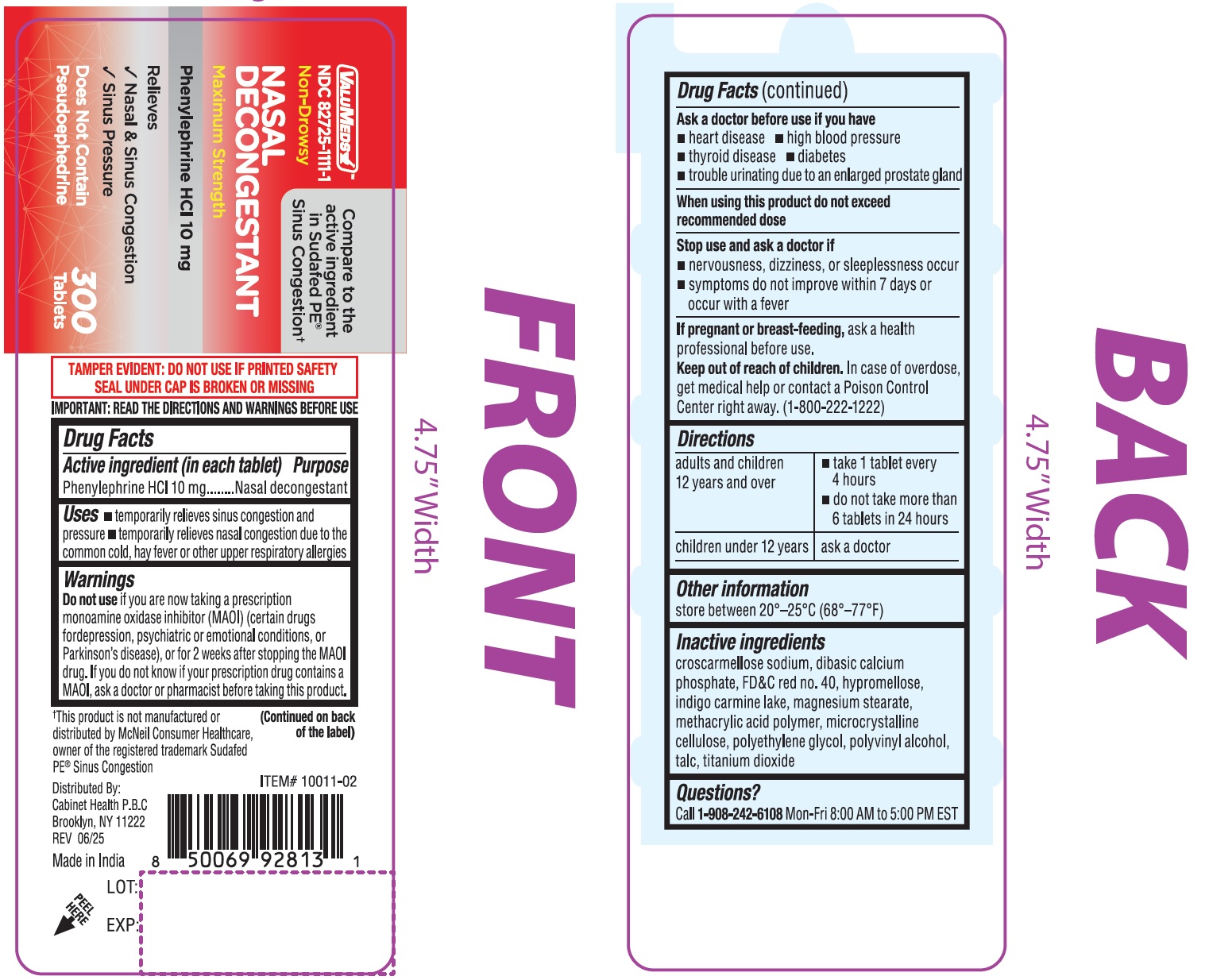
Valumeds Nasal Decongestant, Phenylephrine Hcl
21676b0c-6be2-464c-ae91-92e5f0d0087d
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Phenylephrine HCl 10 mg
Purpose
Nasal decongestant
Medication Information
Purpose
Nasal decongestant
Description
Phenylephrine HCl 10 mg
Uses
• temporarily relieves sinus congestion and pressure •temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
Warnings
Directions
| adults and children 12 years and over |
|
| children under 12 years | ask a doctor |
Do Not Use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs fordepression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains a MAOI, ask a doctor or pharmacist before taking this product.
Drug Facts
Other Information
store between 20°–25°C (68°–77°F)
Package Labeling:
Inactive Ingredients
croscarmellose sodium, dibasic calcium phosphate, FD&C red no. 40, hypromellose, indigo carmine lake, magnesium stearate, methacrylic acid polymer, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, talc, titanium dioxide
Questions Or Comments?
Call 1-908-242-6108Mon-Fri 8:00 AM to 5:00PM EST
When Using This Product
do not exceed recommended dose
Stop Use and Ask A Doctor If
- nervousness, dizziness, or sleeplessness occur
- symptoms do not improve within 7 days or occur with a fever
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Active Ingredient (in Each Tablet)
Phenylephrine HCl 10 mg
Ask A Doctor Before Use If You Have
• heart disease • high blood pressure • thyroid disease • diabetes • trouble urinating due to an enlarged prostate gland
Structured Label Content
Uses
• temporarily relieves sinus congestion and pressure •temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
Purpose
Nasal decongestant
Warnings
Directions
| adults and children 12 years and over |
|
| children under 12 years | ask a doctor |
Do Not Use (Do not use)
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs fordepression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains a MAOI, ask a doctor or pharmacist before taking this product.
Drug Facts
Other Information (Other information)
store between 20°–25°C (68°–77°F)
Package Labeling:
Inactive Ingredients (Inactive ingredients)
croscarmellose sodium, dibasic calcium phosphate, FD&C red no. 40, hypromellose, indigo carmine lake, magnesium stearate, methacrylic acid polymer, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, talc, titanium dioxide
Questions Or Comments? (Questions or comments?)
Call 1-908-242-6108Mon-Fri 8:00 AM to 5:00PM EST
When Using This Product (When using this product)
do not exceed recommended dose
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- nervousness, dizziness, or sleeplessness occur
- symptoms do not improve within 7 days or occur with a fever
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Active Ingredient (in Each Tablet) (Active Ingredient (in each tablet))
Phenylephrine HCl 10 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
• heart disease • high blood pressure • thyroid disease • diabetes • trouble urinating due to an enlarged prostate gland
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:55.623603 · Updated: 2026-03-14T23:11:54.825001