to maintain the erection for successful intercourse (SEP3) is derived for each patient.
8.3 Females and Males of Reproductive Potential
Infertility
Based on the data from 3 studies in adult males, tadalafil decreased sperm concentrations in the study of 10 mg tadalafil for 6 months and the study of 20 mg tadalafil for 9 months. This effect was not seen in the study of 20 mg tadalafil taken for 6 months. There was no adverse effect of tadalafil 10 mg or 20 mg on mean concentrations of testosterone, luteinizing hormone or follicle stimulating hormone. The clinical significance of the decreased sperm concentrations in the two studies is unknown. There have been no studies evaluating the effect of tadalafil on fertility in men [see Clinical Pharmacology ( 12.2)] .
Based on studies in animals, a decrease in spermatogenesis was observed in dogs, but not in rats [see Nonclinical Toxicology ( 13.1)] .
14.2 Tadalafil Tablets for Once Daily Use for Ed (14.2 Tadalafil Tablets for Once Daily Use for ED)
The efficacy and safety of Tadalafil Tablets for once daily use in the treatment of erectile dysfunction has been evaluated in 2 clinical trials of 12-weeks duration and 1 clinical trial of 24-weeks duration, involving a total of 853 patients. Tadalafil Tablets, when taken once daily, were shown to be effective in improving erectile function in men with erectile dysfunction (ED).
Tadalafil Tablets were studied in the general ED population in 2 randomized, multicenter, double-blinded, placebo-controlled, parallel-arm design, primary efficacy and safety studies of 12- and 24-weeks duration, respectively. One of these studies was conducted in the United States and one was conducted in centers outside the US. An additional efficacy and safety study was performed in ED patients with diabetes mellitus. Tadalafil Tablets were taken once daily at doses ranging from 2.5 to 10 mg. Food and alcohol intake were not restricted. Timing of sexual activity was not restricted relative to when patients took Tadalafil Tablets.
4.3 Concomitant Guanylate Cyclase (gc) Stimulators (4.3 Concomitant Guanylate Cyclase (GC) Stimulators)
Do not use Tadalafil Tablets in patients who are using a GC stimulator, such as riociguat. PDE5 inhibitors, including Tadalafil Tablets, may potentiate the hypotensive effects of GC stimulators.
1.3 Erectile Dysfunction and Benign Prostatic Hyperplasia
Tadalafil Tablets are indicated for the treatment of ED and the signs and symptoms of BPH (ED/BPH).
7.2 Potential for Other Drugs to Affect Tadalafil Tablets
17.4 Concomitant Use With Drugs Which Lower Blood Pressure (17.4 Concomitant Use with Drugs Which Lower Blood Pressure)
5.13 Counseling Patients About Sexually Transmitted Diseases
The use of Tadalafil Tablets offers no protection against sexually transmitted diseases. Counseling patients about the protective measures necessary to guard against sexually transmitted diseases, including Human Immunodeficiency Virus (HIV) should be considered.
14.4 Tadalafil Tablets 5 mg for Once Daily Use for Ed and Bph (14.4 Tadalafil Tablets 5 mg for Once Daily Use for ED and BPH)
The efficacy and safety of Tadalafil Tablets for once daily use for the treatment of ED, and the signs and symptoms of BPH, in patients with both conditions was evaluated in one placebo-controlled, multinational, double-blind, parallel-arm study which randomized 606 patients to receive either Tadalafil Tablets 2.5 mg, 5 mg, for once daily use or placebo. ED severity ranged from mild to severe and BPH severity ranged from moderate to severe. The full study population had a mean age of 63 years (range 45 to 83) and was 93% White, 4% Black, 3% other races; 16% were of Hispanic ethnicity. Patients with multiple co-morbid conditions such as diabetes mellitus, hypertension, and other cardiovascular disease were included.
In this study, the co-primary endpoints were total IPSS and the Erectile Function (EF) domain score of the International Index of Erectile Function (IIEF). One of the key secondary endpoints in this study was Question 3 of the Sexual Encounter Profile diary (SEP3). Timing of sexual activity was not restricted relative to when patients took Tadalafil Tablets.
The efficacy results for patients with both ED and BPH, who received either Tadalafil Tablets 5 mg for once daily use or placebo (N=408) are shown in Tables 21and 22and Figure 8.
Tadalafil Tablets 5 mg for once daily use resulted in statistically significant improvements in the total IPSS and in the EF domain of the IIEF questionnaire. Tadalafil Tablets 5 mg for once daily use also resulted in statistically significant improvement in SEP3. Tadalafil Tablets 2.5 mg did not result in statistically significant improvement in the total IPSS.
| Placebo | Tadalafil Tablets 5 mg | p-value | |
| Total Symptom Score (IPSS) | |||
| (N=193) | (N=206) | ||
| Baseline | 18.2 | 18.5 | |
| Change from Baseline to Week 12 | -3.8 | -6.1 | <.001 |
| EF Domain Score (IIEF EF) | |||
| (N=188) | (N=202) | ||
| Baseline | 15.6 | 16.5 | |
| Endpoint | 17.6 | 22.9 | |
| Change from Baseline to Week 12 | 1.9 | 6.5 | <.001 |
| Placebo | Tadalafil Tablets 5 mg | ||
| (N=187) | (N=199) | p-value | |
| Maintenance of Erection (SEP3) | |||
| Baseline | 36% | 43% | |
| Endpoint | 48% | 72% | |
| Change from Baseline to Week 12 | 12% | 32% | <.001 |
Tadalafil Tablets for once daily use resulted in improvement in the IPSS total score at the first scheduled observation (week 2) and throughout the 12 weeks of treatment ( see Figure 8).
Figure 8: Mean IPSS Changes in ED/BPH Patients by Visit in Study L
In this study, the effect of Tadalafil Tablets 5 mg once daily on Q maxwas evaluated as a safety endpoint. Mean Q maxincreased from baseline in both the treatment and placebo groups (Tadalafil Tablets 5 mg: 1.6 mL/sec, placebo: 1.2 mL/sec); however, these changes were not significantly different between groups.
2.1 Tadalafil Tablets for Use As Needed for Erectile Dysfunction (2.1 Tadalafil Tablets for Use as Needed for Erectile Dysfunction)
- The recommended starting dose of Tadalafil Tablets for use as needed in most patients is 10 mg, taken prior to anticipated sexual activity.
- The dose may be increased to 20 mg or decreased to 5 mg, based on individual efficacy and tolerability. The maximum recommended dosing frequency is once per day in most patients.
- Tadalafil Tablets for use as needed was shown to improve erectile function compared to placebo up to 36 hours following dosing. Therefore, when advising patients on optimal use of Tadalafil Tablets, this should be taken into consideration.
2.2 Tadalafil Tablets for Once Daily Use for Erectile Dysfunction
- The recommended starting dose of Tadalafil Tablets for once daily use is 2.5 mg, taken at approximately the same time every day, without regard to timing of sexual activity.
- The Tadalafil Tablets dose for once daily use may be increased to 5 mg, based on individual efficacy and tolerability.
2.3 Tadalafil Tablets for Once Daily Use for Benign Prostatic Hyperplasia
- The recommended dose of Tadalafil Tablets for once daily use is 5 mg, taken at approximately the same time every day.
- When therapy for BPH is initiated with Tadalafil Tablets and finasteride, the recommended dose of Tadalafil Tablets for once daily use is 5 mg, taken at approximately the same time every day for up to 26 weeks.
5.10 Concomitant Use of Potent Inhibitors of Cytochrome P450 3a4 (cyp3a4) (5.10 Concomitant Use of Potent Inhibitors of Cytochrome P450 3A4 (CYP3A4))
Tadalafil Tablets are metabolized predominantly by CYP3A4 in the liver. The dose of Tadalafil Tablets for use as needed should be limited to 10 mg no more than once every 72 hours in patients taking potent inhibitors of CYP3A4 such as ritonavir, ketoconazole, and itraconazole [see Drug Interactions ( 7.2)] . In patients taking potent inhibitors of CYP3A4 and Tadalafil Tablets for once daily use, the maximum recommended dose is 2.5 mg [see Dosage and Administration ( 2.7)] .
5.11 Combination With Other Pde5 Inhibitors Or Erectile Dysfunction Therapies (5.11 Combination With Other PDE5 Inhibitors or Erectile Dysfunction Therapies)
The safety and efficacy of combinations of Tadalafil Tablets and other PDE5 inhibitors or treatments for erectile dysfunction have not been studied. Inform patients not to take Tadalafil Tablets with other PDE5 inhibitors, including ADCIRCA.
5.2 Potential for Drug Interactions When Taking Tadalafil Tablets for Once Daily Use
Physicians should be aware that Tadalafil Tablets for once daily use provides continuous plasma tadalafil levels and should consider this when evaluating the potential for interactions with medications (e.g., nitrates, alpha-blockers, anti-hypertensives and potent inhibitors of CYP3A4) and with substantial consumption of alcohol [see Drug Interactions ( 7.1, 7.2, 7.3)] .
17.5 Potential for Drug Interactions When Taking Tadalafil Tablets for Once Daily Use
Physicians should discuss with patients the clinical implications of continuous exposure to tadalafil when prescribing Tadalafil Tablets for once daily use, especially the potential for interactions with medications (e.g., nitrates, alpha-blockers, antihypertensives and potent inhibitors of cytochrome P450 3A4) and with substantial consumption of alcohol. [see Dosage and Administration ( 2.7), Warnings and Precautions ( 5.6), Drug Interactions ( 7.1, 7.2), Clinical Pharmacology ( 12.2), and Clinical Studies ( 14.2)] .
14.3 Tadalafil Tablets 5 mg for Once Daily Use for Benign Prostatic Hyperplasia (bph) (14.3 Tadalafil Tablets 5 mg for Once Daily Use for Benign Prostatic Hyperplasia (BPH))
The efficacy and safety of Tadalafil Tablets for once daily use for the treatment of the signs and symptoms of BPH was evaluated in 3 randomized, multinational, double-blinded, placebo-controlled, parallel-design, efficacy and safety studies of 12 weeks duration. Two of these studies were in men with BPH and one study was specific to men with both ED and BPH [see Clinical Studies ( 14.4)] . The first study (Study J) randomized 1058 patients to receive either Tadalafil Tablets 2.5 mg, 5 mg, 10 mg or 20 mg for once daily use or placebo. The second study (Study K) randomized 325 patients to receive either Tadalafil Tablets 5 mg for once daily use or placebo. The full study population was 87% White, 2% Black, 11% other races; 15% was of Hispanic ethnicity. Patients with multiple co-morbid conditions such as diabetes mellitus, hypertension, and other cardiovascular disease were included.
The primary efficacy endpoint in the two studies that evaluated the effect of Tadalafil Tablets for the signs and symptoms of BPH was the International Prostate Symptom Score (IPSS), a four week recall questionnaire that was administered at the beginning and end of a placebo run-in period and subsequently at follow-up visits after randomization. The IPSS assesses the severity of irritative (frequency, urgency, nocturia) and obstructive symptoms (incomplete emptying, stopping and starting, weak stream, and pushing or straining), with scores ranging from 0 to 35; higher numeric scores representing greater severity. Maximum urinary flow rate (Q max), an objective measure of urine flow, was assessed as a secondary efficacy endpoint in Study J and as a safety endpoint in Study K.
The results for BPH patients with moderate to severe symptoms and a mean age of 63.2 years (range 44 to 87) who received either Tadalafil Tablets 5 mg for once daily use or placebo (N=748) in Studies J and K are shown in Table 19and Figures 5and 6, respectively.
In each of these 2 trials, Tadalafil Tablets 5 mg for once daily use resulted in statistically significant improvement in the total IPSS compared to placebo. Mean total IPSS showed a decrease starting at the first scheduled observation (4 weeks) in Study K and remained decreased through 12 weeks.
| Study J | Study K | |||||
| Placebo |
Tadalafil Tablets
5 mg |
Placebo |
Tadalafil Tablets
5 mg |
|||
| (N=205) | (N=205) | p-value | (N=164) | (N=160) | p-value | |
| Total Symptom Score (IPSS) | ||||||
| Baseline | 17.1 | 17.3 | 16.6 | 17.1 | ||
| Change from Baseline to Week 12 | -2.2 | -4.8 | <.001 | -3.6 | -5.6 | .004 |
Figure 5: Mean IPSS Changes in BPH Patients by Visit in Study J
Figure 6: Mean IPSS Changes in BPH Patients by Visit in Study K
In Study J, the effect of Tadalafil Tablets 5 mg once daily on maximum urinary flow rate (Q max) was evaluated as a secondary efficacy endpoint. Mean Q maxincreased from baseline in both the treatment and placebo groups (Tadalafil Tablets 5 mg: 1.6 mL/sec, placebo: 1.2 mL/sec); however, these changes were not significantly different between groups.
In Study K, the effect of Tadalafil Tablets 5 mg once daily on Q maxwas evaluated as a safety endpoint. Mean Q maxincreased from baseline in both the treatment and placebo groups (Tadalafil Tablets 5 mg: 1.6 mL/sec, placebo: 1.1 mL/sec); however, these changes were not significantly different between groups.
Efficacy Results in Patients with BPH initiating Tadalafil Tablets and Finasteride– Tadalafil Tablets for once daily use initiated together with finasteride was shown to be effective in treating the signs and symptoms of BPH in men with an enlarged prostate (>30 cc) for up to 26 weeks. This additional double-blinded, parallel-design study of 26 weeks duration randomized 696 men to initiate either Tadalafil Tablets 5 mg with finasteride 5 mg or placebo with finasteride 5 mg. The study population had a mean age of 64 years (range 46-86). Patients with multiple co-morbid conditions such as erectile dysfunction, diabetes mellitus, hypertension, and other cardiovascular disease were included.
Tadalafil Tablets with finasteride demonstrated statistically significant improvement in the signs and symptoms of BPH compared to placebo with finasteride, as measured by the total IPSS at 12 weeks, the primary study endpoint ( see Table 20). Key secondary endpoints demonstrated improvement in total IPSS starting at the first scheduled observation at week 4 (Tadalafil Tablets -4.0, placebo -2.3: p<.001) and the score remained decreased through 26 weeks (Tadalafil Tablets -5.5, placebo -4.5; p=.022). However, the magnitude of the treatment difference between placebo/finasteride and Tadalafil Tablets/finasteride decreased from 1.7 points at Week 4 to 1.0 point at Week 26, as shown in Table 20and in Figure 7. The incremental benefit of Tadalafil Tablets beyond 26 weeks is unknown.
|
aOverall ITT population. |
||||||
|
bMixed model for repeated measurements. |
||||||
|
cUnadjusted mean. |
||||||
| Placebo and finasteride 5 mg | Tadalafil Tablets 5mg and finasteride 5 mg | Treatment difference | ||||
| n | (N=350) a | n | (N=345) a | p-value b | ||
| Total Symptom Score (IPSS) | ||||||
| Baseline c | 349 | 17.4 | 344 | 17.1 | ||
| Change from Baseline to Week 4 b | 340 | -2.3 | 330 | -4.0 | -1.7 | <.001 |
| Change from Baseline to Week 12 b | 318 | -3.8 | 317 | -5.2 | -1.4 | .001 |
| Change from Baseline to Week 26 b | 295 | -4.5 | 308 | -5.5 | -1.0 | .022 |
Figure 7: Mean Total IPSS Changes By Visit in BPH Patients Taking Tadalafil Tablets for Once Daily Use Together With Finasteride
In the 404 patients who had both ED and BPH at baseline, changes in erectile function were assessed as key secondary endpoints using the EF domain of the IIEF questionnaire. Tadalafil Tablets with finasteride (N=203) were compared to placebo with finasteride (N=201). A statistically significant improvement from baseline (Tadalafil Tablets/finasteride 13.7, placebo/finasteride 15.1) was observed at week 4 (Tadalafil Tablets/finasteride 3.7, placebo/finasteride -1.1; p<.001), week 12 (Tadalafil Tablets/finasteride 4.7, placebo/finasteride 0.6; p<.001), and week 26 (Tadalafil Tablets/finasteride 4.7, placebo/finasteride 0.0; p<.001).
5.14 Consideration of Other Urological Conditions Prior to Initiating Treatment for Bph (5.14 Consideration of Other Urological Conditions Prior to Initiating Treatment for BPH)
Prior to initiating treatment with Tadalafil Tablets for BPH, consideration should be given to other urological conditions that may cause similar symptoms. In addition, prostate cancer and BPH may coexist.
2.4 Tadalafil Tablets for Once Daily Use for Erectile Dysfunction and Benign Prostatic Hyperplasia
The recommended dose of Tadalafil Tablets for once daily use is 5 mg, taken at approximately the same time every day, without regard to timing of sexual activity.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:17.818072 · Updated: 2026-03-14T22:14:32.441829