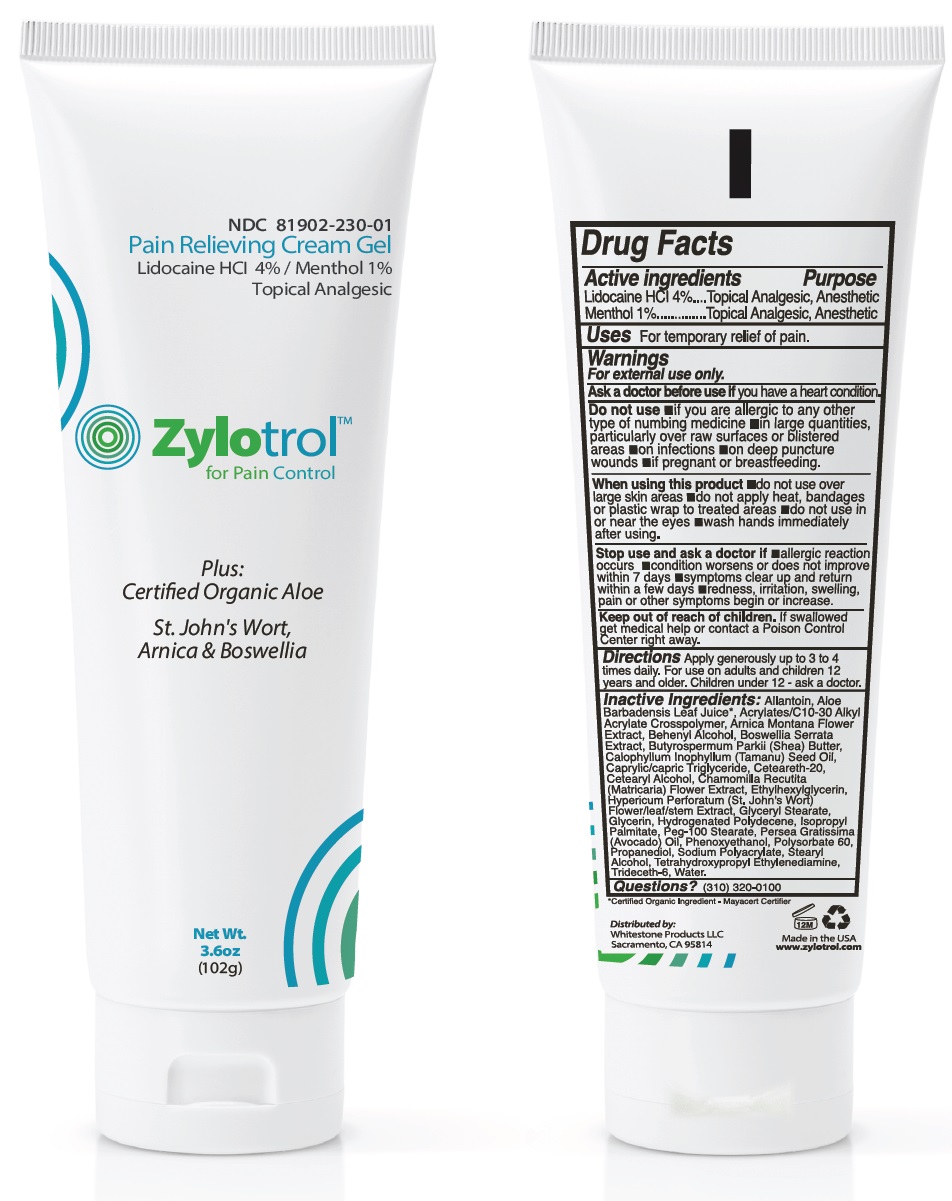
zylotrol pain relieving cream
2301fed7-0fa9-450d-8b1b-006f53235be0
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Topical Analgesic, Anesthetic
Description
Lidocaine HCl 4.0% Menthol 1.0%
Medication Information
Warnings and Precautions
Warnings
For external use only.
Ask a doctor before use ifyou have a heart condition.
Do not use •if you are allergic to any other type of numbing medicine •in large quantities, particularly over raw surfaces or blistered areas •on infections •on deep puncture wounds •if pregnant or breastfeeding.
When using this product •do not use over large skin areas •do not apply heat, bandages or plastic wrap to treated areas •do not use in or near the eyes •wash hands immediately after using.
Stop use and ask a doctor if •allergic reaction occurs •condition worsens or does not improve within 7 days •symptoms clear up and return within a few days •redness, irritation, swelling, pain or other symptoms begin or increase.
Indications and Usage
Uses
For temporarily relief of pain
Purpose
Topical Analgesic, Anesthetic
Dosage and Administration
Directions
Apply generously up to 3 to 4 times daily. For use on adults and children 12 years and older. Children under 12 - ask a doctor.
Description
Lidocaine HCl 4.0% Menthol 1.0%
Section 50565-1
Keep out of reach of children.If swallowed get medical help or contact a Poison Control Center right away.
Section 51727-6
Inactive ingredients:
Allantoin, Aloe Barbadensis Leaf Juice*, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Arnica Montana Flower Extract, Behenyl Alcohol, Boswellia Serrata Extract, Butyrospermum Parkii (Shea) Butter, Calophyllum Inophyllum (Tamanu) Seed Oil, Caprylic/capric Triglyceride, Ceteareth-20, Cetearyl Alcohol, Chamomilla Recutita (Matricaria) Flower Extract, Ethylhexylglycerin, Hypericum Perforatum (St. John's Wort) Flower/leaf/stem Extract, Glyceryl Stearate, Glycerin, Hydrogenated Polydecene, Isopropyl Palmitate, Peg-100 Stearate, Persea Gratissima (Avocado) Oil, Phenoxyethanol, Polysorbate 60, Propanediol, Sodium Polyacrylate, Stearyl Alcohol, Tetrahydroxypropyl Ethylenediamine, Trideceth-6, Water.
*Certified Organic Ingredient - Mayacert Certifier
Section 53413-1
Questions?
(310) 320-0100
Drug Facts
Active Ingredients
Lidocaine HCl 4.0%
Menthol 1.0%
Principal Display Panel Jar Label
Principal Display Panel Tube Label
Structured Label Content
Warnings and Precautions (34071-1)
Warnings
For external use only.
Ask a doctor before use ifyou have a heart condition.
Do not use •if you are allergic to any other type of numbing medicine •in large quantities, particularly over raw surfaces or blistered areas •on infections •on deep puncture wounds •if pregnant or breastfeeding.
When using this product •do not use over large skin areas •do not apply heat, bandages or plastic wrap to treated areas •do not use in or near the eyes •wash hands immediately after using.
Stop use and ask a doctor if •allergic reaction occurs •condition worsens or does not improve within 7 days •symptoms clear up and return within a few days •redness, irritation, swelling, pain or other symptoms begin or increase.
Indications and Usage (34067-9)
Uses
For temporarily relief of pain
Purpose
Topical Analgesic, Anesthetic
Dosage and Administration (34068-7)
Directions
Apply generously up to 3 to 4 times daily. For use on adults and children 12 years and older. Children under 12 - ask a doctor.
Section 50565-1 (50565-1)
Keep out of reach of children.If swallowed get medical help or contact a Poison Control Center right away.
Section 51727-6 (51727-6)
Inactive ingredients:
Allantoin, Aloe Barbadensis Leaf Juice*, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Arnica Montana Flower Extract, Behenyl Alcohol, Boswellia Serrata Extract, Butyrospermum Parkii (Shea) Butter, Calophyllum Inophyllum (Tamanu) Seed Oil, Caprylic/capric Triglyceride, Ceteareth-20, Cetearyl Alcohol, Chamomilla Recutita (Matricaria) Flower Extract, Ethylhexylglycerin, Hypericum Perforatum (St. John's Wort) Flower/leaf/stem Extract, Glyceryl Stearate, Glycerin, Hydrogenated Polydecene, Isopropyl Palmitate, Peg-100 Stearate, Persea Gratissima (Avocado) Oil, Phenoxyethanol, Polysorbate 60, Propanediol, Sodium Polyacrylate, Stearyl Alcohol, Tetrahydroxypropyl Ethylenediamine, Trideceth-6, Water.
*Certified Organic Ingredient - Mayacert Certifier
Section 53413-1 (53413-1)
Questions?
(310) 320-0100
Drug Facts
Active Ingredients (Active ingredients )
Lidocaine HCl 4.0%
Menthol 1.0%
Principal Display Panel Jar Label (PRINCIPAL DISPLAY PANEL-Jar Label)
Principal Display Panel Tube Label (PRINCIPAL DISPLAY PANEL-Tube Label)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:36.618533 · Updated: 2026-03-14T23:03:47.044278