Dermacort
24044b0f-dd1e-4762-82b8-7a28b08c2f77
34390-5
HUMAN OTC DRUG LABEL
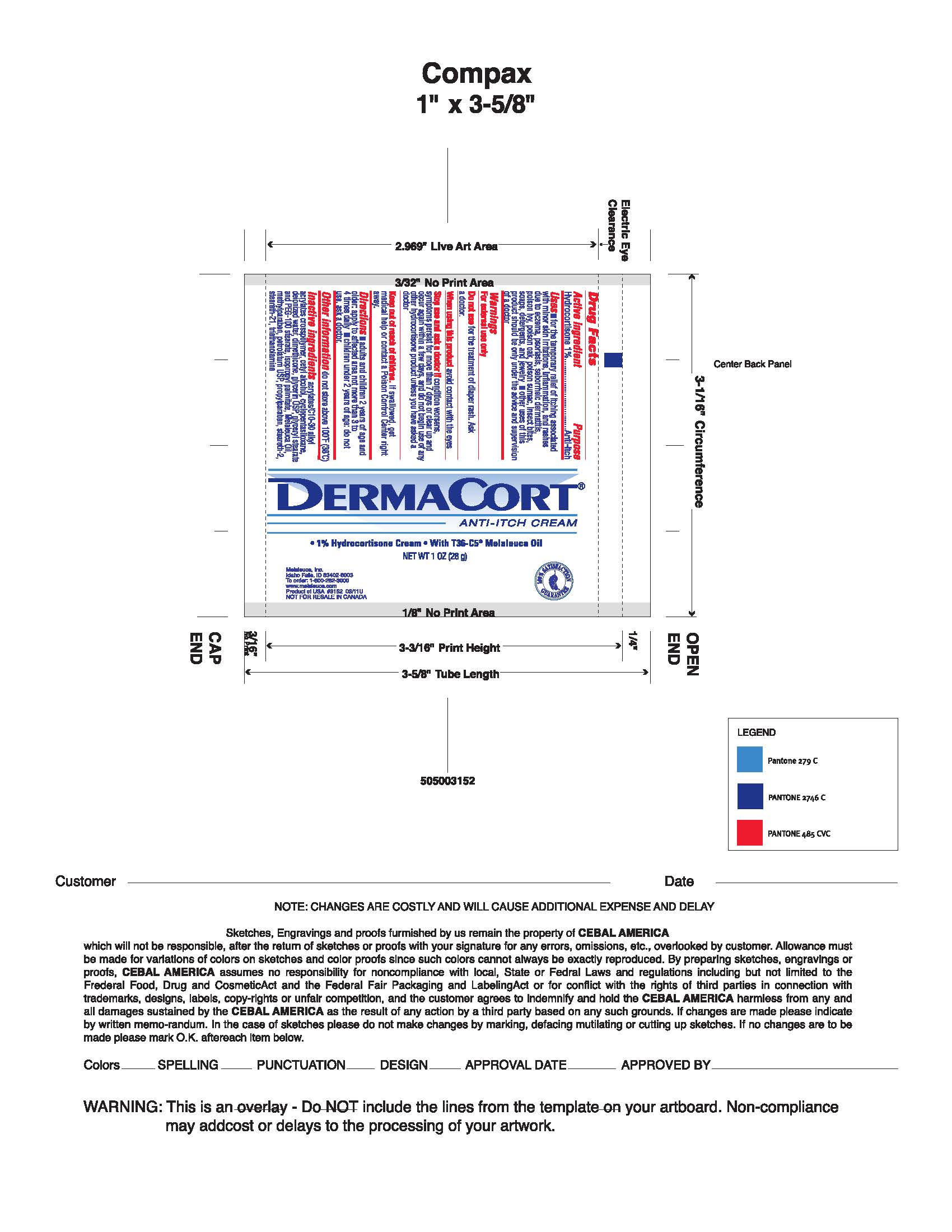
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient Hydrocortisone 1%
Medication Information
Warnings and Precautions
Warnings
For external use only.
Indications and Usage
Uses
- For the temporary relief of itching associated with minor skin irritations, inflammation and rashes due to eczema, psoriasis, , seborrheic dermatitis, poison ivy, poison oak, poison sumac, insect bites, soaps, detergents, and jewelery
- Other use of this product should be only under the advice and supervision of a doctor.
Dosage and Administration
Directions adults and children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily. Children under 2 years of age: Do not use, consult a doctor.
Description
Active ingredient Hydrocortisone 1%
Section 44425-7
Other information Do not store above 100°F.
Section 50565-1
Keep out of reach of children.
In case of accidental ingestion, seek professional assistance or contact a Poison Control Center immediately.
Section 50566-9
Stop use and ask a doctor if conditions worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days, stop use of this product and do not begin use of any other hydrocortisone product unless you have consulted a doctor.
Section 50567-7
When using this product avoid contact with the eyes.
Section 50570-1
Do not use for the treatment of diaper rash. Consult a doctor.
Section 51727-6
Inactive ingredients Acrylates/C10-30 Alkyl Acrylates Crosspolymer, Cetyl Alcohol, Cyclopentasiloxane, Deionized Water, Dimethicone, Glycerin USP, Glyceryl Stearate and PEG-100 Stearate, Isopropyl Palmitate, Melaleuca Oil, Methylparaben, Petrolatum USP, Propylparaben, Serareth-2, Steareth-21, Triethanolamine.
Section 51945-4
Section 55105-1
Purpose
Anti-itch
Section 55106-9
Active ingredient
Hydrocortisone 1%
Structured Label Content
Indications and Usage (34067-9)
Uses
- For the temporary relief of itching associated with minor skin irritations, inflammation and rashes due to eczema, psoriasis, , seborrheic dermatitis, poison ivy, poison oak, poison sumac, insect bites, soaps, detergents, and jewelery
- Other use of this product should be only under the advice and supervision of a doctor.
Dosage and Administration (34068-7)
Directions adults and children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily. Children under 2 years of age: Do not use, consult a doctor.
Warnings and Precautions (34071-1)
Warnings
For external use only.
Section 44425-7 (44425-7)
Other information Do not store above 100°F.
Section 50565-1 (50565-1)
Keep out of reach of children.
In case of accidental ingestion, seek professional assistance or contact a Poison Control Center immediately.
Section 50566-9 (50566-9)
Stop use and ask a doctor if conditions worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days, stop use of this product and do not begin use of any other hydrocortisone product unless you have consulted a doctor.
Section 50567-7 (50567-7)
When using this product avoid contact with the eyes.
Section 50570-1 (50570-1)
Do not use for the treatment of diaper rash. Consult a doctor.
Section 51727-6 (51727-6)
Inactive ingredients Acrylates/C10-30 Alkyl Acrylates Crosspolymer, Cetyl Alcohol, Cyclopentasiloxane, Deionized Water, Dimethicone, Glycerin USP, Glyceryl Stearate and PEG-100 Stearate, Isopropyl Palmitate, Melaleuca Oil, Methylparaben, Petrolatum USP, Propylparaben, Serareth-2, Steareth-21, Triethanolamine.
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Purpose
Anti-itch
Section 55106-9 (55106-9)
Active ingredient
Hydrocortisone 1%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:52.103375 · Updated: 2026-03-14T23:08:14.770901