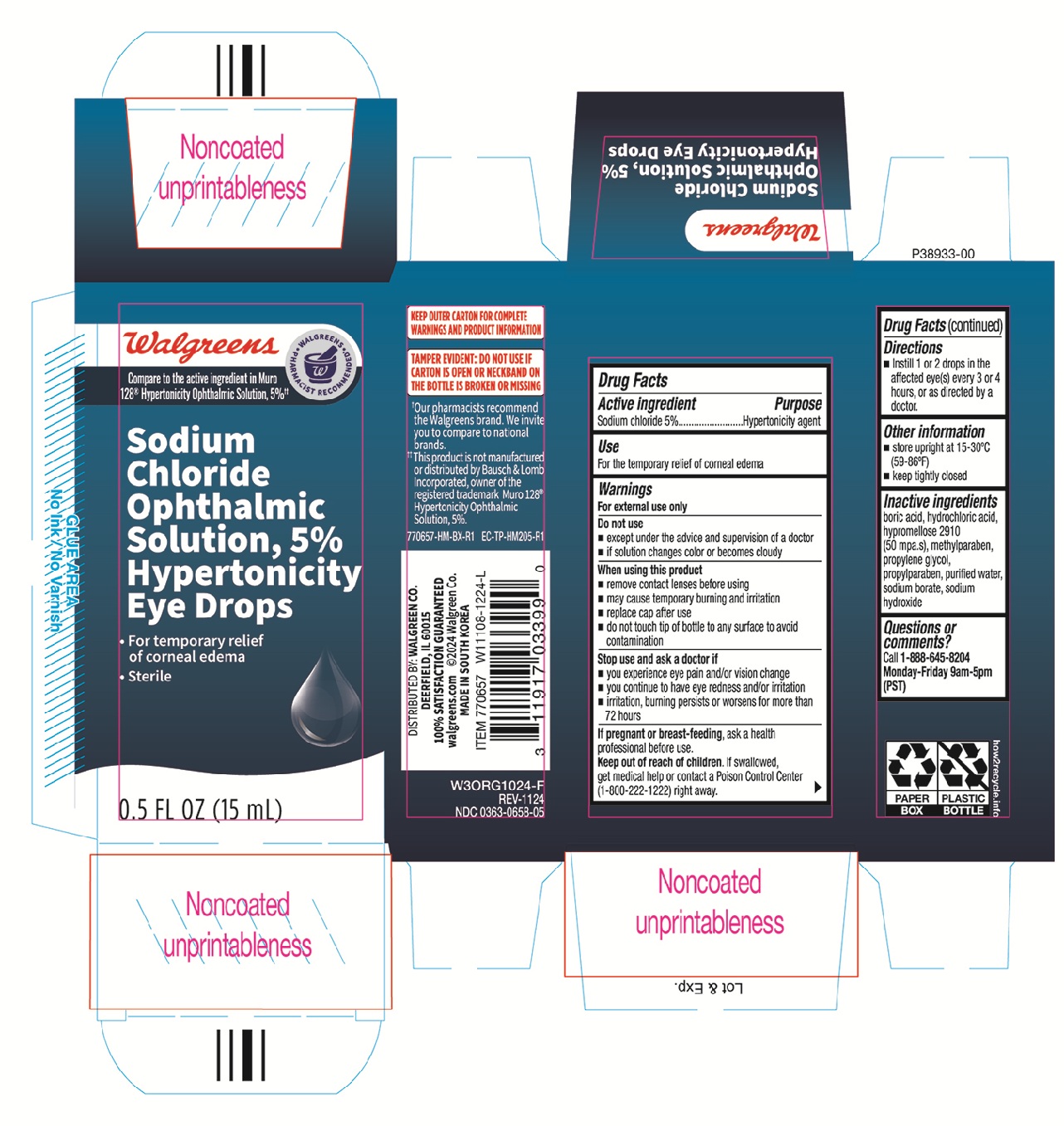
Walgreens Sodium Chloride Ophthalmic Solution 5% Hypertonicity Eye Drops 0.5oz 770657 (nbe Muro128)
21dfc2cf-65ae-c3fb-e063-6294a90a0a94
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient Purpose Sodium chloride 5%.............................Hypertonicity agent
Medication Information
Indications and Usage
Directions
- Instill 1 or 2 drops in the affected eye(s) every 3 or 4 hours, or as directed by a doctor.
Dosage and Administration
Distributed by:
Walgreen Co.
Deerfield, IL 60015
Made in South Korea
Description
Active ingredient Purpose Sodium chloride 5%.............................Hypertonicity agent
Section 44425-7
Other information
- store upright at 15-30°C (59-86°F)
- keep tightly closed
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9
Stop use and ask a doctor if
- you experience eye pain and/or vision change
- you continue to have eye redness and or irritation
- irritation, burning persits or worsens for more than 72 hours
Section 50567-7
When using this product
- remove contact lenses before using
- may cause temporary burning and irritation
- replace cap after use
- do not touch tip of bottle to any surface to avoid contamination
Section 51727-6
Inactive ingredients
boric acid, hydrochloric acid, hypromellose 2910 (50 mpa.s), methylparaben, propylene glycol, propylparaben, purified water, sodium borate, sodium hydroxide
Section 51945-4
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Use
For the temporary relief of corneal edema
Section 55106-9
Active ingredient Purpose
Sodium chloride 5%.............................Hypertonicity agent
Structured Label Content
Indications and Usage (34067-9)
Directions
- Instill 1 or 2 drops in the affected eye(s) every 3 or 4 hours, or as directed by a doctor.
Dosage and Administration (34068-7)
Distributed by:
Walgreen Co.
Deerfield, IL 60015
Made in South Korea
Section 44425-7 (44425-7)
Other information
- store upright at 15-30°C (59-86°F)
- keep tightly closed
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- you experience eye pain and/or vision change
- you continue to have eye redness and or irritation
- irritation, burning persits or worsens for more than 72 hours
Section 50567-7 (50567-7)
When using this product
- remove contact lenses before using
- may cause temporary burning and irritation
- replace cap after use
- do not touch tip of bottle to any surface to avoid contamination
Section 51727-6 (51727-6)
Inactive ingredients
boric acid, hydrochloric acid, hypromellose 2910 (50 mpa.s), methylparaben, propylene glycol, propylparaben, purified water, sodium borate, sodium hydroxide
Section 51945-4 (51945-4)
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Use
For the temporary relief of corneal edema
Section 55106-9 (55106-9)
Active ingredient Purpose
Sodium chloride 5%.............................Hypertonicity agent
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:52.403552 · Updated: 2026-03-14T23:13:06.603726