These Highlights Do Not Include All The Information Needed To Use Mitosol®
244002c6-c61d-43fe-9326-072797113979
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Mitosol ® is an antimetabolite indicated for use as an adjunct to ab externo glaucoma surgery.
Indications and Usage
Mitosol ® is an antimetabolite indicated for use as an adjunct to ab externo glaucoma surgery.
Dosage and Administration
Mitosol ® is intended for topical application to the surgical site of glaucoma filtration surgery. It is not intended for intraocular administration. ( 2 ) • Each vial of Mitosol ® contains 0.2 mg of mitomycin and mannitol in a 1:2 concentration ratio. To reconstitute, add 1 mL of Sterile Water for Injection, then shake to dissolve. If product does not dissolve immediately, allow to stand at room temperature until the product has dissolved into solution. ( 2.2 ) • Fully saturate sponges provided within the Mitosol ® Kit utilizing the entire reconstituted contents of the vial in the manner prescribed in the Instructions for Use. ( 2.3 ) • Apply fully saturated sponges equally to the treatment area, in a single layer, with the use of a surgical forceps. Keep the sponges on the treatment area for two (2) minutes, then remove and return to the Mitosol ® Tray for defined disposal. ( 2.3 )
Warnings and Precautions
• Cell Death : Mitomycin is cytotoxic. Use of mitomycin in concentrations higher than 0.2 mg/mL or use for longer than 2 minutes may lead to unintended corneal and/or scleral damage including thinning or perforation. Direct contact with the corneal endothelium will result in cell death. ( 5.1 ) • Hypotony : The use of mitomycin has been associated with an increased incidence of post-operative hypotony. ( 5.2 ) • Cataract Development : Use in phakic patients has been correlated to a higher incidence of lenticular change and cataract formation. ( 5.3 ) • Embryo-Fetal Toxicity : Can cause fetal harm. Advise of potential risk to a fetus. Verify pregnancy status in females of reproductive potential prior to use. ( 5.4 , 8.1 , 8.3 )
Contraindications
• Hypersensitivity to mitomycin. ( 4.1 )
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Cell Death [see Warnings and Precautions ( 5.1 )] • Hypotony [see Warnings and Precautions ( 5.2 )] • Cataract Formation [see Warnings and Precautions ( 5.3 )]
How Supplied
Mitosol ® (mitomycin for solution) is available in a kit containing: One Vial containing 0.2 mg mitomycin One 1 mL syringe (Sterile Water For Injection) with Safety Connector One Plunger Rod One Vial Adapter with Spike One 1 mL TB Syringe, Luer Lock One Sponge Container Six 3 mm Absorbent Sponges Six 6 mm Absorbent Sponges Six Half Moon Sponges One Instrument Wedge Sponge One Protective Foam Pouch One Chemotherapy Waste Bag One Label, MMC (mitomycin) Three kits are supplied in each carton (NDC 49771-002-03).
Medication Information
Warnings and Precautions
• Cell Death : Mitomycin is cytotoxic. Use of mitomycin in concentrations higher than 0.2 mg/mL or use for longer than 2 minutes may lead to unintended corneal and/or scleral damage including thinning or perforation. Direct contact with the corneal endothelium will result in cell death. ( 5.1 ) • Hypotony : The use of mitomycin has been associated with an increased incidence of post-operative hypotony. ( 5.2 ) • Cataract Development : Use in phakic patients has been correlated to a higher incidence of lenticular change and cataract formation. ( 5.3 ) • Embryo-Fetal Toxicity : Can cause fetal harm. Advise of potential risk to a fetus. Verify pregnancy status in females of reproductive potential prior to use. ( 5.4 , 8.1 , 8.3 )
Indications and Usage
Mitosol ® is an antimetabolite indicated for use as an adjunct to ab externo glaucoma surgery.
Dosage and Administration
Mitosol ® is intended for topical application to the surgical site of glaucoma filtration surgery. It is not intended for intraocular administration. ( 2 ) • Each vial of Mitosol ® contains 0.2 mg of mitomycin and mannitol in a 1:2 concentration ratio. To reconstitute, add 1 mL of Sterile Water for Injection, then shake to dissolve. If product does not dissolve immediately, allow to stand at room temperature until the product has dissolved into solution. ( 2.2 ) • Fully saturate sponges provided within the Mitosol ® Kit utilizing the entire reconstituted contents of the vial in the manner prescribed in the Instructions for Use. ( 2.3 ) • Apply fully saturated sponges equally to the treatment area, in a single layer, with the use of a surgical forceps. Keep the sponges on the treatment area for two (2) minutes, then remove and return to the Mitosol ® Tray for defined disposal. ( 2.3 )
Contraindications
• Hypersensitivity to mitomycin. ( 4.1 )
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Cell Death [see Warnings and Precautions ( 5.1 )] • Hypotony [see Warnings and Precautions ( 5.2 )] • Cataract Formation [see Warnings and Precautions ( 5.3 )]
How Supplied
Mitosol ® (mitomycin for solution) is available in a kit containing: One Vial containing 0.2 mg mitomycin One 1 mL syringe (Sterile Water For Injection) with Safety Connector One Plunger Rod One Vial Adapter with Spike One 1 mL TB Syringe, Luer Lock One Sponge Container Six 3 mm Absorbent Sponges Six 6 mm Absorbent Sponges Six Half Moon Sponges One Instrument Wedge Sponge One Protective Foam Pouch One Chemotherapy Waste Bag One Label, MMC (mitomycin) Three kits are supplied in each carton (NDC 49771-002-03).
Description
Mitosol ® is an antimetabolite indicated for use as an adjunct to ab externo glaucoma surgery.
Mitosol
®
(mitomycin for solution)
0.2 mg/vial
Kit for Ophthalmic Use
Read INSTRUCTIONS FOR USE Before Proceeding
Instructions for Use
|
A. Outer Pack
One Sterile Chemotherapy Waste Bag One Instructions for Use One Package Insert One Inner Tray Two Patient Chart Labels
|
|
|
B. STERILE Inner Tray One Vial Containing 0.2 mg mitomycin (inside protective foam pouch) One 1 mL Syringe (Sterile Water for Injection) with Safety Connector One Plunger Rod One Vial Adaptor with Spike (inside protective foam pouch) One 1 mL TB Syringe, Luer Lock One Sponge Container Containing: • Six 3 mm Absorbent Sponges • Six 6 mm Absorbent Sponges • Six Half Moon Sponges • One Instrument Wedge Sponge One Label, MMC (mitomycin)
This tray and its contents are STERILE. |
|
|
1. Getting Started Non-Sterile Circulating Nurse: Open outer pack. Affect sterile transfer of ALL contents to the sterile field. Sterile Surgical Technician: Open sterile inner tray. |
|
|
2. Reconstituting Mitosol®
a. Remove vial and vial adapter from blue foam pouch. b.
Screw
white plunger rod to rubber plunger of pre‑filled syringe. (Fig. 1) c.
Press firmly
and screw the
blue end
of the vial adapter into the
blue end
of the syringe connector. (Fig. 2)
NOTE: Do not force plunger. Syringe will not operate if vial adapter and syringe connector are not properly connected. Forcing plunger may result in syringe leakage and Mitosol® exposure. |
|
|
d. Stand vial upright on a sturdy, flat surface and push on the vial lid until seated and secure. (Fig. 3)
e. Inject entire contents of sterile water (1 ml) into vial. (Fig. 4) Do not force syringe plunger. See note at step 2. f. IMPORTANT: INVERT VIAL REPEATEDLY to saturate ALL drug product, including that adhering to stopper, then shake until complete reconstitution of Mitosol®. If product does not dissolve immediately, allow to stand at room temperature until the product has dissolved into solution. |
|
|
3. Preparing sponges a.
Invert vial and syringe
and draw full volume of medication into syringe. (Fig. 5) b. Remove all sponges from sponge tray. c. Return to sponge tray only those sponges to be saturated with Mitosol®. d. Unscrew the syringe with safety connector from vial and vial adapter. (Fig. 6) Note: DO NOT remove safety connector from syringe.
e. Place vial and vial adaptor in chemotherapy waste disposal bag (yellow bag), and set bag aside, within sterile field, for additional use. f. Take sponge container from sterile inner tray. g. Screw both syringes into sponge container; the TB syringe to one end, the syringe with reconstituted Mitosol® to the other. |
|
|
h.
Mitosol® must be used within 1 hour of reconstitution: • Inject medication into sponge container, saturating sponges. Reconstituted Mitosol® should remain undisturbed in sponge container for
60 seconds
. (Fig. 7) Do not force syringe plunger. See note at step 2. • If any excess fluid remains, withdraw plunger of TB syringe, drawing excess fluid/air into syringe. |
|
|
4. Using Mitosol®
a. With both syringes connected, the TB syringe to one end, the pre-filled syringe to the other, open sponge container, offering contents to surgeon for placement on surgical site. (Fig. 8) b. Apply saturated sponges to surgical site for two minutes. Remove sponges from eye and copiously irrigate surgical site. c. As used sponges are removed from surgical site, accept used sponges back into sponge container for disposal. Close container lid. d. With syringes still connected to sponge container, remove entire assembly from surgical field in chemotherapy waste disposal bag. |
DISPOSE OF CHEMOTHERAPY WASTE BAG AND ITS CONTENTS AS CHEMOTHERAPY WASTE
US Patents #7,806,265, #8,186,511, #D685,962, #D685,963, #9,205,075, #9,539,241 and #9,649,428; other international patents issued and pending.
A4807998-2
Rev. 07/20
Data
Animal Data
Parenteral administration of mitomycin in animal reproduction studies produced fetal malformations and embryofetal lethality.
Section 42229-5
Absorption
The systemic exposure of mitomycin following ocular administration of Mitosol® in humans is unknown. Based on a comparison of the proposed dose of up to 0.2 mg to intravenous (IV) doses of mitomycin used clinically for treatment of oncologic indications (up to 20 mg/m2), systemic concentrations in humans upon ocular administration are expected to be multiple orders of magnitude lower than those achieved by IV administration.
5.2 Hypotony
The use of mitomycin has been associated with an increased incidence of post-operative hypotony.
Risk Summary
Based on findings in animals and mechanism of action [see Clinical Pharmacology (12.1)], Mitosol® can cause fetal harm when administered to a pregnant woman. There are no available data on Mitosol® use in pregnant women to inform the drug-associated risk. In animal reproduction studies, parenteral administration of mitomycin resulted in teratogenicity (see Data ). Advise pregnant women of the potential risk to a fetus.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% - 4% and 15% - 20%, respectively.
2.4 Stability
Lyophilized Mitosol® stored at 20°C to 25°C (68°F to 77°F) is stable for the shelf life indicated on the package. Avoid excessive heat. Protect from light.
Reconstituted with 1 mL of Sterile Water for Injection at a concentration of 0.2 mg/mL, mitomycin is stable for one (1) hour at room temperature.
11 Description
Mitomycin is an antibiotic isolated from the broth of Streptomyces verticillus Yingtanensis which has been shown to have antimetabolic activity.
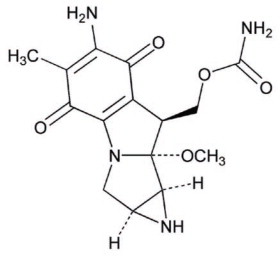
Mitomycin is a blue-violet crystalline powder with the molecular formula of C15H18N4O5 and a molecular weight of 334.33. Its chemical name is 7-amino-9α-methoxymitosane and it has the following structural formula:
Mitosol® is a sterile lyophilized mixture of mitomycin and mannitol, which, when reconstituted with Sterile Water for Injection, provides a solution for application in glaucoma filtration surgery. Mitosol® is supplied in vials containing 0.2 mg of mitomycin. Each vial also contains mannitol 0.4 mg, at a 1:2 ratio of mitomycin to mannitol. Each mL of reconstituted solution contains 0.2 mg mitomycin and has a pH between 5.0 and 8.0.
5.1 Cell Death
Mitomycin is cytotoxic. Use of mitomycin in concentrations higher than 0.2 mg/mL or use for longer than 2 minutes may lead to unintended corneal and/or scleral damage including thinning or perforation. Direct contact with the corneal endothelium will result in cell death.
16.1 How Supplied
Mitosol® (mitomycin for solution) is available in a kit containing:
One Vial containing 0.2 mg mitomycin
One 1 mL syringe (Sterile Water For Injection) with Safety Connector
One Plunger Rod
One Vial Adapter with Spike
One 1 mL TB Syringe, Luer Lock
One Sponge Container
Six 3 mm Absorbent Sponges
Six 6 mm Absorbent Sponges
Six Half Moon Sponges
One Instrument Wedge Sponge
One Protective Foam Pouch
One Chemotherapy Waste Bag
One Label, MMC (mitomycin)
Three kits are supplied in each carton (NDC 49771-002-03).
2.3 Method of Use
Sponges provided within the Mitosol® Kit should be fully saturated with the entire reconstituted contents in the manner prescribed in the Instructions for Use. A treatment area approximating 10mm x 6mm +/- 2mm should be treated with the Mitosol®. Apply fully saturated sponges equally to the treatment area, in a single layer, with the use of a surgical forceps. Keep the sponges on the treatment area for two (2) minutes, then remove and return to the Mitosol® Tray for defined disposal in the Chemotherapy Waste Bag provided.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
No overall differences in safety and effectiveness have been observed between elderly and younger patients.
Pregnancy Testing
Verify pregnancy status in females of reproductive potential prior to using Mitosol®.
14 Clinical Studies
In placebo-controlled studies reported in the medical literature, mitomycin reduced intraocular pressure (IOP) by 3 mmHg in patients with open-angle glaucoma when used as an adjunct to ab externo glaucoma surgery by Month 12.
In studies with a historical control reported in the medical literature, mitomycin reduced intraocular pressure (IOP) by 5 mmHg in patients with open-angle glaucoma when used as an adjunct to ab externo glaucoma surgery by Month 12.
4 Contraindications
-
•Hypersensitivity to mitomycin. (4.1)
6 Adverse Reactions
4.1 Hypersensitivity
Mitosol® is contraindicated in patients that have demonstrated a hypersensitivity to mitomycin in the past.
5.3 Cataract Formation
Use in phakic patients has been correlated to a higher incidence of lenticular change and cataract formation.
1 Indications and Usage
Mitosol® is an antimetabolite indicated for use as an adjunct to ab externo glaucoma surgery.
12.1 Mechanism of Action
Mitosol® inhibits the synthesis of deoxyribonucleic acid (DNA). The guanine and cytosine content correlates with the degree of mitomycin-induced cross-linking. Cellular RNA and protein synthesis may also be suppressed.
5.4 Embryo Fetal Toxicity
5 Warnings and Precautions
-
•Cell Death: Mitomycin is cytotoxic. Use of mitomycin in concentrations higher than 0.2 mg/mL or use for longer than 2 minutes may lead to unintended corneal and/or scleral damage including thinning or perforation. Direct contact with the corneal endothelium will result in cell death. (5.1)
-
•Hypotony: The use of mitomycin has been associated with an increased incidence of post-operative hypotony. (5.2)
-
•Cataract Development: Use in phakic patients has been correlated to a higher incidence of lenticular change and cataract formation. (5.3)
-
•Embryo-Fetal Toxicity: Can cause fetal harm. Advise of potential risk to a fetus. Verify pregnancy status in females of reproductive potential prior to use. (5.4, 8.1, 8.3)
2 Dosage and Administration
Mitosol® is intended for topical application to the surgical site of glaucoma filtration surgery. It is not intended for intraocular administration. (2)
-
•Each vial of Mitosol® contains 0.2 mg of mitomycin and mannitol in a 1:2 concentration ratio. To reconstitute, add 1 mL of Sterile Water for Injection, then shake to dissolve. If product does not dissolve immediately, allow to stand at room temperature until the product has dissolved into solution. (2.2)
-
•Fully saturate sponges provided within the Mitosol® Kit utilizing the entire reconstituted contents of the vial in the manner prescribed in the Instructions for Use. (2.3)
-
•Apply fully saturated sponges equally to the treatment area, in a single layer, with the use of a surgical forceps. Keep the sponges on the treatment area for two (2) minutes, then remove and return to the Mitosol® Tray for defined disposal. (2.3)
2.2 Method of Reconstitution
Each vial of Mitosol® contains 0.2 mg of mitomycin and mannitol in a 1:2 concentration ratio. To reconstitute, add 1 mL of Sterile Water for Injection, then shake to dissolve. If product does not dissolve immediately, allow to stand at room temperature until the product dissolves into solution.
3 Dosage Forms and Strengths
Mitosol® is a sterile lyophilized mixture of mitomycin and mannitol, which, when reconstituted with Sterile Water for Injection, provides a solution for application in glaucoma filtration surgery. Mitosol® is supplied in vials containing 0.2 mg of mitomycin. Each vial also contains mannitol 0.4 mg, at a 1:2 ratio of mitomycin to mannitol. Each mL of reconstituted solution contains 0.2 mg mitomycin and has a pH between 5.0 and 8.0.
6.1 Ophthalmic Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The most frequent adverse reactions to Mitosol® occur locally, as an extension of the pharmacological activity of the drug. These reactions include:
Blebitis: bleb ulceration, chronic bleb leak, encapsulated/cystic bleb, bleb-related infection, wound dehiscence, conjunctival necrosis, thin-walled bleb
Cornea: corneal endothelial damage, epithelial defect, anterior synechiae, superficial punctuate keratitis, Descemet's detachment, induced astigmatism
Endophthalmitis
Hypotony: choroidal reactions (choroidal detachment, choroidal effusion, serous choroidal detachment, suprachoroidal hemorrhage, hypotony maculopathy, presence of supraciliochoroidal fluid, hypoechogenic suprachoroidal effusion)
Inflammation: iritis, fibrin reaction
Lens: cataract development, cataract progression, capsule opacification, capsular constriction and/or capsulotomy rupture, posterior synechiae
Retina: retinal pigment epithelial tear, retinal detachment (serous and rhegatogenous)
Scleritis: wound dehiscence
Vascular: hyphema, central retinal vein occlusion, hemiretinal vein occlusion, retinal hemorrhage, vitreal hemorrhage and blood clot, subconjunctival hemorrhage, disk hemorrhage
Additional Reactions: macular edema, sclera thinning or ulceration, intraocular lens capture, disk swelling, malignant glaucoma, lacrimal drainage system obstruction, ciliary block, corneal vascularization, visual acuity decrease, cystic conjunctival degeneration, upper eyelid retraction, dislocated implants, severe loss of vision.
17 Patient Counseling Information
-
•Instruct patients to discuss with their physician if they are pregnant or if they might become pregnant [see Use in Specific Populations (8.1)].
-
•Instruct patients to discuss with their physician if they have demonstrated a hypersensitivity to mitomycin in the past [see Contraindications (4.1)].
-
•Nursing mothers should be advised that it is not known if Mitosol® is excreted in human milk. Because of the potential for serious adverse reactions in a breastfed child, advise women not to breastfeed during and for 1 week following administration of Mitosol® [see Use in Specific Populations ( 8.2 )].
-
•Patients should be advised of the toxicity of Mitosol® and potential complications.
Manufactured for:
Mobius Therapeutics, LLC
1000 Executive Parkway
Suite 224
St. Louis, MO 63141
Principal Display Panel Vial Label
NDC 49771-002-02
Mitosol®
(mitomycin for solution)
0.2 mg/vial
Lyophilized Mitomycin for
reconstitution
For ophthalmic use
Protect from light.
Single Use Vial
Dose: See Package Insert.
Rx Only
Store at 20°-25°C (68°-77°F).
Manufactured for:
Mobius Therapeutics, LLC
1000 Executive Parkway
Suite 224
St. Louis, MO 63141
Manufactured by:
Intas Pharmaceuticals Ltd.
Ahmedabad-382 210, INDIA.
Mfg. Lic. No.: G/28/1026
10 9480 0 6029283 INL5021
Principal Display Panel Vial Label New
NDC 49771-002-02
Mitosol®
(mitomycin for solution)
0.2 mg/vial
Lyophilized Mitomycin for
reconstitution
For ophthalmic use
Protect from light.
Single Use Vial
Dose: See Package Insert.
Rx Only
Store at 20°-25°C (68°-77°F).
Manufactured for:
Mobius Therapeutics, LLC
1000 Executive Parkway
Suite 224
St. Louis, MO 63141
Lot:
EXP Date
YYYY-MM-DD
2.1 Important Administration Instructions
Mitosol® is intended for topical application to the surgical site of glaucoma filtration surgery. Mitosol® is a cytotoxic drug. It is not intended for intraocular administration. If intraocular administration occurs, cell death leading to corneal infarction, retinal infarction, and ciliary body atrophy may result. Verify pregnancy status in females of reproductive potential prior to using Mitosol®.
Principal Display Panel Outer Kit Package
Mitosol®
(mitomycin for solution)
0.2 mg/vial
Kit for Ophthalmic Use
Manufactured for:
Mobius Therapeutics, LLC
1000 Executive Parkway
Suite 224
St. Louis, MO 63141 USA
+1 314-615-6930
1-877-EYE-MITO (1-877-393-6486)
Rx ONLY
US Patents #7,806,265, #8,186,511, #D685,962,
#D685,963, #9,205,075, #9,539,241 and #9,649,428;
other international patents issued and pending.
©2019 Mobius Therapeutics, LLC
mobius
therapeutics™
A1426418-1
Rev. 8/19
NDC #49771-002-01
Re-Order #MOB.2
Each Mitosol® Kit Contains:
One Chemotherapy Waste Bag
One Instructions for Use
One Package Insert
One Inner Tray
Two Patient Chart Labels
Inner Tray Contains:
One Vial Containing 0.2 mg mitomycin
(inside protective foam pouch)
One 1 mL Syringe (Sterile Water for Injection)
with Safety Connector
One Plunger Rod
One Vial Adaptor with Spike (inside protective foam pouch)
One 1 mL TB Syringe, Luer Lock
One Sponge Container Containing:
Six 3 mm Absorbent Sponges
Six 6 mm Absorbent Sponges
Six Half Moon Sponges
One Instrument Wedge Sponge
One Label, MMC
Contents STERILE in unopened undamaged package.
Storage: Store kits at 20° - 25° C (68° - 77° F). Protect from light.
HD CAUTION: HAZARDOUS DRUG
OBSERVE SPECIAL HANDLING, ADMINISTRATION
AND DISPOSAL REQUIREMENTS
8.3 Females and Males of Reproductive Potential
Mitosol® can cause fetal harm when administered to pregnant women [see Use in Specific Populations (8.1)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Adequate long-term studies in animals to evaluate carcinogenic potential have not been conducted with Mitosol®. Intravenous administration of mitomycin has been found to be carcinogenic in rats and mice. At doses approximating the recommended clinical injectable dose in humans, mitomycin produces a greater than 100 percent increase in tumor incidence in male Sprague-Dawley rats, and a greater than 50 percent increase in tumor incidence in female Swiss mice.
The effect of Mitosol® on fertility is unknown.
Structured Label Content
Mitosol
®
(mitomycin for solution)
0.2 mg/vial
Kit for Ophthalmic Use
Read INSTRUCTIONS FOR USE Before Proceeding
Instructions for Use
|
A. Outer Pack
One Sterile Chemotherapy Waste Bag One Instructions for Use One Package Insert One Inner Tray Two Patient Chart Labels
|
|
|
B. STERILE Inner Tray One Vial Containing 0.2 mg mitomycin (inside protective foam pouch) One 1 mL Syringe (Sterile Water for Injection) with Safety Connector One Plunger Rod One Vial Adaptor with Spike (inside protective foam pouch) One 1 mL TB Syringe, Luer Lock One Sponge Container Containing: • Six 3 mm Absorbent Sponges • Six 6 mm Absorbent Sponges • Six Half Moon Sponges • One Instrument Wedge Sponge One Label, MMC (mitomycin)
This tray and its contents are STERILE. |
|
|
1. Getting Started Non-Sterile Circulating Nurse: Open outer pack. Affect sterile transfer of ALL contents to the sterile field. Sterile Surgical Technician: Open sterile inner tray. |
|
|
2. Reconstituting Mitosol®
a. Remove vial and vial adapter from blue foam pouch. b.
Screw
white plunger rod to rubber plunger of pre‑filled syringe. (Fig. 1) c.
Press firmly
and screw the
blue end
of the vial adapter into the
blue end
of the syringe connector. (Fig. 2)
NOTE: Do not force plunger. Syringe will not operate if vial adapter and syringe connector are not properly connected. Forcing plunger may result in syringe leakage and Mitosol® exposure. |
|
|
d. Stand vial upright on a sturdy, flat surface and push on the vial lid until seated and secure. (Fig. 3)
e. Inject entire contents of sterile water (1 ml) into vial. (Fig. 4) Do not force syringe plunger. See note at step 2. f. IMPORTANT: INVERT VIAL REPEATEDLY to saturate ALL drug product, including that adhering to stopper, then shake until complete reconstitution of Mitosol®. If product does not dissolve immediately, allow to stand at room temperature until the product has dissolved into solution. |
|
|
3. Preparing sponges a.
Invert vial and syringe
and draw full volume of medication into syringe. (Fig. 5) b. Remove all sponges from sponge tray. c. Return to sponge tray only those sponges to be saturated with Mitosol®. d. Unscrew the syringe with safety connector from vial and vial adapter. (Fig. 6) Note: DO NOT remove safety connector from syringe.
e. Place vial and vial adaptor in chemotherapy waste disposal bag (yellow bag), and set bag aside, within sterile field, for additional use. f. Take sponge container from sterile inner tray. g. Screw both syringes into sponge container; the TB syringe to one end, the syringe with reconstituted Mitosol® to the other. |
|
|
h.
Mitosol® must be used within 1 hour of reconstitution: • Inject medication into sponge container, saturating sponges. Reconstituted Mitosol® should remain undisturbed in sponge container for
60 seconds
. (Fig. 7) Do not force syringe plunger. See note at step 2. • If any excess fluid remains, withdraw plunger of TB syringe, drawing excess fluid/air into syringe. |
|
|
4. Using Mitosol®
a. With both syringes connected, the TB syringe to one end, the pre-filled syringe to the other, open sponge container, offering contents to surgeon for placement on surgical site. (Fig. 8) b. Apply saturated sponges to surgical site for two minutes. Remove sponges from eye and copiously irrigate surgical site. c. As used sponges are removed from surgical site, accept used sponges back into sponge container for disposal. Close container lid. d. With syringes still connected to sponge container, remove entire assembly from surgical field in chemotherapy waste disposal bag. |
DISPOSE OF CHEMOTHERAPY WASTE BAG AND ITS CONTENTS AS CHEMOTHERAPY WASTE
US Patents #7,806,265, #8,186,511, #D685,962, #D685,963, #9,205,075, #9,539,241 and #9,649,428; other international patents issued and pending.
A4807998-2
Rev. 07/20
Data
Animal Data
Parenteral administration of mitomycin in animal reproduction studies produced fetal malformations and embryofetal lethality.
Section 42229-5 (42229-5)
Absorption
The systemic exposure of mitomycin following ocular administration of Mitosol® in humans is unknown. Based on a comparison of the proposed dose of up to 0.2 mg to intravenous (IV) doses of mitomycin used clinically for treatment of oncologic indications (up to 20 mg/m2), systemic concentrations in humans upon ocular administration are expected to be multiple orders of magnitude lower than those achieved by IV administration.
5.2 Hypotony
The use of mitomycin has been associated with an increased incidence of post-operative hypotony.
Risk Summary
Based on findings in animals and mechanism of action [see Clinical Pharmacology (12.1)], Mitosol® can cause fetal harm when administered to a pregnant woman. There are no available data on Mitosol® use in pregnant women to inform the drug-associated risk. In animal reproduction studies, parenteral administration of mitomycin resulted in teratogenicity (see Data ). Advise pregnant women of the potential risk to a fetus.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% - 4% and 15% - 20%, respectively.
2.4 Stability
Lyophilized Mitosol® stored at 20°C to 25°C (68°F to 77°F) is stable for the shelf life indicated on the package. Avoid excessive heat. Protect from light.
Reconstituted with 1 mL of Sterile Water for Injection at a concentration of 0.2 mg/mL, mitomycin is stable for one (1) hour at room temperature.
11 Description (11 DESCRIPTION)
Mitomycin is an antibiotic isolated from the broth of Streptomyces verticillus Yingtanensis which has been shown to have antimetabolic activity.
Mitomycin is a blue-violet crystalline powder with the molecular formula of C15H18N4O5 and a molecular weight of 334.33. Its chemical name is 7-amino-9α-methoxymitosane and it has the following structural formula:
Mitosol® is a sterile lyophilized mixture of mitomycin and mannitol, which, when reconstituted with Sterile Water for Injection, provides a solution for application in glaucoma filtration surgery. Mitosol® is supplied in vials containing 0.2 mg of mitomycin. Each vial also contains mannitol 0.4 mg, at a 1:2 ratio of mitomycin to mannitol. Each mL of reconstituted solution contains 0.2 mg mitomycin and has a pH between 5.0 and 8.0.
5.1 Cell Death
Mitomycin is cytotoxic. Use of mitomycin in concentrations higher than 0.2 mg/mL or use for longer than 2 minutes may lead to unintended corneal and/or scleral damage including thinning or perforation. Direct contact with the corneal endothelium will result in cell death.
16.1 How Supplied
Mitosol® (mitomycin for solution) is available in a kit containing:
One Vial containing 0.2 mg mitomycin
One 1 mL syringe (Sterile Water For Injection) with Safety Connector
One Plunger Rod
One Vial Adapter with Spike
One 1 mL TB Syringe, Luer Lock
One Sponge Container
Six 3 mm Absorbent Sponges
Six 6 mm Absorbent Sponges
Six Half Moon Sponges
One Instrument Wedge Sponge
One Protective Foam Pouch
One Chemotherapy Waste Bag
One Label, MMC (mitomycin)
Three kits are supplied in each carton (NDC 49771-002-03).
2.3 Method of Use
Sponges provided within the Mitosol® Kit should be fully saturated with the entire reconstituted contents in the manner prescribed in the Instructions for Use. A treatment area approximating 10mm x 6mm +/- 2mm should be treated with the Mitosol®. Apply fully saturated sponges equally to the treatment area, in a single layer, with the use of a surgical forceps. Keep the sponges on the treatment area for two (2) minutes, then remove and return to the Mitosol® Tray for defined disposal in the Chemotherapy Waste Bag provided.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
No overall differences in safety and effectiveness have been observed between elderly and younger patients.
Pregnancy Testing
Verify pregnancy status in females of reproductive potential prior to using Mitosol®.
14 Clinical Studies (14 CLINICAL STUDIES)
In placebo-controlled studies reported in the medical literature, mitomycin reduced intraocular pressure (IOP) by 3 mmHg in patients with open-angle glaucoma when used as an adjunct to ab externo glaucoma surgery by Month 12.
In studies with a historical control reported in the medical literature, mitomycin reduced intraocular pressure (IOP) by 5 mmHg in patients with open-angle glaucoma when used as an adjunct to ab externo glaucoma surgery by Month 12.
4 Contraindications (4 CONTRAINDICATIONS)
-
•Hypersensitivity to mitomycin. (4.1)
6 Adverse Reactions (6 ADVERSE REACTIONS)
4.1 Hypersensitivity
Mitosol® is contraindicated in patients that have demonstrated a hypersensitivity to mitomycin in the past.
5.3 Cataract Formation
Use in phakic patients has been correlated to a higher incidence of lenticular change and cataract formation.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Mitosol® is an antimetabolite indicated for use as an adjunct to ab externo glaucoma surgery.
12.1 Mechanism of Action
Mitosol® inhibits the synthesis of deoxyribonucleic acid (DNA). The guanine and cytosine content correlates with the degree of mitomycin-induced cross-linking. Cellular RNA and protein synthesis may also be suppressed.
5.4 Embryo Fetal Toxicity (5.4 Embryo-Fetal Toxicity)
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Cell Death: Mitomycin is cytotoxic. Use of mitomycin in concentrations higher than 0.2 mg/mL or use for longer than 2 minutes may lead to unintended corneal and/or scleral damage including thinning or perforation. Direct contact with the corneal endothelium will result in cell death. (5.1)
-
•Hypotony: The use of mitomycin has been associated with an increased incidence of post-operative hypotony. (5.2)
-
•Cataract Development: Use in phakic patients has been correlated to a higher incidence of lenticular change and cataract formation. (5.3)
-
•Embryo-Fetal Toxicity: Can cause fetal harm. Advise of potential risk to a fetus. Verify pregnancy status in females of reproductive potential prior to use. (5.4, 8.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Mitosol® is intended for topical application to the surgical site of glaucoma filtration surgery. It is not intended for intraocular administration. (2)
-
•Each vial of Mitosol® contains 0.2 mg of mitomycin and mannitol in a 1:2 concentration ratio. To reconstitute, add 1 mL of Sterile Water for Injection, then shake to dissolve. If product does not dissolve immediately, allow to stand at room temperature until the product has dissolved into solution. (2.2)
-
•Fully saturate sponges provided within the Mitosol® Kit utilizing the entire reconstituted contents of the vial in the manner prescribed in the Instructions for Use. (2.3)
-
•Apply fully saturated sponges equally to the treatment area, in a single layer, with the use of a surgical forceps. Keep the sponges on the treatment area for two (2) minutes, then remove and return to the Mitosol® Tray for defined disposal. (2.3)
2.2 Method of Reconstitution
Each vial of Mitosol® contains 0.2 mg of mitomycin and mannitol in a 1:2 concentration ratio. To reconstitute, add 1 mL of Sterile Water for Injection, then shake to dissolve. If product does not dissolve immediately, allow to stand at room temperature until the product dissolves into solution.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Mitosol® is a sterile lyophilized mixture of mitomycin and mannitol, which, when reconstituted with Sterile Water for Injection, provides a solution for application in glaucoma filtration surgery. Mitosol® is supplied in vials containing 0.2 mg of mitomycin. Each vial also contains mannitol 0.4 mg, at a 1:2 ratio of mitomycin to mannitol. Each mL of reconstituted solution contains 0.2 mg mitomycin and has a pH between 5.0 and 8.0.
6.1 Ophthalmic Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The most frequent adverse reactions to Mitosol® occur locally, as an extension of the pharmacological activity of the drug. These reactions include:
Blebitis: bleb ulceration, chronic bleb leak, encapsulated/cystic bleb, bleb-related infection, wound dehiscence, conjunctival necrosis, thin-walled bleb
Cornea: corneal endothelial damage, epithelial defect, anterior synechiae, superficial punctuate keratitis, Descemet's detachment, induced astigmatism
Endophthalmitis
Hypotony: choroidal reactions (choroidal detachment, choroidal effusion, serous choroidal detachment, suprachoroidal hemorrhage, hypotony maculopathy, presence of supraciliochoroidal fluid, hypoechogenic suprachoroidal effusion)
Inflammation: iritis, fibrin reaction
Lens: cataract development, cataract progression, capsule opacification, capsular constriction and/or capsulotomy rupture, posterior synechiae
Retina: retinal pigment epithelial tear, retinal detachment (serous and rhegatogenous)
Scleritis: wound dehiscence
Vascular: hyphema, central retinal vein occlusion, hemiretinal vein occlusion, retinal hemorrhage, vitreal hemorrhage and blood clot, subconjunctival hemorrhage, disk hemorrhage
Additional Reactions: macular edema, sclera thinning or ulceration, intraocular lens capture, disk swelling, malignant glaucoma, lacrimal drainage system obstruction, ciliary block, corneal vascularization, visual acuity decrease, cystic conjunctival degeneration, upper eyelid retraction, dislocated implants, severe loss of vision.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
-
•Instruct patients to discuss with their physician if they are pregnant or if they might become pregnant [see Use in Specific Populations (8.1)].
-
•Instruct patients to discuss with their physician if they have demonstrated a hypersensitivity to mitomycin in the past [see Contraindications (4.1)].
-
•Nursing mothers should be advised that it is not known if Mitosol® is excreted in human milk. Because of the potential for serious adverse reactions in a breastfed child, advise women not to breastfeed during and for 1 week following administration of Mitosol® [see Use in Specific Populations ( 8.2 )].
-
•Patients should be advised of the toxicity of Mitosol® and potential complications.
Manufactured for:
Mobius Therapeutics, LLC
1000 Executive Parkway
Suite 224
St. Louis, MO 63141
Principal Display Panel Vial Label (PRINCIPAL DISPLAY PANEL - VIAL LABEL)
NDC 49771-002-02
Mitosol®
(mitomycin for solution)
0.2 mg/vial
Lyophilized Mitomycin for
reconstitution
For ophthalmic use
Protect from light.
Single Use Vial
Dose: See Package Insert.
Rx Only
Store at 20°-25°C (68°-77°F).
Manufactured for:
Mobius Therapeutics, LLC
1000 Executive Parkway
Suite 224
St. Louis, MO 63141
Manufactured by:
Intas Pharmaceuticals Ltd.
Ahmedabad-382 210, INDIA.
Mfg. Lic. No.: G/28/1026
10 9480 0 6029283 INL5021
Principal Display Panel Vial Label New (PRINCIPAL DISPLAY PANEL - VIAL LABEL-New)
NDC 49771-002-02
Mitosol®
(mitomycin for solution)
0.2 mg/vial
Lyophilized Mitomycin for
reconstitution
For ophthalmic use
Protect from light.
Single Use Vial
Dose: See Package Insert.
Rx Only
Store at 20°-25°C (68°-77°F).
Manufactured for:
Mobius Therapeutics, LLC
1000 Executive Parkway
Suite 224
St. Louis, MO 63141
Lot:
EXP Date
YYYY-MM-DD
2.1 Important Administration Instructions
Mitosol® is intended for topical application to the surgical site of glaucoma filtration surgery. Mitosol® is a cytotoxic drug. It is not intended for intraocular administration. If intraocular administration occurs, cell death leading to corneal infarction, retinal infarction, and ciliary body atrophy may result. Verify pregnancy status in females of reproductive potential prior to using Mitosol®.
Principal Display Panel Outer Kit Package (PRINCIPAL DISPLAY PANEL - OUTER KIT PACKAGE)
Mitosol®
(mitomycin for solution)
0.2 mg/vial
Kit for Ophthalmic Use
Manufactured for:
Mobius Therapeutics, LLC
1000 Executive Parkway
Suite 224
St. Louis, MO 63141 USA
+1 314-615-6930
1-877-EYE-MITO (1-877-393-6486)
Rx ONLY
US Patents #7,806,265, #8,186,511, #D685,962,
#D685,963, #9,205,075, #9,539,241 and #9,649,428;
other international patents issued and pending.
©2019 Mobius Therapeutics, LLC
mobius
therapeutics™
A1426418-1
Rev. 8/19
NDC #49771-002-01
Re-Order #MOB.2
Each Mitosol® Kit Contains:
One Chemotherapy Waste Bag
One Instructions for Use
One Package Insert
One Inner Tray
Two Patient Chart Labels
Inner Tray Contains:
One Vial Containing 0.2 mg mitomycin
(inside protective foam pouch)
One 1 mL Syringe (Sterile Water for Injection)
with Safety Connector
One Plunger Rod
One Vial Adaptor with Spike (inside protective foam pouch)
One 1 mL TB Syringe, Luer Lock
One Sponge Container Containing:
Six 3 mm Absorbent Sponges
Six 6 mm Absorbent Sponges
Six Half Moon Sponges
One Instrument Wedge Sponge
One Label, MMC
Contents STERILE in unopened undamaged package.
Storage: Store kits at 20° - 25° C (68° - 77° F). Protect from light.
HD CAUTION: HAZARDOUS DRUG
OBSERVE SPECIAL HANDLING, ADMINISTRATION
AND DISPOSAL REQUIREMENTS
8.3 Females and Males of Reproductive Potential
Mitosol® can cause fetal harm when administered to pregnant women [see Use in Specific Populations (8.1)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Adequate long-term studies in animals to evaluate carcinogenic potential have not been conducted with Mitosol®. Intravenous administration of mitomycin has been found to be carcinogenic in rats and mice. At doses approximating the recommended clinical injectable dose in humans, mitomycin produces a greater than 100 percent increase in tumor incidence in male Sprague-Dawley rats, and a greater than 50 percent increase in tumor incidence in female Swiss mice.
The effect of Mitosol® on fertility is unknown.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:46.076679 · Updated: 2026-03-14T22:25:35.146821