Systane Pro Preservative Free
23b3515f-fbe5-4398-b2df-8b2eccdd01e2
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredients Purpose Propylene Glycol 0.6% Lubricant
Medication Information
Description
Active Ingredients Purpose Propylene Glycol 0.6% Lubricant
Uses
- for the temporary relief of burning and irritation due to dryness of the eye
- for the temporary relief of discomfort due to minor irritations of the eye or to exposure to wind or sun
- for use as a protectant against further irritation or to relieve dryness of the eye
- for use as a lubricant to prevent further irritation or to relieve dryness of the eye
Section 55105-1
| Active Ingredients | Purpose |
| Propylene Glycol 0.6% | Lubricant |
Warnings
For external use only
Directions
- shake well before using
- put 1 or 2 drops in the affected eye(s) as needed
- see side of carton for directions for use
Do Not Use
- if this product changes color
- if you are sensitive to any ingredient in this product
Questions?
In the U.S. call 1-800-757-9195
www.systane.com
[email protected]
Other Information
- store at room temperature
Directions for Use:
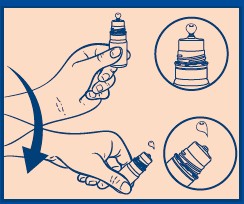
- Hold the bottle just below the cap and twist the cap to open.
- BEFORE FIRST USE, hold the bottle upside down and dispense one drop of product to discard.
- Shake the bottle downward to remove residual product (see picture).
- Place 1 or 2 drops in the affected eye(s) as needed and blink.
-
After use, shake the bottle downward to remove residual product that may be left on the tip (see picture on side panel).
- Replace cap after use.
- Discard 30 days after opening.
Inactive Ingredients
boric acid, dimyristoyl phosphatidylglycerol, hydroxypropyl guar, mineral oil, polyoxyl 40 stearate, sodium hyaluronate, sorbitan tristearate, sorbitol and purified water. May contain hydrochloric acid and/or sodium hydroxide to adjust pH.
Principal Display Panel
Preservative-Free
Systane
®
LUBRICANT EYE DROPS
PRO PF
TRIPLE ACTION
DRY EYE RELIEF
Hydrates, Restores, Protects
Long-Lasting
With Hyaluronate
Preservative-Free Bottle
#1 DOCTOR RECOMMENDED BRANDS1
Alcon
Sterile
10mL (0.34 FL OZ)
LOT: EXP.:
Side Panel:
TAMPER EVIDENT:
For your protection, this bottle has a tamper evident ring around the bottom of the cap. Do not
use if this ring is damaged or missing at the time of purchase.
1Based on a survey of eye care professionals. Data on file.
Alcon Laboratories, Inc.
Fort Worth, TX 76134 USA
Country of Origin United States
U.S. Pat.: www.alconpatents.com
Actual Size
300066249-0524
When Using This Product
- do not touch tip of container to any surface to avoid contamination
- replace cap after each use
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Stop Use and Ask A Doctor If You Experience Any of the Following:
- eye pain
- changes in vision
- continued redness or irritation of the eye
- condition worsens or persists for more than 72 hours
Structured Label Content
Uses
- for the temporary relief of burning and irritation due to dryness of the eye
- for the temporary relief of discomfort due to minor irritations of the eye or to exposure to wind or sun
- for use as a protectant against further irritation or to relieve dryness of the eye
- for use as a lubricant to prevent further irritation or to relieve dryness of the eye
Section 55105-1 (55105-1)
| Active Ingredients | Purpose |
| Propylene Glycol 0.6% | Lubricant |
Warnings
For external use only
Directions
- shake well before using
- put 1 or 2 drops in the affected eye(s) as needed
- see side of carton for directions for use
Do Not Use (Do not use)
- if this product changes color
- if you are sensitive to any ingredient in this product
Questions?
In the U.S. call 1-800-757-9195
www.systane.com
[email protected]
Other Information (Other information)
- store at room temperature
Directions for Use: (Directions for use:)
- Hold the bottle just below the cap and twist the cap to open.
- BEFORE FIRST USE, hold the bottle upside down and dispense one drop of product to discard.
- Shake the bottle downward to remove residual product (see picture).
- Place 1 or 2 drops in the affected eye(s) as needed and blink.
-
After use, shake the bottle downward to remove residual product that may be left on the tip (see picture on side panel).
- Replace cap after use.
- Discard 30 days after opening.
Inactive Ingredients (Inactive ingredients)
boric acid, dimyristoyl phosphatidylglycerol, hydroxypropyl guar, mineral oil, polyoxyl 40 stearate, sodium hyaluronate, sorbitan tristearate, sorbitol and purified water. May contain hydrochloric acid and/or sodium hydroxide to adjust pH.
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
Preservative-Free
Systane
®
LUBRICANT EYE DROPS
PRO PF
TRIPLE ACTION
DRY EYE RELIEF
Hydrates, Restores, Protects
Long-Lasting
With Hyaluronate
Preservative-Free Bottle
#1 DOCTOR RECOMMENDED BRANDS1
Alcon
Sterile
10mL (0.34 FL OZ)
LOT: EXP.:
Side Panel:
TAMPER EVIDENT:
For your protection, this bottle has a tamper evident ring around the bottom of the cap. Do not
use if this ring is damaged or missing at the time of purchase.
1Based on a survey of eye care professionals. Data on file.
Alcon Laboratories, Inc.
Fort Worth, TX 76134 USA
Country of Origin United States
U.S. Pat.: www.alconpatents.com
Actual Size
300066249-0524
When Using This Product (When using this product)
- do not touch tip of container to any surface to avoid contamination
- replace cap after each use
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Stop Use and Ask A Doctor If You Experience Any of the Following: (Stop use and ask a doctor if you experience any of the following:)
- eye pain
- changes in vision
- continued redness or irritation of the eye
- condition worsens or persists for more than 72 hours
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:08.531999 · Updated: 2026-03-14T23:08:44.584027