These Highlights Do Not Include All The Information Needed To Use Fentanyl Transdermal System Safely And Effectively.
242759ef-cb6d-4e3e-9f8d-5e31efa1f289
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Boxed Warning 12/2025 Indications and Usage ( 1 ) 12/2025 Dosage and Administration ( 2.2 , 2.3 ) 12/2025 Warnings and Precautions ( 5.1, 5.2, 5.4, 5.19 , 5.21 ) 12/2025
Indications and Usage
Fentanyl transdermal system is indicated for the management of severe and persistent pain in opioid-tolerant patients, that requires an opioid analgesic and that cannot be adequately treated with alternative options, including immediate-release opioids. Patients considered opioid-tolerant are those who are taking, for one week or longer, at least 60 mg morphine per day, 25 mcg transdermal fentanyl per hour, 30 mg oral oxycodone per day, 8 mg oral hydromorphone per day, 25 mg oral oxymorphone per day, 60 mg oral hydrocodone per day, or an equianalgesic dose of another opioid. Limitations of Use Because of the risks of addiction, abuse, misuse, overdose, and death, which can occur at any dosage or duration and persist over the course of therapy [see Warnings and Precautions ( 5.1 ) ], reserve opioid analgesics, including fentanyl transdermal system, for use in patients for whom alternative treatment options are ineffective, not tolerated, or would be otherwise inadequate to provide sufficient management of pain. Fentanyl transdermal system is not indicated as an as-needed (prn) analgesic.
Dosage and Administration
Fentanyl transdermal system should be prescribed only by healthcare providers who are knowledgeable about the use of extended-release/long-acting opioids and how to mitigate the associated risks. ( 2.1 ) Use the lowest effective dosage for the shortest duration of time consistent with individual patient treatment goals. Reserve titration to higher doses of fentanyl transdermal system for patients in whom lower doses are insufficiently effective and in whom the expected benefits of using a higher dose opioid clearly outweigh the substantial risks. ( 2.1 , 5 ) Initiate the dosing regimen for each patient individually, taking into account the patient's underlying cause and severity of pain, prior analgesic treatment and response, and risk factors for addiction, abuse, and misuse. ( 5.1 ) Respiratory depression can occur at any time during opioid therapy, especially when initiating and following dosage increases with fentanyl transdermal system. Consider this risk when selecting an initial dose and when making dose adjustments. ( 2.1 , 5.2 ) Discuss opioid overdose reversal agents and options for acquiring them with the patient and/or caregiver, both when initiating and renewing treatment with fentanyl transdermal system, especially if the patient has additional risk factors for overdose, or close contacts at risk for exposure and overdose. ( 2.2 , 5.1 , 5.2 , 5.4 ) Initial dose selection: consult conversion instructions. ( 2.3 ) Periodically reassess patients receiving fentanyl transdermal system to evaluate the continued need for opioid analgesics to maintain pain control, for the signs or symptoms of adverse reactions, and for the development of addiction, abuse, or misuse ( 2.4 ) Each transdermal system is intended to be worn continuously for up to 72 hours. ( 2.3 , 2.7) Adhere to instructions concerning administration and disposal of fentanyl transdermal system. ( 2.7 , 2.8 ) Mild to moderate hepatic and renal impairment: Initiate treatment with one half the usual starting dose, titrate slowly, and regularly evaluate for signs of respiratory and central nervous system depression. ( 2.5 , 2.6 ) Do not rapidly reduce or discontinue fentanyl transdermal system in a physically-dependent patient because rapid reduction or abrupt discontinuation of opioid analgesics has resulted in serious withdrawal symptoms, uncontrolled pain, and suicide. ( 2.9 , 5.21 )
Warnings and Precautions
.
Contraindications
Fentanyl transdermal system is contraindicated in: patients who are not opioid-tolerant. the management of acute or intermittent pain, or in patients who require opioid analgesia for a short period of time. the management of post-operative pain, including use after outpatient or day surgeries, (e.g., tonsillectomies). the management of mild pain. patients with significant respiratory depression [see Warnings and Precautions ( 5.12 ) ]. patients with acute or severe bronchial asthma in an unmonitored setting or in the absence of resuscitative equipment [see Warnings and Precautions ( 5.12 ) ]. patients with known or suspected gastrointestinal obstruction, including paralytic ileus [see Warnings and Precautions ( 5.19 ) ]. patients with hypersensitivity to fentanyl (e.g., anaphylaxis) or any components of the transdermal system [see Adverse Reactions ( 6.2 ) ].
Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling: Addiction, Abuse, and Misuse [see Warnings and Precautions ( 5.1 )] Life-Threatening Respiratory Depression [see Warnings and Precautions ( 5.2 )] Accidental Exposure [see Warnings and Precautions ( 5.3 )] Interactions with Benzodiazepines or Other Central Nervous System Depressants [see Warnings and Precautions ( 5.4 )] Neonatal Opioid Withdrawal Syndrome [see Warnings and Precautions ( 5.5 )] Opioid-Induced Hyperalgesia and Allodynia [see Warnings and Precautions ( 5.10 )] Serotonin Syndrome [see Warnings and Precautions ( 5.11 )] Adrenal Insufficiency [see Warnings and Precautions ( 5.13 )] Severe Hypotension [see Warnings and Precautions ( 5.14 )] Gastrointestinal Adverse Reactions [see Warnings and Precautions ( 5.19 )] Seizures [see Warnings and Precautions ( 5.20 )] Withdrawal [see Warnings and Precautions ( 5.21 )]
Drug Interactions
Table 6 includes clinically significant drug interactions with fentanyl transdermal system. Table 6: Clinically Significant Drug Interactions with Fentanyl Transdermal System Inhibitors of CYP3A4 Clinical Impact: The concomitant use of fentanyl transdermal system and CYP3A4 inhibitors can increase the plasma concentration of fentanyl, resulting in increased or prolonged opioid effects particularly when an inhibitor is added after a stable dose of fentanyl transdermal system is achieved [see Warnings and Precautions ( 5.7 ) ]. After stopping a CYP3A4 inhibitor, as the effects of the inhibitor decline, the fentanyl transdermal system plasma concentration will decrease [see Clinical Pharmacology ( 12.3 ) ], resulting in decreased opioid efficacy or a withdrawal syndrome in patients who had developed physical dependence to fentanyl. Intervention: If concomitant use is necessary, consider dosage reduction of fentanyl transdermal system until stable drug effects are achieved. Evaluate patients at frequent intervals for respiratory depression and sedation. If a CYP3A4 inhibitor is discontinued, consider increasing the fentanyl transdermal system dosage until stable drug effects are achieved. Evaluate for signs of opioid withdrawal. Examples: Macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g. ketoconazole), protease inhibitors (e.g., ritonavir), grapefruit juice. CYP3A4 Inducers Clinical Impact: The concomitant use of fentanyl transdermal system and CYP3A4 inducers can decrease the plasma concentration of fentanyl [see Clinical Pharmacology ( 12.3 ) ], resulting in decreased efficacy or onset of a withdrawal syndrome in patients who have developed physical dependence to fentanyl [see Warnings and Precautions ( 5.7 ) ]. After stopping a CYP3A4 inducer, as the effects of the inducer decline, the fentanyl plasma concentration will increase [see Clinical Pharmacology ( 12.3 )] , which could increase or prolong both the therapeutic effects and adverse reactions, and may cause serious respiratory depression. Intervention: If concomitant use is necessary, consider increasing the fentanyl transdermal system dosage until stable drug effects are achieved. Evaluate for signs of opioid withdrawal. If a CYP3A4 inducer is discontinued, consider fentanyl transdermal system dosage reduction and evaluate patients at frequent intervals for signs of respiratory depression and sedation. Examples: Rifampin, carbamazepine, phenytoin Benzodiazepines and Other Central Nervous System (CNS) Depressants Clinical Impact: Due to additive pharmacologic effect, the concomitant use of benzodiazepines or other CNS depressants, including alcohol, can increase the risk of hypotension, respiratory depression, profound sedation, coma, and death. Intervention: Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Inform patients and caregivers of this potential interaction, educate them on the signs and symptoms of respiratory depression (including sedation). If concomitant use is warranted, consider recommending or prescribing an opioid overdose reversal agent [see Dosage and Administration ( 2.2 ), Warnings and Precautions ( 5.1 , 5.2 , 5.4 )] . Examples: Benzodiazepines and other sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, gabapentinoids (gabapentin or pregabalin), other opioids, alcohol. Serotonergic Drugs Clinical Impact: The concomitant use of opioids with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome [see Warnings and Precautions ( 5.11 ) ]. Intervention: If concomitant use is warranted, frequently evaluate the patient, particularly during treatment initiation and dose adjustment. Discontinue fentanyl transdermal system immediately if serotonin syndrome is suspected. Examples: Selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that affect the serotonin neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), monoamine oxidase (MAO) inhibitors (those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue). Monoamine Oxidase Inhibitors (MAOIs) Clinical Impact: MAOI interactions with opioids may manifest as serotonin syndrome [see Warnings and Precautions ( 5.11 ) ] or opioid toxicity (e.g., respiratory depression, coma). Intervention: The use of fentanyl transdermal system is not recommended for patients taking MAOIs or within 14 days of stopping such treatment. Examples: phenelzine, tranylcypromine, linezolid Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics Clinical Impact: May reduce the analgesic effect of fentanyl transdermal system and/or precipitate withdrawal symptoms. Intervention: Avoid concomitant use. Examples: butorphanol, nalbuphine, pentazocine, buprenorphine Muscle Relaxants Clinical Impact: Fentanyl transdermal system may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression. Intervention: Because respiratory depression may be greater than otherwise expected, decrease the dosage of fentanyl transdermal system and/or the muscle relaxant as necessary. Due to the risk of respiratory depression with concomitant use of skeletal muscle relaxants and opioids, consider prescribing an opioid overdose reversal agent [see Dosage and Administration ( 2.2 ), Warnings and Precautions ( 5.2 , 5.4 )] . Examples: cyclobenzaprine, metaxalone Diuretics Clinical Impact: Opioids can reduce the efficacy of diuretics by inducing the release of antidiuretic hormone. Intervention: Evaluate patients for signs of diminished diuresis and/or effects on blood pressure and increase the dosage of the diuretic as needed. Anticholinergic Drugs Clinical Impact: The concomitant use of anticholinergic drugs may increase risk of urinary retention and/or severe constipation, which may lead to paralytic ileus. Intervention: Evaluate patients for signs of urinary retention or reduced gastric motility when fentanyl transdermal system is used concomitantly with anticholinergic drugs.
Storage and Handling
Fentanyl transdermal system is supplied in cartons containing 5 individually packaged systems. See chart for information regarding individual systems. * This lowest strength is designated as 12 mcg/hour (however, the actual strength is 12.5 mcg/hour) to distinguish it from a 125 mcg/hour strength that could be prescribed by using multiple transdermal systems. Fentanyl Transdermal System Strength (mcg/hour) System Size (cm 2 ) Fentanyl Content (mg) NDC Number Fentanyl transdermal system – 12 * 4.72 1.33 47781-423-47 Fentanyl transdermal system – 25 9.44 2.66 47781-424-47 Fentanyl transdermal system – 50 18.88 5.32 47781-426-47 Fentanyl transdermal system – 75 28.32 7.99 47781-427-47 Fentanyl transdermal system – 100 37.76 10.65 47781-428-47 Store in original unopened pouch. Store up to 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature]. Store fentanyl transdermal system securely and dispose of properly [see Patient Counseling Information ( 17 ) ].
How Supplied
Fentanyl transdermal system is supplied in cartons containing 5 individually packaged systems. See chart for information regarding individual systems. * This lowest strength is designated as 12 mcg/hour (however, the actual strength is 12.5 mcg/hour) to distinguish it from a 125 mcg/hour strength that could be prescribed by using multiple transdermal systems. Fentanyl Transdermal System Strength (mcg/hour) System Size (cm 2 ) Fentanyl Content (mg) NDC Number Fentanyl transdermal system – 12 * 4.72 1.33 47781-423-47 Fentanyl transdermal system – 25 9.44 2.66 47781-424-47 Fentanyl transdermal system – 50 18.88 5.32 47781-426-47 Fentanyl transdermal system – 75 28.32 7.99 47781-427-47 Fentanyl transdermal system – 100 37.76 10.65 47781-428-47 Store in original unopened pouch. Store up to 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature]. Store fentanyl transdermal system securely and dispose of properly [see Patient Counseling Information ( 17 ) ].
Medication Information
Warnings and Precautions
.
Indications and Usage
Fentanyl transdermal system is indicated for the management of severe and persistent pain in opioid-tolerant patients, that requires an opioid analgesic and that cannot be adequately treated with alternative options, including immediate-release opioids. Patients considered opioid-tolerant are those who are taking, for one week or longer, at least 60 mg morphine per day, 25 mcg transdermal fentanyl per hour, 30 mg oral oxycodone per day, 8 mg oral hydromorphone per day, 25 mg oral oxymorphone per day, 60 mg oral hydrocodone per day, or an equianalgesic dose of another opioid. Limitations of Use Because of the risks of addiction, abuse, misuse, overdose, and death, which can occur at any dosage or duration and persist over the course of therapy [see Warnings and Precautions ( 5.1 ) ], reserve opioid analgesics, including fentanyl transdermal system, for use in patients for whom alternative treatment options are ineffective, not tolerated, or would be otherwise inadequate to provide sufficient management of pain. Fentanyl transdermal system is not indicated as an as-needed (prn) analgesic.
Dosage and Administration
Fentanyl transdermal system should be prescribed only by healthcare providers who are knowledgeable about the use of extended-release/long-acting opioids and how to mitigate the associated risks. ( 2.1 ) Use the lowest effective dosage for the shortest duration of time consistent with individual patient treatment goals. Reserve titration to higher doses of fentanyl transdermal system for patients in whom lower doses are insufficiently effective and in whom the expected benefits of using a higher dose opioid clearly outweigh the substantial risks. ( 2.1 , 5 ) Initiate the dosing regimen for each patient individually, taking into account the patient's underlying cause and severity of pain, prior analgesic treatment and response, and risk factors for addiction, abuse, and misuse. ( 5.1 ) Respiratory depression can occur at any time during opioid therapy, especially when initiating and following dosage increases with fentanyl transdermal system. Consider this risk when selecting an initial dose and when making dose adjustments. ( 2.1 , 5.2 ) Discuss opioid overdose reversal agents and options for acquiring them with the patient and/or caregiver, both when initiating and renewing treatment with fentanyl transdermal system, especially if the patient has additional risk factors for overdose, or close contacts at risk for exposure and overdose. ( 2.2 , 5.1 , 5.2 , 5.4 ) Initial dose selection: consult conversion instructions. ( 2.3 ) Periodically reassess patients receiving fentanyl transdermal system to evaluate the continued need for opioid analgesics to maintain pain control, for the signs or symptoms of adverse reactions, and for the development of addiction, abuse, or misuse ( 2.4 ) Each transdermal system is intended to be worn continuously for up to 72 hours. ( 2.3 , 2.7) Adhere to instructions concerning administration and disposal of fentanyl transdermal system. ( 2.7 , 2.8 ) Mild to moderate hepatic and renal impairment: Initiate treatment with one half the usual starting dose, titrate slowly, and regularly evaluate for signs of respiratory and central nervous system depression. ( 2.5 , 2.6 ) Do not rapidly reduce or discontinue fentanyl transdermal system in a physically-dependent patient because rapid reduction or abrupt discontinuation of opioid analgesics has resulted in serious withdrawal symptoms, uncontrolled pain, and suicide. ( 2.9 , 5.21 )
Contraindications
Fentanyl transdermal system is contraindicated in: patients who are not opioid-tolerant. the management of acute or intermittent pain, or in patients who require opioid analgesia for a short period of time. the management of post-operative pain, including use after outpatient or day surgeries, (e.g., tonsillectomies). the management of mild pain. patients with significant respiratory depression [see Warnings and Precautions ( 5.12 ) ]. patients with acute or severe bronchial asthma in an unmonitored setting or in the absence of resuscitative equipment [see Warnings and Precautions ( 5.12 ) ]. patients with known or suspected gastrointestinal obstruction, including paralytic ileus [see Warnings and Precautions ( 5.19 ) ]. patients with hypersensitivity to fentanyl (e.g., anaphylaxis) or any components of the transdermal system [see Adverse Reactions ( 6.2 ) ].
Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling: Addiction, Abuse, and Misuse [see Warnings and Precautions ( 5.1 )] Life-Threatening Respiratory Depression [see Warnings and Precautions ( 5.2 )] Accidental Exposure [see Warnings and Precautions ( 5.3 )] Interactions with Benzodiazepines or Other Central Nervous System Depressants [see Warnings and Precautions ( 5.4 )] Neonatal Opioid Withdrawal Syndrome [see Warnings and Precautions ( 5.5 )] Opioid-Induced Hyperalgesia and Allodynia [see Warnings and Precautions ( 5.10 )] Serotonin Syndrome [see Warnings and Precautions ( 5.11 )] Adrenal Insufficiency [see Warnings and Precautions ( 5.13 )] Severe Hypotension [see Warnings and Precautions ( 5.14 )] Gastrointestinal Adverse Reactions [see Warnings and Precautions ( 5.19 )] Seizures [see Warnings and Precautions ( 5.20 )] Withdrawal [see Warnings and Precautions ( 5.21 )]
Drug Interactions
Table 6 includes clinically significant drug interactions with fentanyl transdermal system. Table 6: Clinically Significant Drug Interactions with Fentanyl Transdermal System Inhibitors of CYP3A4 Clinical Impact: The concomitant use of fentanyl transdermal system and CYP3A4 inhibitors can increase the plasma concentration of fentanyl, resulting in increased or prolonged opioid effects particularly when an inhibitor is added after a stable dose of fentanyl transdermal system is achieved [see Warnings and Precautions ( 5.7 ) ]. After stopping a CYP3A4 inhibitor, as the effects of the inhibitor decline, the fentanyl transdermal system plasma concentration will decrease [see Clinical Pharmacology ( 12.3 ) ], resulting in decreased opioid efficacy or a withdrawal syndrome in patients who had developed physical dependence to fentanyl. Intervention: If concomitant use is necessary, consider dosage reduction of fentanyl transdermal system until stable drug effects are achieved. Evaluate patients at frequent intervals for respiratory depression and sedation. If a CYP3A4 inhibitor is discontinued, consider increasing the fentanyl transdermal system dosage until stable drug effects are achieved. Evaluate for signs of opioid withdrawal. Examples: Macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g. ketoconazole), protease inhibitors (e.g., ritonavir), grapefruit juice. CYP3A4 Inducers Clinical Impact: The concomitant use of fentanyl transdermal system and CYP3A4 inducers can decrease the plasma concentration of fentanyl [see Clinical Pharmacology ( 12.3 ) ], resulting in decreased efficacy or onset of a withdrawal syndrome in patients who have developed physical dependence to fentanyl [see Warnings and Precautions ( 5.7 ) ]. After stopping a CYP3A4 inducer, as the effects of the inducer decline, the fentanyl plasma concentration will increase [see Clinical Pharmacology ( 12.3 )] , which could increase or prolong both the therapeutic effects and adverse reactions, and may cause serious respiratory depression. Intervention: If concomitant use is necessary, consider increasing the fentanyl transdermal system dosage until stable drug effects are achieved. Evaluate for signs of opioid withdrawal. If a CYP3A4 inducer is discontinued, consider fentanyl transdermal system dosage reduction and evaluate patients at frequent intervals for signs of respiratory depression and sedation. Examples: Rifampin, carbamazepine, phenytoin Benzodiazepines and Other Central Nervous System (CNS) Depressants Clinical Impact: Due to additive pharmacologic effect, the concomitant use of benzodiazepines or other CNS depressants, including alcohol, can increase the risk of hypotension, respiratory depression, profound sedation, coma, and death. Intervention: Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Inform patients and caregivers of this potential interaction, educate them on the signs and symptoms of respiratory depression (including sedation). If concomitant use is warranted, consider recommending or prescribing an opioid overdose reversal agent [see Dosage and Administration ( 2.2 ), Warnings and Precautions ( 5.1 , 5.2 , 5.4 )] . Examples: Benzodiazepines and other sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, gabapentinoids (gabapentin or pregabalin), other opioids, alcohol. Serotonergic Drugs Clinical Impact: The concomitant use of opioids with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome [see Warnings and Precautions ( 5.11 ) ]. Intervention: If concomitant use is warranted, frequently evaluate the patient, particularly during treatment initiation and dose adjustment. Discontinue fentanyl transdermal system immediately if serotonin syndrome is suspected. Examples: Selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that affect the serotonin neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), monoamine oxidase (MAO) inhibitors (those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue). Monoamine Oxidase Inhibitors (MAOIs) Clinical Impact: MAOI interactions with opioids may manifest as serotonin syndrome [see Warnings and Precautions ( 5.11 ) ] or opioid toxicity (e.g., respiratory depression, coma). Intervention: The use of fentanyl transdermal system is not recommended for patients taking MAOIs or within 14 days of stopping such treatment. Examples: phenelzine, tranylcypromine, linezolid Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics Clinical Impact: May reduce the analgesic effect of fentanyl transdermal system and/or precipitate withdrawal symptoms. Intervention: Avoid concomitant use. Examples: butorphanol, nalbuphine, pentazocine, buprenorphine Muscle Relaxants Clinical Impact: Fentanyl transdermal system may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression. Intervention: Because respiratory depression may be greater than otherwise expected, decrease the dosage of fentanyl transdermal system and/or the muscle relaxant as necessary. Due to the risk of respiratory depression with concomitant use of skeletal muscle relaxants and opioids, consider prescribing an opioid overdose reversal agent [see Dosage and Administration ( 2.2 ), Warnings and Precautions ( 5.2 , 5.4 )] . Examples: cyclobenzaprine, metaxalone Diuretics Clinical Impact: Opioids can reduce the efficacy of diuretics by inducing the release of antidiuretic hormone. Intervention: Evaluate patients for signs of diminished diuresis and/or effects on blood pressure and increase the dosage of the diuretic as needed. Anticholinergic Drugs Clinical Impact: The concomitant use of anticholinergic drugs may increase risk of urinary retention and/or severe constipation, which may lead to paralytic ileus. Intervention: Evaluate patients for signs of urinary retention or reduced gastric motility when fentanyl transdermal system is used concomitantly with anticholinergic drugs.
Storage and Handling
Fentanyl transdermal system is supplied in cartons containing 5 individually packaged systems. See chart for information regarding individual systems. * This lowest strength is designated as 12 mcg/hour (however, the actual strength is 12.5 mcg/hour) to distinguish it from a 125 mcg/hour strength that could be prescribed by using multiple transdermal systems. Fentanyl Transdermal System Strength (mcg/hour) System Size (cm 2 ) Fentanyl Content (mg) NDC Number Fentanyl transdermal system – 12 * 4.72 1.33 47781-423-47 Fentanyl transdermal system – 25 9.44 2.66 47781-424-47 Fentanyl transdermal system – 50 18.88 5.32 47781-426-47 Fentanyl transdermal system – 75 28.32 7.99 47781-427-47 Fentanyl transdermal system – 100 37.76 10.65 47781-428-47 Store in original unopened pouch. Store up to 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature]. Store fentanyl transdermal system securely and dispose of properly [see Patient Counseling Information ( 17 ) ].
How Supplied
Fentanyl transdermal system is supplied in cartons containing 5 individually packaged systems. See chart for information regarding individual systems. * This lowest strength is designated as 12 mcg/hour (however, the actual strength is 12.5 mcg/hour) to distinguish it from a 125 mcg/hour strength that could be prescribed by using multiple transdermal systems. Fentanyl Transdermal System Strength (mcg/hour) System Size (cm 2 ) Fentanyl Content (mg) NDC Number Fentanyl transdermal system – 12 * 4.72 1.33 47781-423-47 Fentanyl transdermal system – 25 9.44 2.66 47781-424-47 Fentanyl transdermal system – 50 18.88 5.32 47781-426-47 Fentanyl transdermal system – 75 28.32 7.99 47781-427-47 Fentanyl transdermal system – 100 37.76 10.65 47781-428-47 Store in original unopened pouch. Store up to 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature]. Store fentanyl transdermal system securely and dispose of properly [see Patient Counseling Information ( 17 ) ].
Description
Boxed Warning 12/2025 Indications and Usage ( 1 ) 12/2025 Dosage and Administration ( 2.2 , 2.3 ) 12/2025 Warnings and Precautions ( 5.1, 5.2, 5.4, 5.19 , 5.21 ) 12/2025
Section 42229-5
Addiction, Abuse, and Misuse
Because the use of fentanyl transdermal system exposes patients and other users to the risks of opioid addiction, abuse, and misuse, which can lead to overdose and death, assess each patient's risk prior to prescribing and reassess all patients regularly for the development of these behaviors and conditions [see Warnings and Precautions ( 5.1) ].
Section 42231-1
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: 12/2025 |
|
|
Medication Guide Fentanyl (fen' ta nil) Transdermal System, CII |
|
Fentanyl Transdermal System is:
|
|
Important information about Fentanyl Transdermal System:
|
|
Do not use Fentanyl Transdermal System if you have:
|
|
| Before applying Fentanyl Transdermal System, tell your healthcare provider if you have a history of: | |
|
|
|
|
Tell your healthcare provider if you:
|
|
When using Fentanyl Transdermal System:
|
|
While using Fentanyl Transdermal System DO NOT:
|
|
The possible side effects of Fentanyl Transdermal System are:
|
|
Get emergency medical help or call 911 right away if you have:
|
|
| These are not all the possible side effects of fentanyl transdermal system. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
For more information go to dailymed.nlm.nih.gov.
Manufactured by: Kindeva Drug Delivery L.P., Northridge, CA 91324 USA; Distributed by: Alvogen, Inc., Morristown, NJ 07960 USA, www.Alvogen.com or call Alvogen, Inc. at 1-866-770-3024. |
Section 43683-2
| Boxed Warning | 12/2025 |
| Indications and Usage ( 1) | 12/2025 |
| Dosage and Administration ( 2.2, 2.3) | 12/2025 |
| Warnings and Precautions ( 5.1, 5.2, 5.4, 5.19, 5.21) | 12/2025 |
Section 51945-4
Principal Display Panel - 12 mcg/h Patch Carton
12
mcg/h
NDC47781- 423-47
FENTANYL Transdermal System CII
In vivodelivery of 12 mcg/h fentanyl for 72 hours
Because serious or life-threatening breathing problems could
result, DO NOT USE FENTANYL TRANSDERMAL SYSTEM:
-
for pain that can be treated with immediate-release opioids or
non-opioid analgesics - for intermittent (on an as-needed basis) pain
- for any postoperative pain
-
unless you are opioid tolerant (have been using other narcotic
opioid medicines)
Each transdermal system contains:1.33 mg fentanyl
DO NOT USE IF SEAL ON POUCH IS BROKEN
KEEP OUT OF REACH OF CHILDREN
Read enclosed Medication Guide for important safety information.
Rx Only
Alvogen ®
five (12 mcg/h) systems
For Opioid-Tolerant
Patients Only
Section 59845-8
Instructions for Use
Fentanyl (fen' ta nil) Transdermal System CII
Be sure that you read, understand, and follow these Instructions for Use before you apply fentanyl transdermal system (patch). Talk to your healthcare provider or pharmacist if you have any questions.
Important information about the fentanyl transdermal system (patch) appearance:
- Fentanyl transdermal system is a rectangular, see-through patch with rounded corners.
-
Fentanyl transdermal system comes in 5 different dosage strengths and sizes:
- 12 mcg/hour
- 25 mcg/hour
- 50 mcg/hour
- 75 mcg/hour
- 100 mcg/hour
- The product name, “FENTANYL”, and dosage strength are printed in green on each patch.
Parts of the Fentanyl Transdermal System patch:
BEFORE APPLYING FENTANYL TRANSDERMAL SYSTEM
- Each fentanyl transdermal system patch is sealed in its own protective pouch. Do not remove a fentanyl transdermal system patch from the pouch until you are ready to use it.
- Do not use a fentanyl transdermal system patch if the pouch seal is broken or the patch is cut, damaged or changed in any way.
- Fentanyl transdermal system patches are available in 5 different dosage strengths and patch sizes. Make sure you have the right dose patch or patches that have been prescribed for you.
APPLYING A FENTANYL TRANSDERMAL PATCH
|
Figure A Figure B Figure C Figure D Figure E |
|
2. Prepare to apply a fentanyl transdermal system patch:
|
|
|
3. Open the pouch: Fold and tear at slit, or cut at slit taking care not to cut the patch. Remove the fentanyl transdermal system patch. Each fentanyl transdermal system patch is sealed in its own protective pouch. Do not remove the fentanyl transdermal system patch from the pouch until you are ready to use it (See Figure F). 4. Peel:Peel off both parts of the release liner from the patch. Each fentanyl transdermal system patch has a clear plastic release liner that can be peeled off in two pieces. This covers the sticky side of the patch. Carefully peel this release liner off and throw the pieces away. Touch the sticky side of the fentanyl transdermal system patch as little as possible(See Figure G). |
Figure F Figure G |
|
5. Press:Press the patch onto the chosen skin site with the palm of your hand and hold there for at least 30 seconds(See Figure H). Make sure it sticks well, especially at the edges.
6. Wash your hands when you have finished applying a fentanyl transdermal system patch. 7. Remove a fentanyl transdermal system patch after wearing it for 3 days (72 hours).
Dispose of the used patch right away. See the section below “
Disposing of a fentanyl transdermal system patch.”
Choose a
differentskin site to apply a new fentanyl transdermal system patch. Repeat Steps 2 through 6 above when applying a new fentanyl transdermal system patch.
DISPOSING OF A FENTANYL TRANSDERMAL SYSTEM PATCH
|
Figure H Figure I Figure J |
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by:
Kindeva Drug Delivery L.P.
Northridge, CA 91324 USA
Distributed by:
Alvogen, Inc.
Morristown, NJ 07960 USA
Revised: 12/2025
3200001799
Bioclusive™ is a trademark of Ethicon, Inc.
Tegaderm™ is a trademark of 3M
9.2 Abuse
Fentanyl transdermal system contains fentanyl, a substance with high potential for misuse and abuse, which can lead to the development of substance use disorder, including addiction [see Warnings and Precautions ( 5.1) ].
Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a healthcare provider or for whom it was not prescribed.
Abuse is the intentional, non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects.
Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance, or physical dependence.
Misuse and abuse of fentanyl transdermal system increases risk of overdose, which may lead to central nervous system and respiratory depression, hypotension, seizures, and death. The risk is increased with concurrent use of fentanyl transdermal system with alcohol and/or other CNS depressants. Abuse of and addiction to opioids in some individuals may not be accompanied by concurrent tolerance and symptoms of physical dependence. In addition, abuse of opioids can occur in the absence of addiction.
All patients treated with opioids require careful and frequent reevaluation for signs of misuse, abuse, and addiction, because use of opioid analgesic products carries the risk of addiction even under appropriate medical use. Patients at high risk of fentanyl transdermal system abuse include those with a history of prolonged use of any opioid, including products containing fentanyl, those with a history of drug or alcohol abuse, or those who use fentanyl transdermal system in combination with other abused drugs.
“Drug seeking” behavior is very common in persons with substance use disorders. Drug-seeking tactics include emergency calls or visits near the end of office hours, refusal to undergo appropriate examination, testing, or referral, repeated “loss” of prescriptions, tampering with prescriptions, and reluctance to provide prior medical records or contact information for other treating healthcare provider(s). “Doctor shopping” (visiting multiple prescribers to obtain additional prescriptions) is common among people who abuse drugs and people with substance use disorder. Preoccupation with achieving adequate pain relief can be appropriate behavior in a patient with inadequate pain control.
Fentanyl transdermal system, like other opioids, can be diverted for nonmedical use into illicit channels of distribution. Careful record-keeping of prescribing information, including quantity, frequency, and renewal requests, as required by state and federal law, is strongly advised.
Proper assessment of the patient, proper prescribing practices, periodic reevaluation of therapy, and proper dispensing and storage are appropriate measures that help to limit abuse of opioid drugs.
10 Overdosage
.
8.1 Pregnancy
.
11 Description
The system contains fentanyl, an opioid agonist, for transdermal administration. The amount of fentanyl released from each system per hour is proportional to the surface area (25 mcg/hour per 9.44 cm 2). The composition per unit area of all transdermal system sizes is identical.
| *Nominal delivery rate is 12.5 mcg/hour per hour | ||
| **Nominal delivery rate per hour | ||
|
Strength
(mcg/hour) |
Size
(cm²) |
Fentanyl Content
(mg) |
| 12* | 4.72 | 1.33 |
| 25** | 9.44 | 2.66 |
| 50** | 18.88 | 5.32 |
| 75** | 28.32 | 7.99 |
| 100** | 37.76 | 10.65 |
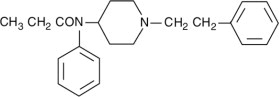
The molecular weight of fentanyl base is 336.5, and the molecular formula is C 22H 28N 2O. The n-octanol: water partition coefficient is 860:1. The pKa is 8.4.
The chemical name is N-Phenyl-N-(1-(2-phenylethyl)-4-piperidinyl) propanamide. The structural formula is:
Fentanyl transdermal system is a rectangular translucent system with rounded corners. The product name, “FENTANYL” and dosage strength are printed in green on each system.
Each system is comprised of a clear polyethylene terephthalate (PET) release liner and two functional layers. Proceeding from the outer surface toward the surface adhering to skin, these functional layers are:
1) a translucent backing layer of ethylene vinyl acetate/polyethylene terephthalate (EVA/PET) film with green print; 2) a drug-in-adhesive layer containing fentanyl, acrylate copolymer adhesive, and methyl laurate. Before use, a clear PET release liner covering the drug-in-adhesive layer is removed and discarded.
9.3 Dependence
Both tolerance and physical dependence can develop during use of opioid therapy.
Tolerance is a physiological state characterized by a reduced response to a drug after repeated administration (i.e., a higher dose of a drug is required to produce the same effect that was once obtained at a lower dose).
Physical dependence is a state that develops as a result of a physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug.
Withdrawal may be precipitated through the administration of drugs with opioid antagonist activity (e.g., naloxone, nalmefene), mixed agonist/antagonist analgesics (e.g., pentazocine, butorphanol, nalbuphine), or partial agonists (e.g., buprenorphine). Physical dependence may not occur to a clinically significant degree until after several days to weeks of continued use.
Do not rapidly reduce or abruptly discontinue fentanyl transdermal system in a patient physically dependent on opioids. Rapid reduction or abrupt discontinuation of fentanyl transdermal system in a patient physically dependent on opioids may lead to serious withdrawal symptoms, uncontrolled pain, and suicide. Rapid reduction or abrupt discontinuation has also been associated with attempts to find other sources of opioid analgesics, which may be confused with drug-seeking for abuse.
When discontinuing fentanyl transdermal system, gradually taper the dosage using a patient-specific plan that considers the following: the dose of fentanyl transdermal system the patient has been taking, the duration of treatment, and the physical and psychological attributes of the patient. To improve the likelihood of a successful taper and minimize withdrawal symptoms, it is important that the opioid tapering schedule is agreed upon by the patient. In patients taking opioids for an extended period of time at high doses, ensure that a multimodal approach to pain management, including mental health support (if needed), is in place prior to initiating an opioid analgesic taper [see Dosage and Administration ( 2.9), and Warnings and Precautions ( 5.21) ].
Infants born to mothers physically dependent on opioids will also be physically dependent and may exhibit respiratory difficulties and withdrawal signs [see Use in Specific Populations ( 8.1) ].
5.21 Withdrawal
Do not rapidly reduce or abruptly discontinue fentanyl transdermal system in a patient physically dependent on opioids. When discontinuing fentanyl transdermal system in a physically dependent patient, gradually taper the dosage. Rapid tapering of fentanyl transdermal system in a patient physically dependent on opioids may lead to a withdrawal syndrome and return of pain [see Dosage and Administration ( 2.9), Drug Abuse and Dependence ( 9.3) ].
Additionally, avoid the use of mixed agonist/antagonist (e.g., pentazocine, nalbuphine, and butorphanol) or partial agonist (e.g., buprenorphine) analgesics in patients who are receiving a full opioid agonist analgesic, including fentanyl transdermal system. In these patients, mixed agonist/antagonist and partial agonist analgesics may reduce the analgesic effect and/or may precipitate withdrawal symptoms [see Drug Interactions ( 7) ].
8.4 Pediatric Use
The safety of fentanyl transdermal system was evaluated in three open-label trials in 289 pediatric patients with chronic pain, 2 years of age through 18 years of age. Starting doses of 25 mcg/h and higher were used by 181 patients who had been on prior daily opioid doses of at least 45 mg/day of oral morphine or an equianalgesic dose of another opioid. Initiation of fentanyl transdermal system therapy in pediatric patients taking less than 60 mg/day of oral morphine or an equianalgesic dose of another opioid has not been evaluated in controlled clinical trials.
The safety and effectiveness of fentanyl transdermal system in children under 2 years of age have not been established.
To guard against excessive exposure to fentanyl transdermal system by young children, advise caregivers to strictly adhere to recommended fentanyl transdermal system application and disposal instructions [see Dosage and Administration ( 2.7), ( 2.8) and Warnings and Precautions ( 5.3) ].
8.5 Geriatric Use
Clinical studies of fentanyl transdermal system did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, use caution when selecting a dosage for an elderly patient, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Data from intravenous studies with fentanyl suggest that the elderly patients may have reduced clearance and a prolonged half-life. Moreover, elderly patients may be more sensitive to the active substance than younger patients. A study conducted with the fentanyl transdermal system patch in elderly patients demonstrated that fentanyl pharmacokinetics did not differ significantly from young adult subjects, although peak serum concentrations tended to be lower and mean half-life values were prolonged to approximately 34 hours [see Clinical Pharmacology ( 12.3) ].
Respiratory depression is the chief risk for elderly patients treated with opioids, and has occurred after large initial doses were administered to patients who were not opioid-tolerant or when opioids were co-administered with other agents that depress respiration. Titrate the dosage of fentanyl transdermal system slowly in geriatric patients and frequently reevaluate the patient for signs of central nervous system and respiratory depression [see Warnings and Precautions ( 5.12) ].
Fentanyl is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
2.3 Initial Dosage
It is safer to underestimate a patient's 24-hour fentanyl dosage and provide rescue medication (e.g. immediate-release opioid) than to overestimate the 24-hour fentanyl dosage and manage an adverse reaction to an overdose. While useful tables of opioid equivalents are readily available, there is inter-patient variability in the potency of opioid drugs and opioid formulations. Frequently reevaluate patients for signs and symptoms of opioid withdrawal and for signs of oversedation/toxicity after converting patients to fentanyl transdermal system.
- Do not initiate treatment with fentanyl transdermal system in opioid nontolerant patients [see Contraindications ( 4)] .
- The recommended starting dose when converting from other opioids to fentanyl transdermal system is intended to minimize the potential for overdosing patients with the first dose.
- When fentanyl transdermal system therapy is initiated, discontinue all opioid analgesics other than those used on an as needed basis for breakthrough pain when appropriate.
- Each fentanyl transdermal system is worn continuously for up to 72 hours [see Dosage and Administration (2.7)].
In a fentanyl transdermal system clinical trial, patients were converted from their prior opioid to fentanyl transdermal system using Table 1as a guide for the initial fentanyl transdermal system dose.
When converting patients from oral or parenteral opioids to fentanyl transdermal system, use Table 1(alternatively use Table 2for adult and pediatric patients taking opioids or doses not listed in Table 1) and consider the following:
- These are not tables of equianalgesic doses.
- The conversion doses in these tables are only for the conversion from one of the listed oral or parenteral opioid analgesics to fentanyl transdermal system.
- The tables cannot be used to convert from fentanyl transdermal system toanother opioid because these conversions will result in an overestimation of the dose of the new opioid (these conversions are conservative) and may result in fatal overdosage.
| 1 Table 1should not be used to convert from fentanyl transdermal system to other therapies because this conversion to fentanyl transdermal system is conservative. Use of Table 1for conversion to other analgesic therapies can overestimate the dose of the new agent. Overdosage of the new analgesic agent is possible [see Dosage and Administration ( 2.9) ]. | ||||
| Current Analgesic | Daily Dosage (mg/day) | |||
| Oral morphine | 60-134 | 135-224 | 225-314 | 315-404 |
| Intramuscular or Intravenous morphine | 10-22 | 23-37 | 38-52 | 53-67 |
| Oral oxycodone | 30-67 | 67.5-112 | 112.5-157 | 157.5-202 |
| Oral codeine | 150-447 | |||
| Oral hydromorphone | 8-17 | 17.1-28 | 28.1-39 | 39.1-51 |
| Intravenous hydromorphone | 1.5-3.4 | 3.5-5.6 | 5.7-7.9 | 8-10 |
| Intramuscular meperidine | 75-165 | 166-278 | 279-390 | 391-503 |
| Oral methadone | 20-44 | 45-74 | 75-104 | 105-134 |
| ⇓ | ⇓ | ⇓ | ⇓ | |
| Recommended fentanyl transdermal system dose | 25 mcg/hour | 50 mcg/hour | 75 mcg/hour | 100 mcg/hour |
Alternatively, for adult and pediatric patients taking opioids or doses not listed in Table 1, use the following methodology when converting patients from oral or parenteral opioids to fentanyl transdermal system:
- Calculate the previous 24-hour analgesic requirement.
- Convert this amount to the equianalgesic oral morphine dose using a reliable reference.
- Refer to Table 2for the range of 24-hour oral morphine doses that are recommended for conversion to each fentanyl transdermal system dose. Use this table to find the calculated 24-hour morphine dose and the corresponding recommended initial fentanyl transdermal system dose.
- Initiate fentanyl transdermal system treatment using the recommended dose and titrate patients upwards (no more frequently than 3 days after the initial dose and every 6 days thereafter) until analgesic efficacy is attained. A 37.5 mcg/hour dose may also be used. For patients that require more than 100 mcg/hour, several transdermal systems may be used.
- Do not use Table 2to convert from fentanyl transdermal system to other therapies because this conversion to fentanyl transdermal system is conservative and will overestimate the dose of the new agent.
| NOTE:In clinical trials, these ranges of daily oral morphine doses were used as a basis for conversion to fentanyl transdermal system. | |
| 1 Table 2should not be used to convert from fentanyl transdermal system to other therapies because this conversion to fentanyl transdermal system is conservative. Use of Table 2for conversion to other analgesic therapies can overestimate the dose of the new agent. Overdosage of the new analgesic agent is possible [see Dosage and Administration ( 2.9) ]. | |
|
Oral 24-
hour
Morphine (mg/day) |
Fentanyl Transdermal System
Dose (mcg/hour) |
| 60-134 | 25 |
| 135-224 | 50 |
| 225-314 | 75 |
| 315-404 | 100 |
| 405-494 | 125 |
| 495-584 | 150 |
| 585-674 | 175 |
| 675-764 | 200 |
| 765-854 | 225 |
| 855-944 | 250 |
| 945-1034 | 275 |
| 1035-1124 | 300 |
An additional intermediate strength 37.5 mcg/hour fentanyl transdermal system is available and may be considered during conversion from prior opioids or dose titration. For example, the 37.5 mcg/hour system could be used before converting or titrating to a 50 mcg/hour system.
The additional 37.5 mcg/hour system was not used in the clinical studies.
For delivery rates in excess of 100 mcg/hour, multiple systems may be used.
14 Clinical Studies
Fentanyl transdermal system as therapy for pain due to cancer has been studied in 153 patients. In this patient population, fentanyl transdermal system has been administered in doses of 25 mcg/h to 600 mcg/h. Individual patients have used fentanyl transdermal system continuously for up to 866 days. At one month after initiation of fentanyl transdermal system therapy, patients generally reported lower pain intensity scores as compared to a pre-study analgesic regimen of oral morphine.
The duration of fentanyl transdermal system use varied in cancer patients; 56% of patients used fentanyl transdermal system for over 30 days, 28% continued treatment for more than 4 months, and 10% used fentanyl transdermal system for more than 1 year.
In the pediatric population, the safety of fentanyl transdermal system has been evaluated in 289 patients with chronic pain 2-18 years of age. The duration of fentanyl transdermal system use varied; 20% of pediatric patients were treated for ≤15 days; 46% for 16-30 days; 16% for 31-60 days; and 17% for at least 61 days. Twenty-five patients were treated with fentanyl transdermal system for at least 4 months and 9 patients for more than 9 months.
4 Contraindications
Fentanyl transdermal system is contraindicated in:
- patients who are not opioid-tolerant.
- the management of acute or intermittent pain, or in patients who require opioid analgesia for a short period of time.
- the management of post-operative pain, including use after outpatient or day surgeries, (e.g., tonsillectomies).
- the management of mild pain.
- patients with significant respiratory depression [see Warnings and Precautions ( 5.12) ].
- patients with acute or severe bronchial asthma in an unmonitored setting or in the absence of resuscitative equipment [see Warnings and Precautions ( 5.12) ].
- patients with known or suspected gastrointestinal obstruction, including paralytic ileus [see Warnings and Precautions ( 5.19) ].
- patients with hypersensitivity to fentanyl (e.g., anaphylaxis) or any components of the transdermal system [see Adverse Reactions ( 6.2) ].
6 Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling:
- Addiction, Abuse, and Misuse [see Warnings and Precautions ( 5.1)]
- Life-Threatening Respiratory Depression [see Warnings and Precautions ( 5.2)]
- Accidental Exposure [see Warnings and Precautions ( 5.3)]
- Interactions with Benzodiazepines or Other Central Nervous System Depressants [see Warnings and Precautions ( 5.4)]
- Neonatal Opioid Withdrawal Syndrome [see Warnings and Precautions ( 5.5)]
- Opioid-Induced Hyperalgesia and Allodynia [see Warnings and Precautions ( 5.10)]
- Serotonin Syndrome [see Warnings and Precautions ( 5.11)]
- Adrenal Insufficiency [see Warnings and Precautions ( 5.13)]
- Severe Hypotension [see Warnings and Precautions ( 5.14)]
- Gastrointestinal Adverse Reactions [see Warnings and Precautions ( 5.19)]
- Seizures [see Warnings and Precautions ( 5.20)]
- Withdrawal [see Warnings and Precautions ( 5.21)]
7 Drug Interactions
Table 6includes clinically significant drug interactions with fentanyl transdermal system.
| Inhibitors of CYP3A4 | |
| Clinical Impact: | The concomitant use of fentanyl transdermal system and CYP3A4 inhibitors can increase the plasma concentration of fentanyl, resulting in increased or prolonged opioid effects particularly when an inhibitor is added after a stable dose of fentanyl transdermal system is achieved [see
Warnings and Precautions (
5.7)
].
After stopping a CYP3A4 inhibitor, as the effects of the inhibitor decline, the fentanyl transdermal system plasma concentration will decrease [see Clinical Pharmacology ( 12.3) ], resulting in decreased opioid efficacy or a withdrawal syndrome in patients who had developed physical dependence to fentanyl. |
| Intervention: | If concomitant use is necessary, consider dosage reduction of fentanyl transdermal system until stable drug effects are achieved. Evaluate patients at frequent intervals for respiratory depression and sedation.
If a CYP3A4 inhibitor is discontinued, consider increasing the fentanyl transdermal system dosage until stable drug effects are achieved. Evaluate for signs of opioid withdrawal. |
| Examples: | Macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g. ketoconazole), protease inhibitors (e.g., ritonavir), grapefruit juice. |
| CYP3A4 Inducers | |
| Clinical Impact: | The concomitant use of fentanyl transdermal system and CYP3A4 inducers can decrease the plasma concentration of fentanyl [see
Clinical Pharmacology (
12.3)
], resulting in decreased efficacy or onset of a withdrawal syndrome in patients who have developed physical dependence to fentanyl [see
Warnings and Precautions (
5.7)
].
After stopping a CYP3A4 inducer, as the effects of the inducer decline, the fentanyl plasma concentration will increase [see Clinical Pharmacology ( 12.3)] , which could increase or prolong both the therapeutic effects and adverse reactions, and may cause serious respiratory depression. |
| Intervention: | If concomitant use is necessary, consider increasing the fentanyl transdermal system dosage until stable drug effects are achieved. Evaluate for signs of opioid withdrawal.
If a CYP3A4 inducer is discontinued, consider fentanyl transdermal system dosage reduction and evaluate patients at frequent intervals for signs of respiratory depression and sedation. |
| Examples: | Rifampin, carbamazepine, phenytoin |
| Benzodiazepines and Other Central Nervous System (CNS) Depressants | |
| Clinical Impact: | Due to additive pharmacologic effect, the concomitant use of benzodiazepines or other CNS depressants, including alcohol, can increase the risk of hypotension, respiratory depression, profound sedation, coma, and death. |
| Intervention: | Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Inform patients and caregivers of this potential interaction, educate them on the signs and symptoms of respiratory depression (including sedation). If concomitant use is warranted, consider recommending or prescribing an opioid overdose reversal agent [see Dosage and Administration ( 2.2), Warnings and Precautions ( 5.1, 5.2, 5.4)] . |
| Examples: | Benzodiazepines and other sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, gabapentinoids (gabapentin or pregabalin), other opioids, alcohol. |
| Serotonergic Drugs | |
| Clinical Impact: | The concomitant use of opioids with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome [see Warnings and Precautions ( 5.11) ]. |
| Intervention: | If concomitant use is warranted, frequently evaluate the patient, particularly during treatment initiation and dose adjustment. Discontinue fentanyl transdermal system immediately if serotonin syndrome is suspected. |
| Examples: | Selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that affect the serotonin neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), monoamine oxidase (MAO) inhibitors (those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue). |
| Monoamine Oxidase Inhibitors (MAOIs) | |
| Clinical Impact: | MAOI interactions with opioids may manifest as serotonin syndrome [see Warnings and Precautions ( 5.11) ] or opioid toxicity (e.g., respiratory depression, coma). |
| Intervention: | The use of fentanyl transdermal system is not recommended for patients taking MAOIs or within 14 days of stopping such treatment. |
| Examples: | phenelzine, tranylcypromine, linezolid |
| Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics | |
| Clinical Impact: | May reduce the analgesic effect of fentanyl transdermal system and/or precipitate withdrawal symptoms. |
| Intervention: | Avoid concomitant use. |
| Examples: | butorphanol, nalbuphine, pentazocine, buprenorphine |
| Muscle Relaxants | |
| Clinical Impact: | Fentanyl transdermal system may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression. |
| Intervention: | Because respiratory depression may be greater than otherwise expected, decrease the dosage of fentanyl transdermal system and/or the muscle relaxant as necessary. Due to the risk of respiratory depression with concomitant use of skeletal muscle relaxants and opioids, consider prescribing an opioid overdose reversal agent [see Dosage and Administration ( 2.2), Warnings and Precautions ( 5.2, 5.4)] . |
| Examples: | cyclobenzaprine, metaxalone |
| Diuretics | |
| Clinical Impact: | Opioids can reduce the efficacy of diuretics by inducing the release of antidiuretic hormone. |
| Intervention: | Evaluate patients for signs of diminished diuresis and/or effects on blood pressure and increase the dosage of the diuretic as needed. |
| Anticholinergic Drugs | |
| Clinical Impact: | The concomitant use of anticholinergic drugs may increase risk of urinary retention and/or severe constipation, which may lead to paralytic ileus. |
| Intervention: | Evaluate patients for signs of urinary retention or reduced gastric motility when fentanyl transdermal system is used concomitantly with anticholinergic drugs. |
5.16 Cardiac Disease
Fentanyl transdermal system may produce bradycardia. Regularly evaluate patients with bradyarrhythmias closely for changes in heart rate, particularly when initiating therapy with fentanyl transdermal system.
8.7 Renal Impairment
The effect of renal impairment on the pharmacokinetics of fentanyl transdermal system has not been fully evaluated. A clinical pharmacology study with intravenous fentanyl in patients undergoing kidney transplantation has shown that patients with high blood urea nitrogen level had low fentanyl clearance. Because there is in-vivoevidence of renal contribution to the elimination of fentanyl transdermal system, renal impairment would be expected to have significant effects on the pharmacokinetics of fentanyl transdermal system. Avoid the use of fentanyl transdermal system in patients with severe renal impairment [see Dosage and Administration ( 2.6), Warnings and Precautions ( 5.18) and Clinical Pharmacology ( 12.3) ].
12.2 Pharmacodynamics
.
12.3 Pharmacokinetics
.
5.18 Renal Impairment
A clinical pharmacology study with intravenous fentanyl in patients undergoing kidney transplantation has shown that patients with high blood urea nitrogen level had low fentanyl clearance. Because of the long half-life of fentanyl when administered as fentanyl transdermal system, avoid the use of fentanyl transdermal system in patients with severe renal impairment. Insufficient information exists to make precise dosing recommendations regarding the use of fentanyl transdermal system in patients with impaired renal function. Therefore, to avoid starting patients with mild to moderate renal impairment on too high of a dose, start with one half of the usual dosage of fentanyl transdermal system. Regularly evaluate for signs of sedation and respiratory depression, including at each dosage increase [see Dosage and Administration ( 2.6), Use in Specific Populations ( 8.7) and Clinical Pharmacology ( 12.3) ].
8.6 Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of fentanyl transdermal system has not been fully evaluated. A clinical pharmacology study with fentanyl transdermal system in patients with cirrhosis has shown that systemic fentanyl exposure increased in these patients. Because there is in-vitroand in-vivoevidence of extensive hepatic contribution to the elimination of fentanyl transdermal system, hepatic impairment would be expected to have significant effects on the pharmacokinetics of fentanyl transdermal system. Avoid use of fentanyl transdermal system in patients with severe hepatic impairment [see Dosage and Administration ( 2.5), Warnings and Precautions ( 5.17) and Clinical Pharmacology 12.3) ].
1 Indications and Usage
Fentanyl transdermal system is indicated for the management of severe and persistent pain in opioid-tolerant patients, that requires an opioid analgesic and that cannot be adequately treated with alternative options, including immediate-release opioids.
Patients considered opioid-tolerant are those who are taking, for one week or longer, at least 60 mg morphine per day, 25 mcg transdermal fentanyl per hour, 30 mg oral oxycodone per day, 8 mg oral hydromorphone per day, 25 mg oral oxymorphone per day, 60 mg oral hydrocodone per day, or an equianalgesic dose of another opioid.
Limitations of Use
- Because of the risks of addiction, abuse, misuse, overdose, and death, which can occur at any dosage or duration and persist over the course of therapy [see Warnings and Precautions ( 5.1) ], reserve opioid analgesics, including fentanyl transdermal system, for use in patients for whom alternative treatment options are ineffective, not tolerated, or would be otherwise inadequate to provide sufficient management of pain.
- Fentanyl transdermal system is not indicated as an as-needed (prn) analgesic.
5.14 Severe Hypotension
Fentanyl transdermal system may cause severe hypotension including orthostatic hypotension and syncope in ambulatory patients. There is an increased risk in patients whose ability to maintain blood pressure has already been compromised by a reduced blood volume or concurrent administration of certain CNS depressant drugs (e.g., phenothiazines or general anesthetics) [see Drug Interactions ( 7) ]. Regularly evaluate these patients for signs of hypotension after initiating or titrating the dosage of fentanyl transdermal system. In patients with circulatory shock, fentanyl transdermal system may cause vasodilation that can further reduce cardiac output and blood pressure. Avoid the use of fentanyl transdermal system in patients with circulatory shock.
5.17 Hepatic Impairment
A clinical pharmacology study with fentanyl transdermal system in patients with cirrhosis has shown that systemic fentanyl exposure increased in these patients. Because of the long half-life of fentanyl when administered as fentanyl transdermal system and hepatic metabolism of fentanyl, avoid use of fentanyl transdermal system in patients with severe hepatic impairment. Insufficient information exists to make precise dosing recommendations regarding the use of fentanyl transdermal system in patients with impaired hepatic function. Therefore, to avoid starting patients with mild to moderate hepatic impairment on too high of a dose, start with one half of the usual dosage of fentanyl transdermal system. Regularly evaluate for signs of sedation and respiratory depression, including at each dosage increase [see Dosage and Administration ( 2.5), Use in Specific Populations ( 8.6) and Clinical Pharmacology ( 12.3) ].
5.3 Accidental Exposure
A considerable amount of active fentanyl remains in fentanyl transdermal system even after use as directed. Death and other serious medical problems have occurred when children and adults were accidentally exposed to fentanyl transdermal system. Accidental or deliberate application or ingestion by a child or adolescent will cause respiratory depression, and has resulted in deaths. Placing fentanyl transdermal system in the mouth, chewing it, swallowing it, or using it in ways other than indicated may cause choking or overdose that could result in death. Improper disposal of fentanyl transdermal system in the trash has resulted in accidental exposures and deaths.
Advise patients about strict adherence to the recommended handling and disposal instructions in order to prevent accidental exposure to fentanyl transdermal system [see Dosage and Administration ( 2.7), ( 2.8) ]. Exposure to fentanyl transdermal system patches discarded in the trash by children have been reported and have resulted in deaths.
12 Clinical Pharmacology
.
12.1 Mechanism of Action
Fentanyl is an opioid agonist. Fentanyl interacts predominately with the opioid mu-receptor. These mu-binding sites are distributed in the human brain, spinal cord, and other tissues.
9.1 Controlled Substance
Fentanyl transdermal system contains fentanyl, a Schedule II controlled substance.
2.8 Disposal Instructions
Failure to properly dispose of fentanyl transdermal system has resulted in accidental exposures and deaths, including deaths of children [see Warnings and Precautions ( 5.3) ].
Instruct patients to dispose of used patches immediately upon removal by folding the adhesive side of the patch to itself, then flushing down the toilet.
Instruct patients to remove unused patches from their pouches, remove the release liners, fold the patches so that the adhesive side of the patch adheres to itself, and to immediately flush the patches down the toilet.
Instruct patients to dispose of any patches remaining from a prescription as soon as they are no longer needed.
13 Non Clinical Toxicology
.
5 Warnings and Precautions
.
5.13 Adrenal Insufficiency
Cases of adrenal insufficiency have been reported with opioid use, more often following greater than one month of use. Presentation of adrenal insufficiency may include non-specific symptoms and signs including nausea, vomiting, anorexia, fatigue, weakness, dizziness, and low blood pressure. If adrenal insufficiency is suspected, confirm the diagnosis with diagnostic testing as soon as possible. If adrenal insufficiency is diagnosed, treat with physiologic replacement doses of corticosteroids. Wean the patient off of the opioid to allow adrenal function to recover and continue corticosteroid treatment until adrenal function recovers. Other opioids may be tried as some cases reported use of a different opioid without recurrence of adrenal insufficiency. The information available does not identify any particular opioids as being more likely to be associated with adrenal insufficiency.
2 Dosage and Administration
9 Drug Abuse and Dependence
.
3 Dosage Forms and Strengths
Transdermal system:
- Fentanyl Transdermal System 12 mcg/hour* Transdermal System (system size 4.72 cm 2)
- Fentanyl Transdermal System 25 mcg/hour Transdermal System (system size 9.44 cm 2)
- Fentanyl Transdermal System 50 mcg/hour Transdermal System (system size 18.88 cm 2)
- Fentanyl Transdermal System 75 mcg/hour Transdermal System (system size 28.32 cm 2)
- Fentanyl Transdermal System 100 mcg/hour Transdermal System (system size 37.76 cm 2)
- This lowest strength is designated as 12 mcg/hour (however, the actual strength is 12.5 mcg/hour) to distinguish it from a possible 125 mcg/hour dosage that could be prescribed by using multiple transdermal systems.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of fentanyl transdermal system. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac Disorders: tachycardia, bradycardia
Eye Disorders: vision blurred
Gastrointestinal Disorders: ileus, dyspepsia
General Disorders and Administration Site Conditions: pyrexia, application site erosion and application site ulcer
Investigations: weight decreased
Nervous System Disorders: convulsions (including clonic convulsions and grand mal convulsion), amnesia, depressed level of consciousness, loss of consciousness
Psychiatric Disorders: agitation
Respiratory, Thoracic, and Mediastinal Disorders: respiratory distress, apnea, bradypnea, hypoventilation, dyspnea
Vascular Disorders: hypotension, hypertension
Serotonin syndrome: cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of opioids with serotonergic drugs.
Adrenal insufficiency: cases of adrenal insufficiency have been reported with opioid use, more often following greater than one month of use.
Anaphylaxis: anaphylaxis, including anaphylactic shock, has been reported with ingredients contained in fentanyl transdermal system.
Androgen deficiency: cases of androgen deficiency have occurred with use of opioids for an extended period of time. [see Clinical Pharmacology ( 12.2) ].
Hyperalgesia and Allodynia: Cases of hyperalgesia and allodynia have been reported with opioid therapy of any duration [see Warnings and Precautions ( 5.10) ].
Hypoglycemia: Cases of hypoglycemia have been reported in patients taking opioids. Most reports were in patients with at least one predisposing risk factor (e.g., diabetes).
Opioid-induced esophageal dysfunction (OIED): Cases of OIED have been reported in patients taking opioids, and may occur more frequently in patients taking higher doses of opioids, and/or in patients taking opioids longer term [see Warnings and Precautions (5.19)] .
Adverse Reactions from Observational Studies
A prospective, observational cohort study estimated the risks of addiction, abuse, and misuse in patients initiating long-term use of Schedule II opioid analgesics between 2017 and 2021. Study participants included in one or more analyses had been enrolled in selected insurance plans or health systems for a least one year, were free of at least one outcome at baseline, completed a minimum number of follow-up assessments, and either: 1) filled multiple extended-release/long-acting opioid analgesic prescriptions during a 90-day period (n=978); or 2) filled any Schedule II opioid analgesic prescriptions covering at least 70 of 90 days (n=1,244). Those included also had no dispensing of the qualifying opioids in the previous 6 months.
Over 12 months:
- approximately 1% to 6% of participants across the two cohorts newly met criteria for addiction, as assessed with two validated interview-based measures of moderate-to-severe opioid use disorder based on Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria, and
- approximately 9% and 22% of participants across the two cohorts newly met criteria for prescritpion opioid abuse and misuse [defined in Drug Abuse and Dependence (9.2) ], respectively, as measured with a validated self-reported instrument.
A retrospective, observational cohort study estimated the risk of opioid-involved overdose or opioid overdose-related death in patients with new long-term use of Schedule II opioid analgesics from 2006 through 2016 (n=220,249). Included patients had been enrolled in either one of two commercial insurance programs, one managed care program, or one Medicaid program for at least 9 months. New long-term use was defined as having Schedule II opioid analgesic prescriptions covering at least 70 days' supply over the 3 months prior to study entry and none during the preceding 6 months. Patients were excluded if they had an opioid-involved overdose in the 9 months prior to study entry. Overdose was measured using a validated medical code-based algorithm with linkage to the National Death Index database. The 5-year cumumlative incidence estimates for opioid-involved overdose or opioid overdose-related death ranged from approximately 1.5% to 4% across study sites, counting only the first event during follow-up. Approximately 17% of first opioid overdoses observed over the entire study period (5 years to 11 years, depending on the study site) were fatal. Higher baseline opioid dose was the strongest and most consistent predictor of opioid-involved overdose or opioid overdose-related death. Study exclusion criteria may have selected patients at lower risk of overdose, and substantial loss to follow-up (approximately 80%) also may have biased estimates.
The risk estimates from the studies described above may not be generalizable to all patients receiving opioid analgesics, such as those with exposures shorter or longer than the duration evaluated in the studies.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of fentanyl transdermal system was evaluated in 216 patients who took at least one dose of fentanyl transdermal system in a multicenter, double-blind, randomized, placebo-controlled clinical trial of fentanyl transdermal system. This trial examined patients over 40 years of age with severe pain induced by osteoarthritis of the hip or knee and who were in need of and waiting for joint replacement.
The most common adverse reactions (≥5%) in a double-blind, randomized, placebo-controlled clinical trial in patients with severe pain were nausea, vomiting, somnolence, dizziness, insomnia, constipation, hyperhidrosis, fatigue, feeling cold, and anorexia. Other common adverse reactions (≥5%) reported in clinical trials in patients with chronic malignant or nonmalignant pain were headache and diarrhea. Adverse reactions reported for ≥1% of fentanyl transdermal system-treated patients and with an incidence greater than placebo-treated patients are shown in Table 3.
The most common adverse reactions that were associated with discontinuation in patients with pain (causing discontinuation in ≥1% of patients) were depression, dizziness, somnolence, headache, nausea, vomiting, constipation, hyperhidrosis, and fatigue.
|
System/Organ Class
Adverse Reaction |
Fentanyl Transdermal System
% (N=216) |
Placebo
% (N=200) |
| Cardiac disorders | ||
| Palpitations | 4 | 1 |
| Ear and labyrinth disorders | ||
| Vertigo | 2 | 1 |
| Gastrointestinal disorders | ||
| Nausea | 41 | 17 |
| Vomiting | 26 | 3 |
| Constipation | 9 | 1 |
| Abdominal pain upper | 3 | 2 |
| Dry mouth | 2 | 0 |
| General disorders and administration site conditions | ||
| Fatigue | 6 | 3 |
| Feeling cold | 6 | 2 |
| Malaise | 4 | 1 |
| Asthenia | 2 | 0 |
| Edema peripheral | 1 | 1 |
| Metabolism and nutrition disorders | ||
| Anorexia | 5 | 0 |
| Musculoskeletal and connective tissue disorders | ||
| Muscle spasms | 4 | 2 |
| Nervous system disorders | ||
| Somnolence | 19 | 3 |
| Dizziness | 10 | 4 |
| Psychiatric disorders | ||
| Insomnia | 10 | 7 |
| Depression | 1 | 0 |
| Skin and subcutaneous tissue disorders | ||
| Hyperhidrosis | 6 | 1 |
| Pruritus | 3 | 2 |
| Rash | 2 | 1 |
Adverse reactions not reported in Table 3that were reported by ≥1% of fentanyl transdermal system-treated adult and pediatric patients (N=1854) in 11 controlled and uncontrolled clinical trials of fentanyl transdermal system used for the treatment of chronic malignant or nonmalignant pain are shown in Table 4.
|
System/Organ Class Adverse Reaction |
Fentanyl Transdermal System
% (N=1854) |
| Gastrointestinal disorders | |
| Diarrhea | 10 |
| Abdominal pain | 3 |
| Immune system disorders | |
| Hypersensitivity | 1 |
| Nervous system disorders | |
| Headache | 12 |
| Tremor | 3 |
| Paresthesia | 2 |
| Psychiatric disorders | |
| Anxiety | 3 |
| Confusional state | 2 |
| Hallucination | 1 |
| Renal and urinary disorders | |
| Urinary retention | 1 |
| Skin and subcutaneous tissue disorders | |
| Erythema | 1 |
The following adverse reactions occurred in adult and pediatric patients with an overall frequency of <1% and are listed in descending frequency within each System/Organ Class:
Cardiac disorders: cyanosis
Eye disorders: miosis
Gastrointestinal disorders: subileus
General disorders and administration site conditions: application site reaction, influenza-like illness, application site hypersensitivity, drug withdrawal syndrome, application site dermatitis
Musculoskeletal and connective tissue disorders: muscle twitching
Nervous system disorders: hypoesthesia
Psychiatric disorders: disorientation, euphoric mood
Reproductive system and breast disorders: erectile dysfunction, sexual dysfunction
Respiratory, thoracic and mediastinal disorders: respiratory depression
Skin and subcutaneous tissue disorders: eczema, dermatitis allergic, dermatitis contact
8 Use in Specific Populations
.
5.1 Addiction, Abuse, and Misuse
Fentanyl transdermal system contains fentanyl, an opioid agonist and a Schedule II controlled substance. As an opioid, fentanyl transdermal system exposes users to the risks of addiction, abuse, and misuse.
Although the risk of addiction in any individual is unknown, it can occur in patients appropriately prescribed fentanyl transdermal system. Addiction can occur at recommended doses and if the drug is misused or abused. The risk of opioid-related overdose or overdose-related death is increased with higher opioid doses, and this risk persists over the course of therapy. In postmarketing studies, addiction, abuse, misuse, and fatal and non-fatal opioid overdose were observed in patients with long-term opioid use [see Adverse Reactions (6.2)].
Assess each patient's risk for opioid addiction, abuse, or misuse prior to prescribing fentanyl transdermal system, and reassess all patients receiving fentanyl transdermal system for the development of these behaviors and conditions. Risks are increased in patients with a personal or family history of substance abuse (including drug or alcohol abuse or addiction) or mental illness (e.g., major depression). The potential for these risks should not, however, prevent the proper management of pain in any given patient. Patients at increased risk may be prescribed opioids such as fentanyl transdermal system, but use in such patients necessitates intensive counseling about the risks and proper use of fentanyl transdermal system along with frequent reevaluation for signs of addiction, abuse, and misuse. Consider recommending or prescribing an opioid overdose reversal agent [see Dosage and Administration ( 2.2), Warnings and Precautions ( 5.2) ].
Abuse or misuse of fentanyl transdermal system by placing it in the mouth, chewing it, swallowing it, or using it in ways other than indicated may cause choking, overdose, and death [see Overdosage ( 10) ].
Opioids are sought for nonmedical use and are subject to diversion from legitimate prescribed use. Consider these risks when prescribing or dispensing fentanyl transdermal system. Strategies to reduce these risks include prescribing the drug in the smallest appropriate quantity and advising the patient on careful storage of the drug during the course of treatment and the proper disposal of unused drug. Contact local state professional licensing board or state-controlled substances authority for information on how to prevent and detect abuse or diversion of this product.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling ( Medication Guideand Instructions for Use).
16 How Supplied/storage and Handling
Fentanyl transdermal system is supplied in cartons containing 5 individually packaged systems. See chart for information regarding individual systems.
|
* This lowest strength is designated as 12 mcg/hour (however, the actual strength is 12.5 mcg/hour) to distinguish it from a 125 mcg/hour strength that could be prescribed by using multiple transdermal systems. |
|||
|
Fentanyl Transdermal System Strength
(mcg/hour) |
System
Size (cm 2 ) |
Fentanyl
Content (mg) |
NDC
Number |
| Fentanyl transdermal system – 12 * | 4.72 | 1.33 | 47781-423-47 |
| Fentanyl transdermal system – 25 | 9.44 | 2.66 | 47781-424-47 |
| Fentanyl transdermal system – 50 | 18.88 | 5.32 | 47781-426-47 |
| Fentanyl transdermal system – 75 | 28.32 | 7.99 | 47781-427-47 |
| Fentanyl transdermal system – 100 | 37.76 | 10.65 | 47781-428-47 |
Store in original unopened pouch. Store up to 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
Store fentanyl transdermal system securely and dispose of properly [see Patient Counseling Information ( 17) ].
5.5 Neonatal Opioid Withdrawal Syndrome
Use of fentanyl transdermal system for an extended period of time during pregnancy can result in withdrawal in the neonate. Neonatal opioid withdrawal syndrome, unlike opioid withdrawal syndrome in adults, may be life-threatening if not recognized and treated, and requires management according to protocols developed by neonatology experts. Observe newborns for signs of neonatal opioid withdrawal syndrome and manage accordingly. Advise pregnant women using opioids for an extended period of time of the risk of neonatal opioid withdrawal syndrome and ensure that appropriate treatment will be available [see Use in Specific Populations ( 8.1), Patient Counseling Information ( 17) ].
2.4 Titration and Maintenance of Therapy
Individually titrate fentanyl transdermal system to a dose that provides adequate analgesia and minimizes adverse reactions. Continually reevaluate patients receiving fentanyl transdermal system to assess the maintenance of pain control, signs and symptoms of opioid withdrawal, and other adverse reactions, as well as to reassess for the development of addiction, abuse, or misuse [see Warnings and Precautions ( 5.1, 5.21) ]. Frequent communication is important among the prescriber, other members of the healthcare team, the patient, and the caregiver/family during periods of changing analgesic requirements, including initial titration. During use of opioid therapy for an extended period of time, periodically reassess the continued need for use of opioid analgesics.
Patients who experience breakthrough pain may require a dosage adjustment of fentanyl transdermal system, or may need rescue medication with an appropriate dose of an immediate-release analgesic. If the level of pain increases after dosage stabilization, attempt to identify the source of increased pain before increasing the fentanyl transdermal system dosage.
The dosing interval for fentanyl transdermal system is 72 hours. Do not increase the fentanyl transdermal system dose for the first time until at least 3 days after the initial application. Titrate the dose based on the daily dose of supplemental opioid analgesics required by the patient on the second or third day of the initial application.
It may take up to 6 days for fentanyl levels to reach equilibrium on a new dose [see Clinical Pharmacology ( 12.3) ]. Therefore, evaluate patients for further titration after no less than two 3-day applications before any further increase in dosage is made.
Base dosage increments on the daily dosage of supplementary opioids, using the ratio of 45 mg/24 hours of oral morphine to a 12 mcg/hour increase in fentanyl transdermal system dose.
If after increasing the dosage, unacceptable opioid-related adverse reactions are observed, (including an increase in pain after dosage increase), consider reducing the dosage [see Warnings and Precautions ( 5)] . Adjust the dose to obtain an appropriate balance between management of pain and opioid-related adverse reactions.
A small proportion of adult patients may not achieve adequate analgesia using a 72-hour dosing interval and may require systems to be applied at 48 hours rather than at 72 hours, only if adequate pain control cannot be achieved using a 72-hour regimen. An increase in the fentanyl transdermal system dose should be evaluated before changing dosing intervals in order to maintain patients on a 72-hour regimen.
Dosing intervals less than every 72 hours were not studied in children and adolescents and are not recommended.
5.2 Life Threatening Respiratory Depression
Serious, life-threatening, or fatal respiratory depression has been reported with the use of opioids, even when used as recommended. Respiratory depression, if not immediately recognized and treated, may lead to respiratory arrest and death. Management of respiratory depression may include close observation, supportive measures, and use of opioid overdose reversal agents, depending on the patient's clinical status [see Overdosage ( 10) ]. Carbon dioxide (CO 2) retention from opioid-induced respiratory depression can exacerbate the sedating effects of opioids.
Fentanyl transdermal system is indicated only in opioid tolerant patients because of the risk for respiratory depression and death. While serious, life-threatening, or fatal respiratory depression can occur at any time during the use of fentanyl transdermal system, the risk is greatest during the initiation of therapy or following a dosage increase.
To reduce the risk of respiratory depression, proper dosing and titration of fentanyl transdermal system are essential [see Dosage and Administration ( 2) ]. Overestimating the fentanyl transdermal system dosage when converting patients from another opioid product can result in fatal overdose with the first dose.
Accidental exposure to fentanyl transdermal system, especially in children, can result in respiratory depression and death due to an overdose of fentanyl.
Educate patients and caregivers on how to recognize respiratory depression and emphasize the importance of calling 911 or getting emergency medical help right away in the event of a known or suspected overdose [see Patient Counseling Information ( 17) ].
Opioids can cause sleep-related breathing disorders including central sleep apnea (CSA) and sleep-related hypoxemia. Opioid use increases the risk of CSA in a dose-dependent fashion. In patients who present with CSA, consider decreasing the opioid dosage using best practices for opioid taper [see Dosage and Administration ( 2.9) ].
5.19 Risks of Gastrointestinal Complications
Fentanyl transdermal system is contraindicated in patients with known or suspected gastrointestinal obstruction, including paralytic ileus.
The fentanyl in fentanyl transdermal system may cause spasm of the sphincter of Oddi. Opioids may cause increases in serum amylase. Regularly evaluate patients with biliary tract disease, including acute pancreatitis for worsening symptoms.
Cases of opioid-induced esophageal dysfuntion (OIED) have been reported in patients taking opioids. The risk of OIED may increase as the dose and/or duration of opioids increases. Regularly evaluate patients for signs and sypmtoms of OIED (e.g. dysphagia, regurgitation, non-cardiac chest pain), and if necessary, adjust opioid therapy as clinically appropriate [see Clinical Pharmacology (12.2)].
5.22 Risks of Driving and Operating Machinery
Fentanyl transdermal system may impair the mental or physical abilities required for the performance of potentially dangerous activities, such as driving a car or operating machinery. Warn patients not to drive or operate dangerous machinery unless they are tolerant to the effects of the fentanyl transdermal system and know how they will react to the medication [see Patient Counseling Information ( 17) ].
5.10 Opioid Induced Hyperalgesia and Allodynia
Opioid-Induced Hyperalgesia (OIH) occurs when an opioid analgesic paradoxically causes an increase in pain, or an increase in sensitivity to pain. This condition differs from tolerance, which is the need for increasing doses of opioids to maintain a defined effect [see Dependence ( 9.3) ]. Symptoms of OIH include (but may not be limited to) increased levels of pain upon opioid dosage increase, decreased levels of pain upon opioid dosage decrease, or pain from ordinarily non-painful stimuli (allodynia). These symptoms may suggest OIH only if there is no evidence of underlying disease progression, opioid tolerance, opioid withdrawal, or addictive behavior.
Cases of OIH have been reported, both with short-term and longer-term use of opioid analgesics. Though the mechanism of OIH is not fully understood, multiple biochemical pathways have been implicated. Medical literature suggests a strong biologic plausibility between opioid analgesics and OIH and allodynia. If a patient is suspected to be experiencing OIH, carefully consider appropriately decreasing the dose of the current opioid analgesic, or opioid rotation (safely switching the patient to a different opioid moiety) [see Dosage and Administration ( 2.9) , Warnings and Precautions ( 5.21) ].
2.7 Administration of Fentanyl Transdermal System
FENTANYL TRANSDERMAL SYSTEM PATCHES ARE FOR TRANSDERMAL USE ONLY.
Proper handling of fentanyl transdermal system is necessary in order to prevent serious adverse outcomes, including death, associated with accidental secondary exposure to fentanyl transdermal system [see Warnings and Precautions ( 5.3) ].
Application and Handling Instructions
- Patients should apply fentanyl transdermal system to intact, non-irritated, and non-irradiated skin on a flat surface such as the chest, back, flank, or upper arm. In young children and persons with cognitive impairment, adhesion should be monitored and the upper back is the preferred location to minimize the potential of inappropriate patch removal. Hair at the application site may be clipped (not shaved) prior to system application. If the site of fentanyl transdermal system application must be cleansed prior to application of the patch, do so with clear water. Do not use soaps, oils, lotions, alcohol, or any other agents that might irritate the skin or alter its characteristics. Allow the skin to dry completely prior to patch application.
- Patients should apply fentanyl transdermal system immediately upon removal from the sealed package. The patch must not be altered (e.g., cut) in any way prior to application. Fentanyl transdermal system should not be used if the pouch seal is broken or if the patch is cut or damaged.
- The transdermal system is pressed firmly in place with the palm of the hand for 30 seconds, making sure the contact is complete, especially around the edges.
- Each fentanyl transdermal system patch may be worn continuously for up to 72 hours. The next patch is applied to a different skin site after removal of the previous transdermal system.
- If problems with adhesion of the fentanyl transdermal system patch occur, the edges of the patch may be taped with first-aid tape. If problems with adhesion persist, the patch may be overlayed with a transparent adhesive film dressing.
- If the patch falls off before 72 hours, dispose of it by folding in half and flushing down the toilet. A new patch may be applied to a different skin site.
- Patients (or caregivers who apply fentanyl transdermal system) should wash their hands immediately with soap and water after applying fentanyl transdermal system.
- Contact with unwashed or unclothed application sites can result in secondary exposure to fentanyl transdermal system and should be avoided. Examples of accidental exposure include transfer of a fentanyl transdermal system patch from an adult's body to a child while hugging, sharing the same bed as the patient, accidental sitting on a patch and possible accidental exposure of a caregiver's skin to the medication in the patch while applying or removing the patch.
- Instruct patients, family members, and caregivers to keep patches in a secure location out of the reach of children and of others for whom fentanyl transdermal system was not prescribed.
Avoidance of Heat
Instruct patients to avoid exposing the fentanyl transdermal system application site and surrounding area to direct external heat sources, such as heating pads or electric blankets, heat or tanning lamps, sunbathing, hot baths, saunas, hot tubs, and heated water beds, while wearing the system [see Warnings and Precautions ( 5.8) ].
2.1 Important Dosage and Administration Instructions
- Fentanyl transdermal system should be prescribed only by healthcare professionals who are knowledgeable about the use of extended-release/long-acting opioids and how to mitigate the associated risks.
- Due to the risk of respiratory depression, fentanyl transdermal system is only indicated for use in patients who are already opioid-tolerant. Discontinue or taper all other extended-release opioids when beginning fentanyl transdermal system therapy. As fentanyl transdermal system is only for use in opioid-tolerant patients, do not begin any patient on fentanyl transdermal system as the first opioid [see Indications and Usage ( 1)] .
- Use the lowest effective dosage for the shortest duration of time consistent with individual patient treatment goals [see Warnings and Precautions ( 5)] . Because the risk of overdose increases as opioid doses increase, reserve titration to higher doses of fentanyl transdermal system for patients in whom lower doses are insufficiently effective and in whom the expected benefits of using a higher dose opioid clearly outweigh the substantial risks.
- Initiate the dosing regimen for each patient individually, taking into account the patient's underlying cause and severity of pain, prior analgesic treatment and response, and risk factors for addiction, abuse, and misuse [see Warnings and Precautions ( 5.1)] .
- Respiratory depression can occur at any time during opioid therapy, especially when initiating and following dosage increases with fentanyl transdermal system. Consider this risk when selecting an initial dose and when making dose adjustments [see Warnings and Precautions ( 5)] .
- Each fentanyl transdermal system is worn continuously for up to 72 hours [see Dosage and Administration (2.7)].
2.6 Dosage Modifications in Patients With Renal Impairment
Avoid the use of fentanyl transdermal system in patients with severe renal impairment. In patients with mild to moderate renal impairment, start with one half of the usual dosage of fentanyl transdermal system. Closely monitor for signs of respiratory and central nervous system depression, including at each dosage increase [see Warnings and Precautions ( 5.18), Use in Specific Populations ( 8.7) and Clinical Pharmacology ( 12.3) ].
2.5 Dosage Modifications in Patients With Hepatic Impairment
Avoid the use of fentanyl transdermal system in patients with severe hepatic impairment. In patients with mild to moderate hepatic impairment, start with one half of the usual dosage of fentanyl transdermal system. Closely monitor for signs of respiratory and central nervous system depression, including at each dosage increase [see Warnings and Precautions ( 5.17), Use in Specific Populations ( 8.6) and Clinical Pharmacology ( 12.3) ].
13.1 Carcinogenesis, Mutagenesis, and Impairment of Fertility
.
5.11 Serotonin Syndrome With Concomitant Use of Serotonergic Drugs
Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of fentanyl transdermal system with serotonergic drugs. Serotonergic drugs include selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that affect the serotonergic neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), and drugs that impair metabolism of serotonin (including MAO inhibitors, both those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue) [see Drug Interactions ( 7)] . This may occur within the recommended dosage range. Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination, rigidity), and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). The onset of symptoms generally occurs within several hours to a few days of concomitant use, but may occur later than that. Discontinue fentanyl transdermal system immediately if serotonin syndrome is suspected.
5.20 Increased Risk of Seizures in Patients With Seizure Disorders
The fentanyl in fentanyl transdermal system may increase the frequency of seizures in patients with seizure disorders, and may increase the risk of seizures occurring in other clinical settings associated with seizures. Regularly evaluate patients with a history of seizure disorders for worsened seizure control during fentanyl transdermal system therapy.
5.6 Opioid Analgesic Risk Evaluation and Mitigation Strategy (rems)
To ensure that the benefits of opioid analgesics outweigh the risks of addiction, abuse, and misuse, the Food and Drug Administration (FDA) has required a Risk Evaluation and Mitigation Strategy (REMS) for these products. Under the requirements of the REMS, drug companies with approved opioid analgesic products must make REMS-compliant education programs available to healthcare providers. Healthcare providers are strongly encouraged to do all of the following:
- Complete a REMS-compliant education programoffered by an accredited provider of continuing education (CE) or another education program that includes all the elements of the FDA Education Blueprint for Health Care Providers Involved in the Management or Support of Patients with Pain.
- Discuss the safe use, serious risks, and proper storage and disposal of opioid analgesics with patients and/or their caregivers every time these medicines are prescribed. The Patient Counseling Guide (PCG) can be obtained at this link: www.fda.gov/OpioidAnalgesicREMSPCG.
- Emphasize to patients and their caregivers the importance of reading the Medication Guide that they will receive from their pharmacist every time an opioid analgesic is dispensed to them.
- Consider using other tools to improve patient, household, and community safety, such as patient-prescriber agreements that reinforce patient-prescriber responsibilities.
To obtain further information on the opioid analgesic REMS and for a list of accredited REMS CME/CE, call 1-800-503-0784, or log on to www.opioidanalgesicrems.com. The FDA Blueprint can be found at www.fda.gov/OpioidAnalgesicREMSBlueprint.
2.9 Safe Reduction Or Discontinuation of Fentanyl Transdermal System
Do not rapidly reduce or abruptly discontinue fentanyl transdermal system in patients who may be physically dependent on opioids. Rapid reduction or abrupt discontinuation of opioid analgesics in patients who are physically dependent on opioids has resulted in serious withdrawal symptoms, uncontrolled pain, and suicide. Rapid reduction or abrupt discontinuation has also been associated with attempts to find other sources of opioid analgesics, which may be confused with drug-seeking for abuse. Patients may also attempt to treat their pain or withdrawal symptoms with illicit opioids, such as heroin, and other substances.
When a decision has been made to decrease the dose or discontinue therapy in an opioid-dependent patient taking fentanyl transdermal system, there are a variety of factors that should be considered, including the total daily dose of opioid (including fentanyl transdermal system) the patient has been taking, the duration of treatment, the type of pain being treated, and the physical and psychological attributes of the patient. It is important to ensure ongoing care of the patient and to agree on an appropriate tapering schedule and follow-up plan so that patient and provider goals and expectations are clear and realistic. When opioid analgesics are being discontinued due to a suspected substance use disorder, evaluate and treat the patient, or refer for evaluation and treatment of the substance use disorder. Treatment should include evidence-based approaches, such as medication assisted treatment of opioid use disorder. Complex patients with comorbid pain and substance use disorders may benefit from referral to a specialist.
There are no standard opioid tapering schedules that are suitable for all patients. Good clinical practice dictates a patient-specific plan to taper the dose of the opioid gradually. For patients on fentanyl transdermal system who are physically opioid-dependent, initiate the taper by a small enough increment (e.g., no greater than 25% of the total daily dose) to avoid withdrawal symptoms, and proceed with dose-lowering at an interval of every 2 to 4 weeks. Patients who have been taking opioids for briefer periods of time may tolerate a more rapid taper.
It may be necessary to provide the patient with a lower dosage strength to accomplish a successful taper. Reassess the patient frequently to manage pain and withdrawal symptoms, should they emerge. Common withdrawal symptoms include restlessness, lacrimation, rhinorrhea, yawning, perspiration, chills, myalgia, and mydriasis. Other signs and symptoms also may develop, including irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory rate, or heart rate. If withdrawal symptoms arise, it may be necessary to pause the taper for a period of time or raise the dose of the opioid analgesic to the previous dose, and then proceed with a slower taper. In addition, evaluate patients for any changes in mood, emergence of suicidal thoughts, or use of other substances.
When managing patients taking opioid analgesics, particularly those who have been treated for an extended period of time, and/or with high doses for chronic pain, ensure that a multimodal approach to pain management, including mental health support (if needed), is in place prior to initiating an opioid analgesic taper. A multimodal approach to pain management may optimize the treatment of chronic pain, as well as assist with the successful tapering of the opioid analgesic [see Warnings and Precautions ( 5.21), Drug Abuse and Dependence ( 9.3) ].
5.9 Risk of Increased Fentanyl Absorption With Elevated Body Temperature
Based on a pharmacokinetic model, serum fentanyl concentrations could theoretically increase by approximately one-third for patients with a body temperature of 40°C (104°F) due to temperature-dependent increases in fentanyl released from the system and increased skin permeability. Monitor patients wearing fentanyl transdermal systems who develop fever closely for sedation and respiratory depression and reduce the fentanyl transdermal system dose, if necessary. Warn patients to avoid strenuous exertion that leads to increased core body temperature while wearing fentanyl transdermal system to avoid the risk of potential overdose and death.
5.8 Risk of Increased Fentanyl Absorption With Application of External Heat
Exposure to heat may increase fentanyl absorption and there have been reports of overdose and death as a result of exposure to heat. A clinical pharmacology study conducted in healthy adult subjects has shown that the application of heat over the fentanyl transdermal system increased fentanyl exposure [see Clinical Pharmacology ( 12.3) ].
Warn patients to avoid exposing the fentanyl transdermal system application site and surrounding area to direct external heat sources [see Dosage and Administration ( 2.7)] .
5.4 Risks From Concomitant Use With Benzodiazepines Or Other Cns Depressants
Profound sedation, respiratory depression, coma, and death may result from the concomitant use of fentanyl transdermal system with benzodiazepines and/or other CNS depressants, including alcohol (e.g., non-benzodiazepine sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, gabapentinoids [gabapentin or pregabalin], and other opioids). Because of these risks, reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate.
Observational studies have demonstrated that concomitant use of opioid analgesics and benzodiazepines increases the risk of drug-related mortality compared to use of opioid analgesics alone. Because of similar pharmacological properties, it is reasonable to expect similar risk with the concomitant use of other CNS depressant drugs with opioid analgesics [see Drug Interactions ( 7) ].
If the decision is made to prescribe a benzodiazepine or other CNS depressant concomitantly with an opioid analgesic, prescribe the lowest effective dosages and minimum durations of concomitant use. In patients already receiving an opioid analgesic, prescribe a lower initial dose of the benzodiazepine or other CNS depressant than indicated in the absence of an opioid, and titrate based on clinical response. If an opioid analgesic is initiated in a patient already taking a benzodiazepine or other CNS depressant, prescribe a lower initial dose of the opioid analgesic, and titrate based on clinical response. Inform patients and caregivers of this potential interaction, educate them on the signs and symptoms of respiratory depression (including sedation).
If concomitant use is warranted, consider recommending or prescribing an opioid overdose reversal agent [see Dosage and Administration ( 2.2), Warnings and Precautions ( 5.2), Overdosage (10)] .
Advise both patients and caregivers about the risks of respiratory depression and sedation when fentanyl transdermal system is used with benzodiazepines or other CNS depressants (including alcohol and illicit drugs). Advise patients not to drive or operate heavy machinery until the effects of concomitant use of the benzodiazepine or other CNS depressant have been determined. Screen patients for risk of substance use disorders, including opioid abuse and misuse, and warn them of the risk for overdose and death associated with the use of additional CNS depressants including alcohol and illicit drugs [see Drug Interactions ( 7), Patient Counseling Information ( 17) ].
Warning: Serious and Life Threatening Risks From Use of Fentanyl Transdermal System
.
5.7 Risks of Concomitant Use Or Discontinuation of Cytochrome P450 3a4 Inhibitors and Inducers
Concomitant use of fentanyl transdermal system with a CYP3A4 inhibitor, such as macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g., ketoconazole), and protease inhibitors (e.g., ritonavir), may increase plasma concentrations of fentanyl and prolong opioid adverse reactions, which may cause potentially fatal respiratory depression [see Warnings and Precautions ( 5.2) ], particularly when an inhibitor is added after a stable dose of fentanyl transdermal system is achieved. Similarly, discontinuation of a CYP3A4 inducer, such as rifampin, carbamazepine, and phenytoin, in fentanyl transdermal system-treated patients may increase fentanyl plasma concentrations and prolong opioid adverse reactions. When using fentanyl transdermal system with CYP3A4 inhibitors or discontinuing CYP3A4 inducers in fentanyl transdermal system-treated patients, evaluate patients at frequent intervals and consider dosage reduction of fentanyl transdermal system until stable drug effects are achieved [see Dosage and Administration ( 2.4), Drug Interactions ( 7) ].
Concomitant use of fentanyl transdermal system with CYP3A4 inducers or discontinuation of a CYP3A4 inhibitor could decrease fentanyl transdermal system plasma concentrations, decrease opioid efficacy or, possibly, lead to a withdrawal syndrome in a patient who had developed physical dependence to fentanyl. When using fentanyl transdermal system with CYP3A4 inducers or discontinuing CYP3A4 inhibitors, monitor patients closely at frequent intervals and consider increasing the opioid dosage if needed to maintain adequate analgesia or if symptoms of opioid withdrawal occur [see Drug Interactions ( 7) ].
2.2 Patient Access to An Opioid Overdose Reversal Agent for the Emergency Treatment of Opioid Overdose
Inform patients and caregivers about opioid overdose reversal agents (e.g. naloxone, nalmefene). Discuss the importance of having access to an opioid overdose reversal agent, especially if the patient has risk factors for overdose (e.g. concomitant use of CNS depressants, a history of opioid use disorder, or prior opioid overdose) or if there are household members (including children) or other close contacts at risk for accidental ingestion or opioid overdose. The presence of risk factors for overdose should not prevent the management of pain in any patient [see Warnings and Precautions ( 5.1, 5.2, 5.4) ].
Discuss the options for obtaining an opioid overdose reversal agent (e.g. prescription, over-the-counter, or as part of a community-based program) [see Warnings and Precautions (5.2)].
There are important differences among the opioid overdose reversal agents, such as route of administration, product strength, approved patient age range, and pharmacokinetics. Be familiar with these differences, as outlined in the approved labeling for those products, prior to recommending or prescribing such an agent.
5.15 Risks of Use in Patients With Increased Intracranial Pressure, Brain Tumors, Head Injury, Or Impaired Consciousness
In patients who may be susceptible to the intracranial effects of CO 2retention (e.g., those with evidence of increased intracranial pressure or brain tumors), fentanyl transdermal system may reduce respiratory drive, and the resultant CO 2retention can further increase intracranial pressure. Monitor such patients for signs of sedation and respiratory depression, particularly when initiating therapy with fentanyl transdermal system.
Opioids may also obscure the clinical course in a patient with a head injury. Avoid the use of fentanyl transdermal system in patients with impaired consciousness or coma.
5.12 Life Threatening Respiratory Depression in Patients With Chronic Pulmonary Disease Or in Elderly, Cachectic, Or Debilitated Patients
The use of fentanyl transdermal systems in patients with acute or severe bronchial asthma in an unmonitored setting or in the absence of resuscitative equipment is contraindicated.
Structured Label Content
Section 42229-5 (42229-5)
Addiction, Abuse, and Misuse
Because the use of fentanyl transdermal system exposes patients and other users to the risks of opioid addiction, abuse, and misuse, which can lead to overdose and death, assess each patient's risk prior to prescribing and reassess all patients regularly for the development of these behaviors and conditions [see Warnings and Precautions ( 5.1) ].
Section 42231-1 (42231-1)
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: 12/2025 |
|
|
Medication Guide Fentanyl (fen' ta nil) Transdermal System, CII |
|
Fentanyl Transdermal System is:
|
|
Important information about Fentanyl Transdermal System:
|
|
Do not use Fentanyl Transdermal System if you have:
|
|
| Before applying Fentanyl Transdermal System, tell your healthcare provider if you have a history of: | |
|
|
|
|
Tell your healthcare provider if you:
|
|
When using Fentanyl Transdermal System:
|
|
While using Fentanyl Transdermal System DO NOT:
|
|
The possible side effects of Fentanyl Transdermal System are:
|
|
Get emergency medical help or call 911 right away if you have:
|
|
| These are not all the possible side effects of fentanyl transdermal system. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
For more information go to dailymed.nlm.nih.gov.
Manufactured by: Kindeva Drug Delivery L.P., Northridge, CA 91324 USA; Distributed by: Alvogen, Inc., Morristown, NJ 07960 USA, www.Alvogen.com or call Alvogen, Inc. at 1-866-770-3024. |
Section 43683-2 (43683-2)
| Boxed Warning | 12/2025 |
| Indications and Usage ( 1) | 12/2025 |
| Dosage and Administration ( 2.2, 2.3) | 12/2025 |
| Warnings and Precautions ( 5.1, 5.2, 5.4, 5.19, 5.21) | 12/2025 |
Section 51945-4 (51945-4)
Principal Display Panel - 12 mcg/h Patch Carton
12
mcg/h
NDC47781- 423-47
FENTANYL Transdermal System CII
In vivodelivery of 12 mcg/h fentanyl for 72 hours
Because serious or life-threatening breathing problems could
result, DO NOT USE FENTANYL TRANSDERMAL SYSTEM:
-
for pain that can be treated with immediate-release opioids or
non-opioid analgesics - for intermittent (on an as-needed basis) pain
- for any postoperative pain
-
unless you are opioid tolerant (have been using other narcotic
opioid medicines)
Each transdermal system contains:1.33 mg fentanyl
DO NOT USE IF SEAL ON POUCH IS BROKEN
KEEP OUT OF REACH OF CHILDREN
Read enclosed Medication Guide for important safety information.
Rx Only
Alvogen ®
five (12 mcg/h) systems
For Opioid-Tolerant
Patients Only
Section 59845-8 (59845-8)
Instructions for Use
Fentanyl (fen' ta nil) Transdermal System CII
Be sure that you read, understand, and follow these Instructions for Use before you apply fentanyl transdermal system (patch). Talk to your healthcare provider or pharmacist if you have any questions.
Important information about the fentanyl transdermal system (patch) appearance:
- Fentanyl transdermal system is a rectangular, see-through patch with rounded corners.
-
Fentanyl transdermal system comes in 5 different dosage strengths and sizes:
- 12 mcg/hour
- 25 mcg/hour
- 50 mcg/hour
- 75 mcg/hour
- 100 mcg/hour
- The product name, “FENTANYL”, and dosage strength are printed in green on each patch.
Parts of the Fentanyl Transdermal System patch:
BEFORE APPLYING FENTANYL TRANSDERMAL SYSTEM
- Each fentanyl transdermal system patch is sealed in its own protective pouch. Do not remove a fentanyl transdermal system patch from the pouch until you are ready to use it.
- Do not use a fentanyl transdermal system patch if the pouch seal is broken or the patch is cut, damaged or changed in any way.
- Fentanyl transdermal system patches are available in 5 different dosage strengths and patch sizes. Make sure you have the right dose patch or patches that have been prescribed for you.
APPLYING A FENTANYL TRANSDERMAL PATCH
|
Figure A Figure B Figure C Figure D Figure E |
|
2. Prepare to apply a fentanyl transdermal system patch:
|
|
|
3. Open the pouch: Fold and tear at slit, or cut at slit taking care not to cut the patch. Remove the fentanyl transdermal system patch. Each fentanyl transdermal system patch is sealed in its own protective pouch. Do not remove the fentanyl transdermal system patch from the pouch until you are ready to use it (See Figure F). 4. Peel:Peel off both parts of the release liner from the patch. Each fentanyl transdermal system patch has a clear plastic release liner that can be peeled off in two pieces. This covers the sticky side of the patch. Carefully peel this release liner off and throw the pieces away. Touch the sticky side of the fentanyl transdermal system patch as little as possible(See Figure G). |
Figure F Figure G |
|
5. Press:Press the patch onto the chosen skin site with the palm of your hand and hold there for at least 30 seconds(See Figure H). Make sure it sticks well, especially at the edges.
6. Wash your hands when you have finished applying a fentanyl transdermal system patch. 7. Remove a fentanyl transdermal system patch after wearing it for 3 days (72 hours).
Dispose of the used patch right away. See the section below “
Disposing of a fentanyl transdermal system patch.”
Choose a
differentskin site to apply a new fentanyl transdermal system patch. Repeat Steps 2 through 6 above when applying a new fentanyl transdermal system patch.
DISPOSING OF A FENTANYL TRANSDERMAL SYSTEM PATCH
|
Figure H Figure I Figure J |
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by:
Kindeva Drug Delivery L.P.
Northridge, CA 91324 USA
Distributed by:
Alvogen, Inc.
Morristown, NJ 07960 USA
Revised: 12/2025
3200001799
Bioclusive™ is a trademark of Ethicon, Inc.
Tegaderm™ is a trademark of 3M
9.2 Abuse
Fentanyl transdermal system contains fentanyl, a substance with high potential for misuse and abuse, which can lead to the development of substance use disorder, including addiction [see Warnings and Precautions ( 5.1) ].
Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a healthcare provider or for whom it was not prescribed.
Abuse is the intentional, non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects.
Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance, or physical dependence.
Misuse and abuse of fentanyl transdermal system increases risk of overdose, which may lead to central nervous system and respiratory depression, hypotension, seizures, and death. The risk is increased with concurrent use of fentanyl transdermal system with alcohol and/or other CNS depressants. Abuse of and addiction to opioids in some individuals may not be accompanied by concurrent tolerance and symptoms of physical dependence. In addition, abuse of opioids can occur in the absence of addiction.
All patients treated with opioids require careful and frequent reevaluation for signs of misuse, abuse, and addiction, because use of opioid analgesic products carries the risk of addiction even under appropriate medical use. Patients at high risk of fentanyl transdermal system abuse include those with a history of prolonged use of any opioid, including products containing fentanyl, those with a history of drug or alcohol abuse, or those who use fentanyl transdermal system in combination with other abused drugs.
“Drug seeking” behavior is very common in persons with substance use disorders. Drug-seeking tactics include emergency calls or visits near the end of office hours, refusal to undergo appropriate examination, testing, or referral, repeated “loss” of prescriptions, tampering with prescriptions, and reluctance to provide prior medical records or contact information for other treating healthcare provider(s). “Doctor shopping” (visiting multiple prescribers to obtain additional prescriptions) is common among people who abuse drugs and people with substance use disorder. Preoccupation with achieving adequate pain relief can be appropriate behavior in a patient with inadequate pain control.
Fentanyl transdermal system, like other opioids, can be diverted for nonmedical use into illicit channels of distribution. Careful record-keeping of prescribing information, including quantity, frequency, and renewal requests, as required by state and federal law, is strongly advised.
Proper assessment of the patient, proper prescribing practices, periodic reevaluation of therapy, and proper dispensing and storage are appropriate measures that help to limit abuse of opioid drugs.
10 Overdosage (10 OVERDOSAGE)
.
8.1 Pregnancy
.
11 Description (11 DESCRIPTION)
The system contains fentanyl, an opioid agonist, for transdermal administration. The amount of fentanyl released from each system per hour is proportional to the surface area (25 mcg/hour per 9.44 cm 2). The composition per unit area of all transdermal system sizes is identical.
| *Nominal delivery rate is 12.5 mcg/hour per hour | ||
| **Nominal delivery rate per hour | ||
|
Strength
(mcg/hour) |
Size
(cm²) |
Fentanyl Content
(mg) |
| 12* | 4.72 | 1.33 |
| 25** | 9.44 | 2.66 |
| 50** | 18.88 | 5.32 |
| 75** | 28.32 | 7.99 |
| 100** | 37.76 | 10.65 |
The molecular weight of fentanyl base is 336.5, and the molecular formula is C 22H 28N 2O. The n-octanol: water partition coefficient is 860:1. The pKa is 8.4.
The chemical name is N-Phenyl-N-(1-(2-phenylethyl)-4-piperidinyl) propanamide. The structural formula is:
Fentanyl transdermal system is a rectangular translucent system with rounded corners. The product name, “FENTANYL” and dosage strength are printed in green on each system.
Each system is comprised of a clear polyethylene terephthalate (PET) release liner and two functional layers. Proceeding from the outer surface toward the surface adhering to skin, these functional layers are:
1) a translucent backing layer of ethylene vinyl acetate/polyethylene terephthalate (EVA/PET) film with green print; 2) a drug-in-adhesive layer containing fentanyl, acrylate copolymer adhesive, and methyl laurate. Before use, a clear PET release liner covering the drug-in-adhesive layer is removed and discarded.
9.3 Dependence
Both tolerance and physical dependence can develop during use of opioid therapy.
Tolerance is a physiological state characterized by a reduced response to a drug after repeated administration (i.e., a higher dose of a drug is required to produce the same effect that was once obtained at a lower dose).
Physical dependence is a state that develops as a result of a physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug.
Withdrawal may be precipitated through the administration of drugs with opioid antagonist activity (e.g., naloxone, nalmefene), mixed agonist/antagonist analgesics (e.g., pentazocine, butorphanol, nalbuphine), or partial agonists (e.g., buprenorphine). Physical dependence may not occur to a clinically significant degree until after several days to weeks of continued use.
Do not rapidly reduce or abruptly discontinue fentanyl transdermal system in a patient physically dependent on opioids. Rapid reduction or abrupt discontinuation of fentanyl transdermal system in a patient physically dependent on opioids may lead to serious withdrawal symptoms, uncontrolled pain, and suicide. Rapid reduction or abrupt discontinuation has also been associated with attempts to find other sources of opioid analgesics, which may be confused with drug-seeking for abuse.
When discontinuing fentanyl transdermal system, gradually taper the dosage using a patient-specific plan that considers the following: the dose of fentanyl transdermal system the patient has been taking, the duration of treatment, and the physical and psychological attributes of the patient. To improve the likelihood of a successful taper and minimize withdrawal symptoms, it is important that the opioid tapering schedule is agreed upon by the patient. In patients taking opioids for an extended period of time at high doses, ensure that a multimodal approach to pain management, including mental health support (if needed), is in place prior to initiating an opioid analgesic taper [see Dosage and Administration ( 2.9), and Warnings and Precautions ( 5.21) ].
Infants born to mothers physically dependent on opioids will also be physically dependent and may exhibit respiratory difficulties and withdrawal signs [see Use in Specific Populations ( 8.1) ].
5.21 Withdrawal
Do not rapidly reduce or abruptly discontinue fentanyl transdermal system in a patient physically dependent on opioids. When discontinuing fentanyl transdermal system in a physically dependent patient, gradually taper the dosage. Rapid tapering of fentanyl transdermal system in a patient physically dependent on opioids may lead to a withdrawal syndrome and return of pain [see Dosage and Administration ( 2.9), Drug Abuse and Dependence ( 9.3) ].
Additionally, avoid the use of mixed agonist/antagonist (e.g., pentazocine, nalbuphine, and butorphanol) or partial agonist (e.g., buprenorphine) analgesics in patients who are receiving a full opioid agonist analgesic, including fentanyl transdermal system. In these patients, mixed agonist/antagonist and partial agonist analgesics may reduce the analgesic effect and/or may precipitate withdrawal symptoms [see Drug Interactions ( 7) ].
8.4 Pediatric Use
The safety of fentanyl transdermal system was evaluated in three open-label trials in 289 pediatric patients with chronic pain, 2 years of age through 18 years of age. Starting doses of 25 mcg/h and higher were used by 181 patients who had been on prior daily opioid doses of at least 45 mg/day of oral morphine or an equianalgesic dose of another opioid. Initiation of fentanyl transdermal system therapy in pediatric patients taking less than 60 mg/day of oral morphine or an equianalgesic dose of another opioid has not been evaluated in controlled clinical trials.
The safety and effectiveness of fentanyl transdermal system in children under 2 years of age have not been established.
To guard against excessive exposure to fentanyl transdermal system by young children, advise caregivers to strictly adhere to recommended fentanyl transdermal system application and disposal instructions [see Dosage and Administration ( 2.7), ( 2.8) and Warnings and Precautions ( 5.3) ].
8.5 Geriatric Use
Clinical studies of fentanyl transdermal system did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, use caution when selecting a dosage for an elderly patient, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Data from intravenous studies with fentanyl suggest that the elderly patients may have reduced clearance and a prolonged half-life. Moreover, elderly patients may be more sensitive to the active substance than younger patients. A study conducted with the fentanyl transdermal system patch in elderly patients demonstrated that fentanyl pharmacokinetics did not differ significantly from young adult subjects, although peak serum concentrations tended to be lower and mean half-life values were prolonged to approximately 34 hours [see Clinical Pharmacology ( 12.3) ].
Respiratory depression is the chief risk for elderly patients treated with opioids, and has occurred after large initial doses were administered to patients who were not opioid-tolerant or when opioids were co-administered with other agents that depress respiration. Titrate the dosage of fentanyl transdermal system slowly in geriatric patients and frequently reevaluate the patient for signs of central nervous system and respiratory depression [see Warnings and Precautions ( 5.12) ].
Fentanyl is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
2.3 Initial Dosage
It is safer to underestimate a patient's 24-hour fentanyl dosage and provide rescue medication (e.g. immediate-release opioid) than to overestimate the 24-hour fentanyl dosage and manage an adverse reaction to an overdose. While useful tables of opioid equivalents are readily available, there is inter-patient variability in the potency of opioid drugs and opioid formulations. Frequently reevaluate patients for signs and symptoms of opioid withdrawal and for signs of oversedation/toxicity after converting patients to fentanyl transdermal system.
- Do not initiate treatment with fentanyl transdermal system in opioid nontolerant patients [see Contraindications ( 4)] .
- The recommended starting dose when converting from other opioids to fentanyl transdermal system is intended to minimize the potential for overdosing patients with the first dose.
- When fentanyl transdermal system therapy is initiated, discontinue all opioid analgesics other than those used on an as needed basis for breakthrough pain when appropriate.
- Each fentanyl transdermal system is worn continuously for up to 72 hours [see Dosage and Administration (2.7)].
In a fentanyl transdermal system clinical trial, patients were converted from their prior opioid to fentanyl transdermal system using Table 1as a guide for the initial fentanyl transdermal system dose.
When converting patients from oral or parenteral opioids to fentanyl transdermal system, use Table 1(alternatively use Table 2for adult and pediatric patients taking opioids or doses not listed in Table 1) and consider the following:
- These are not tables of equianalgesic doses.
- The conversion doses in these tables are only for the conversion from one of the listed oral or parenteral opioid analgesics to fentanyl transdermal system.
- The tables cannot be used to convert from fentanyl transdermal system toanother opioid because these conversions will result in an overestimation of the dose of the new opioid (these conversions are conservative) and may result in fatal overdosage.
| 1 Table 1should not be used to convert from fentanyl transdermal system to other therapies because this conversion to fentanyl transdermal system is conservative. Use of Table 1for conversion to other analgesic therapies can overestimate the dose of the new agent. Overdosage of the new analgesic agent is possible [see Dosage and Administration ( 2.9) ]. | ||||
| Current Analgesic | Daily Dosage (mg/day) | |||
| Oral morphine | 60-134 | 135-224 | 225-314 | 315-404 |
| Intramuscular or Intravenous morphine | 10-22 | 23-37 | 38-52 | 53-67 |
| Oral oxycodone | 30-67 | 67.5-112 | 112.5-157 | 157.5-202 |
| Oral codeine | 150-447 | |||
| Oral hydromorphone | 8-17 | 17.1-28 | 28.1-39 | 39.1-51 |
| Intravenous hydromorphone | 1.5-3.4 | 3.5-5.6 | 5.7-7.9 | 8-10 |
| Intramuscular meperidine | 75-165 | 166-278 | 279-390 | 391-503 |
| Oral methadone | 20-44 | 45-74 | 75-104 | 105-134 |
| ⇓ | ⇓ | ⇓ | ⇓ | |
| Recommended fentanyl transdermal system dose | 25 mcg/hour | 50 mcg/hour | 75 mcg/hour | 100 mcg/hour |
Alternatively, for adult and pediatric patients taking opioids or doses not listed in Table 1, use the following methodology when converting patients from oral or parenteral opioids to fentanyl transdermal system:
- Calculate the previous 24-hour analgesic requirement.
- Convert this amount to the equianalgesic oral morphine dose using a reliable reference.
- Refer to Table 2for the range of 24-hour oral morphine doses that are recommended for conversion to each fentanyl transdermal system dose. Use this table to find the calculated 24-hour morphine dose and the corresponding recommended initial fentanyl transdermal system dose.
- Initiate fentanyl transdermal system treatment using the recommended dose and titrate patients upwards (no more frequently than 3 days after the initial dose and every 6 days thereafter) until analgesic efficacy is attained. A 37.5 mcg/hour dose may also be used. For patients that require more than 100 mcg/hour, several transdermal systems may be used.
- Do not use Table 2to convert from fentanyl transdermal system to other therapies because this conversion to fentanyl transdermal system is conservative and will overestimate the dose of the new agent.
| NOTE:In clinical trials, these ranges of daily oral morphine doses were used as a basis for conversion to fentanyl transdermal system. | |
| 1 Table 2should not be used to convert from fentanyl transdermal system to other therapies because this conversion to fentanyl transdermal system is conservative. Use of Table 2for conversion to other analgesic therapies can overestimate the dose of the new agent. Overdosage of the new analgesic agent is possible [see Dosage and Administration ( 2.9) ]. | |
|
Oral 24-
hour
Morphine (mg/day) |
Fentanyl Transdermal System
Dose (mcg/hour) |
| 60-134 | 25 |
| 135-224 | 50 |
| 225-314 | 75 |
| 315-404 | 100 |
| 405-494 | 125 |
| 495-584 | 150 |
| 585-674 | 175 |
| 675-764 | 200 |
| 765-854 | 225 |
| 855-944 | 250 |
| 945-1034 | 275 |
| 1035-1124 | 300 |
An additional intermediate strength 37.5 mcg/hour fentanyl transdermal system is available and may be considered during conversion from prior opioids or dose titration. For example, the 37.5 mcg/hour system could be used before converting or titrating to a 50 mcg/hour system.
The additional 37.5 mcg/hour system was not used in the clinical studies.
For delivery rates in excess of 100 mcg/hour, multiple systems may be used.
14 Clinical Studies (14 CLINICAL STUDIES)
Fentanyl transdermal system as therapy for pain due to cancer has been studied in 153 patients. In this patient population, fentanyl transdermal system has been administered in doses of 25 mcg/h to 600 mcg/h. Individual patients have used fentanyl transdermal system continuously for up to 866 days. At one month after initiation of fentanyl transdermal system therapy, patients generally reported lower pain intensity scores as compared to a pre-study analgesic regimen of oral morphine.
The duration of fentanyl transdermal system use varied in cancer patients; 56% of patients used fentanyl transdermal system for over 30 days, 28% continued treatment for more than 4 months, and 10% used fentanyl transdermal system for more than 1 year.
In the pediatric population, the safety of fentanyl transdermal system has been evaluated in 289 patients with chronic pain 2-18 years of age. The duration of fentanyl transdermal system use varied; 20% of pediatric patients were treated for ≤15 days; 46% for 16-30 days; 16% for 31-60 days; and 17% for at least 61 days. Twenty-five patients were treated with fentanyl transdermal system for at least 4 months and 9 patients for more than 9 months.
4 Contraindications (4 CONTRAINDICATIONS)
Fentanyl transdermal system is contraindicated in:
- patients who are not opioid-tolerant.
- the management of acute or intermittent pain, or in patients who require opioid analgesia for a short period of time.
- the management of post-operative pain, including use after outpatient or day surgeries, (e.g., tonsillectomies).
- the management of mild pain.
- patients with significant respiratory depression [see Warnings and Precautions ( 5.12) ].
- patients with acute or severe bronchial asthma in an unmonitored setting or in the absence of resuscitative equipment [see Warnings and Precautions ( 5.12) ].
- patients with known or suspected gastrointestinal obstruction, including paralytic ileus [see Warnings and Precautions ( 5.19) ].
- patients with hypersensitivity to fentanyl (e.g., anaphylaxis) or any components of the transdermal system [see Adverse Reactions ( 6.2) ].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed elsewhere in the labeling:
- Addiction, Abuse, and Misuse [see Warnings and Precautions ( 5.1)]
- Life-Threatening Respiratory Depression [see Warnings and Precautions ( 5.2)]
- Accidental Exposure [see Warnings and Precautions ( 5.3)]
- Interactions with Benzodiazepines or Other Central Nervous System Depressants [see Warnings and Precautions ( 5.4)]
- Neonatal Opioid Withdrawal Syndrome [see Warnings and Precautions ( 5.5)]
- Opioid-Induced Hyperalgesia and Allodynia [see Warnings and Precautions ( 5.10)]
- Serotonin Syndrome [see Warnings and Precautions ( 5.11)]
- Adrenal Insufficiency [see Warnings and Precautions ( 5.13)]
- Severe Hypotension [see Warnings and Precautions ( 5.14)]
- Gastrointestinal Adverse Reactions [see Warnings and Precautions ( 5.19)]
- Seizures [see Warnings and Precautions ( 5.20)]
- Withdrawal [see Warnings and Precautions ( 5.21)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Table 6includes clinically significant drug interactions with fentanyl transdermal system.
| Inhibitors of CYP3A4 | |
| Clinical Impact: | The concomitant use of fentanyl transdermal system and CYP3A4 inhibitors can increase the plasma concentration of fentanyl, resulting in increased or prolonged opioid effects particularly when an inhibitor is added after a stable dose of fentanyl transdermal system is achieved [see
Warnings and Precautions (
5.7)
].
After stopping a CYP3A4 inhibitor, as the effects of the inhibitor decline, the fentanyl transdermal system plasma concentration will decrease [see Clinical Pharmacology ( 12.3) ], resulting in decreased opioid efficacy or a withdrawal syndrome in patients who had developed physical dependence to fentanyl. |
| Intervention: | If concomitant use is necessary, consider dosage reduction of fentanyl transdermal system until stable drug effects are achieved. Evaluate patients at frequent intervals for respiratory depression and sedation.
If a CYP3A4 inhibitor is discontinued, consider increasing the fentanyl transdermal system dosage until stable drug effects are achieved. Evaluate for signs of opioid withdrawal. |
| Examples: | Macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g. ketoconazole), protease inhibitors (e.g., ritonavir), grapefruit juice. |
| CYP3A4 Inducers | |
| Clinical Impact: | The concomitant use of fentanyl transdermal system and CYP3A4 inducers can decrease the plasma concentration of fentanyl [see
Clinical Pharmacology (
12.3)
], resulting in decreased efficacy or onset of a withdrawal syndrome in patients who have developed physical dependence to fentanyl [see
Warnings and Precautions (
5.7)
].
After stopping a CYP3A4 inducer, as the effects of the inducer decline, the fentanyl plasma concentration will increase [see Clinical Pharmacology ( 12.3)] , which could increase or prolong both the therapeutic effects and adverse reactions, and may cause serious respiratory depression. |
| Intervention: | If concomitant use is necessary, consider increasing the fentanyl transdermal system dosage until stable drug effects are achieved. Evaluate for signs of opioid withdrawal.
If a CYP3A4 inducer is discontinued, consider fentanyl transdermal system dosage reduction and evaluate patients at frequent intervals for signs of respiratory depression and sedation. |
| Examples: | Rifampin, carbamazepine, phenytoin |
| Benzodiazepines and Other Central Nervous System (CNS) Depressants | |
| Clinical Impact: | Due to additive pharmacologic effect, the concomitant use of benzodiazepines or other CNS depressants, including alcohol, can increase the risk of hypotension, respiratory depression, profound sedation, coma, and death. |
| Intervention: | Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Inform patients and caregivers of this potential interaction, educate them on the signs and symptoms of respiratory depression (including sedation). If concomitant use is warranted, consider recommending or prescribing an opioid overdose reversal agent [see Dosage and Administration ( 2.2), Warnings and Precautions ( 5.1, 5.2, 5.4)] . |
| Examples: | Benzodiazepines and other sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, gabapentinoids (gabapentin or pregabalin), other opioids, alcohol. |
| Serotonergic Drugs | |
| Clinical Impact: | The concomitant use of opioids with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome [see Warnings and Precautions ( 5.11) ]. |
| Intervention: | If concomitant use is warranted, frequently evaluate the patient, particularly during treatment initiation and dose adjustment. Discontinue fentanyl transdermal system immediately if serotonin syndrome is suspected. |
| Examples: | Selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that affect the serotonin neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), monoamine oxidase (MAO) inhibitors (those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue). |
| Monoamine Oxidase Inhibitors (MAOIs) | |
| Clinical Impact: | MAOI interactions with opioids may manifest as serotonin syndrome [see Warnings and Precautions ( 5.11) ] or opioid toxicity (e.g., respiratory depression, coma). |
| Intervention: | The use of fentanyl transdermal system is not recommended for patients taking MAOIs or within 14 days of stopping such treatment. |
| Examples: | phenelzine, tranylcypromine, linezolid |
| Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics | |
| Clinical Impact: | May reduce the analgesic effect of fentanyl transdermal system and/or precipitate withdrawal symptoms. |
| Intervention: | Avoid concomitant use. |
| Examples: | butorphanol, nalbuphine, pentazocine, buprenorphine |
| Muscle Relaxants | |
| Clinical Impact: | Fentanyl transdermal system may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression. |
| Intervention: | Because respiratory depression may be greater than otherwise expected, decrease the dosage of fentanyl transdermal system and/or the muscle relaxant as necessary. Due to the risk of respiratory depression with concomitant use of skeletal muscle relaxants and opioids, consider prescribing an opioid overdose reversal agent [see Dosage and Administration ( 2.2), Warnings and Precautions ( 5.2, 5.4)] . |
| Examples: | cyclobenzaprine, metaxalone |
| Diuretics | |
| Clinical Impact: | Opioids can reduce the efficacy of diuretics by inducing the release of antidiuretic hormone. |
| Intervention: | Evaluate patients for signs of diminished diuresis and/or effects on blood pressure and increase the dosage of the diuretic as needed. |
| Anticholinergic Drugs | |
| Clinical Impact: | The concomitant use of anticholinergic drugs may increase risk of urinary retention and/or severe constipation, which may lead to paralytic ileus. |
| Intervention: | Evaluate patients for signs of urinary retention or reduced gastric motility when fentanyl transdermal system is used concomitantly with anticholinergic drugs. |
5.16 Cardiac Disease
Fentanyl transdermal system may produce bradycardia. Regularly evaluate patients with bradyarrhythmias closely for changes in heart rate, particularly when initiating therapy with fentanyl transdermal system.
8.7 Renal Impairment
The effect of renal impairment on the pharmacokinetics of fentanyl transdermal system has not been fully evaluated. A clinical pharmacology study with intravenous fentanyl in patients undergoing kidney transplantation has shown that patients with high blood urea nitrogen level had low fentanyl clearance. Because there is in-vivoevidence of renal contribution to the elimination of fentanyl transdermal system, renal impairment would be expected to have significant effects on the pharmacokinetics of fentanyl transdermal system. Avoid the use of fentanyl transdermal system in patients with severe renal impairment [see Dosage and Administration ( 2.6), Warnings and Precautions ( 5.18) and Clinical Pharmacology ( 12.3) ].
12.2 Pharmacodynamics
.
12.3 Pharmacokinetics
.
5.18 Renal Impairment
A clinical pharmacology study with intravenous fentanyl in patients undergoing kidney transplantation has shown that patients with high blood urea nitrogen level had low fentanyl clearance. Because of the long half-life of fentanyl when administered as fentanyl transdermal system, avoid the use of fentanyl transdermal system in patients with severe renal impairment. Insufficient information exists to make precise dosing recommendations regarding the use of fentanyl transdermal system in patients with impaired renal function. Therefore, to avoid starting patients with mild to moderate renal impairment on too high of a dose, start with one half of the usual dosage of fentanyl transdermal system. Regularly evaluate for signs of sedation and respiratory depression, including at each dosage increase [see Dosage and Administration ( 2.6), Use in Specific Populations ( 8.7) and Clinical Pharmacology ( 12.3) ].
8.6 Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of fentanyl transdermal system has not been fully evaluated. A clinical pharmacology study with fentanyl transdermal system in patients with cirrhosis has shown that systemic fentanyl exposure increased in these patients. Because there is in-vitroand in-vivoevidence of extensive hepatic contribution to the elimination of fentanyl transdermal system, hepatic impairment would be expected to have significant effects on the pharmacokinetics of fentanyl transdermal system. Avoid use of fentanyl transdermal system in patients with severe hepatic impairment [see Dosage and Administration ( 2.5), Warnings and Precautions ( 5.17) and Clinical Pharmacology 12.3) ].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Fentanyl transdermal system is indicated for the management of severe and persistent pain in opioid-tolerant patients, that requires an opioid analgesic and that cannot be adequately treated with alternative options, including immediate-release opioids.
Patients considered opioid-tolerant are those who are taking, for one week or longer, at least 60 mg morphine per day, 25 mcg transdermal fentanyl per hour, 30 mg oral oxycodone per day, 8 mg oral hydromorphone per day, 25 mg oral oxymorphone per day, 60 mg oral hydrocodone per day, or an equianalgesic dose of another opioid.
Limitations of Use
- Because of the risks of addiction, abuse, misuse, overdose, and death, which can occur at any dosage or duration and persist over the course of therapy [see Warnings and Precautions ( 5.1) ], reserve opioid analgesics, including fentanyl transdermal system, for use in patients for whom alternative treatment options are ineffective, not tolerated, or would be otherwise inadequate to provide sufficient management of pain.
- Fentanyl transdermal system is not indicated as an as-needed (prn) analgesic.
5.14 Severe Hypotension
Fentanyl transdermal system may cause severe hypotension including orthostatic hypotension and syncope in ambulatory patients. There is an increased risk in patients whose ability to maintain blood pressure has already been compromised by a reduced blood volume or concurrent administration of certain CNS depressant drugs (e.g., phenothiazines or general anesthetics) [see Drug Interactions ( 7) ]. Regularly evaluate these patients for signs of hypotension after initiating or titrating the dosage of fentanyl transdermal system. In patients with circulatory shock, fentanyl transdermal system may cause vasodilation that can further reduce cardiac output and blood pressure. Avoid the use of fentanyl transdermal system in patients with circulatory shock.
5.17 Hepatic Impairment
A clinical pharmacology study with fentanyl transdermal system in patients with cirrhosis has shown that systemic fentanyl exposure increased in these patients. Because of the long half-life of fentanyl when administered as fentanyl transdermal system and hepatic metabolism of fentanyl, avoid use of fentanyl transdermal system in patients with severe hepatic impairment. Insufficient information exists to make precise dosing recommendations regarding the use of fentanyl transdermal system in patients with impaired hepatic function. Therefore, to avoid starting patients with mild to moderate hepatic impairment on too high of a dose, start with one half of the usual dosage of fentanyl transdermal system. Regularly evaluate for signs of sedation and respiratory depression, including at each dosage increase [see Dosage and Administration ( 2.5), Use in Specific Populations ( 8.6) and Clinical Pharmacology ( 12.3) ].
5.3 Accidental Exposure
A considerable amount of active fentanyl remains in fentanyl transdermal system even after use as directed. Death and other serious medical problems have occurred when children and adults were accidentally exposed to fentanyl transdermal system. Accidental or deliberate application or ingestion by a child or adolescent will cause respiratory depression, and has resulted in deaths. Placing fentanyl transdermal system in the mouth, chewing it, swallowing it, or using it in ways other than indicated may cause choking or overdose that could result in death. Improper disposal of fentanyl transdermal system in the trash has resulted in accidental exposures and deaths.
Advise patients about strict adherence to the recommended handling and disposal instructions in order to prevent accidental exposure to fentanyl transdermal system [see Dosage and Administration ( 2.7), ( 2.8) ]. Exposure to fentanyl transdermal system patches discarded in the trash by children have been reported and have resulted in deaths.
12 Clinical Pharmacology (12 CLINICAL PHARMACOLOGY)
.
12.1 Mechanism of Action
Fentanyl is an opioid agonist. Fentanyl interacts predominately with the opioid mu-receptor. These mu-binding sites are distributed in the human brain, spinal cord, and other tissues.
9.1 Controlled Substance
Fentanyl transdermal system contains fentanyl, a Schedule II controlled substance.
2.8 Disposal Instructions
Failure to properly dispose of fentanyl transdermal system has resulted in accidental exposures and deaths, including deaths of children [see Warnings and Precautions ( 5.3) ].
Instruct patients to dispose of used patches immediately upon removal by folding the adhesive side of the patch to itself, then flushing down the toilet.
Instruct patients to remove unused patches from their pouches, remove the release liners, fold the patches so that the adhesive side of the patch adheres to itself, and to immediately flush the patches down the toilet.
Instruct patients to dispose of any patches remaining from a prescription as soon as they are no longer needed.
13 Non Clinical Toxicology (13 NON-CLINICAL TOXICOLOGY)
.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
.
5.13 Adrenal Insufficiency
Cases of adrenal insufficiency have been reported with opioid use, more often following greater than one month of use. Presentation of adrenal insufficiency may include non-specific symptoms and signs including nausea, vomiting, anorexia, fatigue, weakness, dizziness, and low blood pressure. If adrenal insufficiency is suspected, confirm the diagnosis with diagnostic testing as soon as possible. If adrenal insufficiency is diagnosed, treat with physiologic replacement doses of corticosteroids. Wean the patient off of the opioid to allow adrenal function to recover and continue corticosteroid treatment until adrenal function recovers. Other opioids may be tried as some cases reported use of a different opioid without recurrence of adrenal insufficiency. The information available does not identify any particular opioids as being more likely to be associated with adrenal insufficiency.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
9 Drug Abuse and Dependence (9 DRUG ABUSE AND DEPENDENCE)
.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Transdermal system:
- Fentanyl Transdermal System 12 mcg/hour* Transdermal System (system size 4.72 cm 2)
- Fentanyl Transdermal System 25 mcg/hour Transdermal System (system size 9.44 cm 2)
- Fentanyl Transdermal System 50 mcg/hour Transdermal System (system size 18.88 cm 2)
- Fentanyl Transdermal System 75 mcg/hour Transdermal System (system size 28.32 cm 2)
- Fentanyl Transdermal System 100 mcg/hour Transdermal System (system size 37.76 cm 2)
- This lowest strength is designated as 12 mcg/hour (however, the actual strength is 12.5 mcg/hour) to distinguish it from a possible 125 mcg/hour dosage that could be prescribed by using multiple transdermal systems.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of fentanyl transdermal system. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac Disorders: tachycardia, bradycardia
Eye Disorders: vision blurred
Gastrointestinal Disorders: ileus, dyspepsia
General Disorders and Administration Site Conditions: pyrexia, application site erosion and application site ulcer
Investigations: weight decreased
Nervous System Disorders: convulsions (including clonic convulsions and grand mal convulsion), amnesia, depressed level of consciousness, loss of consciousness
Psychiatric Disorders: agitation
Respiratory, Thoracic, and Mediastinal Disorders: respiratory distress, apnea, bradypnea, hypoventilation, dyspnea
Vascular Disorders: hypotension, hypertension
Serotonin syndrome: cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of opioids with serotonergic drugs.
Adrenal insufficiency: cases of adrenal insufficiency have been reported with opioid use, more often following greater than one month of use.
Anaphylaxis: anaphylaxis, including anaphylactic shock, has been reported with ingredients contained in fentanyl transdermal system.
Androgen deficiency: cases of androgen deficiency have occurred with use of opioids for an extended period of time. [see Clinical Pharmacology ( 12.2) ].
Hyperalgesia and Allodynia: Cases of hyperalgesia and allodynia have been reported with opioid therapy of any duration [see Warnings and Precautions ( 5.10) ].
Hypoglycemia: Cases of hypoglycemia have been reported in patients taking opioids. Most reports were in patients with at least one predisposing risk factor (e.g., diabetes).
Opioid-induced esophageal dysfunction (OIED): Cases of OIED have been reported in patients taking opioids, and may occur more frequently in patients taking higher doses of opioids, and/or in patients taking opioids longer term [see Warnings and Precautions (5.19)] .
Adverse Reactions from Observational Studies
A prospective, observational cohort study estimated the risks of addiction, abuse, and misuse in patients initiating long-term use of Schedule II opioid analgesics between 2017 and 2021. Study participants included in one or more analyses had been enrolled in selected insurance plans or health systems for a least one year, were free of at least one outcome at baseline, completed a minimum number of follow-up assessments, and either: 1) filled multiple extended-release/long-acting opioid analgesic prescriptions during a 90-day period (n=978); or 2) filled any Schedule II opioid analgesic prescriptions covering at least 70 of 90 days (n=1,244). Those included also had no dispensing of the qualifying opioids in the previous 6 months.
Over 12 months:
- approximately 1% to 6% of participants across the two cohorts newly met criteria for addiction, as assessed with two validated interview-based measures of moderate-to-severe opioid use disorder based on Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria, and
- approximately 9% and 22% of participants across the two cohorts newly met criteria for prescritpion opioid abuse and misuse [defined in Drug Abuse and Dependence (9.2) ], respectively, as measured with a validated self-reported instrument.
A retrospective, observational cohort study estimated the risk of opioid-involved overdose or opioid overdose-related death in patients with new long-term use of Schedule II opioid analgesics from 2006 through 2016 (n=220,249). Included patients had been enrolled in either one of two commercial insurance programs, one managed care program, or one Medicaid program for at least 9 months. New long-term use was defined as having Schedule II opioid analgesic prescriptions covering at least 70 days' supply over the 3 months prior to study entry and none during the preceding 6 months. Patients were excluded if they had an opioid-involved overdose in the 9 months prior to study entry. Overdose was measured using a validated medical code-based algorithm with linkage to the National Death Index database. The 5-year cumumlative incidence estimates for opioid-involved overdose or opioid overdose-related death ranged from approximately 1.5% to 4% across study sites, counting only the first event during follow-up. Approximately 17% of first opioid overdoses observed over the entire study period (5 years to 11 years, depending on the study site) were fatal. Higher baseline opioid dose was the strongest and most consistent predictor of opioid-involved overdose or opioid overdose-related death. Study exclusion criteria may have selected patients at lower risk of overdose, and substantial loss to follow-up (approximately 80%) also may have biased estimates.
The risk estimates from the studies described above may not be generalizable to all patients receiving opioid analgesics, such as those with exposures shorter or longer than the duration evaluated in the studies.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of fentanyl transdermal system was evaluated in 216 patients who took at least one dose of fentanyl transdermal system in a multicenter, double-blind, randomized, placebo-controlled clinical trial of fentanyl transdermal system. This trial examined patients over 40 years of age with severe pain induced by osteoarthritis of the hip or knee and who were in need of and waiting for joint replacement.
The most common adverse reactions (≥5%) in a double-blind, randomized, placebo-controlled clinical trial in patients with severe pain were nausea, vomiting, somnolence, dizziness, insomnia, constipation, hyperhidrosis, fatigue, feeling cold, and anorexia. Other common adverse reactions (≥5%) reported in clinical trials in patients with chronic malignant or nonmalignant pain were headache and diarrhea. Adverse reactions reported for ≥1% of fentanyl transdermal system-treated patients and with an incidence greater than placebo-treated patients are shown in Table 3.
The most common adverse reactions that were associated with discontinuation in patients with pain (causing discontinuation in ≥1% of patients) were depression, dizziness, somnolence, headache, nausea, vomiting, constipation, hyperhidrosis, and fatigue.
|
System/Organ Class
Adverse Reaction |
Fentanyl Transdermal System
% (N=216) |
Placebo
% (N=200) |
| Cardiac disorders | ||
| Palpitations | 4 | 1 |
| Ear and labyrinth disorders | ||
| Vertigo | 2 | 1 |
| Gastrointestinal disorders | ||
| Nausea | 41 | 17 |
| Vomiting | 26 | 3 |
| Constipation | 9 | 1 |
| Abdominal pain upper | 3 | 2 |
| Dry mouth | 2 | 0 |
| General disorders and administration site conditions | ||
| Fatigue | 6 | 3 |
| Feeling cold | 6 | 2 |
| Malaise | 4 | 1 |
| Asthenia | 2 | 0 |
| Edema peripheral | 1 | 1 |
| Metabolism and nutrition disorders | ||
| Anorexia | 5 | 0 |
| Musculoskeletal and connective tissue disorders | ||
| Muscle spasms | 4 | 2 |
| Nervous system disorders | ||
| Somnolence | 19 | 3 |
| Dizziness | 10 | 4 |
| Psychiatric disorders | ||
| Insomnia | 10 | 7 |
| Depression | 1 | 0 |
| Skin and subcutaneous tissue disorders | ||
| Hyperhidrosis | 6 | 1 |
| Pruritus | 3 | 2 |
| Rash | 2 | 1 |
Adverse reactions not reported in Table 3that were reported by ≥1% of fentanyl transdermal system-treated adult and pediatric patients (N=1854) in 11 controlled and uncontrolled clinical trials of fentanyl transdermal system used for the treatment of chronic malignant or nonmalignant pain are shown in Table 4.
|
System/Organ Class Adverse Reaction |
Fentanyl Transdermal System
% (N=1854) |
| Gastrointestinal disorders | |
| Diarrhea | 10 |
| Abdominal pain | 3 |
| Immune system disorders | |
| Hypersensitivity | 1 |
| Nervous system disorders | |
| Headache | 12 |
| Tremor | 3 |
| Paresthesia | 2 |
| Psychiatric disorders | |
| Anxiety | 3 |
| Confusional state | 2 |
| Hallucination | 1 |
| Renal and urinary disorders | |
| Urinary retention | 1 |
| Skin and subcutaneous tissue disorders | |
| Erythema | 1 |
The following adverse reactions occurred in adult and pediatric patients with an overall frequency of <1% and are listed in descending frequency within each System/Organ Class:
Cardiac disorders: cyanosis
Eye disorders: miosis
Gastrointestinal disorders: subileus
General disorders and administration site conditions: application site reaction, influenza-like illness, application site hypersensitivity, drug withdrawal syndrome, application site dermatitis
Musculoskeletal and connective tissue disorders: muscle twitching
Nervous system disorders: hypoesthesia
Psychiatric disorders: disorientation, euphoric mood
Reproductive system and breast disorders: erectile dysfunction, sexual dysfunction
Respiratory, thoracic and mediastinal disorders: respiratory depression
Skin and subcutaneous tissue disorders: eczema, dermatitis allergic, dermatitis contact
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
.
5.1 Addiction, Abuse, and Misuse
Fentanyl transdermal system contains fentanyl, an opioid agonist and a Schedule II controlled substance. As an opioid, fentanyl transdermal system exposes users to the risks of addiction, abuse, and misuse.
Although the risk of addiction in any individual is unknown, it can occur in patients appropriately prescribed fentanyl transdermal system. Addiction can occur at recommended doses and if the drug is misused or abused. The risk of opioid-related overdose or overdose-related death is increased with higher opioid doses, and this risk persists over the course of therapy. In postmarketing studies, addiction, abuse, misuse, and fatal and non-fatal opioid overdose were observed in patients with long-term opioid use [see Adverse Reactions (6.2)].
Assess each patient's risk for opioid addiction, abuse, or misuse prior to prescribing fentanyl transdermal system, and reassess all patients receiving fentanyl transdermal system for the development of these behaviors and conditions. Risks are increased in patients with a personal or family history of substance abuse (including drug or alcohol abuse or addiction) or mental illness (e.g., major depression). The potential for these risks should not, however, prevent the proper management of pain in any given patient. Patients at increased risk may be prescribed opioids such as fentanyl transdermal system, but use in such patients necessitates intensive counseling about the risks and proper use of fentanyl transdermal system along with frequent reevaluation for signs of addiction, abuse, and misuse. Consider recommending or prescribing an opioid overdose reversal agent [see Dosage and Administration ( 2.2), Warnings and Precautions ( 5.2) ].
Abuse or misuse of fentanyl transdermal system by placing it in the mouth, chewing it, swallowing it, or using it in ways other than indicated may cause choking, overdose, and death [see Overdosage ( 10) ].
Opioids are sought for nonmedical use and are subject to diversion from legitimate prescribed use. Consider these risks when prescribing or dispensing fentanyl transdermal system. Strategies to reduce these risks include prescribing the drug in the smallest appropriate quantity and advising the patient on careful storage of the drug during the course of treatment and the proper disposal of unused drug. Contact local state professional licensing board or state-controlled substances authority for information on how to prevent and detect abuse or diversion of this product.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling ( Medication Guideand Instructions for Use).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Fentanyl transdermal system is supplied in cartons containing 5 individually packaged systems. See chart for information regarding individual systems.
|
* This lowest strength is designated as 12 mcg/hour (however, the actual strength is 12.5 mcg/hour) to distinguish it from a 125 mcg/hour strength that could be prescribed by using multiple transdermal systems. |
|||
|
Fentanyl Transdermal System Strength
(mcg/hour) |
System
Size (cm 2 ) |
Fentanyl
Content (mg) |
NDC
Number |
| Fentanyl transdermal system – 12 * | 4.72 | 1.33 | 47781-423-47 |
| Fentanyl transdermal system – 25 | 9.44 | 2.66 | 47781-424-47 |
| Fentanyl transdermal system – 50 | 18.88 | 5.32 | 47781-426-47 |
| Fentanyl transdermal system – 75 | 28.32 | 7.99 | 47781-427-47 |
| Fentanyl transdermal system – 100 | 37.76 | 10.65 | 47781-428-47 |
Store in original unopened pouch. Store up to 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
Store fentanyl transdermal system securely and dispose of properly [see Patient Counseling Information ( 17) ].
5.5 Neonatal Opioid Withdrawal Syndrome
Use of fentanyl transdermal system for an extended period of time during pregnancy can result in withdrawal in the neonate. Neonatal opioid withdrawal syndrome, unlike opioid withdrawal syndrome in adults, may be life-threatening if not recognized and treated, and requires management according to protocols developed by neonatology experts. Observe newborns for signs of neonatal opioid withdrawal syndrome and manage accordingly. Advise pregnant women using opioids for an extended period of time of the risk of neonatal opioid withdrawal syndrome and ensure that appropriate treatment will be available [see Use in Specific Populations ( 8.1), Patient Counseling Information ( 17) ].
2.4 Titration and Maintenance of Therapy
Individually titrate fentanyl transdermal system to a dose that provides adequate analgesia and minimizes adverse reactions. Continually reevaluate patients receiving fentanyl transdermal system to assess the maintenance of pain control, signs and symptoms of opioid withdrawal, and other adverse reactions, as well as to reassess for the development of addiction, abuse, or misuse [see Warnings and Precautions ( 5.1, 5.21) ]. Frequent communication is important among the prescriber, other members of the healthcare team, the patient, and the caregiver/family during periods of changing analgesic requirements, including initial titration. During use of opioid therapy for an extended period of time, periodically reassess the continued need for use of opioid analgesics.
Patients who experience breakthrough pain may require a dosage adjustment of fentanyl transdermal system, or may need rescue medication with an appropriate dose of an immediate-release analgesic. If the level of pain increases after dosage stabilization, attempt to identify the source of increased pain before increasing the fentanyl transdermal system dosage.
The dosing interval for fentanyl transdermal system is 72 hours. Do not increase the fentanyl transdermal system dose for the first time until at least 3 days after the initial application. Titrate the dose based on the daily dose of supplemental opioid analgesics required by the patient on the second or third day of the initial application.
It may take up to 6 days for fentanyl levels to reach equilibrium on a new dose [see Clinical Pharmacology ( 12.3) ]. Therefore, evaluate patients for further titration after no less than two 3-day applications before any further increase in dosage is made.
Base dosage increments on the daily dosage of supplementary opioids, using the ratio of 45 mg/24 hours of oral morphine to a 12 mcg/hour increase in fentanyl transdermal system dose.
If after increasing the dosage, unacceptable opioid-related adverse reactions are observed, (including an increase in pain after dosage increase), consider reducing the dosage [see Warnings and Precautions ( 5)] . Adjust the dose to obtain an appropriate balance between management of pain and opioid-related adverse reactions.
A small proportion of adult patients may not achieve adequate analgesia using a 72-hour dosing interval and may require systems to be applied at 48 hours rather than at 72 hours, only if adequate pain control cannot be achieved using a 72-hour regimen. An increase in the fentanyl transdermal system dose should be evaluated before changing dosing intervals in order to maintain patients on a 72-hour regimen.
Dosing intervals less than every 72 hours were not studied in children and adolescents and are not recommended.
5.2 Life Threatening Respiratory Depression (5.2 Life-Threatening Respiratory Depression)
Serious, life-threatening, or fatal respiratory depression has been reported with the use of opioids, even when used as recommended. Respiratory depression, if not immediately recognized and treated, may lead to respiratory arrest and death. Management of respiratory depression may include close observation, supportive measures, and use of opioid overdose reversal agents, depending on the patient's clinical status [see Overdosage ( 10) ]. Carbon dioxide (CO 2) retention from opioid-induced respiratory depression can exacerbate the sedating effects of opioids.
Fentanyl transdermal system is indicated only in opioid tolerant patients because of the risk for respiratory depression and death. While serious, life-threatening, or fatal respiratory depression can occur at any time during the use of fentanyl transdermal system, the risk is greatest during the initiation of therapy or following a dosage increase.
To reduce the risk of respiratory depression, proper dosing and titration of fentanyl transdermal system are essential [see Dosage and Administration ( 2) ]. Overestimating the fentanyl transdermal system dosage when converting patients from another opioid product can result in fatal overdose with the first dose.
Accidental exposure to fentanyl transdermal system, especially in children, can result in respiratory depression and death due to an overdose of fentanyl.
Educate patients and caregivers on how to recognize respiratory depression and emphasize the importance of calling 911 or getting emergency medical help right away in the event of a known or suspected overdose [see Patient Counseling Information ( 17) ].
Opioids can cause sleep-related breathing disorders including central sleep apnea (CSA) and sleep-related hypoxemia. Opioid use increases the risk of CSA in a dose-dependent fashion. In patients who present with CSA, consider decreasing the opioid dosage using best practices for opioid taper [see Dosage and Administration ( 2.9) ].
5.19 Risks of Gastrointestinal Complications
Fentanyl transdermal system is contraindicated in patients with known or suspected gastrointestinal obstruction, including paralytic ileus.
The fentanyl in fentanyl transdermal system may cause spasm of the sphincter of Oddi. Opioids may cause increases in serum amylase. Regularly evaluate patients with biliary tract disease, including acute pancreatitis for worsening symptoms.
Cases of opioid-induced esophageal dysfuntion (OIED) have been reported in patients taking opioids. The risk of OIED may increase as the dose and/or duration of opioids increases. Regularly evaluate patients for signs and sypmtoms of OIED (e.g. dysphagia, regurgitation, non-cardiac chest pain), and if necessary, adjust opioid therapy as clinically appropriate [see Clinical Pharmacology (12.2)].
5.22 Risks of Driving and Operating Machinery
Fentanyl transdermal system may impair the mental or physical abilities required for the performance of potentially dangerous activities, such as driving a car or operating machinery. Warn patients not to drive or operate dangerous machinery unless they are tolerant to the effects of the fentanyl transdermal system and know how they will react to the medication [see Patient Counseling Information ( 17) ].
5.10 Opioid Induced Hyperalgesia and Allodynia
Opioid-Induced Hyperalgesia (OIH) occurs when an opioid analgesic paradoxically causes an increase in pain, or an increase in sensitivity to pain. This condition differs from tolerance, which is the need for increasing doses of opioids to maintain a defined effect [see Dependence ( 9.3) ]. Symptoms of OIH include (but may not be limited to) increased levels of pain upon opioid dosage increase, decreased levels of pain upon opioid dosage decrease, or pain from ordinarily non-painful stimuli (allodynia). These symptoms may suggest OIH only if there is no evidence of underlying disease progression, opioid tolerance, opioid withdrawal, or addictive behavior.
Cases of OIH have been reported, both with short-term and longer-term use of opioid analgesics. Though the mechanism of OIH is not fully understood, multiple biochemical pathways have been implicated. Medical literature suggests a strong biologic plausibility between opioid analgesics and OIH and allodynia. If a patient is suspected to be experiencing OIH, carefully consider appropriately decreasing the dose of the current opioid analgesic, or opioid rotation (safely switching the patient to a different opioid moiety) [see Dosage and Administration ( 2.9) , Warnings and Precautions ( 5.21) ].
2.7 Administration of Fentanyl Transdermal System
FENTANYL TRANSDERMAL SYSTEM PATCHES ARE FOR TRANSDERMAL USE ONLY.
Proper handling of fentanyl transdermal system is necessary in order to prevent serious adverse outcomes, including death, associated with accidental secondary exposure to fentanyl transdermal system [see Warnings and Precautions ( 5.3) ].
Application and Handling Instructions
- Patients should apply fentanyl transdermal system to intact, non-irritated, and non-irradiated skin on a flat surface such as the chest, back, flank, or upper arm. In young children and persons with cognitive impairment, adhesion should be monitored and the upper back is the preferred location to minimize the potential of inappropriate patch removal. Hair at the application site may be clipped (not shaved) prior to system application. If the site of fentanyl transdermal system application must be cleansed prior to application of the patch, do so with clear water. Do not use soaps, oils, lotions, alcohol, or any other agents that might irritate the skin or alter its characteristics. Allow the skin to dry completely prior to patch application.
- Patients should apply fentanyl transdermal system immediately upon removal from the sealed package. The patch must not be altered (e.g., cut) in any way prior to application. Fentanyl transdermal system should not be used if the pouch seal is broken or if the patch is cut or damaged.
- The transdermal system is pressed firmly in place with the palm of the hand for 30 seconds, making sure the contact is complete, especially around the edges.
- Each fentanyl transdermal system patch may be worn continuously for up to 72 hours. The next patch is applied to a different skin site after removal of the previous transdermal system.
- If problems with adhesion of the fentanyl transdermal system patch occur, the edges of the patch may be taped with first-aid tape. If problems with adhesion persist, the patch may be overlayed with a transparent adhesive film dressing.
- If the patch falls off before 72 hours, dispose of it by folding in half and flushing down the toilet. A new patch may be applied to a different skin site.
- Patients (or caregivers who apply fentanyl transdermal system) should wash their hands immediately with soap and water after applying fentanyl transdermal system.
- Contact with unwashed or unclothed application sites can result in secondary exposure to fentanyl transdermal system and should be avoided. Examples of accidental exposure include transfer of a fentanyl transdermal system patch from an adult's body to a child while hugging, sharing the same bed as the patient, accidental sitting on a patch and possible accidental exposure of a caregiver's skin to the medication in the patch while applying or removing the patch.
- Instruct patients, family members, and caregivers to keep patches in a secure location out of the reach of children and of others for whom fentanyl transdermal system was not prescribed.
Avoidance of Heat
Instruct patients to avoid exposing the fentanyl transdermal system application site and surrounding area to direct external heat sources, such as heating pads or electric blankets, heat or tanning lamps, sunbathing, hot baths, saunas, hot tubs, and heated water beds, while wearing the system [see Warnings and Precautions ( 5.8) ].
2.1 Important Dosage and Administration Instructions
- Fentanyl transdermal system should be prescribed only by healthcare professionals who are knowledgeable about the use of extended-release/long-acting opioids and how to mitigate the associated risks.
- Due to the risk of respiratory depression, fentanyl transdermal system is only indicated for use in patients who are already opioid-tolerant. Discontinue or taper all other extended-release opioids when beginning fentanyl transdermal system therapy. As fentanyl transdermal system is only for use in opioid-tolerant patients, do not begin any patient on fentanyl transdermal system as the first opioid [see Indications and Usage ( 1)] .
- Use the lowest effective dosage for the shortest duration of time consistent with individual patient treatment goals [see Warnings and Precautions ( 5)] . Because the risk of overdose increases as opioid doses increase, reserve titration to higher doses of fentanyl transdermal system for patients in whom lower doses are insufficiently effective and in whom the expected benefits of using a higher dose opioid clearly outweigh the substantial risks.
- Initiate the dosing regimen for each patient individually, taking into account the patient's underlying cause and severity of pain, prior analgesic treatment and response, and risk factors for addiction, abuse, and misuse [see Warnings and Precautions ( 5.1)] .
- Respiratory depression can occur at any time during opioid therapy, especially when initiating and following dosage increases with fentanyl transdermal system. Consider this risk when selecting an initial dose and when making dose adjustments [see Warnings and Precautions ( 5)] .
- Each fentanyl transdermal system is worn continuously for up to 72 hours [see Dosage and Administration (2.7)].
2.6 Dosage Modifications in Patients With Renal Impairment (2.6 Dosage Modifications in Patients with Renal Impairment)
Avoid the use of fentanyl transdermal system in patients with severe renal impairment. In patients with mild to moderate renal impairment, start with one half of the usual dosage of fentanyl transdermal system. Closely monitor for signs of respiratory and central nervous system depression, including at each dosage increase [see Warnings and Precautions ( 5.18), Use in Specific Populations ( 8.7) and Clinical Pharmacology ( 12.3) ].
2.5 Dosage Modifications in Patients With Hepatic Impairment (2.5 Dosage Modifications in Patients with Hepatic Impairment)
Avoid the use of fentanyl transdermal system in patients with severe hepatic impairment. In patients with mild to moderate hepatic impairment, start with one half of the usual dosage of fentanyl transdermal system. Closely monitor for signs of respiratory and central nervous system depression, including at each dosage increase [see Warnings and Precautions ( 5.17), Use in Specific Populations ( 8.6) and Clinical Pharmacology ( 12.3) ].
13.1 Carcinogenesis, Mutagenesis, and Impairment of Fertility
.
5.11 Serotonin Syndrome With Concomitant Use of Serotonergic Drugs (5.11 Serotonin Syndrome with Concomitant Use of Serotonergic Drugs)
Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of fentanyl transdermal system with serotonergic drugs. Serotonergic drugs include selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that affect the serotonergic neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), and drugs that impair metabolism of serotonin (including MAO inhibitors, both those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue) [see Drug Interactions ( 7)] . This may occur within the recommended dosage range. Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination, rigidity), and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). The onset of symptoms generally occurs within several hours to a few days of concomitant use, but may occur later than that. Discontinue fentanyl transdermal system immediately if serotonin syndrome is suspected.
5.20 Increased Risk of Seizures in Patients With Seizure Disorders (5.20 Increased Risk of Seizures in Patients with Seizure Disorders)
The fentanyl in fentanyl transdermal system may increase the frequency of seizures in patients with seizure disorders, and may increase the risk of seizures occurring in other clinical settings associated with seizures. Regularly evaluate patients with a history of seizure disorders for worsened seizure control during fentanyl transdermal system therapy.
5.6 Opioid Analgesic Risk Evaluation and Mitigation Strategy (rems) (5.6 Opioid Analgesic Risk Evaluation and Mitigation Strategy (REMS))
To ensure that the benefits of opioid analgesics outweigh the risks of addiction, abuse, and misuse, the Food and Drug Administration (FDA) has required a Risk Evaluation and Mitigation Strategy (REMS) for these products. Under the requirements of the REMS, drug companies with approved opioid analgesic products must make REMS-compliant education programs available to healthcare providers. Healthcare providers are strongly encouraged to do all of the following:
- Complete a REMS-compliant education programoffered by an accredited provider of continuing education (CE) or another education program that includes all the elements of the FDA Education Blueprint for Health Care Providers Involved in the Management or Support of Patients with Pain.
- Discuss the safe use, serious risks, and proper storage and disposal of opioid analgesics with patients and/or their caregivers every time these medicines are prescribed. The Patient Counseling Guide (PCG) can be obtained at this link: www.fda.gov/OpioidAnalgesicREMSPCG.
- Emphasize to patients and their caregivers the importance of reading the Medication Guide that they will receive from their pharmacist every time an opioid analgesic is dispensed to them.
- Consider using other tools to improve patient, household, and community safety, such as patient-prescriber agreements that reinforce patient-prescriber responsibilities.
To obtain further information on the opioid analgesic REMS and for a list of accredited REMS CME/CE, call 1-800-503-0784, or log on to www.opioidanalgesicrems.com. The FDA Blueprint can be found at www.fda.gov/OpioidAnalgesicREMSBlueprint.
2.9 Safe Reduction Or Discontinuation of Fentanyl Transdermal System (2.9 Safe Reduction or Discontinuation of Fentanyl Transdermal System)
Do not rapidly reduce or abruptly discontinue fentanyl transdermal system in patients who may be physically dependent on opioids. Rapid reduction or abrupt discontinuation of opioid analgesics in patients who are physically dependent on opioids has resulted in serious withdrawal symptoms, uncontrolled pain, and suicide. Rapid reduction or abrupt discontinuation has also been associated with attempts to find other sources of opioid analgesics, which may be confused with drug-seeking for abuse. Patients may also attempt to treat their pain or withdrawal symptoms with illicit opioids, such as heroin, and other substances.
When a decision has been made to decrease the dose or discontinue therapy in an opioid-dependent patient taking fentanyl transdermal system, there are a variety of factors that should be considered, including the total daily dose of opioid (including fentanyl transdermal system) the patient has been taking, the duration of treatment, the type of pain being treated, and the physical and psychological attributes of the patient. It is important to ensure ongoing care of the patient and to agree on an appropriate tapering schedule and follow-up plan so that patient and provider goals and expectations are clear and realistic. When opioid analgesics are being discontinued due to a suspected substance use disorder, evaluate and treat the patient, or refer for evaluation and treatment of the substance use disorder. Treatment should include evidence-based approaches, such as medication assisted treatment of opioid use disorder. Complex patients with comorbid pain and substance use disorders may benefit from referral to a specialist.
There are no standard opioid tapering schedules that are suitable for all patients. Good clinical practice dictates a patient-specific plan to taper the dose of the opioid gradually. For patients on fentanyl transdermal system who are physically opioid-dependent, initiate the taper by a small enough increment (e.g., no greater than 25% of the total daily dose) to avoid withdrawal symptoms, and proceed with dose-lowering at an interval of every 2 to 4 weeks. Patients who have been taking opioids for briefer periods of time may tolerate a more rapid taper.
It may be necessary to provide the patient with a lower dosage strength to accomplish a successful taper. Reassess the patient frequently to manage pain and withdrawal symptoms, should they emerge. Common withdrawal symptoms include restlessness, lacrimation, rhinorrhea, yawning, perspiration, chills, myalgia, and mydriasis. Other signs and symptoms also may develop, including irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory rate, or heart rate. If withdrawal symptoms arise, it may be necessary to pause the taper for a period of time or raise the dose of the opioid analgesic to the previous dose, and then proceed with a slower taper. In addition, evaluate patients for any changes in mood, emergence of suicidal thoughts, or use of other substances.
When managing patients taking opioid analgesics, particularly those who have been treated for an extended period of time, and/or with high doses for chronic pain, ensure that a multimodal approach to pain management, including mental health support (if needed), is in place prior to initiating an opioid analgesic taper. A multimodal approach to pain management may optimize the treatment of chronic pain, as well as assist with the successful tapering of the opioid analgesic [see Warnings and Precautions ( 5.21), Drug Abuse and Dependence ( 9.3) ].
5.9 Risk of Increased Fentanyl Absorption With Elevated Body Temperature (5.9 Risk of Increased Fentanyl Absorption with Elevated Body Temperature)
Based on a pharmacokinetic model, serum fentanyl concentrations could theoretically increase by approximately one-third for patients with a body temperature of 40°C (104°F) due to temperature-dependent increases in fentanyl released from the system and increased skin permeability. Monitor patients wearing fentanyl transdermal systems who develop fever closely for sedation and respiratory depression and reduce the fentanyl transdermal system dose, if necessary. Warn patients to avoid strenuous exertion that leads to increased core body temperature while wearing fentanyl transdermal system to avoid the risk of potential overdose and death.
5.8 Risk of Increased Fentanyl Absorption With Application of External Heat (5.8 Risk of Increased Fentanyl Absorption with Application of External Heat)
Exposure to heat may increase fentanyl absorption and there have been reports of overdose and death as a result of exposure to heat. A clinical pharmacology study conducted in healthy adult subjects has shown that the application of heat over the fentanyl transdermal system increased fentanyl exposure [see Clinical Pharmacology ( 12.3) ].
Warn patients to avoid exposing the fentanyl transdermal system application site and surrounding area to direct external heat sources [see Dosage and Administration ( 2.7)] .
5.4 Risks From Concomitant Use With Benzodiazepines Or Other Cns Depressants (5.4 Risks from Concomitant Use with Benzodiazepines or Other CNS Depressants)
Profound sedation, respiratory depression, coma, and death may result from the concomitant use of fentanyl transdermal system with benzodiazepines and/or other CNS depressants, including alcohol (e.g., non-benzodiazepine sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, gabapentinoids [gabapentin or pregabalin], and other opioids). Because of these risks, reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate.
Observational studies have demonstrated that concomitant use of opioid analgesics and benzodiazepines increases the risk of drug-related mortality compared to use of opioid analgesics alone. Because of similar pharmacological properties, it is reasonable to expect similar risk with the concomitant use of other CNS depressant drugs with opioid analgesics [see Drug Interactions ( 7) ].
If the decision is made to prescribe a benzodiazepine or other CNS depressant concomitantly with an opioid analgesic, prescribe the lowest effective dosages and minimum durations of concomitant use. In patients already receiving an opioid analgesic, prescribe a lower initial dose of the benzodiazepine or other CNS depressant than indicated in the absence of an opioid, and titrate based on clinical response. If an opioid analgesic is initiated in a patient already taking a benzodiazepine or other CNS depressant, prescribe a lower initial dose of the opioid analgesic, and titrate based on clinical response. Inform patients and caregivers of this potential interaction, educate them on the signs and symptoms of respiratory depression (including sedation).
If concomitant use is warranted, consider recommending or prescribing an opioid overdose reversal agent [see Dosage and Administration ( 2.2), Warnings and Precautions ( 5.2), Overdosage (10)] .
Advise both patients and caregivers about the risks of respiratory depression and sedation when fentanyl transdermal system is used with benzodiazepines or other CNS depressants (including alcohol and illicit drugs). Advise patients not to drive or operate heavy machinery until the effects of concomitant use of the benzodiazepine or other CNS depressant have been determined. Screen patients for risk of substance use disorders, including opioid abuse and misuse, and warn them of the risk for overdose and death associated with the use of additional CNS depressants including alcohol and illicit drugs [see Drug Interactions ( 7), Patient Counseling Information ( 17) ].
Warning: Serious and Life Threatening Risks From Use of Fentanyl Transdermal System (WARNING: SERIOUS AND LIFE-THREATENING RISKS FROM USE OF FENTANYL TRANSDERMAL SYSTEM)
.
5.7 Risks of Concomitant Use Or Discontinuation of Cytochrome P450 3a4 Inhibitors and Inducers (5.7 Risks of Concomitant Use or Discontinuation of Cytochrome P450 3A4 Inhibitors and Inducers)
Concomitant use of fentanyl transdermal system with a CYP3A4 inhibitor, such as macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g., ketoconazole), and protease inhibitors (e.g., ritonavir), may increase plasma concentrations of fentanyl and prolong opioid adverse reactions, which may cause potentially fatal respiratory depression [see Warnings and Precautions ( 5.2) ], particularly when an inhibitor is added after a stable dose of fentanyl transdermal system is achieved. Similarly, discontinuation of a CYP3A4 inducer, such as rifampin, carbamazepine, and phenytoin, in fentanyl transdermal system-treated patients may increase fentanyl plasma concentrations and prolong opioid adverse reactions. When using fentanyl transdermal system with CYP3A4 inhibitors or discontinuing CYP3A4 inducers in fentanyl transdermal system-treated patients, evaluate patients at frequent intervals and consider dosage reduction of fentanyl transdermal system until stable drug effects are achieved [see Dosage and Administration ( 2.4), Drug Interactions ( 7) ].
Concomitant use of fentanyl transdermal system with CYP3A4 inducers or discontinuation of a CYP3A4 inhibitor could decrease fentanyl transdermal system plasma concentrations, decrease opioid efficacy or, possibly, lead to a withdrawal syndrome in a patient who had developed physical dependence to fentanyl. When using fentanyl transdermal system with CYP3A4 inducers or discontinuing CYP3A4 inhibitors, monitor patients closely at frequent intervals and consider increasing the opioid dosage if needed to maintain adequate analgesia or if symptoms of opioid withdrawal occur [see Drug Interactions ( 7) ].
2.2 Patient Access to An Opioid Overdose Reversal Agent for the Emergency Treatment of Opioid Overdose (2.2 Patient Access to an Opioid Overdose Reversal Agent for the Emergency Treatment of Opioid Overdose)
Inform patients and caregivers about opioid overdose reversal agents (e.g. naloxone, nalmefene). Discuss the importance of having access to an opioid overdose reversal agent, especially if the patient has risk factors for overdose (e.g. concomitant use of CNS depressants, a history of opioid use disorder, or prior opioid overdose) or if there are household members (including children) or other close contacts at risk for accidental ingestion or opioid overdose. The presence of risk factors for overdose should not prevent the management of pain in any patient [see Warnings and Precautions ( 5.1, 5.2, 5.4) ].
Discuss the options for obtaining an opioid overdose reversal agent (e.g. prescription, over-the-counter, or as part of a community-based program) [see Warnings and Precautions (5.2)].
There are important differences among the opioid overdose reversal agents, such as route of administration, product strength, approved patient age range, and pharmacokinetics. Be familiar with these differences, as outlined in the approved labeling for those products, prior to recommending or prescribing such an agent.
5.15 Risks of Use in Patients With Increased Intracranial Pressure, Brain Tumors, Head Injury, Or Impaired Consciousness (5.15 Risks of Use in Patients with Increased Intracranial Pressure, Brain Tumors, Head Injury, or Impaired Consciousness)
In patients who may be susceptible to the intracranial effects of CO 2retention (e.g., those with evidence of increased intracranial pressure or brain tumors), fentanyl transdermal system may reduce respiratory drive, and the resultant CO 2retention can further increase intracranial pressure. Monitor such patients for signs of sedation and respiratory depression, particularly when initiating therapy with fentanyl transdermal system.
Opioids may also obscure the clinical course in a patient with a head injury. Avoid the use of fentanyl transdermal system in patients with impaired consciousness or coma.
5.12 Life Threatening Respiratory Depression in Patients With Chronic Pulmonary Disease Or in Elderly, Cachectic, Or Debilitated Patients (5.12 Life-Threatening Respiratory Depression in Patients with Chronic Pulmonary Disease or in Elderly, Cachectic, or Debilitated Patients)
The use of fentanyl transdermal systems in patients with acute or severe bronchial asthma in an unmonitored setting or in the absence of resuscitative equipment is contraindicated.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:46.412644 · Updated: 2026-03-14T22:51:19.879041