Erythromycin Topical Solution, Usp 2%
21b64a3f-2c95-1c1a-e063-6294a90a0ea0
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
For Topical Use Only Not for Ophthalmic Use Rx Only
Indications and Usage
Erythromycin topical solution, 2% is indicated for the topical treatment of acne vulgaris.
Dosage and Administration
Erythromycin topical solution, 2% should be applied over the affected areas twice a day (morning and evening) after the skin is thoroughly washed with warm water and soap and patted dry. Acne lesions on the face, neck, shoulders, chest, and back may be treated in this manner. This medication should be applied with applicator top. If fingertips are used, wash hands after application. Drying and peeling may be controlled by reducing the frequency of applications.
Contraindications
Erythromycin topical solution, 2% is contraindicated in those individuals who have shown hypersensitivity to any of its components.
Adverse Reactions
The following local adverse reactions have been reported occasionally: peeling, dryness, itching, erythema, and oiliness. Irritation of the eyes and tenderness of the skin have also been reported with topical use of erythromycin. Generalized urticarial reactions possibly related to the use of erythromycin, which required systemic steroid therapy have been reported. To report SUSPECTED ADVERSE REACTIONS, contact Saptalis Pharmaceuticals, LLC at 1-833-727-8254 or FDA at 1-800‑FDA‑1088 or www.fda.gov/medwatch.
How Supplied
Erythromycin topical solution, USP 2% is available as follows: 60 mL bottle with applicator (NDC 71656-030-60) Store at 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Keep container tightly closed.
Medication Information
Indications and Usage
Erythromycin topical solution, 2% is indicated for the topical treatment of acne vulgaris.
Dosage and Administration
Erythromycin topical solution, 2% should be applied over the affected areas twice a day (morning and evening) after the skin is thoroughly washed with warm water and soap and patted dry. Acne lesions on the face, neck, shoulders, chest, and back may be treated in this manner.
This medication should be applied with applicator top. If fingertips are used, wash hands after application. Drying and peeling may be controlled by reducing the frequency of applications.
Contraindications
Erythromycin topical solution, 2% is contraindicated in those individuals who have shown hypersensitivity to any of its components.
Adverse Reactions
The following local adverse reactions have been reported occasionally: peeling, dryness, itching, erythema, and oiliness. Irritation of the eyes and tenderness of the skin have also been reported with topical use of erythromycin. Generalized urticarial reactions possibly related to the use of erythromycin, which required systemic steroid therapy have been reported.
To report SUSPECTED ADVERSE REACTIONS, contact Saptalis Pharmaceuticals, LLC at 1-833-727-8254 or FDA at 1-800‑FDA‑1088 or www.fda.gov/medwatch.
How Supplied
Erythromycin topical solution, USP 2% is available as follows:
60 mL bottle with applicator (NDC 71656-030-60)
Store at 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Keep container tightly closed.
Description
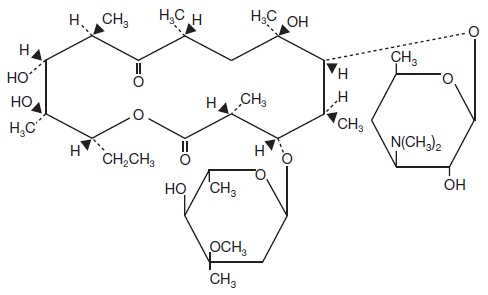
Erythromycin topical solution, USP 2% contains erythromycin, USP for topical dermatologic use. Erythromycin is a macrolide antibiotic produced from a strain of Saccaropolyspora erythraea(formerly Streptomyces erythreus). It is a base and readily forms salts with acids. Chemically, erythromycin is: (3 R*,4 S*,5 S*,6 R*,7 R*,9 R*,11 R*,12 R*,13 S*,14 R*)-4-[(2,6-Dideoxy-3- C-methyl-3- 0-methyl-α-L- ribo-hexopyranosyl)oxy]-14-ethyl-7,12,13-trihydroxy -3,5,7,9,11,13-hexamethyl-6-[[3,4,6-trideoxy-3-(dimethylamino)-ß-D- xylo-hexopyranosyl]oxy]oxacyclotetradecane-2,10-dione. It has the following structural formula:
Molecular Formula: C 37H 67NO 13
Molecular Weight: 733.94
Erythromycin, USP is a white or slightly yellow, crystalline powder that is soluble in water, alcohol, chloroform and in ether.
Each mL of erythromycin topical solution, USP 2% contains 20 mg of erythromycin base in a vehicle consisting of alcohol (66%), citric acid, and propylene glycol.
Section 42229-5
For Topical Use Only
Not for Ophthalmic Use
Rx Only
Warnings
Pseudomembranous colitis has been reported with nearly all antibacterial agents, including erythromycin, and may range in severity from mild to life-threatening. Therefore, it is important to consider this diagnosis in patients who present with diarrhea subsequent to the administration of antibacterial agents.
Treatment with antibacterial agents alters the normal flora of the colon and may permit overgrowth of clostridia. Studies indicate that a toxin produced by Clostridium difficileis one primary cause of “antibiotic-associated colitis”.
After the diagnosis of pseudomembranous colitis has been established, therapeutic measures should be initiated. Mild cases of pseudomembranous colitis usually respond to drug discontinuation alone. In moderate to severe cases, consideration should be given to management with fluids and electrolytes, protein supplementation and treatment with an antibacterial drug clinically effective against C. difficilecolitis.
General
For topical use only; not for ophthalmic use. Concomitant topical acne therapy should be used with caution because a possible cumulative irritancy effect may occur, especially with the use of peeling, desquamating or abrasive agents.
The use of antibiotic agents may be associated with the overgrowth of antibiotic-resistant organisms. If this occurs, discontinue use and take appropriate measures.
Avoid contact with eyes and all mucous membranes.
Microbiology
Erythromycin acts by inhibition of protein synthesis in susceptible organisms by reversibly binding to 50S ribosomal subunits, thereby inhibiting translocation of aminoacyl transfer-RNA and inhibiting polypeptide synthesis. Antagonism has been demonstrated in vitrobetween erythromycin, lincomycin, chloramphenicol, and clindamycin.
Pediatric Use
Safety and effectiveness of this product in pediatric patients have not been established.
Nursing Mothers
It is not known whether erythromycin is excreted in human milk after topical application. However, erythromycin is excreted in human milk following oral and parenteral erythromycin administration. Therefore, caution should be exercised when erythromycin is administered to a nursing woman.
Clinical Pharmacology
The exact mechanism by which erythromycin reduces lesions of acne vulgaris is not fully known; however, the effect appears to be due in part to the antibacterial activity of the drug.
Information for Patients
Patients using erythromycin topical solution, 2% should receive the following information and instructions:
- This medication is to be used as directed by the physician. It is for external use only. Avoid contact with the eyes, nose, mouth, and all mucous membranes.
- This medication should not be used for any disorder other than that for which it was prescribed.
- Patients should not use any other topical acne medication unless otherwise directed by their physician.
- Patients should report to their physician any signs of local adverse reactions.
Principal Display Panel Carton
NDC 71656-030-60
Erythromycin Topical Solution, USP 2%
For Topical Use Only
Not for Ophthalmic Use
Rx only
60 mL
Principal Display Panel Label
NDC 71656-030-60
Erythromycin Topical Solutino, USP 2%
For Topical Use Only
Not for Ophthalmic Use
Rx only
60 mL
Carcinogenesis, Mutagenesis, Impairment of Fertility
No animal studies have been performed to evaluate the carcinogenic and mutagenic potential or effects on fertility of topical erythromycin. However, long-term (2 years) oral studies in rats with erythromycin ethylsuccinate and erythromycin base did not provide evidence of tumorigenicity. There was no apparent effect on male or female fertility in rats fed erythromycin (base) at levels up to 0.25% of diet.
Structured Label Content
Section 42229-5 (42229-5)
For Topical Use Only
Not for Ophthalmic Use
Rx Only
Warnings (WARNINGS)
Pseudomembranous colitis has been reported with nearly all antibacterial agents, including erythromycin, and may range in severity from mild to life-threatening. Therefore, it is important to consider this diagnosis in patients who present with diarrhea subsequent to the administration of antibacterial agents.
Treatment with antibacterial agents alters the normal flora of the colon and may permit overgrowth of clostridia. Studies indicate that a toxin produced by Clostridium difficileis one primary cause of “antibiotic-associated colitis”.
After the diagnosis of pseudomembranous colitis has been established, therapeutic measures should be initiated. Mild cases of pseudomembranous colitis usually respond to drug discontinuation alone. In moderate to severe cases, consideration should be given to management with fluids and electrolytes, protein supplementation and treatment with an antibacterial drug clinically effective against C. difficilecolitis.
General
For topical use only; not for ophthalmic use. Concomitant topical acne therapy should be used with caution because a possible cumulative irritancy effect may occur, especially with the use of peeling, desquamating or abrasive agents.
The use of antibiotic agents may be associated with the overgrowth of antibiotic-resistant organisms. If this occurs, discontinue use and take appropriate measures.
Avoid contact with eyes and all mucous membranes.
Description (DESCRIPTION)
Erythromycin topical solution, USP 2% contains erythromycin, USP for topical dermatologic use. Erythromycin is a macrolide antibiotic produced from a strain of Saccaropolyspora erythraea(formerly Streptomyces erythreus). It is a base and readily forms salts with acids. Chemically, erythromycin is: (3 R*,4 S*,5 S*,6 R*,7 R*,9 R*,11 R*,12 R*,13 S*,14 R*)-4-[(2,6-Dideoxy-3- C-methyl-3- 0-methyl-α-L- ribo-hexopyranosyl)oxy]-14-ethyl-7,12,13-trihydroxy -3,5,7,9,11,13-hexamethyl-6-[[3,4,6-trideoxy-3-(dimethylamino)-ß-D- xylo-hexopyranosyl]oxy]oxacyclotetradecane-2,10-dione. It has the following structural formula:
Molecular Formula: C 37H 67NO 13
Molecular Weight: 733.94
Erythromycin, USP is a white or slightly yellow, crystalline powder that is soluble in water, alcohol, chloroform and in ether.
Each mL of erythromycin topical solution, USP 2% contains 20 mg of erythromycin base in a vehicle consisting of alcohol (66%), citric acid, and propylene glycol.
How Supplied (HOW SUPPLIED)
Erythromycin topical solution, USP 2% is available as follows:
60 mL bottle with applicator (NDC 71656-030-60)
Store at 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Keep container tightly closed.
Microbiology
Erythromycin acts by inhibition of protein synthesis in susceptible organisms by reversibly binding to 50S ribosomal subunits, thereby inhibiting translocation of aminoacyl transfer-RNA and inhibiting polypeptide synthesis. Antagonism has been demonstrated in vitrobetween erythromycin, lincomycin, chloramphenicol, and clindamycin.
Pediatric Use
Safety and effectiveness of this product in pediatric patients have not been established.
Adverse Reactions (ADVERSE REACTIONS)
The following local adverse reactions have been reported occasionally: peeling, dryness, itching, erythema, and oiliness. Irritation of the eyes and tenderness of the skin have also been reported with topical use of erythromycin. Generalized urticarial reactions possibly related to the use of erythromycin, which required systemic steroid therapy have been reported.
To report SUSPECTED ADVERSE REACTIONS, contact Saptalis Pharmaceuticals, LLC at 1-833-727-8254 or FDA at 1-800‑FDA‑1088 or www.fda.gov/medwatch.
Contraindications (CONTRAINDICATIONS)
Erythromycin topical solution, 2% is contraindicated in those individuals who have shown hypersensitivity to any of its components.
Nursing Mothers
It is not known whether erythromycin is excreted in human milk after topical application. However, erythromycin is excreted in human milk following oral and parenteral erythromycin administration. Therefore, caution should be exercised when erythromycin is administered to a nursing woman.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
The exact mechanism by which erythromycin reduces lesions of acne vulgaris is not fully known; however, the effect appears to be due in part to the antibacterial activity of the drug.
Indications and Usage (INDICATIONS AND USAGE)
Erythromycin topical solution, 2% is indicated for the topical treatment of acne vulgaris.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Erythromycin topical solution, 2% should be applied over the affected areas twice a day (morning and evening) after the skin is thoroughly washed with warm water and soap and patted dry. Acne lesions on the face, neck, shoulders, chest, and back may be treated in this manner.
This medication should be applied with applicator top. If fingertips are used, wash hands after application. Drying and peeling may be controlled by reducing the frequency of applications.
Information for Patients
Patients using erythromycin topical solution, 2% should receive the following information and instructions:
- This medication is to be used as directed by the physician. It is for external use only. Avoid contact with the eyes, nose, mouth, and all mucous membranes.
- This medication should not be used for any disorder other than that for which it was prescribed.
- Patients should not use any other topical acne medication unless otherwise directed by their physician.
- Patients should report to their physician any signs of local adverse reactions.
Principal Display Panel Carton (Principal display panel-Carton)
NDC 71656-030-60
Erythromycin Topical Solution, USP 2%
For Topical Use Only
Not for Ophthalmic Use
Rx only
60 mL
Principal Display Panel Label (Principal display panel-Label )
NDC 71656-030-60
Erythromycin Topical Solutino, USP 2%
For Topical Use Only
Not for Ophthalmic Use
Rx only
60 mL
Carcinogenesis, Mutagenesis, Impairment of Fertility
No animal studies have been performed to evaluate the carcinogenic and mutagenic potential or effects on fertility of topical erythromycin. However, long-term (2 years) oral studies in rats with erythromycin ethylsuccinate and erythromycin base did not provide evidence of tumorigenicity. There was no apparent effect on male or female fertility in rats fed erythromycin (base) at levels up to 0.25% of diet.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:42:50.051561 · Updated: 2026-03-14T22:11:10.971269