Diclofenac Sodium Topical Gel
22f4b8aa-902f-a860-e063-6394a90ada26
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Diclofenac sodium topical gel is indicated for the topical treatment of actinic keratoses (AK).
Dosage and Administration
Use the lowest effective dosage for the shortest duration consistent with individual patient treatment goals [see Warnings and Precautions (5) ] . Apply diclofenac sodium topical gel gently to lesion areas twice daily to adequately cover each lesion. Use 0.5 g of gel (pea size) on each 5 cm × 5 cm lesion site. The recommended duration of therapy is from 60 days to 90 days. Complete healing of the lesion(s) or optimal therapeutic effect may not be evident for up to 30 days following cessation of therapy. Lesions that do not respond to therapy should be re-evaluated and management reconsidered. Avoid contact of diclofenac sodium topical gel with eyes and mucous membranes.
Contraindications
Diclofenac sodium topical gel is contraindicated in the following patients: With known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to diclofenac or any components of the drug product [ see Warnings and Precautions (5.1 , 5.3 , 5.10) and Description (11) ]. With the history of asthma, urticaria, or other allergic type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients [ see Warnings and Precautions (5.1 , 5.2) ]. Application on damaged skin resulting from any etiology, including exudative dermatitis, eczema, infected lesions, burns or wounds [ see Warnings and Precautions (5.3) ]. In the setting of coronary bypass graft (CABG) surgery [ see Warnings and Precautions (5.4) ].
Warnings and Precautions
Anaphylactic Reactions: Seek emergency help if an anaphylactic reaction occurs. ( 5.1 ) Exacerbation of Asthma Related to Aspirin Sensitivity: Diclofenac sodium topical gel is contraindicated in patients with aspirin-sensitive asthma. Monitor patients with pre-existing asthma (without aspirin sensitivity). ( 5.2 ) Serious Skin Reactions: Discontinue diclofenac sodium topical gel at first appearance of skin rash or other signs of hypersensitivity. ( 5.3 , 5.15 ) Hepatoxicity: Inform patients of warning signs and symptoms of hepatotoxicity. Discontinue if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop. ( 5.6 ) Hypertension: Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure. ( 5.7 , 7 ) Heart Failure and Edema: Avoid use of diclofenac sodium topical gel in patients with severe heart failure. ( 5.8 ) Renal Toxicity: Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of diclofenac sodium topical gel in patients with advanced renal disease. ( 5.9 ) Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue diclofenac sodium topical gel and evaluate clinically. ( 5.10 ) Fetal Toxicity: Limit use of NSAIDs, including diclofenac sodium topical gel, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal renal dysfunction and premature closure of the fetal ductus arteriosus. ( 5.11 , 8.1 ) Hematologic Toxicity: Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia. ( 5.12 , 7 ). Photosensitivity: Avoid exposure of treated area(s) to natural or artificial sunlight. ( 5.15 ) Exposure to Eyes and Mucosal Membranes: Avoid contact of diclofenac sodium topical gel with eyes and mucosal membranes. ( 5.16 ) Oral Nonsteroidal Anti-inflammatory Drugs: Avoid concurrent use with oral NSAIDs. ( 5.17 )
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: Anaphylactic Reactions [ see Warnings and Precautions (5.1) ] Exacerbation of Asthma Related to Aspirin Sensitivity [ see Warnings and Precautions (5.2) ] Serious Skin Reactions [ see Warnings and Precautions (5.3) ] Cardiovascular Thrombotic Events [ see Warnings and Precautions (5.4) ] GI Bleeding, Ulceration and Perforation [ see Warnings and Precautions (5.5) ] Hepatotoxicity [ see Warnings and Precautions (5.6) ] Hypertension [ see Warnings and Precautions (5.7) ] Heart Failure and Edema [ see Warnings and Precautions (5.8) ] Renal Toxicity and Hyperkalemia [ see Warnings and Precautions (5.9) ] DRESS [ see Warnings and Precautions (5.10) ] Hematologic Toxicity [ see Warnings and Precautions (5.12) ] Photosensitivity [ see Warnings and Precautions (5.15) ]
Drug Interactions
See Table 2 for clinically significant drug interactions with diclofenac. Table 2: Clinically Significant Drug Interactions with Diclofenac Drugs That Interfere with Hemostasis Clinical Impact: Diclofenac and anticoagulants such as warfarin have a synergistic effect on bleeding. The concomitant use of diclofenac and anticoagulants have an increased risk of serious bleeding compared to the use of either drug alone. Serotonin release by platelets plays an important role in hemostasis. Case-control and cohort epidemiological studies showed that concomitant use of drugs that interfere with serotonin reuptake and an NSAID may potentiate the risk of bleeding more than an NSAID alone. Intervention: Monitor patients with concomitant use of diclofenac sodium topical gel with anticoagulants (e.g., warfarin), antiplatelet agents (e.g., aspirin), selective serotonin reuptake inhibitors (SSRIs), and serotonin norepinephrine reuptake inhibitors (SNRIs) for signs of bleeding [see Warnings and Precautions (5.5) ]. Aspirin Clinical Impact: In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.5) ]. Intervention: Concomitant use of diclofenac sodium topical gel and analgesic doses of aspirin is not generally recommended because of the increased risk of bleeding [see Warnings and Precautions (5.12) ] . Diclofenac sodium topical gel is not a substitute for low dose aspirin for cardiovascular protection. ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers Clinical Impact: NSAIDs may diminish the antihypertensive effect of angiotensin converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), or beta-blockers (including propranolol). In patients who are elderly, volume-depleted (including those on diuretic therapy), or have renal impairment, co-administration of an NSAID with ACE inhibitors or ARBs may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Intervention: During concomitant use of diclofenac sodium topical gel and ACE-inhibitors, ARBs, or beta-blockers, monitor blood pressure to ensure that the desired blood pressure is obtained. During concomitant use of diclofenac sodium topical gel and ACE-inhibitors or ARBs in patients who are elderly, volume-depleted, or have impaired renal function, monitor for signs of worsening renal function [see Warnings and Precautions (5.9) ]. Diuretics Clinical Impact: Clinical studies, as well as post-marketing observations, showed that NSAIDs reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. Intervention: During concomitant use of diclofenac sodium topical gel with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects [see Warnings and Precautions (5.9) ]. Digoxin Clinical Impact: The concomitant use of diclofenac with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. Intervention: During concomitant use of diclofenac sodium topical gel and digoxin, monitor serum digoxin levels. Lithium Clinical Impact: NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis. Intervention: During concomitant use of diclofenac sodium topical gel and lithium, monitor patients for signs of lithium toxicity. Methotrexate Clinical Impact: Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction) Intervention: During concomitant use of diclofenac sodium topical gel and methotrexate, monitor patients for methotrexate toxicity. Cyclosporine Clinical Impact: Concomitant use of diclofenac sodium topical gel and cyclosporine may increase cyclosporine's nephrotoxicity. Intervention: During concomitant use of diclofenac sodium topical gel and cyclosporine, monitor patients for signs of worsening renal function. NSAIDs and Salicylates Clinical Impact: Concomitant use of diclofenac sodium topical gel with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity [see Warnings and Precautions (5.5) ]. Intervention: The concomitant use of diclofenac sodium topical gel with other NSAIDs or salicylates is not recommended. Pemetrexed Clinical Impact: Concomitant use of diclofenac sodium topical gel and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). Intervention: During concomitant use of diclofenac sodium topical gel and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. NSAIDs with short elimination half-lives (e.g., diclofenac, indomethacin) should be avoided for a period of two days before, the day of, and two days following administration of pemetrexed. In the absence of data regarding potential interaction between pemetrexed and NSAIDs with longer half-lives (e.g., meloxicam, nabumetone), patients taking these NSAIDs should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration.
How Supplied
Diclofenac Sodium Topical Gel, 3% is available 100 g (NDC 68071-3688-1) tubes. Each gram of topical gel contains 30 mg of diclofenac sodium.
Storage and Handling
Diclofenac Sodium Topical Gel, 3% is available 100 g (NDC 68071-3688-1) tubes. Each gram of topical gel contains 30 mg of diclofenac sodium.
Description
WARNING: RISK OF SERIOUS CARDIOVASCULAR EVENTS AND GASTROINTESTINAL EVENTS See full prescribing information for complete boxed warning. Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use. ( 5.4 ) Diclofenac sodium topical gel is contraindicated in the setting of coronary artery bypass graft (CABG) surgery. ( 4 , 5.4 ) NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events ( 5.5 )
Medication Information
Warnings and Precautions
Anaphylactic Reactions: Seek emergency help if an anaphylactic reaction occurs. ( 5.1 ) Exacerbation of Asthma Related to Aspirin Sensitivity: Diclofenac sodium topical gel is contraindicated in patients with aspirin-sensitive asthma. Monitor patients with pre-existing asthma (without aspirin sensitivity). ( 5.2 ) Serious Skin Reactions: Discontinue diclofenac sodium topical gel at first appearance of skin rash or other signs of hypersensitivity. ( 5.3 , 5.15 ) Hepatoxicity: Inform patients of warning signs and symptoms of hepatotoxicity. Discontinue if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop. ( 5.6 ) Hypertension: Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure. ( 5.7 , 7 ) Heart Failure and Edema: Avoid use of diclofenac sodium topical gel in patients with severe heart failure. ( 5.8 ) Renal Toxicity: Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of diclofenac sodium topical gel in patients with advanced renal disease. ( 5.9 ) Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue diclofenac sodium topical gel and evaluate clinically. ( 5.10 ) Fetal Toxicity: Limit use of NSAIDs, including diclofenac sodium topical gel, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal renal dysfunction and premature closure of the fetal ductus arteriosus. ( 5.11 , 8.1 ) Hematologic Toxicity: Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia. ( 5.12 , 7 ). Photosensitivity: Avoid exposure of treated area(s) to natural or artificial sunlight. ( 5.15 ) Exposure to Eyes and Mucosal Membranes: Avoid contact of diclofenac sodium topical gel with eyes and mucosal membranes. ( 5.16 ) Oral Nonsteroidal Anti-inflammatory Drugs: Avoid concurrent use with oral NSAIDs. ( 5.17 )
Indications and Usage
Diclofenac sodium topical gel is indicated for the topical treatment of actinic keratoses (AK).
Dosage and Administration
Use the lowest effective dosage for the shortest duration consistent with individual patient treatment goals [see Warnings and Precautions (5) ] . Apply diclofenac sodium topical gel gently to lesion areas twice daily to adequately cover each lesion. Use 0.5 g of gel (pea size) on each 5 cm × 5 cm lesion site. The recommended duration of therapy is from 60 days to 90 days. Complete healing of the lesion(s) or optimal therapeutic effect may not be evident for up to 30 days following cessation of therapy. Lesions that do not respond to therapy should be re-evaluated and management reconsidered. Avoid contact of diclofenac sodium topical gel with eyes and mucous membranes.
Contraindications
Diclofenac sodium topical gel is contraindicated in the following patients: With known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to diclofenac or any components of the drug product [ see Warnings and Precautions (5.1 , 5.3 , 5.10) and Description (11) ]. With the history of asthma, urticaria, or other allergic type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients [ see Warnings and Precautions (5.1 , 5.2) ]. Application on damaged skin resulting from any etiology, including exudative dermatitis, eczema, infected lesions, burns or wounds [ see Warnings and Precautions (5.3) ]. In the setting of coronary bypass graft (CABG) surgery [ see Warnings and Precautions (5.4) ].
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: Anaphylactic Reactions [ see Warnings and Precautions (5.1) ] Exacerbation of Asthma Related to Aspirin Sensitivity [ see Warnings and Precautions (5.2) ] Serious Skin Reactions [ see Warnings and Precautions (5.3) ] Cardiovascular Thrombotic Events [ see Warnings and Precautions (5.4) ] GI Bleeding, Ulceration and Perforation [ see Warnings and Precautions (5.5) ] Hepatotoxicity [ see Warnings and Precautions (5.6) ] Hypertension [ see Warnings and Precautions (5.7) ] Heart Failure and Edema [ see Warnings and Precautions (5.8) ] Renal Toxicity and Hyperkalemia [ see Warnings and Precautions (5.9) ] DRESS [ see Warnings and Precautions (5.10) ] Hematologic Toxicity [ see Warnings and Precautions (5.12) ] Photosensitivity [ see Warnings and Precautions (5.15) ]
Drug Interactions
See Table 2 for clinically significant drug interactions with diclofenac. Table 2: Clinically Significant Drug Interactions with Diclofenac Drugs That Interfere with Hemostasis Clinical Impact: Diclofenac and anticoagulants such as warfarin have a synergistic effect on bleeding. The concomitant use of diclofenac and anticoagulants have an increased risk of serious bleeding compared to the use of either drug alone. Serotonin release by platelets plays an important role in hemostasis. Case-control and cohort epidemiological studies showed that concomitant use of drugs that interfere with serotonin reuptake and an NSAID may potentiate the risk of bleeding more than an NSAID alone. Intervention: Monitor patients with concomitant use of diclofenac sodium topical gel with anticoagulants (e.g., warfarin), antiplatelet agents (e.g., aspirin), selective serotonin reuptake inhibitors (SSRIs), and serotonin norepinephrine reuptake inhibitors (SNRIs) for signs of bleeding [see Warnings and Precautions (5.5) ]. Aspirin Clinical Impact: In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.5) ]. Intervention: Concomitant use of diclofenac sodium topical gel and analgesic doses of aspirin is not generally recommended because of the increased risk of bleeding [see Warnings and Precautions (5.12) ] . Diclofenac sodium topical gel is not a substitute for low dose aspirin for cardiovascular protection. ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers Clinical Impact: NSAIDs may diminish the antihypertensive effect of angiotensin converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), or beta-blockers (including propranolol). In patients who are elderly, volume-depleted (including those on diuretic therapy), or have renal impairment, co-administration of an NSAID with ACE inhibitors or ARBs may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Intervention: During concomitant use of diclofenac sodium topical gel and ACE-inhibitors, ARBs, or beta-blockers, monitor blood pressure to ensure that the desired blood pressure is obtained. During concomitant use of diclofenac sodium topical gel and ACE-inhibitors or ARBs in patients who are elderly, volume-depleted, or have impaired renal function, monitor for signs of worsening renal function [see Warnings and Precautions (5.9) ]. Diuretics Clinical Impact: Clinical studies, as well as post-marketing observations, showed that NSAIDs reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. Intervention: During concomitant use of diclofenac sodium topical gel with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects [see Warnings and Precautions (5.9) ]. Digoxin Clinical Impact: The concomitant use of diclofenac with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. Intervention: During concomitant use of diclofenac sodium topical gel and digoxin, monitor serum digoxin levels. Lithium Clinical Impact: NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis. Intervention: During concomitant use of diclofenac sodium topical gel and lithium, monitor patients for signs of lithium toxicity. Methotrexate Clinical Impact: Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction) Intervention: During concomitant use of diclofenac sodium topical gel and methotrexate, monitor patients for methotrexate toxicity. Cyclosporine Clinical Impact: Concomitant use of diclofenac sodium topical gel and cyclosporine may increase cyclosporine's nephrotoxicity. Intervention: During concomitant use of diclofenac sodium topical gel and cyclosporine, monitor patients for signs of worsening renal function. NSAIDs and Salicylates Clinical Impact: Concomitant use of diclofenac sodium topical gel with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity [see Warnings and Precautions (5.5) ]. Intervention: The concomitant use of diclofenac sodium topical gel with other NSAIDs or salicylates is not recommended. Pemetrexed Clinical Impact: Concomitant use of diclofenac sodium topical gel and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). Intervention: During concomitant use of diclofenac sodium topical gel and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. NSAIDs with short elimination half-lives (e.g., diclofenac, indomethacin) should be avoided for a period of two days before, the day of, and two days following administration of pemetrexed. In the absence of data regarding potential interaction between pemetrexed and NSAIDs with longer half-lives (e.g., meloxicam, nabumetone), patients taking these NSAIDs should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration.
Storage and Handling
Diclofenac Sodium Topical Gel, 3% is available 100 g (NDC 68071-3688-1) tubes. Each gram of topical gel contains 30 mg of diclofenac sodium.
How Supplied
Diclofenac Sodium Topical Gel, 3% is available 100 g (NDC 68071-3688-1) tubes. Each gram of topical gel contains 30 mg of diclofenac sodium.
Description
WARNING: RISK OF SERIOUS CARDIOVASCULAR EVENTS AND GASTROINTESTINAL EVENTS See full prescribing information for complete boxed warning. Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use. ( 5.4 ) Diclofenac sodium topical gel is contraindicated in the setting of coronary artery bypass graft (CABG) surgery. ( 4 , 5.4 ) NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events ( 5.5 )
Section 42229-5
Cardiovascular Thrombotic Events
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use [see Warnings and Precautions (5.4)].
- Diclofenac sodium topical gel is contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications (4)and Warnings and Precautions (5.4)].
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | |
| Revised: August 2023 | |
| Dispense with Medication Guide available at: https://www.taro.com/usa-medication-guides | |
|
Medication Guide
Diclofenac (dye-KLOE-fen-ak) Sodium Topical Gel, 3% |
|
| What is the most important information I should know about diclofenac sodium topical gel and medicines called Nonsteroidal Anti-inflammatory Drugs (NSAIDs)? | |
NSAIDs can cause serious side effects, including:
Do not take or use NSAIDs right before or after a heart surgery called a "coronary artery bypass graft (CABG)". Avoid taking NSAIDs after a recent heart attack unless your healthcare provider tells you to. You may have an increased risk of another heart attack if you take or use NSAIDs after a recent heart attack.
The risk of getting an ulcer or bleeding increases with:
|
|
NSAIDs should only be used:
|
|
|
What is diclofenac sodium topical gel?
Diclofenac sodium topical gel is an NSAID that is used on the skin (topical) to treat a skin condition called actinic keratosis. Diclofenac sodium topical gel is not for use in children. |
|
Do not use diclofenac sodium topical gel:
|
|
Before using diclofenac sodium topical gel, tell your healthcare provider about all of your medical conditions, including if you:
|
|
| Tell your healthcare provider about all of the medicines you take, including prescription or over-the-counter medicines, vitamins, or herbal supplements.NSAIDs and some other medicines can interact with each other and cause serious side effects. Do not start taking any new medicine without talking to your healthcare provider first. | |
How should I use diclofenac sodium topical gel?
|
|
What should I avoid while using diclofenac sodium topical gel?
|
|
| What are the possible side effects of diclofenac sodium topical gel? | |
| Diclofenac sodium topical gel and other NSAIDs can cause serious side effects, including: | |
See "
What is the most important information I should know about diclofenac sodium topical gel and medicines called Nonsteroidal Anti-inflammatory Drugs (NSAIDs)?"
|
|
| Other side effects of NSAIDs include:stomach pain, constipation, diarrhea, gas, heartburn, nausea, vomiting, and dizziness. | |
| Get emergency help right away if you get any of the following symptoms: | |
|
|
| Stop using diclofenac sodium topical gel and call your healthcare provider right away if you get any of the following symptoms: | |
|
|
|
Application site skin reactions are common with diclofenac sodium topical gel including:skin redness, itching, rash, dry skin, scaling, and peeling.
If you take too much NSAID, call your healthcare provider or get medical help right away. Diclofenac sodium topical gel may cause fertility problems in females, which may affect your ability to have a child. Talk to your healthcare provider if this a concern for you. These are not all of the possible side effects of NSAIDs. For more information, ask your healthcare provider or pharmacist about NSAIDs. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
| Other information about NSAIDs | |
|
|
How should I store diclofenac sodium topical gel?
|
|
| Keep diclofenac sodium topical gel and all medicines out of the reach of children. | |
| General information about the safe and effective use of diclofenac sodium topical gel. | |
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use diclofenac sodium topical gel for a condition for which it was not prescribed. Do not give diclofenac sodium topical gel to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information about diclofenac sodium topical gel, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about diclofenac sodium topical gel that is written for health professionals. | |
| What are the ingredients in diclofenac sodium topical gel? | |
| Active ingredient:diclofenac sodium | |
| Inactive ingredients:benzyl alcohol, hyaluronate sodium, polyethylene glycol monomethyl ether, and purified water. | |
| Manufactured by: Taro Pharmaceuticals Inc., Brampton, Ontario, Canada L6T 1C1 | |
| Distributed by: Taro Pharmaceuticals U.S.A., Inc., Hawthorne, NY 10532 | |
| For more information, call 1-866-923-4914. |
Section 44425-7
Storage: Store at 20° to 25°C (68° to 77°F)[see USP Controlled Room Temperature]. Protect from heat. Avoid freezing.
10 Overdosage
Symptoms following acute NSAID overdosages have been typically limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which have been generally reversible with supportive care. Gastrointestinal bleeding, hypertension, acute renal failure, respiratory depression, and coma have been reported. [see Warnings and Precautions (5.4, 5.5, 5.7, 5.9)].
Manage patients with symptomatic and supportive care following an NSAID overdosage. There are no specific antidotes. Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may not be useful due to high protein binding.
In the event of oral ingestion, resulting in significant systemic side effects, it is recommended that the stomach be emptied by vomiting or lavage. In addition to supportive measures, the use of oral activated charcoal may help to reduce the absorption of diclofenac.
For additional information about overdosage treatment, call a poison control center (1-800-222-1222).
11 Description
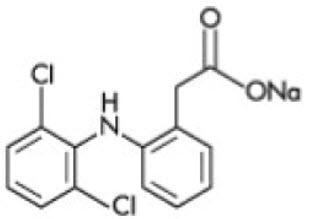
Diclofenac Sodium Topical Gel, 3%, intended for dermatologic use, contains the active ingredient, diclofenac sodium, in a clear, transparent, colorless to slightly yellow gel base. Diclofenac sodium is a white to slightly yellow crystalline powder. It is freely soluble in methanol, soluble in ethanol, sparingly soluble in water, slightly soluble in acetone, and partially insoluble in ether. The chemical name for diclofenac sodium is:
Sodium [ o-(2,6-dichloranilino) phenyl] acetate
Diclofenac sodium has a molecular weight of 318.13.
The CAS number is CAS-15307-79-6. The structural formula is represented below:
Diclofenac Sodium Topical Gel, 3% also contains benzyl alcohol, hyaluronate sodium, polyethylene glycol monomethyl ether, and purified water.
1 g of diclofenac sodium topical gel contains 30 mg of the active substance, diclofenac sodium.
5.7 Hypertension
NSAIDs, including diclofenac sodium topical gel, can lead to new onset of hypertension or worsening of pre-existing hypertension, either of which may contribute to the increased incidence of CV events. Patients taking angiotensin converting enzyme (ACE) inhibitors, thiazide diuretics, or loop diuretics may have impaired response to these therapies when taking NSAIDs [see Drug Interactions (7)].
Monitor blood pressure (BP) during the initiation of NSAID treatment and throughout the course of therapy.
8.4 Pediatric Use
Actinic keratoses is not a condition seen within the pediatric population. Diclofenac sodium topical gel should not be used by children.
8.5 Geriatric Use
Elderly patients, compared to younger patients, are at greater risk for NSAID-associated serious cardiovascular, gastrointestinal, and/or renal adverse reactions. If the anticipated benefit for the elderly patient outweighs these potential risks, start dosing at the low end of the dosing range, and monitor patients for adverse effects [see Warnings and Precautions (5.4, 5.5, 5.6, 5.9, 5.14)].
Of the 211 subjects treated with diclofenac sodium topical gel in controlled clinical trials, 143 subjects were 65 years of age and over. Of those 143 subjects, 55 subjects were 75 years of age and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
5.6 Hepatotoxicity
In clinical trials with diclofenac sodium topical gel, 2 to 3% of subjects had elevations of liver function tests (LFTs) [see Clinical Trials Experience (6.1)]. To minimize the potential risk for an adverse liver-related event in patients treated with diclofenac sodium topical gel, use the lowest effective dose for the shortest duration possible. Exercise caution when prescribing diclofenac sodium topical gel with concomitant drugs that are known to be potentially hepatotoxic (e.g., acetaminophen, antibiotics, anti-epileptics).
Physicians should measure transaminases at baseline and periodically in patients receiving long-term therapy with diclofenac, because severe hepatotoxicity may develop without a prodrome of distinguishing symptoms. The optimum times for making the first and subsequent transaminase measurements are not known. Based on clinical trial data and postmarketing experiences, transaminases should be monitored within 4 to 8 weeks after initiating treatment with diclofenac. However, severe hepatic reactions can occur at any time during treatment with diclofenac.
If abnormal liver tests persist or worsen, if clinical signs and/or symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, abdominal pain, diarrhea, dark urine, etc.), diclofenac sodium topical should be discontinued immediately.
Inform patients of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, diarrhea, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), discontinue diclofenac sodium topical gel immediately, and perform a clinical evaluation of the patient.
14 Clinical Studies
Clinical trials were conducted involving a total of 427 patients (213 treated with diclofenac sodium topical gel and 214 with a gel vehicle). Each patient had no fewer than five AK lesions in a major body area, which was defined as one of five 5 cm × 5 cm regions: scalp, forehead, face, forearm and hand. Up to three major body areas were studied in any patient. All patients were 18 years of age or older (male and female) with no clinically significant medical problems outside of the AK lesions and had undergone a 60-day washout period from disallowed medications (masoprocol, 5-fluorouracil, cyclosporine, retinoids, trichloroacetic acid/lactic acid/peel, 50% glycolic acid peel) and hyaluronan-containing cosmetics. Patients were excluded from participation for reasons of known or suspected hypersensitivity to any diclofenac sodium topical gel ingredient, pregnancy, allergies to aspirin or other nonsteroidal anti-inflammatory drugs (NSAIDs), or other dermatological conditions which might affect the absorption of the study medication. Application of dermatologic products such as sunscreens, cosmetics, and other drug products was not permitted. Patients were instructed to apply a small amount of diclofenac sodium topical gel (approximately 0.5 g) onto the affected skin, using their fingers, and gently smoothing the gel over the lesion. In addition, all patients were instructed to avoid sun exposure. Complete clearing of the AK lesions 30 days after completion of treatment was the primary efficacy variable. No long-term patient follow-ups, after the 30-day assessments, were performed for the detection of recurrence.
| Diclofenac Sodium Topical Gel | Vehicle | p-value | |
|---|---|---|---|
| Study 1 90 days treatment | 27/58 (47%) | 11/59 (19%) | <0.001 |
| Study 2 90 days treatment | 18/53 (34%) | 10/55 (18%) | 0.061 |
| Study 3 60 days treatment | 15/48 (31%) | 5/49 (10%) | 0.021 |
| 30 days treatment | 7/49 (14%) | 2/49 (4%) | 0.221 |
| Scalp | Forehead | Face | Arm/Forearm | Back of Hand | |
|---|---|---|---|---|---|
| Study 1
90 days treatment |
|||||
| Diclofenac Sodium Topical Gel | 1/4 (25%) | 17/30 (57%) | 9/17 (53%) | 4/12 (33%) | 6/16 (38%) |
| Vehicle | 3/9 (33%) | 8/24 (33%) | 5/17 (29%) | 4/12 (33%) | 0/14 (0) |
| p-value | 0.7646 | 0.0908 | 0.1682 | 1.000 | 0.0650 |
| Study 2
90 days treatment |
|||||
| Diclofenac Sodium Topical Gel | 2/6 (33%) | 9/19 (47%) | 4/5 (80%) | 5/8 (63%) | 1/17 (6%) |
| Vehicle | 0/4 (0) | 6/22 (27%) | 2/8 (25%) | 0/5 (0) | 3/16 (19%) |
| p-value | 0.4235 | 0.1870 | 0.0727 | 0.0888 | 0.2818 |
| Study 3
60 days treatment |
|||||
| Diclofenac Sodium Topical Gel | 3/7 (43%) | 13/31 (42%) | 10/19 (53%) | 0/1 (0) | 2/8 (25%) |
| Vehicle | 0/6 (0) | 5/36 (14%) | 2/13 (15%) | 0/2 (0) | 1/9 (11%) |
| p-value | 0.2271 | 0.0153 | 0.0433 | - | 0.4637 |
| 30 days treatment | |||||
| Diclofenac Sodium Topical Gel | 2/5 (40%) | 4/29 (14%) | 3/14 (21%) | 0/0 (0) | 0/9 (0) |
| Vehicle | 0/5 (0) | 2/29 (7%) | 2/18 (11%) | 0/1 (0) | 1/9 (11%) |
| p-value | 0.2299 | 0.3748 | 0.4322 | - | 0.6521 |
| All data combined | |||||
| Diclofenac Sodium Topical Gel | 8/22 (36%) | 43/109 (39%) | 26/55 (47%) | 9/21 (43%) | 9/50 (18%) |
| Vehicle | 3/24 (13%) | 21/111 (19%) | 11/56 (20%) | 4/20 (20%) | 5/48 (10%) |
| p-value | 0.0903 | 0.0013 | 0.0016 | 0.2043 | 0.3662 |
4 Contraindications
Diclofenac sodium topical gel is contraindicated in the following patients:
- With known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to diclofenac or any components of the drug product [ see Warnings and Precautions (5.1, 5.3, 5.10)and Description (11) ].
- With the history of asthma, urticaria, or other allergic type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients [ see Warnings and Precautions (5.1, 5.2) ].
- Application on damaged skin resulting from any etiology, including exudative dermatitis, eczema, infected lesions, burns or wounds [ see Warnings and Precautions (5.3) ].
- In the setting of coronary bypass graft (CABG) surgery [ see Warnings and Precautions (5.4) ].
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Anaphylactic Reactions [ see Warnings and Precautions (5.1) ]
- Exacerbation of Asthma Related to Aspirin Sensitivity [ see Warnings and Precautions (5.2) ]
- Serious Skin Reactions [ see Warnings and Precautions (5.3) ]
- Cardiovascular Thrombotic Events [ see Warnings and Precautions (5.4) ]
- GI Bleeding, Ulceration and Perforation [ see Warnings and Precautions (5.5) ]
- Hepatotoxicity [ see Warnings and Precautions (5.6) ]
- Hypertension [ see Warnings and Precautions (5.7) ]
- Heart Failure and Edema [ see Warnings and Precautions (5.8) ]
- Renal Toxicity and Hyperkalemia [ see Warnings and Precautions (5.9) ]
- DRESS [ see Warnings and Precautions (5.10) ]
- Hematologic Toxicity [ see Warnings and Precautions (5.12) ]
- Photosensitivity [ see Warnings and Precautions (5.15) ]
7 Drug Interactions
See Table 2 for clinically significant drug interactions with diclofenac.
| Drugs That Interfere with Hemostasis | |
| Clinical Impact: |
|
| Intervention: |
|
| Aspirin | |
| Clinical Impact: |
|
| Intervention: |
|
| ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers | |
| Clinical Impact: |
|
| Intervention: |
|
| Diuretics | |
| Clinical Impact: |
|
| Intervention: |
|
| Digoxin | |
| Clinical Impact: |
|
| Intervention: |
|
| Lithium | |
| Clinical Impact: |
|
| Intervention: |
|
| Methotrexate | |
| Clinical Impact: |
|
| Intervention: |
|
| Cyclosporine | |
| Clinical Impact: |
|
| Intervention: |
|
| NSAIDs and Salicylates | |
| Clinical Impact: |
|
| Intervention: |
|
| Pemetrexed | |
| Clinical Impact: |
|
| Intervention: |
|
12.2 Pharmacodynamics
The pharmacodynamics of diclofenac sodium topical gel in the treatment of actinic keratosis has not been assessed.
5.15 Photosensitivity
Patients should minimize or avoid exposure to natural or artificial sunlight (tanning beds or UVA/B treatment) while using diclofenac sodium topical gel. If patients need to be outdoors while using diclofenac sodium topical gel, they should wear loose-fitting clothes that protect skin from sun exposure and discuss other sun protection measures with their physician. Advise patients to discontinue treatment with diclofenac sodium topical gel at the first evidence of sunburn.
1 Indications and Usage
Diclofenac sodium topical gel is indicated for the topical treatment of actinic keratoses (AK).
12.1 Mechanism of Action
The mechanism of action of diclofenac sodium in the treatment of actinic keratoses (AK) is unknown.
5.12 Hematologic Toxicity
Anemia has occurred in NSAID-treated patients. This may be due to occult or gross blood loss, fluid retention, or an incompletely described effect on erythropoiesis. If a patient treated with diclofenac sodium topical gel has any signs or symptoms of anemia, monitor hemoglobin or hematocrit.
NSAIDs, including diclofenac sodium topical gel, may increase the risk of bleeding events. Co-morbid conditions such as coagulation disorders, concomitant use of warfarin, other anticoagulants, antiplatelet agents (e.g., aspirin), serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) may increase this risk. Monitor these patients for signs of bleeding [see Drug Interactions (7)].
5 Warnings and Precautions
- Anaphylactic Reactions: Seek emergency help if an anaphylactic reaction occurs. ( 5.1)
- Exacerbation of Asthma Related to Aspirin Sensitivity: Diclofenac sodium topical gel is contraindicated in patients with aspirin-sensitive asthma. Monitor patients with pre-existing asthma (without aspirin sensitivity). ( 5.2)
- Serious Skin Reactions: Discontinue diclofenac sodium topical gel at first appearance of skin rash or other signs of hypersensitivity. ( 5.3, 5.15)
- Hepatoxicity: Inform patients of warning signs and symptoms of hepatotoxicity. Discontinue if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop. ( 5.6)
- Hypertension: Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure. ( 5.7, 7)
- Heart Failure and Edema: Avoid use of diclofenac sodium topical gel in patients with severe heart failure. ( 5.8)
- Renal Toxicity: Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of diclofenac sodium topical gel in patients with advanced renal disease. ( 5.9)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue diclofenac sodium topical gel and evaluate clinically. ( 5.10)
- Fetal Toxicity: Limit use of NSAIDs, including diclofenac sodium topical gel, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal renal dysfunction and premature closure of the fetal ductus arteriosus. ( 5.11, 8.1)
- Hematologic Toxicity: Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia. ( 5.12, 7).
- Photosensitivity: Avoid exposure of treated area(s) to natural or artificial sunlight. ( 5.15)
- Exposure to Eyes and Mucosal Membranes: Avoid contact of diclofenac sodium topical gel with eyes and mucosal membranes. ( 5.16)
- Oral Nonsteroidal Anti-inflammatory Drugs: Avoid concurrent use with oral NSAIDs. ( 5.17)
5.1 Anaphylactic Reactions
Diclofenac has been associated with anaphylactic reactions in patients with and without known hypersensitivity to diclofenac and in patients with aspirin-sensitive asthma [ see Contraindications (4)and Warnings and Precautions (5.2) ].
Seek emergency help if an anaphylactic reaction occurs.
5.14 Laboratory Monitoring
Because serious GI bleeding, hepatotoxicity, and renal injury can occur without warning symptoms or signs, consider monitoring patients on long-term NSAID treatment with a CBC and a chemistry profile periodically [see Warnings and Precautions (5.5, 5.6, 5.9)] .
5.3 Serious Skin Reactions
NSAIDs, including diclofenac, can cause serious skin adverse reactions such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. These serious events may occur without warning. Inform patients about the signs and symptoms of serious skin reactions, and to discontinue the use of diclofenac sodium topical gel at the first appearance of skin rash or any other sign of hypersensitivity. Diclofenac sodium topical gel is contraindicated in patients with previous serious skin reactions to NSAIDs. Do not apply diclofenac sodium topical gel to open skin wounds, infections, or exfoliative dermatitis, as it may affect absorption and tolerability of the drug [ see Contraindications (4) ].
2 Dosage and Administration
Use the lowest effective dosage for the shortest duration consistent with individual patient treatment goals [see Warnings and Precautions (5)] .
Apply diclofenac sodium topical gel gently to lesion areas twice daily to adequately cover each lesion. Use 0.5 g of gel (pea size) on each 5 cm × 5 cm lesion site. The recommended duration of therapy is from 60 days to 90 days. Complete healing of the lesion(s) or optimal therapeutic effect may not be evident for up to 30 days following cessation of therapy. Lesions that do not respond to therapy should be re-evaluated and management reconsidered. Avoid contact of diclofenac sodium topical gel with eyes and mucous membranes.
5.8 Heart Failure and Edema
The Coxib and traditional NSAID Trialists' Collaboration meta-analysis of randomized controlled trials demonstrated an approximately two-fold increase in hospitalizations for heart failure in COX-2 selective-treated patients and nonselective NSAID-treated patients compared to placebo-treated patients. In a Danish National Registry study of patients with heart failure, NSAID use increased the risk of MI, hospitalization for heart failure, and death.
Additionally, fluid retention and edema have been observed in some patients treated with NSAIDs. Use of diclofenac may blunt the CV effects of several therapeutic agents used to treat these medical conditions [e.g., diuretics, ACE inhibitors, or angiotensin receptor blockers (ARBs)].
Avoid the use of diclofenac sodium topical gel in patients with severe heart failure unless the benefits are expected to outweigh the risk of worsening heart failure. If diclofenac sodium topical gel is used in patients with severe heart failure, monitor patients for signs of worsening heart failure.
3 Dosage Forms and Strengths
Topical gel, 3%. Each gram of diclofenac sodium topical gel contains 30 mg of diclofenac sodium in a clear, transparent, colorless to slightly yellow gel base. Diclofenac sodium topical gel is supplied in 50 g and 100 g tubes.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of diclofenac sodium topical gel and other topical diclofenac products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse reactions from diclofenac sodium topical gel: burning sensation, hypersensitivity.
Adverse reactions from other topical diclofenac products: hypoesthesia, gait disturbance, musculoskeletal stiffness.
8 Use in Specific Populations
Infertility: NSAIDs are associated with reversible infertility. Consider withdrawal of diclofenac sodium topical gel in women who have difficulties conceiving. ( 8.3)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Of the 423 subjects evaluable for safety in adequate and well-controlled trials, 211 were treated with diclofenac sodium topical gel drug product and 212 were treated with a vehicle gel. Eighty-seven percent (87%) of the diclofenac sodium topical gel-treated subjects (183 subjects) and 84% of the vehicle-treated subjects (178 subjects) experienced one or more adverse events (AEs) during the trials. The majority of these reactions were mild to moderate in severity and resolved upon discontinuation of therapy.
Of the 211 subjects treated with diclofenac sodium topical gel, 172 (82%) experienced AEs involving skin and the application site compared to 160 (75%) vehicle-treated subjects. Application site reactions (ASRs) were the most frequent AEs in both diclofenac sodium topical gel- and vehicle-treated groups. Of note, four reactions, contact dermatitis, rash, dry skin and exfoliation (scaling) were significantly more prevalent in the diclofenac sodium topical gel group than in the vehicle-treated subjects.
Eighteen percent of diclofenac sodium topical gel-treated subjects and 4% of vehicle-treated subjects discontinued from the clinical trials due to adverse events (whether considered related to treatment or not). These discontinuations were mainly due to skin irritation or related cutaneous adverse reactions.
Table 1 below presents the AEs reported at an incidence of >1% for subjects treated with either diclofenac sodium topical gel or vehicle (60- and 90-day treatment groups) during the phase 3 trials.
| 60-day Treatment | 90-day Treatment | |||
|---|---|---|---|---|
| Diclofenac Sodium Topical Gel (%)
N=48 |
Gel Vehicle
(%) N=49 |
Diclofenac Sodium Topical Gel (%)
N=114 |
Gel Vehicle
(%) N=114 |
|
| BODY AS A WHOLE | 21 | 20 | 20 | 18 |
| Abdominal Pain | 2 | 0 | 1 | 0 |
| Accidental Injury | 0 | 0 | 4 | 2 |
| Allergic Reaction | 0 | 0 | 1 | 3 |
| Asthenia | 0 | 0 | 2 | 0 |
| Back Pain | 4 | 0 | 2 | 2 |
| Chest Pain | 2 | 0 | 1 | 0 |
| Chills | 0 | 2 | 0 | 0 |
| Flu Syndrome | 10 | 6 | 1 | 4 |
| Headache | 0 | 6 | 7 | 6 |
| Infection | 4 | 6 | 4 | 5 |
| Neck Pain | 0 | 0 | 2 | 0 |
| Pain | 2 | 0 | 2 | 2 |
| CARDIOVASCULAR SYSTEM | 2 | 4 | 3 | 1 |
| Hypertension | 2 | 0 | 1 | 0 |
| Migraine | 0 | 2 | 1 | 0 |
| Phlebitis | 0 | 2 | 0 | 0 |
| DIGESTIVE SYSTEM | 4 | 0 | 6 | 8 |
| Constipation | 0 | 0 | 0 | 2 |
| Diarrhea | 2 | 0 | 2 | 3 |
| Dyspepsia | 2 | 0 | 3 | 4 |
| METABOLIC AND NUTRITIONAL DISORDERS | 2 | 8 | 7 | 2 |
| Creatine Phosphokinase Increased | 0 | 0 | 4 | 1 |
| Creatinine Increased | 2 | 2 | 0 | 1 |
| Edema | 0 | 2 | 0 | 0 |
| Hypercholesteremia | 0 | 2 | 1 | 0 |
| Hyperglycemia | 0 | 2 | 1 | 0 |
| SGOT Increased | 0 | 0 | 3 | 0 |
| SGPT Increased | 0 | 0 | 2 | 0 |
| MUSCULOSKELETAL SYSTEM | 4 | 0 | 3 | 4 |
| Arthralgia | 2 | 0 | 0 | 2 |
| Arthrosis | 2 | 0 | 0 | 0 |
| Myalgia | 2 | 0 | 3 | 1 |
| NERVOUS SYSTEM | 2 | 2 | 2 | 5 |
| Anxiety | 0 | 2 | 0 | 1 |
| Dizziness | 0 | 0 | 0 | 4 |
| Hypokinesia | 2 | 0 | 0 | 0 |
| RESPIRATORY SYSTEM | 8 | 8 | 7 | 6 |
| Asthma | 2 | 0 | 0 | 0 |
| Dyspnea | 2 | 0 | 2 | 0 |
| Pharyngitis | 2 | 8 | 2 | 4 |
| Pneumonia | 2 | 0 | 0 | 1 |
| Rhinitis | 2 | 2 | 2 | 2 |
| Sinusitis | 0 | 0 | 2 | 0 |
| SKIN AND APPENDAGES | 75 | 86 | 86 | 71 |
| Acne | 0 | 2 | 0 | 1 |
| Application Site Reaction | 75 | 71 | 84 | 70 |
| Acne | 0 | 4 | 1 | 0 |
| Alopecia | 2 | 0 | 1 | 1 |
| Contact Dermatitis | 19 | 4 | 33 | 4 |
| Dry Skin | 27 | 12 | 25 | 17 |
| Edema | 4 | 0 | 3 | 0 |
| Exfoliation | 6 | 4 | 24 | 13 |
| Hyperesthesia | 0 | 0 | 3 | 1 |
| Pain | 15 | 22 | 26 | 30 |
| Paresthesia | 8 | 4 | 20 | 20 |
| Photosensitivity Reaction | 0 | 2 | 3 | 0 |
| Pruritus | 31 | 59 | 52 | 45 |
| Rash | 35 | 20 | 46 | 17 |
| Vesiculobullous Rash | 0 | 0 | 4 | 1 |
| Contact Dermatitis | 2 | 0 | 0 | 0 |
| Dry Skin | 0 | 4 | 3 | 0 |
| Herpes Simplex | 0 | 2 | 0 | 0 |
| Maculopapular Rash | 0 | 2 | 0 | 0 |
| Pain | 2 | 2 | 1 | 0 |
| Pruritus | 4 | 6 | 4 | 1 |
| Rash | 2 | 10 | 4 | 0 |
| Skin Carcinoma | 0 | 6 | 2 | 2 |
| Skin Nodule | 0 | 2 | 0 | 0 |
| Skin Ulcer | 2 | 0 | 1 | 0 |
| SPECIAL SENSES | 2 | 0 | 4 | 2 |
| Conjunctivitis | 2 | 0 | 4 | 1 |
| Eye Pain | 0 | 2 | 2 | 0 |
| UROGENITAL SYSTEM | 0 | 0 | 4 | 5 |
| Hematuria | 0 | 0 | 2 | 1 |
| OTHER | 0 | 0 | 0 | 3 |
| Procedure | 0 | 0 | 0 | 3 |
Skin and Appendages Adverse Events Reported for Diclofenac Sodium Topical Gel at Less Than 1% Incidence in the Phase 3 Studies:
skin hypertrophy, paresthesia, seborrhea, urticaria, application site reactions (skin carcinoma, hypertonia, skin hypertrophy lacrimation disorder, maculopapular rash, purpuric rash, vasodilation).
Adverse Reactions Reported for Oral Diclofenac Dosage Form (not Topical Diclofenac Sodium Gel):*Incidence Greater than 1% marked with asterisk.
Body as a Whole:abdominal pain or cramps*, headache*, fluid retention*, abdominal distention*, malaise, swelling of lips and tongue, photosensitivity, anaphylaxis, anaphylactoid reactions, chest pain.
Cardiovascular:hypertension, congestive heart failure, palpitations, flushing, tachycardia, premature ventricular contractions, myocardial infarction, hypotension.
Digestive:diarrhea*, indigestion*, nausea*, constipation*, flatulence*, liver test abnormalities*, PUB*, i.e., peptic ulcer, with or without bleeding and/or perforation, or bleeding without ulcer, vomiting, jaundice, melena, esophageal lesions, aphthous stomatitis, dry mouth and mucous membranes, bloody diarrhea, hepatitis, hepatic necrosis, cirrhosis, hepatorenal syndrome, appetite change, pancreatitis with or without concomitant hepatitis, colitis, intestinal perforation.
Hemic and Lymphatic:hemoglobin decrease, leukopenia, thrombocytopenia, eosinophilia, hemolytic anemia, aplastic anemia, agranulocytosis, purpura, allergic purpura, bruising.
Metabolic and Nutritional Disorders:azotemia, hypoglycemia, weight loss.
Nervous System:dizziness*, insomnia, drowsiness, depression, diplopia, anxiety, irritability, aseptic meningitis, convulsions, paresthesia, memory disturbance, nightmares, tremor, tic, abnormal coordination, disorientation, psychotic reaction.
Respiratory:epistaxis, asthma, laryngeal edema, dyspnea, hyperventilation, edema of pharynx.
Skin and Appendages:rash*, pruritus*, alopecia, urticaria, eczema, dermatitis, bullous eruption, erythema multiforme major, angioedema, Stevens-Johnson syndrome, excess perspiration, exfoliative dermatitis.
Special Senses:tinnitus*, blurred vision, taste disorder, reversible and irreversible hearing loss, scotoma, vitreous floaters, night blindness, amblyopia.
Urogenital:nephrotic syndrome, proteinuria, oliguria, interstitial nephritis, papillary necrosis, acute renal failure, urinary frequency, nocturia, hematuria, impotence, vaginal bleeding.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide) that accompanies each prescription dispensed, as well as the Directions for Use on the product packaging. Inform patients, families, or their caregivers of the following information before initiating therapy with diclofenac sodium topical gel and periodically during the course of ongoing therapy.
16 How Supplied/storage and Handling
Diclofenac Sodium Topical Gel, 3% is available 100 g (NDC 68071-3688-1) tubes. Each gram of topical gel contains 30 mg of diclofenac sodium.
5.4 Cardiovascular Thrombotic Events
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, including myocardial infarction (MI) and stroke, which can be fatal. Based on available data, it is unclear that the risk for CV thrombotic events is similar for all NSAIDs. The relative increase in serious CV thrombotic events over baseline conferred by NSAID use appears to be similar in those with and without known CV disease or risk factors for CV disease. However, patients with known CV disease or risk factors had a higher absolute incidence of excess serious CV thrombotic events, due to their increased baseline rate. Some observational studies found that this increased risk of serious CV thrombotic events began as early as the first weeks of treatment. The increase in CV thrombotic risk has been observed most consistently at higher doses.
To minimize the potential risk for an adverse CV event in NSAID-treated patients, use the lowest effective dose for the shortest duration possible. Physicians and patients should remain alert for the development of such events, throughout the entire treatment course, even in the absence of previous CV symptoms. Patients should be informed about the symptoms of serious CV events and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as diclofenac, increases the risk of serious gastrointestinal (GI) events.
5.13 Masking of Inflammation and Fever
The pharmacological activity of diclofenac sodium topical gel in reducing inflammation, and possibly fever, may diminish the utility of diagnostic signs in detecting infections.
5.16 Exposure to Eyes and Mucosal Membranes
Avoid contact of diclofenac sodium topical gel with eyes and mucosa. Advise patients that if contact in the eye, or mucosal membranes occurs, immediately wash out the eye or mucosal membranes with water or saline and consult a physician if irritation persists for more than an hour.
Principal Display Panel 100 G Tube Carton
5.17 Oral Nonsteroidal Anti Inflammatory Drugs
Concomitant use of oral and topical NSAIDs may result in a higher rate of hemorrhage, more frequent abnormal creatinine, urea and hemoglobin. Do not use diclofenac sodium topical gel in combination with an oral NSAID unless the benefit outweighs the risk and periodic laboratory evaluations are conducted.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
There did not appear to be any increase in drug-related neoplasms following daily topical applications of diclofenac sodium topical gel for 2 years at concentrations up to 0.035% diclofenac sodium and 2.5% hyaluronate sodium in albino mice.
When administered orally for 2 years, diclofenac showed no evidence of carcinogenic potential in rats given diclofenac sodium at up to 2 mg/kg/day (3 times the MRHD based on BSA comparison), or in mice given diclofenac sodium at up to 0.3 mg/kg/day in males and 1 mg/kg/day in females (25% and 83%, respectively, of the MRHD based on BSA comparison).
Diclofenac was not genotoxic in in vitropoint mutation assays in mammalian mouse lymphoma cells and Ames microbial test systems, or when tested in mammalian in vivoassays including dominant lethal and male germinal epithelial chromosomal studies in mice, and nucleus anomaly and chromosomal aberration studies in Chinese hamsters. It was also negative in the transformation assay utilizing BALB/3T3 mouse embryo cells.
Fertility studies have not been conducted with diclofenac sodium topical gel. Diclofenac sodium showed no evidence of impairment of fertility after oral treatment with 4 mg/kg/day (7 times the MRHD based on BSA comparison) in male or female rats.
5.2 Exacerbation of Asthma Related to Aspirin Sensitivity
A subpopulation of patients with asthma may have aspirin-sensitive asthma which may include chronic rhinosinusitis complicated by nasal polyps; severe, potentially fatal bronchospasm; and/or intolerance to aspirin and other NSAIDs. Because cross-reactivity between aspirin and other NSAIDs has been reported in such aspirin-sensitive patients, diclofenac sodium topical gel is contraindicated in patients with this form of aspirin sensitivity. When diclofenac sodium topical gel is used in patients with preexisting asthma (without known aspirin sensitivity), monitor patients for changes in the signs and symptoms of asthma.
5.5 Gastrointestinal Bleeding, Ulceration, and Perforation
NSAIDs, including diclofenac, cause serious GI adverse events including inflammation, bleeding, ulceration, and perforation of the esophagus, stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with NSAIDs.
Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occurred in approximately 1% of patients treated for 3 to 6 months, and in about 2% to 4% of patients treated for one year. However, even short-term NSAID therapy is not without risk.
5.10 Drug Reaction With Eosinophilia and Systemic Symptoms (dress)
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported in patients taking NSAIDs such as diclofenac sodium topical gel. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, discontinue diclofenac sodium topical gel and evaluate the patient immediately.
Warning: Risk of Serious Cardiovascular and Gastrointestinal Events
WARNING: RISK OF SERIOUS CARDIOVASCULAR EVENTS AND GASTROINTESTINAL EVENTS
See full prescribing information for complete boxed warning.
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use. ( 5.4)
- Diclofenac sodium topical gel is contraindicated in the setting of coronary artery bypass graft (CABG) surgery. ( 4, 5.4)
- NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events ( 5.5)
Structured Label Content
Section 42229-5 (42229-5)
Cardiovascular Thrombotic Events
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use [see Warnings and Precautions (5.4)].
- Diclofenac sodium topical gel is contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications (4)and Warnings and Precautions (5.4)].
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | |
| Revised: August 2023 | |
| Dispense with Medication Guide available at: https://www.taro.com/usa-medication-guides | |
|
Medication Guide
Diclofenac (dye-KLOE-fen-ak) Sodium Topical Gel, 3% |
|
| What is the most important information I should know about diclofenac sodium topical gel and medicines called Nonsteroidal Anti-inflammatory Drugs (NSAIDs)? | |
NSAIDs can cause serious side effects, including:
Do not take or use NSAIDs right before or after a heart surgery called a "coronary artery bypass graft (CABG)". Avoid taking NSAIDs after a recent heart attack unless your healthcare provider tells you to. You may have an increased risk of another heart attack if you take or use NSAIDs after a recent heart attack.
The risk of getting an ulcer or bleeding increases with:
|
|
NSAIDs should only be used:
|
|
|
What is diclofenac sodium topical gel?
Diclofenac sodium topical gel is an NSAID that is used on the skin (topical) to treat a skin condition called actinic keratosis. Diclofenac sodium topical gel is not for use in children. |
|
Do not use diclofenac sodium topical gel:
|
|
Before using diclofenac sodium topical gel, tell your healthcare provider about all of your medical conditions, including if you:
|
|
| Tell your healthcare provider about all of the medicines you take, including prescription or over-the-counter medicines, vitamins, or herbal supplements.NSAIDs and some other medicines can interact with each other and cause serious side effects. Do not start taking any new medicine without talking to your healthcare provider first. | |
How should I use diclofenac sodium topical gel?
|
|
What should I avoid while using diclofenac sodium topical gel?
|
|
| What are the possible side effects of diclofenac sodium topical gel? | |
| Diclofenac sodium topical gel and other NSAIDs can cause serious side effects, including: | |
See "
What is the most important information I should know about diclofenac sodium topical gel and medicines called Nonsteroidal Anti-inflammatory Drugs (NSAIDs)?"
|
|
| Other side effects of NSAIDs include:stomach pain, constipation, diarrhea, gas, heartburn, nausea, vomiting, and dizziness. | |
| Get emergency help right away if you get any of the following symptoms: | |
|
|
| Stop using diclofenac sodium topical gel and call your healthcare provider right away if you get any of the following symptoms: | |
|
|
|
Application site skin reactions are common with diclofenac sodium topical gel including:skin redness, itching, rash, dry skin, scaling, and peeling.
If you take too much NSAID, call your healthcare provider or get medical help right away. Diclofenac sodium topical gel may cause fertility problems in females, which may affect your ability to have a child. Talk to your healthcare provider if this a concern for you. These are not all of the possible side effects of NSAIDs. For more information, ask your healthcare provider or pharmacist about NSAIDs. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
| Other information about NSAIDs | |
|
|
How should I store diclofenac sodium topical gel?
|
|
| Keep diclofenac sodium topical gel and all medicines out of the reach of children. | |
| General information about the safe and effective use of diclofenac sodium topical gel. | |
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use diclofenac sodium topical gel for a condition for which it was not prescribed. Do not give diclofenac sodium topical gel to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information about diclofenac sodium topical gel, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about diclofenac sodium topical gel that is written for health professionals. | |
| What are the ingredients in diclofenac sodium topical gel? | |
| Active ingredient:diclofenac sodium | |
| Inactive ingredients:benzyl alcohol, hyaluronate sodium, polyethylene glycol monomethyl ether, and purified water. | |
| Manufactured by: Taro Pharmaceuticals Inc., Brampton, Ontario, Canada L6T 1C1 | |
| Distributed by: Taro Pharmaceuticals U.S.A., Inc., Hawthorne, NY 10532 | |
| For more information, call 1-866-923-4914. |
Section 44425-7 (44425-7)
Storage: Store at 20° to 25°C (68° to 77°F)[see USP Controlled Room Temperature]. Protect from heat. Avoid freezing.
10 Overdosage (10 OVERDOSAGE)
Symptoms following acute NSAID overdosages have been typically limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which have been generally reversible with supportive care. Gastrointestinal bleeding, hypertension, acute renal failure, respiratory depression, and coma have been reported. [see Warnings and Precautions (5.4, 5.5, 5.7, 5.9)].
Manage patients with symptomatic and supportive care following an NSAID overdosage. There are no specific antidotes. Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may not be useful due to high protein binding.
In the event of oral ingestion, resulting in significant systemic side effects, it is recommended that the stomach be emptied by vomiting or lavage. In addition to supportive measures, the use of oral activated charcoal may help to reduce the absorption of diclofenac.
For additional information about overdosage treatment, call a poison control center (1-800-222-1222).
11 Description (11 DESCRIPTION)
Diclofenac Sodium Topical Gel, 3%, intended for dermatologic use, contains the active ingredient, diclofenac sodium, in a clear, transparent, colorless to slightly yellow gel base. Diclofenac sodium is a white to slightly yellow crystalline powder. It is freely soluble in methanol, soluble in ethanol, sparingly soluble in water, slightly soluble in acetone, and partially insoluble in ether. The chemical name for diclofenac sodium is:
Sodium [ o-(2,6-dichloranilino) phenyl] acetate
Diclofenac sodium has a molecular weight of 318.13.
The CAS number is CAS-15307-79-6. The structural formula is represented below:
Diclofenac Sodium Topical Gel, 3% also contains benzyl alcohol, hyaluronate sodium, polyethylene glycol monomethyl ether, and purified water.
1 g of diclofenac sodium topical gel contains 30 mg of the active substance, diclofenac sodium.
5.7 Hypertension
NSAIDs, including diclofenac sodium topical gel, can lead to new onset of hypertension or worsening of pre-existing hypertension, either of which may contribute to the increased incidence of CV events. Patients taking angiotensin converting enzyme (ACE) inhibitors, thiazide diuretics, or loop diuretics may have impaired response to these therapies when taking NSAIDs [see Drug Interactions (7)].
Monitor blood pressure (BP) during the initiation of NSAID treatment and throughout the course of therapy.
8.4 Pediatric Use
Actinic keratoses is not a condition seen within the pediatric population. Diclofenac sodium topical gel should not be used by children.
8.5 Geriatric Use
Elderly patients, compared to younger patients, are at greater risk for NSAID-associated serious cardiovascular, gastrointestinal, and/or renal adverse reactions. If the anticipated benefit for the elderly patient outweighs these potential risks, start dosing at the low end of the dosing range, and monitor patients for adverse effects [see Warnings and Precautions (5.4, 5.5, 5.6, 5.9, 5.14)].
Of the 211 subjects treated with diclofenac sodium topical gel in controlled clinical trials, 143 subjects were 65 years of age and over. Of those 143 subjects, 55 subjects were 75 years of age and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
5.6 Hepatotoxicity
In clinical trials with diclofenac sodium topical gel, 2 to 3% of subjects had elevations of liver function tests (LFTs) [see Clinical Trials Experience (6.1)]. To minimize the potential risk for an adverse liver-related event in patients treated with diclofenac sodium topical gel, use the lowest effective dose for the shortest duration possible. Exercise caution when prescribing diclofenac sodium topical gel with concomitant drugs that are known to be potentially hepatotoxic (e.g., acetaminophen, antibiotics, anti-epileptics).
Physicians should measure transaminases at baseline and periodically in patients receiving long-term therapy with diclofenac, because severe hepatotoxicity may develop without a prodrome of distinguishing symptoms. The optimum times for making the first and subsequent transaminase measurements are not known. Based on clinical trial data and postmarketing experiences, transaminases should be monitored within 4 to 8 weeks after initiating treatment with diclofenac. However, severe hepatic reactions can occur at any time during treatment with diclofenac.
If abnormal liver tests persist or worsen, if clinical signs and/or symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, abdominal pain, diarrhea, dark urine, etc.), diclofenac sodium topical should be discontinued immediately.
Inform patients of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, diarrhea, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), discontinue diclofenac sodium topical gel immediately, and perform a clinical evaluation of the patient.
14 Clinical Studies (14 CLINICAL STUDIES)
Clinical trials were conducted involving a total of 427 patients (213 treated with diclofenac sodium topical gel and 214 with a gel vehicle). Each patient had no fewer than five AK lesions in a major body area, which was defined as one of five 5 cm × 5 cm regions: scalp, forehead, face, forearm and hand. Up to three major body areas were studied in any patient. All patients were 18 years of age or older (male and female) with no clinically significant medical problems outside of the AK lesions and had undergone a 60-day washout period from disallowed medications (masoprocol, 5-fluorouracil, cyclosporine, retinoids, trichloroacetic acid/lactic acid/peel, 50% glycolic acid peel) and hyaluronan-containing cosmetics. Patients were excluded from participation for reasons of known or suspected hypersensitivity to any diclofenac sodium topical gel ingredient, pregnancy, allergies to aspirin or other nonsteroidal anti-inflammatory drugs (NSAIDs), or other dermatological conditions which might affect the absorption of the study medication. Application of dermatologic products such as sunscreens, cosmetics, and other drug products was not permitted. Patients were instructed to apply a small amount of diclofenac sodium topical gel (approximately 0.5 g) onto the affected skin, using their fingers, and gently smoothing the gel over the lesion. In addition, all patients were instructed to avoid sun exposure. Complete clearing of the AK lesions 30 days after completion of treatment was the primary efficacy variable. No long-term patient follow-ups, after the 30-day assessments, were performed for the detection of recurrence.
| Diclofenac Sodium Topical Gel | Vehicle | p-value | |
|---|---|---|---|
| Study 1 90 days treatment | 27/58 (47%) | 11/59 (19%) | <0.001 |
| Study 2 90 days treatment | 18/53 (34%) | 10/55 (18%) | 0.061 |
| Study 3 60 days treatment | 15/48 (31%) | 5/49 (10%) | 0.021 |
| 30 days treatment | 7/49 (14%) | 2/49 (4%) | 0.221 |
| Scalp | Forehead | Face | Arm/Forearm | Back of Hand | |
|---|---|---|---|---|---|
| Study 1
90 days treatment |
|||||
| Diclofenac Sodium Topical Gel | 1/4 (25%) | 17/30 (57%) | 9/17 (53%) | 4/12 (33%) | 6/16 (38%) |
| Vehicle | 3/9 (33%) | 8/24 (33%) | 5/17 (29%) | 4/12 (33%) | 0/14 (0) |
| p-value | 0.7646 | 0.0908 | 0.1682 | 1.000 | 0.0650 |
| Study 2
90 days treatment |
|||||
| Diclofenac Sodium Topical Gel | 2/6 (33%) | 9/19 (47%) | 4/5 (80%) | 5/8 (63%) | 1/17 (6%) |
| Vehicle | 0/4 (0) | 6/22 (27%) | 2/8 (25%) | 0/5 (0) | 3/16 (19%) |
| p-value | 0.4235 | 0.1870 | 0.0727 | 0.0888 | 0.2818 |
| Study 3
60 days treatment |
|||||
| Diclofenac Sodium Topical Gel | 3/7 (43%) | 13/31 (42%) | 10/19 (53%) | 0/1 (0) | 2/8 (25%) |
| Vehicle | 0/6 (0) | 5/36 (14%) | 2/13 (15%) | 0/2 (0) | 1/9 (11%) |
| p-value | 0.2271 | 0.0153 | 0.0433 | - | 0.4637 |
| 30 days treatment | |||||
| Diclofenac Sodium Topical Gel | 2/5 (40%) | 4/29 (14%) | 3/14 (21%) | 0/0 (0) | 0/9 (0) |
| Vehicle | 0/5 (0) | 2/29 (7%) | 2/18 (11%) | 0/1 (0) | 1/9 (11%) |
| p-value | 0.2299 | 0.3748 | 0.4322 | - | 0.6521 |
| All data combined | |||||
| Diclofenac Sodium Topical Gel | 8/22 (36%) | 43/109 (39%) | 26/55 (47%) | 9/21 (43%) | 9/50 (18%) |
| Vehicle | 3/24 (13%) | 21/111 (19%) | 11/56 (20%) | 4/20 (20%) | 5/48 (10%) |
| p-value | 0.0903 | 0.0013 | 0.0016 | 0.2043 | 0.3662 |
4 Contraindications (4 CONTRAINDICATIONS)
Diclofenac sodium topical gel is contraindicated in the following patients:
- With known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to diclofenac or any components of the drug product [ see Warnings and Precautions (5.1, 5.3, 5.10)and Description (11) ].
- With the history of asthma, urticaria, or other allergic type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients [ see Warnings and Precautions (5.1, 5.2) ].
- Application on damaged skin resulting from any etiology, including exudative dermatitis, eczema, infected lesions, burns or wounds [ see Warnings and Precautions (5.3) ].
- In the setting of coronary bypass graft (CABG) surgery [ see Warnings and Precautions (5.4) ].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Anaphylactic Reactions [ see Warnings and Precautions (5.1) ]
- Exacerbation of Asthma Related to Aspirin Sensitivity [ see Warnings and Precautions (5.2) ]
- Serious Skin Reactions [ see Warnings and Precautions (5.3) ]
- Cardiovascular Thrombotic Events [ see Warnings and Precautions (5.4) ]
- GI Bleeding, Ulceration and Perforation [ see Warnings and Precautions (5.5) ]
- Hepatotoxicity [ see Warnings and Precautions (5.6) ]
- Hypertension [ see Warnings and Precautions (5.7) ]
- Heart Failure and Edema [ see Warnings and Precautions (5.8) ]
- Renal Toxicity and Hyperkalemia [ see Warnings and Precautions (5.9) ]
- DRESS [ see Warnings and Precautions (5.10) ]
- Hematologic Toxicity [ see Warnings and Precautions (5.12) ]
- Photosensitivity [ see Warnings and Precautions (5.15) ]
7 Drug Interactions (7 DRUG INTERACTIONS)
See Table 2 for clinically significant drug interactions with diclofenac.
| Drugs That Interfere with Hemostasis | |
| Clinical Impact: |
|
| Intervention: |
|
| Aspirin | |
| Clinical Impact: |
|
| Intervention: |
|
| ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers | |
| Clinical Impact: |
|
| Intervention: |
|
| Diuretics | |
| Clinical Impact: |
|
| Intervention: |
|
| Digoxin | |
| Clinical Impact: |
|
| Intervention: |
|
| Lithium | |
| Clinical Impact: |
|
| Intervention: |
|
| Methotrexate | |
| Clinical Impact: |
|
| Intervention: |
|
| Cyclosporine | |
| Clinical Impact: |
|
| Intervention: |
|
| NSAIDs and Salicylates | |
| Clinical Impact: |
|
| Intervention: |
|
| Pemetrexed | |
| Clinical Impact: |
|
| Intervention: |
|
12.2 Pharmacodynamics
The pharmacodynamics of diclofenac sodium topical gel in the treatment of actinic keratosis has not been assessed.
5.15 Photosensitivity
Patients should minimize or avoid exposure to natural or artificial sunlight (tanning beds or UVA/B treatment) while using diclofenac sodium topical gel. If patients need to be outdoors while using diclofenac sodium topical gel, they should wear loose-fitting clothes that protect skin from sun exposure and discuss other sun protection measures with their physician. Advise patients to discontinue treatment with diclofenac sodium topical gel at the first evidence of sunburn.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Diclofenac sodium topical gel is indicated for the topical treatment of actinic keratoses (AK).
12.1 Mechanism of Action
The mechanism of action of diclofenac sodium in the treatment of actinic keratoses (AK) is unknown.
5.12 Hematologic Toxicity
Anemia has occurred in NSAID-treated patients. This may be due to occult or gross blood loss, fluid retention, or an incompletely described effect on erythropoiesis. If a patient treated with diclofenac sodium topical gel has any signs or symptoms of anemia, monitor hemoglobin or hematocrit.
NSAIDs, including diclofenac sodium topical gel, may increase the risk of bleeding events. Co-morbid conditions such as coagulation disorders, concomitant use of warfarin, other anticoagulants, antiplatelet agents (e.g., aspirin), serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) may increase this risk. Monitor these patients for signs of bleeding [see Drug Interactions (7)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Anaphylactic Reactions: Seek emergency help if an anaphylactic reaction occurs. ( 5.1)
- Exacerbation of Asthma Related to Aspirin Sensitivity: Diclofenac sodium topical gel is contraindicated in patients with aspirin-sensitive asthma. Monitor patients with pre-existing asthma (without aspirin sensitivity). ( 5.2)
- Serious Skin Reactions: Discontinue diclofenac sodium topical gel at first appearance of skin rash or other signs of hypersensitivity. ( 5.3, 5.15)
- Hepatoxicity: Inform patients of warning signs and symptoms of hepatotoxicity. Discontinue if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop. ( 5.6)
- Hypertension: Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure. ( 5.7, 7)
- Heart Failure and Edema: Avoid use of diclofenac sodium topical gel in patients with severe heart failure. ( 5.8)
- Renal Toxicity: Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of diclofenac sodium topical gel in patients with advanced renal disease. ( 5.9)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue diclofenac sodium topical gel and evaluate clinically. ( 5.10)
- Fetal Toxicity: Limit use of NSAIDs, including diclofenac sodium topical gel, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal renal dysfunction and premature closure of the fetal ductus arteriosus. ( 5.11, 8.1)
- Hematologic Toxicity: Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia. ( 5.12, 7).
- Photosensitivity: Avoid exposure of treated area(s) to natural or artificial sunlight. ( 5.15)
- Exposure to Eyes and Mucosal Membranes: Avoid contact of diclofenac sodium topical gel with eyes and mucosal membranes. ( 5.16)
- Oral Nonsteroidal Anti-inflammatory Drugs: Avoid concurrent use with oral NSAIDs. ( 5.17)
5.1 Anaphylactic Reactions
Diclofenac has been associated with anaphylactic reactions in patients with and without known hypersensitivity to diclofenac and in patients with aspirin-sensitive asthma [ see Contraindications (4)and Warnings and Precautions (5.2) ].
Seek emergency help if an anaphylactic reaction occurs.
5.14 Laboratory Monitoring
Because serious GI bleeding, hepatotoxicity, and renal injury can occur without warning symptoms or signs, consider monitoring patients on long-term NSAID treatment with a CBC and a chemistry profile periodically [see Warnings and Precautions (5.5, 5.6, 5.9)] .
5.3 Serious Skin Reactions
NSAIDs, including diclofenac, can cause serious skin adverse reactions such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. These serious events may occur without warning. Inform patients about the signs and symptoms of serious skin reactions, and to discontinue the use of diclofenac sodium topical gel at the first appearance of skin rash or any other sign of hypersensitivity. Diclofenac sodium topical gel is contraindicated in patients with previous serious skin reactions to NSAIDs. Do not apply diclofenac sodium topical gel to open skin wounds, infections, or exfoliative dermatitis, as it may affect absorption and tolerability of the drug [ see Contraindications (4) ].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Use the lowest effective dosage for the shortest duration consistent with individual patient treatment goals [see Warnings and Precautions (5)] .
Apply diclofenac sodium topical gel gently to lesion areas twice daily to adequately cover each lesion. Use 0.5 g of gel (pea size) on each 5 cm × 5 cm lesion site. The recommended duration of therapy is from 60 days to 90 days. Complete healing of the lesion(s) or optimal therapeutic effect may not be evident for up to 30 days following cessation of therapy. Lesions that do not respond to therapy should be re-evaluated and management reconsidered. Avoid contact of diclofenac sodium topical gel with eyes and mucous membranes.
5.8 Heart Failure and Edema
The Coxib and traditional NSAID Trialists' Collaboration meta-analysis of randomized controlled trials demonstrated an approximately two-fold increase in hospitalizations for heart failure in COX-2 selective-treated patients and nonselective NSAID-treated patients compared to placebo-treated patients. In a Danish National Registry study of patients with heart failure, NSAID use increased the risk of MI, hospitalization for heart failure, and death.
Additionally, fluid retention and edema have been observed in some patients treated with NSAIDs. Use of diclofenac may blunt the CV effects of several therapeutic agents used to treat these medical conditions [e.g., diuretics, ACE inhibitors, or angiotensin receptor blockers (ARBs)].
Avoid the use of diclofenac sodium topical gel in patients with severe heart failure unless the benefits are expected to outweigh the risk of worsening heart failure. If diclofenac sodium topical gel is used in patients with severe heart failure, monitor patients for signs of worsening heart failure.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Topical gel, 3%. Each gram of diclofenac sodium topical gel contains 30 mg of diclofenac sodium in a clear, transparent, colorless to slightly yellow gel base. Diclofenac sodium topical gel is supplied in 50 g and 100 g tubes.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of diclofenac sodium topical gel and other topical diclofenac products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse reactions from diclofenac sodium topical gel: burning sensation, hypersensitivity.
Adverse reactions from other topical diclofenac products: hypoesthesia, gait disturbance, musculoskeletal stiffness.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Infertility: NSAIDs are associated with reversible infertility. Consider withdrawal of diclofenac sodium topical gel in women who have difficulties conceiving. ( 8.3)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Of the 423 subjects evaluable for safety in adequate and well-controlled trials, 211 were treated with diclofenac sodium topical gel drug product and 212 were treated with a vehicle gel. Eighty-seven percent (87%) of the diclofenac sodium topical gel-treated subjects (183 subjects) and 84% of the vehicle-treated subjects (178 subjects) experienced one or more adverse events (AEs) during the trials. The majority of these reactions were mild to moderate in severity and resolved upon discontinuation of therapy.
Of the 211 subjects treated with diclofenac sodium topical gel, 172 (82%) experienced AEs involving skin and the application site compared to 160 (75%) vehicle-treated subjects. Application site reactions (ASRs) were the most frequent AEs in both diclofenac sodium topical gel- and vehicle-treated groups. Of note, four reactions, contact dermatitis, rash, dry skin and exfoliation (scaling) were significantly more prevalent in the diclofenac sodium topical gel group than in the vehicle-treated subjects.
Eighteen percent of diclofenac sodium topical gel-treated subjects and 4% of vehicle-treated subjects discontinued from the clinical trials due to adverse events (whether considered related to treatment or not). These discontinuations were mainly due to skin irritation or related cutaneous adverse reactions.
Table 1 below presents the AEs reported at an incidence of >1% for subjects treated with either diclofenac sodium topical gel or vehicle (60- and 90-day treatment groups) during the phase 3 trials.
| 60-day Treatment | 90-day Treatment | |||
|---|---|---|---|---|
| Diclofenac Sodium Topical Gel (%)
N=48 |
Gel Vehicle
(%) N=49 |
Diclofenac Sodium Topical Gel (%)
N=114 |
Gel Vehicle
(%) N=114 |
|
| BODY AS A WHOLE | 21 | 20 | 20 | 18 |
| Abdominal Pain | 2 | 0 | 1 | 0 |
| Accidental Injury | 0 | 0 | 4 | 2 |
| Allergic Reaction | 0 | 0 | 1 | 3 |
| Asthenia | 0 | 0 | 2 | 0 |
| Back Pain | 4 | 0 | 2 | 2 |
| Chest Pain | 2 | 0 | 1 | 0 |
| Chills | 0 | 2 | 0 | 0 |
| Flu Syndrome | 10 | 6 | 1 | 4 |
| Headache | 0 | 6 | 7 | 6 |
| Infection | 4 | 6 | 4 | 5 |
| Neck Pain | 0 | 0 | 2 | 0 |
| Pain | 2 | 0 | 2 | 2 |
| CARDIOVASCULAR SYSTEM | 2 | 4 | 3 | 1 |
| Hypertension | 2 | 0 | 1 | 0 |
| Migraine | 0 | 2 | 1 | 0 |
| Phlebitis | 0 | 2 | 0 | 0 |
| DIGESTIVE SYSTEM | 4 | 0 | 6 | 8 |
| Constipation | 0 | 0 | 0 | 2 |
| Diarrhea | 2 | 0 | 2 | 3 |
| Dyspepsia | 2 | 0 | 3 | 4 |
| METABOLIC AND NUTRITIONAL DISORDERS | 2 | 8 | 7 | 2 |
| Creatine Phosphokinase Increased | 0 | 0 | 4 | 1 |
| Creatinine Increased | 2 | 2 | 0 | 1 |
| Edema | 0 | 2 | 0 | 0 |
| Hypercholesteremia | 0 | 2 | 1 | 0 |
| Hyperglycemia | 0 | 2 | 1 | 0 |
| SGOT Increased | 0 | 0 | 3 | 0 |
| SGPT Increased | 0 | 0 | 2 | 0 |
| MUSCULOSKELETAL SYSTEM | 4 | 0 | 3 | 4 |
| Arthralgia | 2 | 0 | 0 | 2 |
| Arthrosis | 2 | 0 | 0 | 0 |
| Myalgia | 2 | 0 | 3 | 1 |
| NERVOUS SYSTEM | 2 | 2 | 2 | 5 |
| Anxiety | 0 | 2 | 0 | 1 |
| Dizziness | 0 | 0 | 0 | 4 |
| Hypokinesia | 2 | 0 | 0 | 0 |
| RESPIRATORY SYSTEM | 8 | 8 | 7 | 6 |
| Asthma | 2 | 0 | 0 | 0 |
| Dyspnea | 2 | 0 | 2 | 0 |
| Pharyngitis | 2 | 8 | 2 | 4 |
| Pneumonia | 2 | 0 | 0 | 1 |
| Rhinitis | 2 | 2 | 2 | 2 |
| Sinusitis | 0 | 0 | 2 | 0 |
| SKIN AND APPENDAGES | 75 | 86 | 86 | 71 |
| Acne | 0 | 2 | 0 | 1 |
| Application Site Reaction | 75 | 71 | 84 | 70 |
| Acne | 0 | 4 | 1 | 0 |
| Alopecia | 2 | 0 | 1 | 1 |
| Contact Dermatitis | 19 | 4 | 33 | 4 |
| Dry Skin | 27 | 12 | 25 | 17 |
| Edema | 4 | 0 | 3 | 0 |
| Exfoliation | 6 | 4 | 24 | 13 |
| Hyperesthesia | 0 | 0 | 3 | 1 |
| Pain | 15 | 22 | 26 | 30 |
| Paresthesia | 8 | 4 | 20 | 20 |
| Photosensitivity Reaction | 0 | 2 | 3 | 0 |
| Pruritus | 31 | 59 | 52 | 45 |
| Rash | 35 | 20 | 46 | 17 |
| Vesiculobullous Rash | 0 | 0 | 4 | 1 |
| Contact Dermatitis | 2 | 0 | 0 | 0 |
| Dry Skin | 0 | 4 | 3 | 0 |
| Herpes Simplex | 0 | 2 | 0 | 0 |
| Maculopapular Rash | 0 | 2 | 0 | 0 |
| Pain | 2 | 2 | 1 | 0 |
| Pruritus | 4 | 6 | 4 | 1 |
| Rash | 2 | 10 | 4 | 0 |
| Skin Carcinoma | 0 | 6 | 2 | 2 |
| Skin Nodule | 0 | 2 | 0 | 0 |
| Skin Ulcer | 2 | 0 | 1 | 0 |
| SPECIAL SENSES | 2 | 0 | 4 | 2 |
| Conjunctivitis | 2 | 0 | 4 | 1 |
| Eye Pain | 0 | 2 | 2 | 0 |
| UROGENITAL SYSTEM | 0 | 0 | 4 | 5 |
| Hematuria | 0 | 0 | 2 | 1 |
| OTHER | 0 | 0 | 0 | 3 |
| Procedure | 0 | 0 | 0 | 3 |
Skin and Appendages Adverse Events Reported for Diclofenac Sodium Topical Gel at Less Than 1% Incidence in the Phase 3 Studies:
skin hypertrophy, paresthesia, seborrhea, urticaria, application site reactions (skin carcinoma, hypertonia, skin hypertrophy lacrimation disorder, maculopapular rash, purpuric rash, vasodilation).
Adverse Reactions Reported for Oral Diclofenac Dosage Form (not Topical Diclofenac Sodium Gel):*Incidence Greater than 1% marked with asterisk.
Body as a Whole:abdominal pain or cramps*, headache*, fluid retention*, abdominal distention*, malaise, swelling of lips and tongue, photosensitivity, anaphylaxis, anaphylactoid reactions, chest pain.
Cardiovascular:hypertension, congestive heart failure, palpitations, flushing, tachycardia, premature ventricular contractions, myocardial infarction, hypotension.
Digestive:diarrhea*, indigestion*, nausea*, constipation*, flatulence*, liver test abnormalities*, PUB*, i.e., peptic ulcer, with or without bleeding and/or perforation, or bleeding without ulcer, vomiting, jaundice, melena, esophageal lesions, aphthous stomatitis, dry mouth and mucous membranes, bloody diarrhea, hepatitis, hepatic necrosis, cirrhosis, hepatorenal syndrome, appetite change, pancreatitis with or without concomitant hepatitis, colitis, intestinal perforation.
Hemic and Lymphatic:hemoglobin decrease, leukopenia, thrombocytopenia, eosinophilia, hemolytic anemia, aplastic anemia, agranulocytosis, purpura, allergic purpura, bruising.
Metabolic and Nutritional Disorders:azotemia, hypoglycemia, weight loss.
Nervous System:dizziness*, insomnia, drowsiness, depression, diplopia, anxiety, irritability, aseptic meningitis, convulsions, paresthesia, memory disturbance, nightmares, tremor, tic, abnormal coordination, disorientation, psychotic reaction.
Respiratory:epistaxis, asthma, laryngeal edema, dyspnea, hyperventilation, edema of pharynx.
Skin and Appendages:rash*, pruritus*, alopecia, urticaria, eczema, dermatitis, bullous eruption, erythema multiforme major, angioedema, Stevens-Johnson syndrome, excess perspiration, exfoliative dermatitis.
Special Senses:tinnitus*, blurred vision, taste disorder, reversible and irreversible hearing loss, scotoma, vitreous floaters, night blindness, amblyopia.
Urogenital:nephrotic syndrome, proteinuria, oliguria, interstitial nephritis, papillary necrosis, acute renal failure, urinary frequency, nocturia, hematuria, impotence, vaginal bleeding.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide) that accompanies each prescription dispensed, as well as the Directions for Use on the product packaging. Inform patients, families, or their caregivers of the following information before initiating therapy with diclofenac sodium topical gel and periodically during the course of ongoing therapy.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Diclofenac Sodium Topical Gel, 3% is available 100 g (NDC 68071-3688-1) tubes. Each gram of topical gel contains 30 mg of diclofenac sodium.
5.4 Cardiovascular Thrombotic Events
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, including myocardial infarction (MI) and stroke, which can be fatal. Based on available data, it is unclear that the risk for CV thrombotic events is similar for all NSAIDs. The relative increase in serious CV thrombotic events over baseline conferred by NSAID use appears to be similar in those with and without known CV disease or risk factors for CV disease. However, patients with known CV disease or risk factors had a higher absolute incidence of excess serious CV thrombotic events, due to their increased baseline rate. Some observational studies found that this increased risk of serious CV thrombotic events began as early as the first weeks of treatment. The increase in CV thrombotic risk has been observed most consistently at higher doses.
To minimize the potential risk for an adverse CV event in NSAID-treated patients, use the lowest effective dose for the shortest duration possible. Physicians and patients should remain alert for the development of such events, throughout the entire treatment course, even in the absence of previous CV symptoms. Patients should be informed about the symptoms of serious CV events and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as diclofenac, increases the risk of serious gastrointestinal (GI) events.
5.13 Masking of Inflammation and Fever
The pharmacological activity of diclofenac sodium topical gel in reducing inflammation, and possibly fever, may diminish the utility of diagnostic signs in detecting infections.
5.16 Exposure to Eyes and Mucosal Membranes
Avoid contact of diclofenac sodium topical gel with eyes and mucosa. Advise patients that if contact in the eye, or mucosal membranes occurs, immediately wash out the eye or mucosal membranes with water or saline and consult a physician if irritation persists for more than an hour.
Principal Display Panel 100 G Tube Carton (PRINCIPAL DISPLAY PANEL - 100 g Tube Carton)
5.17 Oral Nonsteroidal Anti Inflammatory Drugs (5.17 Oral Nonsteroidal Anti-inflammatory Drugs)
Concomitant use of oral and topical NSAIDs may result in a higher rate of hemorrhage, more frequent abnormal creatinine, urea and hemoglobin. Do not use diclofenac sodium topical gel in combination with an oral NSAID unless the benefit outweighs the risk and periodic laboratory evaluations are conducted.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
There did not appear to be any increase in drug-related neoplasms following daily topical applications of diclofenac sodium topical gel for 2 years at concentrations up to 0.035% diclofenac sodium and 2.5% hyaluronate sodium in albino mice.
When administered orally for 2 years, diclofenac showed no evidence of carcinogenic potential in rats given diclofenac sodium at up to 2 mg/kg/day (3 times the MRHD based on BSA comparison), or in mice given diclofenac sodium at up to 0.3 mg/kg/day in males and 1 mg/kg/day in females (25% and 83%, respectively, of the MRHD based on BSA comparison).
Diclofenac was not genotoxic in in vitropoint mutation assays in mammalian mouse lymphoma cells and Ames microbial test systems, or when tested in mammalian in vivoassays including dominant lethal and male germinal epithelial chromosomal studies in mice, and nucleus anomaly and chromosomal aberration studies in Chinese hamsters. It was also negative in the transformation assay utilizing BALB/3T3 mouse embryo cells.
Fertility studies have not been conducted with diclofenac sodium topical gel. Diclofenac sodium showed no evidence of impairment of fertility after oral treatment with 4 mg/kg/day (7 times the MRHD based on BSA comparison) in male or female rats.
5.2 Exacerbation of Asthma Related to Aspirin Sensitivity
A subpopulation of patients with asthma may have aspirin-sensitive asthma which may include chronic rhinosinusitis complicated by nasal polyps; severe, potentially fatal bronchospasm; and/or intolerance to aspirin and other NSAIDs. Because cross-reactivity between aspirin and other NSAIDs has been reported in such aspirin-sensitive patients, diclofenac sodium topical gel is contraindicated in patients with this form of aspirin sensitivity. When diclofenac sodium topical gel is used in patients with preexisting asthma (without known aspirin sensitivity), monitor patients for changes in the signs and symptoms of asthma.
5.5 Gastrointestinal Bleeding, Ulceration, and Perforation
NSAIDs, including diclofenac, cause serious GI adverse events including inflammation, bleeding, ulceration, and perforation of the esophagus, stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with NSAIDs.
Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occurred in approximately 1% of patients treated for 3 to 6 months, and in about 2% to 4% of patients treated for one year. However, even short-term NSAID therapy is not without risk.
5.10 Drug Reaction With Eosinophilia and Systemic Symptoms (dress) (5.10 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS))
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported in patients taking NSAIDs such as diclofenac sodium topical gel. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, discontinue diclofenac sodium topical gel and evaluate the patient immediately.
Warning: Risk of Serious Cardiovascular and Gastrointestinal Events (WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS)
WARNING: RISK OF SERIOUS CARDIOVASCULAR EVENTS AND GASTROINTESTINAL EVENTS
See full prescribing information for complete boxed warning.
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use. ( 5.4)
- Diclofenac sodium topical gel is contraindicated in the setting of coronary artery bypass graft (CABG) surgery. ( 4, 5.4)
- NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events ( 5.5)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:01.657548 · Updated: 2026-03-14T22:12:30.161895