These Highlights Do Not Include All The Information Needed To Use Topamax ®
21628112-0c47-11df-95b3-498d55d89593
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
TOPAMAX is indicated for: Epilepsy: initial monotherapy for the treatment of partial-onset or primary generalized tonic-clonic seizures in patients 2 years of age and older ( 1.1 ); adjunctive therapy for the treatment of partial-onset seizures, primary generalized tonic-clonic seizures, or seizures associated with Lennox-Gastaut syndrome in patients 2 years of age and older ( 1.2 ) Preventive treatment of migraine in patients 12 years of age and older ( 1.3 )
Indications and Usage
TOPAMAX is indicated for: Epilepsy: initial monotherapy for the treatment of partial-onset or primary generalized tonic-clonic seizures in patients 2 years of age and older ( 1.1 ); adjunctive therapy for the treatment of partial-onset seizures, primary generalized tonic-clonic seizures, or seizures associated with Lennox-Gastaut syndrome in patients 2 years of age and older ( 1.2 ) Preventive treatment of migraine in patients 12 years of age and older ( 1.3 )
Dosage and Administration
TOPAMAX initial dose, titration, and recommended maintenance dose varies by indication and age group. See Full Prescribing Information for recommended dosage, and dosing considerations in patients with renal impairment, geriatric patients, and patients undergoing hemodialysis ( 2.1 , 2.2 , 2.3 , 2.4 , 2.5 , 2.6 )
Warnings and Precautions
Acute myopia and secondary angle closure glaucoma: can lead to permanent visual loss; discontinue TOPAMAX as soon as possible ( 5.1 ) Visual field defects: consider discontinuation of TOPAMAX ( 5.2 ) Oligohidrosis and hyperthermia: monitor decreased sweating and increased body temperature, especially in pediatric patients ( 5.3 ) Metabolic acidosis: baseline and periodic measurement of serum bicarbonate is recommended; consider dose reduction or discontinuation of TOPAMAX if clinically appropriate ( 5.4 ) Suicidal behavior and ideation: antiepileptic drugs increase the risk of suicidal behavior or ideation ( 5.5 ) Cognitive/neuropsychiatric adverse reactions: use caution when operating machinery including cars; depression and mood problems may occur ( 5.6 ) Fetal Toxicity: use during pregnancy can cause major congenital malformations, including but not limited to cleft lip and/or palate, and being small for gestational age ( 5.7 ) Withdrawal of AEDs: withdraw TOPAMAX gradually ( 5.8 ) Decrease in Bone Mineral Density: has been shown to decrease bone mineral density and bone mineral content in pediatric patients ( 5.9 ) Negative effects on growth (height and weight): may slow height increase and weight gain; carefully monitor children receiving prolonged therapy ( 5.10 ) Serious skin reactions: If SJS or TEN is suspected, discontinue TOPAMAX ( 5.11 ) Hyperammonemia/encephalopathy: measure ammonia if encephalopathic symptoms occur ( 5.12 ) Kidney stones: avoid use with other carbonic anhydrase inhibitors, drugs causing metabolic acidosis, or in patients on a ketogenic diet ( 5.13 ) Hypothermia has been reported with and without hyperammonemia during topiramate treatment with concomitant valproic acid use ( 5.14 )
Contraindications
None.
Adverse Reactions
TOPAMAX can cause cognitive/neuropsychiatric adverse reactions. The most frequent of these can be classified into three general categories: 1) Cognitive-related dysfunction (e.g., confusion, psychomotor slowing, difficulty with concentration/attention, difficulty with memory, speech or language problems, particularly word-finding difficulties); 2) Psychiatric/behavioral disturbances (e.g., depression or mood problems); and 3) Somnolence or fatigue.
Drug Interactions
Contraceptives: decreased contraceptive efficacy and increased breakthrough bleeding, especially at doses greater than 200 mg/day ( 7.4 ) Monitor lithium levels if lithium is used with high-dose TOPAMAX ( 7.7 )
Medication Information
Warnings and Precautions
Acute myopia and secondary angle closure glaucoma: can lead to permanent visual loss; discontinue TOPAMAX as soon as possible ( 5.1 ) Visual field defects: consider discontinuation of TOPAMAX ( 5.2 ) Oligohidrosis and hyperthermia: monitor decreased sweating and increased body temperature, especially in pediatric patients ( 5.3 ) Metabolic acidosis: baseline and periodic measurement of serum bicarbonate is recommended; consider dose reduction or discontinuation of TOPAMAX if clinically appropriate ( 5.4 ) Suicidal behavior and ideation: antiepileptic drugs increase the risk of suicidal behavior or ideation ( 5.5 ) Cognitive/neuropsychiatric adverse reactions: use caution when operating machinery including cars; depression and mood problems may occur ( 5.6 ) Fetal Toxicity: use during pregnancy can cause major congenital malformations, including but not limited to cleft lip and/or palate, and being small for gestational age ( 5.7 ) Withdrawal of AEDs: withdraw TOPAMAX gradually ( 5.8 ) Decrease in Bone Mineral Density: has been shown to decrease bone mineral density and bone mineral content in pediatric patients ( 5.9 ) Negative effects on growth (height and weight): may slow height increase and weight gain; carefully monitor children receiving prolonged therapy ( 5.10 ) Serious skin reactions: If SJS or TEN is suspected, discontinue TOPAMAX ( 5.11 ) Hyperammonemia/encephalopathy: measure ammonia if encephalopathic symptoms occur ( 5.12 ) Kidney stones: avoid use with other carbonic anhydrase inhibitors, drugs causing metabolic acidosis, or in patients on a ketogenic diet ( 5.13 ) Hypothermia has been reported with and without hyperammonemia during topiramate treatment with concomitant valproic acid use ( 5.14 )
Indications and Usage
TOPAMAX is indicated for: Epilepsy: initial monotherapy for the treatment of partial-onset or primary generalized tonic-clonic seizures in patients 2 years of age and older ( 1.1 ); adjunctive therapy for the treatment of partial-onset seizures, primary generalized tonic-clonic seizures, or seizures associated with Lennox-Gastaut syndrome in patients 2 years of age and older ( 1.2 ) Preventive treatment of migraine in patients 12 years of age and older ( 1.3 )
Dosage and Administration
TOPAMAX initial dose, titration, and recommended maintenance dose varies by indication and age group. See Full Prescribing Information for recommended dosage, and dosing considerations in patients with renal impairment, geriatric patients, and patients undergoing hemodialysis ( 2.1 , 2.2 , 2.3 , 2.4 , 2.5 , 2.6 )
Contraindications
None.
Adverse Reactions
TOPAMAX can cause cognitive/neuropsychiatric adverse reactions. The most frequent of these can be classified into three general categories: 1) Cognitive-related dysfunction (e.g., confusion, psychomotor slowing, difficulty with concentration/attention, difficulty with memory, speech or language problems, particularly word-finding difficulties); 2) Psychiatric/behavioral disturbances (e.g., depression or mood problems); and 3) Somnolence or fatigue.
Drug Interactions
Contraceptives: decreased contraceptive efficacy and increased breakthrough bleeding, especially at doses greater than 200 mg/day ( 7.4 ) Monitor lithium levels if lithium is used with high-dose TOPAMAX ( 7.7 )
Description
TOPAMAX is indicated for: Epilepsy: initial monotherapy for the treatment of partial-onset or primary generalized tonic-clonic seizures in patients 2 years of age and older ( 1.1 ); adjunctive therapy for the treatment of partial-onset seizures, primary generalized tonic-clonic seizures, or seizures associated with Lennox-Gastaut syndrome in patients 2 years of age and older ( 1.2 ) Preventive treatment of migraine in patients 12 years of age and older ( 1.3 )
Section 42229-5
Adults and Pediatric Patients 10 Years of Age and Older
The recommended dose for TOPAMAX monotherapy in adults and pediatric patients 10 years of age and older is 400 mg/day in two divided doses. The dose should be achieved by titration according to the following schedule (Table 1):
| Morning Dose | Evening Dose | |
|---|---|---|
| Week 1 | 25 mg | 25 mg |
| Week 2 | 50 mg | 50 mg |
| Week 3 | 75 mg | 75 mg |
| Week 4 | 100 mg | 100 mg |
| Week 5 | 150 mg | 150 mg |
| Week 6 | 200 mg | 200 mg |
Section 42231-1
| MEDICATION GUIDE
TOPAMAX ®(TOE-PA-MAX) (topiramate) TABLETS, for oral use TOPAMAX ®(TOE-PA-MAX) (topiramate capsules) SPRINKLE CAPSULES, for oral use |
||||
|---|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Revised: 3/2025 | |||
|
What is the most important information I should know about TOPAMAX?
TOPAMAX may cause eye problems. Serious eye problems include:
TOPAMAX can increase the level of acid in your blood (metabolic acidosis).If left untreated, metabolic acidosis can cause brittle or soft bones (osteoporosis, osteomalacia, osteopenia), kidney stones, can slow the rate of growth in children, and may possibly harm your baby if you are pregnant. Metabolic acidosis can happen with or without symptoms. Sometimes people with metabolic acidosis will: |
||||
|
|
|||
| Your healthcare provider should do a blood test to measure the level of acid in your blood before and during your treatment with TOPAMAX. If you are pregnant, you should talk to your healthcare provider about whether you have metabolic acidosis.
Like other antiepileptic drugs, TOPAMAX may cause suicidal thoughts or actions in a very small number of people, about 1 in 500. Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you: |
||||
|
|
|||
Do not stop TOPAMAX without first talking to a healthcare provider.
TOPAMAX may slow height increase and weight gain in children and adolescents when used over a long period. |
||||
|
What is TOPAMAX?
TOPAMAX is a prescription medicine used:
|
||||
Before taking TOPAMAX, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take:
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist each time you get a new medicine. Do not start a new medicine without talking with your healthcare provider. |
||||
How should I take TOPAMAX?
|
||||
What should I avoid while taking TOPAMAX?
|
||||
|
What are the possible side effects of TOPAMAX?
TOPAMAX may cause serious side effects including : See " What is the most important information I should know about TOPAMAX? "
The most common side effects of TOPAMAX include: |
||||
|
|
|
||
| Tell your healthcare provider about any side effect that bothers you or that does not go away. These are not all the possible side effects of TOPAMAX. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Janssen Pharmaceuticals, Inc. at 1-800-526-7736. | ||||
How should I store TOPAMAX?
|
||||
|
General information about the safe and effective use of TOPAMAX.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use TOPAMAX for a condition for which it was not prescribed. Do not give TOPAMAX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about TOPAMAX that is written for health professionals. |
||||
|
What are the ingredients in TOPAMAX?
Active ingredient:topiramate Inactive ingredients:
© Johnson & Johnson and its affiliates 2025 For more information, go to www.topamax.com or call 1-800-526-7736. |
7.7 Lithium
An increase in systemic exposure of lithium following TOPAMAX doses of up to 600 mg/day can occur. Lithium levels should be monitored when co-administered with high-dose TOPAMAX [see Clinical Pharmacology (12.3)] .
1.3 Migraine
TOPAMAX is indicated for the preventive treatment of migraine in patients 12 years of age and older.
10 Overdosage
Overdoses of TOPAMAX have been reported. Signs and symptoms included convulsions, drowsiness, speech disturbance, blurred vision, diplopia, impaired mentation, lethargy, abnormal coordination, stupor, hypotension, abdominal pain, agitation, dizziness and depression. The clinical consequences were not severe in most cases, but deaths have been reported after overdoses involving TOPAMAX.
TOPAMAX overdose has resulted in severe metabolic acidosis [see Warnings and Precautions (5.4)] .
A patient who ingested a dose of TOPAMAX between 96 and 110 g was admitted to a hospital with a coma lasting 20 to 24 hours followed by full recovery after 3 to 4 days.
In the event of overdose, TOPAMAX should be discontinued and general supportive treatment given until clinical toxicity has been diminished or resolved. Hemodialysis is an effective means of removing topiramate from the body.
11 Description
Topiramate is a sulfamate-substituted monosaccharide. TOPAMAX ®(topiramate) Tablets are available as 25 mg, 50 mg, 100 mg, and 200 mg round tablets for oral administration. TOPAMAX (topiramate capsules) Sprinkle Capsules are available as 15 mg and 25 mg sprinkle capsules for oral administration as whole capsules or opened and sprinkled onto soft food.
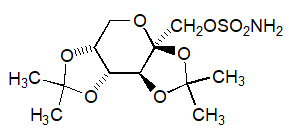
Topiramate is a white crystalline powder with a bitter taste. Topiramate is most soluble in alkaline solutions containing sodium hydroxide or sodium phosphate and having a pH of 9 to 10. It is freely soluble in acetone, chloroform, dimethylsulfoxide, and ethanol. The solubility in water is 9.8 mg/mL. Its saturated solution has a pH of 6.3. Topiramate has the molecular formula C 12H 21NO 8S and a molecular weight of 339.36. Topiramate is designated chemically as 2,3:4,5-Di- O-isopropylidene-β-D-fructopyranose sulfamate and has the following structural formula:
TOPAMAX Tablets contain the following inactive ingredients: carnauba wax, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, pregelatinized starch, purified water, sodium starch glycolate, synthetic iron oxide, and titanium dioxide.
TOPAMAX Sprinkle Capsules contain topiramate-coated beads in a hard gelatin capsule. The inactive ingredients are black pharmaceutical ink, cellulose acetate, gelatin, povidone, sodium lauryl sulfate, sorbitan monolaurate, sugar spheres (sucrose and starch) and titanium dioxide.
7.6 Pioglitazone
A decrease in the exposure of pioglitazone and its active metabolites were noted with the concurrent use of pioglitazone and TOPAMAX in a clinical trial. The clinical relevance of these observations is unknown; however, when TOPAMAX is added to pioglitazone therapy or pioglitazone is added to TOPAMAX therapy, careful attention should be given to the routine monitoring of patients for adequate control of their diabetic disease state [see Clinical Pharmacology (12.3)] .
7.8 Amitriptyline
Some patients may experience a large increase in amitriptyline concentration in the presence of TOPAMAX and any adjustments in amitriptyline dose should be made according to the patient's clinical response and not on the basis of plasma levels [see Clinical Pharmacology (12.3)] .
8.5 Geriatric Use
In clinical trials, 3% of patients were over age 60. No age-related differences in effectiveness or adverse effects were evident. However, clinical studies of topiramate did not include sufficient numbers of subjects age 65 and over to determine whether they respond differently than younger subjects. Dosage adjustment may be necessary for elderly with age-related renal impairment (creatinine clearance rate <70 mL/min/1.73 m 2) resulting in reduced clearance [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)] .
5.13 Kidney Stones
TOPAMAX increases the risk of kidney stones. During adjunctive epilepsy trials, the risk for kidney stones in TOPAMAX-treated adults was 1.5%, an incidence about 2 to 4 times greater than expected in a similar, untreated population. As in the general population, the incidence of stone formation among TOPAMAX-treated patients was higher in men. Kidney stones have also been reported in pediatric patients taking TOPAMAX for epilepsy or migraine. During long-term (up to 1 year) TOPAMAX treatment in an open-label extension study of 284 pediatric patients 1–24 months old with epilepsy, 7% developed kidney or bladder stones. TOPAMAX is not approved for treatment of epilepsy in pediatric patients less than 2 years old [see Use in Specific Populations (8.4)].
TOPAMAX is a carbonic anhydrase inhibitor. Carbonic anhydrase inhibitors can promote stone formation by reducing urinary citrate excretion and by increasing urinary pH [see Warnings and Precautions (5.4)] . The concomitant use of TOPAMAX with any other drug producing metabolic acidosis, or potentially in patients on a ketogenic diet, may create a physiological environment that increases the risk of kidney stone formation, and should therefore be avoided.
Increased fluid intake increases the urinary output, lowering the concentration of substances involved in stone formation. Hydration is recommended to reduce new stone formation.
An increase in urinary calcium and a marked decrease in urinary citrate was observed in TOPAMAX-treated pediatric patients in a one-year active-controlled study [see Use in Specific Populations (8.4)] . This increased ratio of urinary calcium/citrate increases the risk of kidney stones and/or nephrocalcinosis.
5.7 Fetal Toxicity
TOPAMAX can cause fetal harm when administered to a pregnant woman. Data from pregnancy registries indicate that infants exposed to topiramate in uterohave an increased risk of major congenital malformations, including but not limited to cleft lip and/or cleft palate (oral clefts), and of being small for gestational age (SGA). When multiple species of pregnant animals received topiramate at clinically relevant doses, structural malformations, including craniofacial defects, and reduced fetal weights occurred in offspring [see Use in Specific Populations (8.1)] .
Consider the benefits and the risks of TOPAMAX when administering this drug in women of childbearing potential, particularly when TOPAMAX is considered for a condition not usually associated with permanent injury or death [see Use in Specific Populations (8.1), Patient Counseling Information (17)] . TOPAMAX should be used during pregnancy only if the potential benefit outweighs the potential risk. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to a fetus [see Use in Specific Populations (8.1)] .
7.4 Contraceptives
The possibility of decreased contraceptive efficacy and increased breakthrough bleeding may occur in patients taking contraceptive products with TOPAMAX. Patients taking estrogen-containing or progestin-only contraceptives should be asked to report any change in their bleeding patterns. Contraceptive efficacy can be decreased even in the absence of breakthrough bleeding [see Clinical Pharmacology (12.3)].
14 Clinical Studies
The studies described in the following sections were conducted using TOPAMAX (topiramate) Tablets.
4 Contraindications
None.
6 Adverse Reactions
The following serious adverse reactions are discussed in more detail in other sections of the labeling:
- Acute Myopia and Secondary Angle Closure Glaucoma [see Warnings and Precautions (5.1)]
- Visual Field Defects [see Warnings and Precautions (5.2)]
- Oligohidrosis and Hyperthermia [see Warnings and Precautions (5.3)]
- Metabolic Acidosis [see Warnings and Precautions (5.4)]
- Suicidal Behavior and Ideation [see Warnings and Precautions (5.5)]
- Cognitive/Neuropsychiatric Adverse Reactions [see Warnings and Precautions (5.6)]
- Decrease of Bone Mineral Density [see Warnings and Precautions (5.9)]
- Negative Effects on Growth (Height and Weight) [see Warnings and Precautions (5.10)]
- Serious Skin Reactions [see Warnings and Precautions (5.11)]
- Hyperammonemia and Encephalopathy (Without and With Concomitant Valproic Acid [VPA] Use) [see Warnings and Precautions (5.12)]
- Kidney Stones [see Warnings and Precautions (5.13)]
- Hypothermia with Concomitant Valproic Acid (VPA) Use [see Warnings and Precautions (5.14)]
The data described in the following sections were obtained using TOPAMAX Tablets.
7 Drug Interactions
7.3 Cns Depressants
Concomitant administration of TOPAMAX and alcohol or other CNS depressant drugs has not been evaluated in clinical studies. Because of the potential of topiramate to cause CNS depression, as well as other cognitive and/or neuropsychiatric adverse reactions, TOPAMAX should be used with extreme caution if used in combination with alcohol and other CNS depressants.
8.6 Renal Impairment
The clearance of topiramate is reduced in patients with moderate (creatinine clearance 30 to 69 mL/min/1.73 m 2) and severe (creatinine clearance <30 mL/min/1.73 m 2) renal impairment. A dosage adjustment is recommended in patients with moderate or severe renal impairment [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)] .
12.2 Pharmacodynamics
Topiramate has anticonvulsant activity in rat and mouse maximal electroshock seizure (MES) tests. Topiramate is only weakly effective in blocking clonic seizures induced by the GABA Areceptor antagonist, pentylenetetrazole. Topiramate is also effective in rodent models of epilepsy, which include tonic and absence-like seizures in the spontaneous epileptic rat (SER) and tonic and clonic seizures induced in rats by kindling of the amygdala or by global ischemia.
Changes (increases and decreases) from baseline in vital signs (systolic blood pressure-SBP, diastolic blood pressure-DBP, pulse) occurred more frequently in pediatric patients (6 to 17 years) treated with various daily doses of topiramate (50 mg, 100 mg, 200 mg, 2 to 3 mg/kg) than in patients treated with placebo in controlled trials for the preventive treatment of migraine. The most notable changes were SBP <90 mm Hg, DBP <50 mm Hg, SBP or DBP increases or decreases ≥20 mm Hg, and pulse increases or decreases ≥30 beats per minute. These changes were often dose-related, and were most frequently associated with the greatest treatment difference at the 200 mg dose level. Systematic collection of orthostatic vital signs has not been conducted. The clinical significance of these various changes in vital signs has not been clearly established.
12.3 Pharmacokinetics
The sprinkle formulation is bioequivalent to the immediate-release tablet formulation and, therefore, may be substituted as a therapeutic equivalent.
Absorption of topiramate is rapid, with peak plasma concentrations occurring at approximately 2 hours following a 400 mg oral dose. The relative bioavailability of topiramate from the tablet formulation is about 80% compared to a solution. The bioavailability of topiramate is not affected by food.
The pharmacokinetics of topiramate are linear with dose proportional increases in plasma concentration over the dose range studied (200 to 800 mg/day). The mean plasma elimination half-life is 21 hours after single or multiple doses. Steady-state is thus reached in about 4 days in patients with normal renal function. Topiramate is 15% to 41% bound to human plasma proteins over the blood concentration range of 0.5 to 250 µg/mL. The fraction bound decreased as blood concentration increased.
Carbamazepine and phenytoin do not alter the binding of topiramate. Sodium valproate, at 500 µg/mL (a concentration 5 to 10 times higher than considered therapeutic for valproate) decreased the protein binding of topiramate from 23% to 13%. Topiramate does not influence the binding of sodium valproate.
5.4 Metabolic Acidosis
TOPAMAX can cause hyperchloremic, non-anion gap, metabolic acidosis (i.e., decreased serum bicarbonate below the normal reference range in the absence of chronic respiratory alkalosis). This metabolic acidosis is caused by renal bicarbonate loss due to carbonic anhydrase inhibition by TOPAMAX. TOPAMAX-induced metabolic acidosis can occur at any time during treatment. Bicarbonate decrements are usually mild-moderate (average decrease of 4 mEq/L at daily doses of 400 mg in adults and at approximately 6 mg/kg/day in pediatric patients); rarely, patients can experience severe decrements to values below 10 mEq/L. Conditions or therapies that predispose patients to acidosis (such as renal disease, severe respiratory disorders, status epilepticus, diarrhea, ketogenic diet, or specific drugs) may be additive to the bicarbonate lowering effects of TOPAMAX.
Metabolic acidosis was commonly observed in adult and pediatric patients treated with TOPAMAX in clinical trials. The incidence of decreased serum bicarbonate in pediatric trials, for adjunctive treatment of Lennox-Gastaut syndrome or refractory partial-onset seizures was as high as 67% for TOPAMAX (at approximately 6 mg/kg/day), and 10% for placebo. The incidence of a markedly abnormally low serum bicarbonate (i.e., absolute value < 17 mEq/L and ≥5 mEq/L decrease from pretreatment) in these trials was up to 11%, compared to ≤ 2% for placebo.
Manifestations of acute or chronic metabolic acidosis may include hyperventilation, nonspecific symptoms such as fatigue and anorexia, or more severe sequelae including cardiac arrhythmias or stupor. Chronic, untreated metabolic acidosis may increase the risk for nephrolithiasis or nephrocalcinosis, and may also result in osteomalacia (referred to as rickets in pediatric patients) and/or osteoporosis with an increased risk for fractures [see Warnings and Precautions (5.9, 5.13)] . A one-year, active-controlled study of pediatric patients treated with TOPAMAX demonstrated that TOPAMAX decreased lumbar spine bone mineral density and that this lumbar spine bone mineral density decrease was correlated (using change from baseline for lumbar spine Z score at final visit versus lowest post-treatment serum bicarbonate) with decreased serum bicarbonate, a reflection of metabolic acidosis [see Warnings and Precautions (5.9), Use in Specific Populations (8.4)] . Chronic metabolic acidosis in pediatric patients may also reduce growth rates, which may decrease the maximal height achieved. Long-term, open-label treatment of pediatric patients 1 to 24 months old with intractable partial epilepsy, for up to 1 year, showed reductions from baseline in length, weight, and head circumference compared to age and sex-matched normative data, although these patients with epilepsy are likely to have different growth rates than normal 1 to 24 month old pediatrics. Reductions in length and weight were correlated to the degree of acidosis [see Use in Specific Populations (8.4)] . TOPAMAX treatment that causes metabolic acidosis during pregnancy can possibly produce adverse effects on the fetus and might also cause metabolic acidosis in the neonate from possible transfer of topiramate to the fetus [see Warnings and Precautions (5.7), Use in Specific Populations (8.1)] .
1 Indications and Usage
TOPAMAX is indicated for:
- Epilepsy: initial monotherapy for the treatment of partial-onset or primary generalized tonic-clonic seizures in patients 2 years of age and older ( 1.1); adjunctive therapy for the treatment of partial-onset seizures, primary generalized tonic-clonic seizures, or seizures associated with Lennox-Gastaut syndrome in patients 2 years of age and older ( 1.2)
- Preventive treatment of migraine in patients 12 years of age and older ( 1.3)
7.1 Antiepileptic Drugs
Concomitant administration of phenytoin or carbamazepine with TOPAMAX resulted in a clinically significant decrease in plasma concentrations of topiramate when compared to TOPAMAX given alone. A dosage adjustment may be needed [see Dosage and Administration (2.1), Clinical Pharmacology (12.3)].
Concomitant administration of valproic acid and TOPAMAX has been associated with hypothermia and hyperammonemia with and without encephalopathy. Examine blood ammonia levels in patients in whom the onset of hypothermia has been reported [see Warnings and Precautions (5.12, 5.14), Clinical Pharmacology (12.3)] .
1.1 Monotherapy Epilepsy
TOPAMAX is indicated as initial monotherapy for the treatment of partial-onset or primary generalized tonic-clonic seizures in patients 2 years of age and older.
12.1 Mechanism of Action
The precise mechanisms by which topiramate exerts its anticonvulsant and preventive migraine effects are unknown; however, preclinical studies have revealed four properties that may contribute to topiramate's efficacy for epilepsy and the preventive treatment of migraine. Electrophysiological and biochemical evidence suggests that topiramate, at pharmacologically relevant concentrations, blocks voltage-dependent sodium channels, augments the activity of the neurotransmitter gamma-aminobutyrate at some subtypes of the GABA-A receptor, antagonizes the AMPA/kainate subtype of the glutamate receptor, and inhibits the carbonic anhydrase enzyme, particularly isozymes II and IV.
5.2 Visual Field Defects
Visual field defects (independent of elevated intraocular pressure) have been reported in clinical trials and in postmarketing experience in patients receiving topiramate. In clinical trials, most of these events were reversible after topiramate discontinuation. If visual problems occur at any time during topiramate treatment, consideration should be given to discontinuing the drug.
5 Warnings and Precautions
- Acute myopia and secondary angle closure glaucoma: can lead to permanent visual loss; discontinue TOPAMAX as soon as possible ( 5.1)
- Visual field defects: consider discontinuation of TOPAMAX ( 5.2)
- Oligohidrosis and hyperthermia: monitor decreased sweating and increased body temperature, especially in pediatric patients ( 5.3)
- Metabolic acidosis: baseline and periodic measurement of serum bicarbonate is recommended; consider dose reduction or discontinuation of TOPAMAX if clinically appropriate ( 5.4)
- Suicidal behavior and ideation: antiepileptic drugs increase the risk of suicidal behavior or ideation ( 5.5)
- Cognitive/neuropsychiatric adverse reactions: use caution when operating machinery including cars; depression and mood problems may occur ( 5.6)
- Fetal Toxicity: use during pregnancy can cause major congenital malformations, including but not limited to cleft lip and/or palate, and being small for gestational age ( 5.7)
- Withdrawal of AEDs: withdraw TOPAMAX gradually ( 5.8)
- Decrease in Bone Mineral Density: has been shown to decrease bone mineral density and bone mineral content in pediatric patients ( 5.9)
- Negative effects on growth (height and weight): may slow height increase and weight gain; carefully monitor children receiving prolonged therapy ( 5.10)
- Serious skin reactions: If SJS or TEN is suspected, discontinue TOPAMAX ( 5.11)
- Hyperammonemia/encephalopathy: measure ammonia if encephalopathic symptoms occur ( 5.12)
- Kidney stones: avoid use with other carbonic anhydrase inhibitors, drugs causing metabolic acidosis, or in patients on a ketogenic diet ( 5.13)
- Hypothermia has been reported with and without hyperammonemia during topiramate treatment with concomitant valproic acid use ( 5.14)
2 Dosage and Administration
TOPAMAX initial dose, titration, and recommended maintenance dose varies by indication and age group. See Full Prescribing Information for recommended dosage, and dosing considerations in patients with renal impairment, geriatric patients, and patients undergoing hemodialysis ( 2.1, 2.2, 2.3, 2.4, 2.5, 2.6)
5.11 Serious Skin Reactions
Serious skin reactions (Stevens-Johnson Syndrome [SJS] and Toxic Epidermal Necrolysis [TEN]) have been reported in patients receiving topiramate. TOPAMAX should be discontinued at the first sign of a rash, unless the rash is clearly not drug-related. If signs or symptoms suggest SJS/TEN, use of this drug should not be resumed and alternative therapy should be considered. Inform patients about the signs of serious skin reactions.
3 Dosage Forms and Strengths
TOPAMAX Tablets are available as debossed, coated, round tablets in the following strengths and colors:
25 mg cream (debossed "OMN" on one side; "25" on the other)
50 mg light-yellow (debossed "OMN" on one side; "50" on the other)
100 mg yellow (debossed "OMN" on one side; "100" on the other)
200 mg salmon (debossed "OMN" on one side; "200" on the other)
TOPAMAX Sprinkle Capsules contain small, white to off-white spheres. The gelatin capsules are white and clear.
They are marked as follows:
15 mg capsule with "TOP" and "15 mg" on the side
25 mg capsule with "TOP" and "25 mg" on the side
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of TOPAMAX. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as a Whole-General Disorders:oligohydrosis and hyperthermia [see Warnings and Precautions (5.3)] , hyperammonemia, hyperammonemic encephalopathy [see Warnings and Precautions (5.12)], hypothermia with concomitant valproic acid [see Warnings and Precautions (5.14)]
Gastrointestinal System Disorders:hepatic failure (including fatalities), hepatitis, pancreatitis
Skin and Appendage Disorders:bullous skin reactions (including erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis) [see Warnings and Precautions (5.11)] , pemphigus
Urinary System Disorders:kidney stones, nephrocalcinosis [see Warnings and Precautions (5.4, 5.13)]
Vision Disorders:acute myopia, secondary angle closure glaucoma [see Warnings and Precautions (5.1)] , maculopathy
Hematological Disorders:decrease of the International Normalized Ratio (INR) or prothrombin time when given concomitantly with vitamin K antagonist anticoagulant medications such as warfarin.
2.4 Administration Information
TOPAMAX can be taken without regard to meals.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the incidence of adverse reactions observed in the clinical trials of a drug cannot be directly compared to the incidence of adverse reactions in the clinical trials of another drug, and may not reflect the incidence of adverse reactions observed in practice.
7.5 Hydrochlorothiazide (hctz)
Topiramate C maxand AUC increased when HCTZ was added to TOPAMAX. The clinical significance of this change is unknown. The addition of HCTZ to TOPAMAX may require a decrease in the TOPAMAX dose [see Clinical Pharmacology (12.3)] .
1.2 Adjunctive Therapy Epilepsy
TOPAMAX is indicated as adjunctive therapy for the treatment of partial-onset seizures, primary generalized tonic-clonic seizures, and seizures associated with Lennox-Gastaut syndrome in patients 2 years of age and older.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.3 Oligohidrosis and Hyperthermia
Oligohidrosis (decreased sweating), infrequently resulting in hospitalization, has been reported in association with TOPAMAX use. Decreased sweating and an elevation in body temperature above normal characterized these cases. Some of the cases were reported after exposure to elevated environmental temperatures.
The majority of the reports have been in pediatric patients. Patients (especially pediatric patients) treated with TOPAMAX should be monitored closely for evidence of decreased sweating and increased body temperature, especially in hot weather. Caution should be used when TOPAMAX is given with other drugs that predispose patients to heat-related disorders; these drugs include, but are not limited to, other carbonic anhydrase inhibitors and drugs with anticholinergic activity.
5.5 Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including TOPAMAX, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% CI:1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence rate of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5 to 100 years) in the clinical trials analyzed.
Table 4 shows absolute and relative risk by indication for all evaluated AEDs.
| Indication | Placebo Patients with Events per 1000 Patients | Drug Patients with Events per 1000 Patients | Relative Risk:
Incidence of Events in Drug Patients/Incidence in Placebo Patients |
Risk Difference:
Additional Drug Patients with Events per 1000 Patients |
|---|---|---|---|---|
| Epilepsy | 1.0 | 3.4 | 3.5 | 2.4 |
| Psychiatric | 5.7 | 8.5 | 1.5 | 2.9 |
| Other | 1.0 | 1.8 | 1.9 | 0.9 |
| Total | 2.4 | 4.3 | 1.8 | 1.9 |
The relative risk for suicidal thoughts or behavior was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
Anyone considering prescribing TOPAMAX or any other AED must balance the risk of suicidal thoughts or behavior with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
5.9 Decrease in Bone Mineral Density
Results of a one-year active-controlled study in pediatric patients (N=63) demonstrated negative effects of TOPAMAX monotherapy on bone mineral acquisition via statistically significant decreases in bone mineral density (BMD) measured in lumbar spine and in total body less head [see Use in Specific Populations (8.4)] . Twenty-one percent of TOPAMAX-treated patients experienced clinically important reductions in BMD (Z score change from baseline of –0.5 or greater) compared to 0 patients in the control group. Although decreases in BMD occurred across all pediatric age subgroups, patients 6 to 9 years of age were most commonly affected. The sample size and study duration were too small to determine if fracture risk is increased. Decreased BMD in the lumbar spine was correlated with decreased serum bicarbonate, which commonly occurs with TOPAMAX treatment and reflects metabolic acidosis, a known cause of increased bone resorption [see Warnings and Precautions (5.4)] . Although small decreases in some markers of bone metabolism (e.g., serum alkaline phosphatase, calcium, phosphorus, and 1,25-dihydroxyvitamin D) occurred in TOPAMAX-treated patients, more significant decreases in serum parathyroid hormone and 25-hydroxyvitamin D, hormones involved in bone metabolism, were observed, along with an increased excretion of urinary calcium .
8.7 Patients Undergoing Hemodialysis
Topiramate is cleared by hemodialysis at a rate that is 4 to 6 times greater than in a normal individual. A dosage adjustment may be required [see Dosage and Administration (2.6), Clinical Pharmacology (12.3)].
5.8 Withdrawal of Antiepileptic Drugs
In patients with or without a history of seizures or epilepsy, antiepileptic drugs, including TOPAMAX, should be gradually withdrawn to minimize the potential for seizures or increased seizure frequency [see Clinical Studies (14)] . In situations where rapid withdrawal of TOPAMAX is medically required, appropriate monitoring is recommended.
7.2 Other Carbonic Anhydrase Inhibitors
Concomitant use of topiramate, a carbonic anhydrase inhibitor, with any other carbonic anhydrase inhibitor (e.g., zonisamide or acetazolamide) may increase the severity of metabolic acidosis and may also increase the risk of kidney stone formation. Therefore, patients given TOPAMAX concomitantly with another carbonic anhydrase inhibitor should be monitored particularly closely for the appearance or worsening of metabolic acidosis [see Clinical Pharmacology (12.3)] .
2.5 Dosing in Patients With Renal Impairment
In patients with renal impairment (creatinine clearance less than 70 mL/min/1.73 m 2), one-half of the usual adult dose of TOPAMAX is recommended [see Use in Specific Populations (8.5, 8.6), Clinical Pharmacology (12.3)] .
2.6 Dosing in Patients Undergoing Hemodialysis
To avoid rapid drops in topiramate plasma concentration during hemodialysis, a supplemental dose of TOPAMAX may be required. The actual adjustment should take into account 1) the duration of dialysis period, 2) the clearance rate of the dialysis system being used, and 3) the effective renal clearance of topiramate in the patient being dialyzed [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)] .
5.6 Cognitive/neuropsychiatric Adverse Reactions
TOPAMAX can cause cognitive/neuropsychiatric adverse reactions. The most frequent of these can be classified into three general categories: 1) Cognitive-related dysfunction (e.g., confusion, psychomotor slowing, difficulty with concentration/attention, difficulty with memory, speech or language problems, particularly word-finding difficulties); 2) Psychiatric/behavioral disturbances (e.g., depression or mood problems); and 3) Somnolence or fatigue.
2.3 Dosing for the Preventive Treatment of Migraine
The recommended total daily dose of TOPAMAX as treatment for patients 12 years of age and older for the preventive treatment of migraine is 100 mg/day administered in two divided doses (Table 3). The recommended titration rate for TOPAMAX for the preventive treatment of migraine is as follows:
| Morning Dose | Evening Dose | |
|---|---|---|
| Week 1 | None | 25 mg |
| Week 2 | 25 mg | 25 mg |
| Week 3 | 25 mg | 50 mg |
| Week 4 | 50 mg | 50 mg |
Dose and titration rate should be guided by clinical outcome. If required, longer intervals between dose adjustments can be used.
5.10 Negative Effects On Growth (height and Weight)
Results of a one-year active-controlled study of pediatric patients (N=63) demonstrated negative effects of TOPAMAX monotherapy on growth (i.e., height and weight) [see Use in Specific Populations (8.4)] . Although continued growth was observed in both treatment groups, the TOPAMAX group showed statistically significant reductions in mean annual change from baseline in body weight compared to the control group. A similar trend of attenuation in height velocity and height change from baseline was also observed in the TOPAMAX group compared to the control group. Negative effects on weight and height were seen across all TOPAMAX age subgroups. Growth (height and weight) of children receiving prolonged TOPAMAX therapy should be carefully monitored.
5.14 Hypothermia With Concomitant Valproic Acid Use
Hypothermia, defined as a drop in body core temperature to <35 °C (95 °F), has been reported in association with topiramate use with concomitant valproic acid both in conjunction with hyperammonemia and in the absence of hyperammonemia. This adverse reaction in patients using concomitant topiramate and valproate can occur after starting topiramate treatment or after increasing the daily dose of topiramate [see Drug Interactions (7.1)] . Consideration should be given to stopping TOPAMAX or valproate in patients who develop hypothermia, which may be manifested by a variety of clinical abnormalities including lethargy, confusion, coma, and significant alterations in other major organ systems such as the cardiovascular and respiratory systems. Clinical management and assessment should include examination of blood ammonia levels.
Principal Display Panel 25 Mg Tablet Bottle Label
NDC 50458-639-65
TopAMAX®
(topiramate) Tablets
25 mg
CAUTION: Verify Prescription Before Dispensing
Attention: Dispense the enclosed Medication
Guide to each patient.
Rx only
60 tablets
Johnson
&Johnson
Principal Display Panel 50 Mg Tablet Bottle Label
NDC 50458-640-65
TopAMAX®
(topiramate) Tablets
50 mg
CAUTION: Verify Prescription Before Dispensing
Attention: Dispense the enclosed Medication
Guide to each patient.
Rx only
60 tablets
Johnson
&Johnson
Principal Display Panel 100 Mg Tablet Bottle Label
NDC 50458-641-65
TopAMAX®
(topiramate) Tablets
100 mg
CAUTION: Verify Prescription Before Dispensing
Attention: Dispense the enclosed Medication
Guide to each patient.
Rx only
60 tablets
Johnson
&Johnson
Principal Display Panel 15 Mg Capsule Bottle Label
NDC 50458-647-65
Rx only
TopAMAX®
(topiramate)
Sprinkle Capsules
15 mg
Attention: Dispense the enclosed Medication
Guide to each patient.
60 capsules
Johnson
&Johnson
Principal Display Panel 200 Mg Tablet Bottle Label
NDC 50458-642-65
TopAMAX®
(topiramate) Tablets
200 mg
CAUTION: Verify Prescription Before Dispensing
Attention: Dispense the enclosed Medication
Guide to each patient.
Rx only
60 tablets
Johnson
&Johnson
Principal Display Panel 25 Mg Capsule Bottle Label
NDC 50458-645-65
Rx only
TopAMAX®
(topiramate)
Sprinkle Capsules
25 mg
Attention: Dispense the enclosed Medication
Guide to each patient.
60 capsules
Johnson
&Johnson
5.1 Acute Myopia and Secondary Angle Closure Glaucoma Syndrome
A syndrome consisting of acute myopia associated with secondary angle closure glaucoma has been reported in patients receiving TOPAMAX. Symptoms include acute onset of decreased visual acuity and/or ocular pain. Ophthalmologic findings can include some or all of the following: myopia, mydriasis, anterior chamber shallowing, ocular hyperemia (redness), choroidal detachments, retinal pigment epithelial detachments, macular striae, and increased intraocular pressure. This syndrome may be associated with supraciliary effusion resulting in anterior displacement of the lens and iris, with secondary angle closure glaucoma. Symptoms typically occur within 1 month of initiating TOPAMAX therapy. In contrast to primary narrow angle glaucoma, which is rare under 40 years of age, secondary angle closure glaucoma associated with topiramate has been reported in pediatric patients as well as adults. The primary treatment to reverse symptoms is discontinuation of TOPAMAX as rapidly as possible, according to the judgment of the treating physician. Other measures, in conjunction with discontinuation of TOPAMAX, may be helpful.
Elevated intraocular pressure of any etiology, if left untreated, can lead to serious sequelae including permanent vision loss.
5.12 Hyperammonemia and Encephalopathy (without and With Concomitant Valproic Acid Use)
Topiramate treatment can cause hyperammonemia with or without encephalopathy [see Adverse Reactions (6.2)] . The risk for hyperammonemia with topiramate appears dose-related. Hyperammonemia has been reported more frequently when topiramate is used concomitantly with valproic acid. Postmarketing cases of hyperammonemia with or without encephalopathy have been reported with topiramate and valproic acid in patients who previously tolerated either drug alone [see Drug Interactions (7.1)] .
Clinical symptoms of hyperammonemic encephalopathy often include acute alterations in level of consciousness and/or cognitive function with lethargy and/or vomiting. In most cases, hyperammonemic encephalopathy abated with discontinuation of treatment.
The incidence of hyperammonemia in pediatric patients 12 to 17 years of age in the preventive treatment of migraine trials was 26% in patients taking TOPAMAX monotherapy at 100 mg/day, and 14% in patients taking TOPAMAX at 50 mg/day, compared to 9% in patients taking placebo. There was also an increased incidence of markedly increased hyperammonemia at the 100 mg dose.
Dose-related hyperammonemia was also seen in pediatric patients 1 to 24 months of age treated with TOPAMAX and concomitant valproic acid for partial-onset epilepsy and this was not due to a pharmacokinetic interaction.
In some patients, hyperammonemia can be asymptomatic.
Structured Label Content
Section 42229-5 (42229-5)
Adults and Pediatric Patients 10 Years of Age and Older
The recommended dose for TOPAMAX monotherapy in adults and pediatric patients 10 years of age and older is 400 mg/day in two divided doses. The dose should be achieved by titration according to the following schedule (Table 1):
| Morning Dose | Evening Dose | |
|---|---|---|
| Week 1 | 25 mg | 25 mg |
| Week 2 | 50 mg | 50 mg |
| Week 3 | 75 mg | 75 mg |
| Week 4 | 100 mg | 100 mg |
| Week 5 | 150 mg | 150 mg |
| Week 6 | 200 mg | 200 mg |
Section 42231-1 (42231-1)
| MEDICATION GUIDE
TOPAMAX ®(TOE-PA-MAX) (topiramate) TABLETS, for oral use TOPAMAX ®(TOE-PA-MAX) (topiramate capsules) SPRINKLE CAPSULES, for oral use |
||||
|---|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Revised: 3/2025 | |||
|
What is the most important information I should know about TOPAMAX?
TOPAMAX may cause eye problems. Serious eye problems include:
TOPAMAX can increase the level of acid in your blood (metabolic acidosis).If left untreated, metabolic acidosis can cause brittle or soft bones (osteoporosis, osteomalacia, osteopenia), kidney stones, can slow the rate of growth in children, and may possibly harm your baby if you are pregnant. Metabolic acidosis can happen with or without symptoms. Sometimes people with metabolic acidosis will: |
||||
|
|
|||
| Your healthcare provider should do a blood test to measure the level of acid in your blood before and during your treatment with TOPAMAX. If you are pregnant, you should talk to your healthcare provider about whether you have metabolic acidosis.
Like other antiepileptic drugs, TOPAMAX may cause suicidal thoughts or actions in a very small number of people, about 1 in 500. Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you: |
||||
|
|
|||
Do not stop TOPAMAX without first talking to a healthcare provider.
TOPAMAX may slow height increase and weight gain in children and adolescents when used over a long period. |
||||
|
What is TOPAMAX?
TOPAMAX is a prescription medicine used:
|
||||
Before taking TOPAMAX, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take:
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist each time you get a new medicine. Do not start a new medicine without talking with your healthcare provider. |
||||
How should I take TOPAMAX?
|
||||
What should I avoid while taking TOPAMAX?
|
||||
|
What are the possible side effects of TOPAMAX?
TOPAMAX may cause serious side effects including : See " What is the most important information I should know about TOPAMAX? "
The most common side effects of TOPAMAX include: |
||||
|
|
|
||
| Tell your healthcare provider about any side effect that bothers you or that does not go away. These are not all the possible side effects of TOPAMAX. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Janssen Pharmaceuticals, Inc. at 1-800-526-7736. | ||||
How should I store TOPAMAX?
|
||||
|
General information about the safe and effective use of TOPAMAX.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use TOPAMAX for a condition for which it was not prescribed. Do not give TOPAMAX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about TOPAMAX that is written for health professionals. |
||||
|
What are the ingredients in TOPAMAX?
Active ingredient:topiramate Inactive ingredients:
© Johnson & Johnson and its affiliates 2025 For more information, go to www.topamax.com or call 1-800-526-7736. |
7.7 Lithium
An increase in systemic exposure of lithium following TOPAMAX doses of up to 600 mg/day can occur. Lithium levels should be monitored when co-administered with high-dose TOPAMAX [see Clinical Pharmacology (12.3)] .
1.3 Migraine
TOPAMAX is indicated for the preventive treatment of migraine in patients 12 years of age and older.
10 Overdosage (10 OVERDOSAGE)
Overdoses of TOPAMAX have been reported. Signs and symptoms included convulsions, drowsiness, speech disturbance, blurred vision, diplopia, impaired mentation, lethargy, abnormal coordination, stupor, hypotension, abdominal pain, agitation, dizziness and depression. The clinical consequences were not severe in most cases, but deaths have been reported after overdoses involving TOPAMAX.
TOPAMAX overdose has resulted in severe metabolic acidosis [see Warnings and Precautions (5.4)] .
A patient who ingested a dose of TOPAMAX between 96 and 110 g was admitted to a hospital with a coma lasting 20 to 24 hours followed by full recovery after 3 to 4 days.
In the event of overdose, TOPAMAX should be discontinued and general supportive treatment given until clinical toxicity has been diminished or resolved. Hemodialysis is an effective means of removing topiramate from the body.
11 Description (11 DESCRIPTION)
Topiramate is a sulfamate-substituted monosaccharide. TOPAMAX ®(topiramate) Tablets are available as 25 mg, 50 mg, 100 mg, and 200 mg round tablets for oral administration. TOPAMAX (topiramate capsules) Sprinkle Capsules are available as 15 mg and 25 mg sprinkle capsules for oral administration as whole capsules or opened and sprinkled onto soft food.
Topiramate is a white crystalline powder with a bitter taste. Topiramate is most soluble in alkaline solutions containing sodium hydroxide or sodium phosphate and having a pH of 9 to 10. It is freely soluble in acetone, chloroform, dimethylsulfoxide, and ethanol. The solubility in water is 9.8 mg/mL. Its saturated solution has a pH of 6.3. Topiramate has the molecular formula C 12H 21NO 8S and a molecular weight of 339.36. Topiramate is designated chemically as 2,3:4,5-Di- O-isopropylidene-β-D-fructopyranose sulfamate and has the following structural formula:
TOPAMAX Tablets contain the following inactive ingredients: carnauba wax, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, pregelatinized starch, purified water, sodium starch glycolate, synthetic iron oxide, and titanium dioxide.
TOPAMAX Sprinkle Capsules contain topiramate-coated beads in a hard gelatin capsule. The inactive ingredients are black pharmaceutical ink, cellulose acetate, gelatin, povidone, sodium lauryl sulfate, sorbitan monolaurate, sugar spheres (sucrose and starch) and titanium dioxide.
7.6 Pioglitazone
A decrease in the exposure of pioglitazone and its active metabolites were noted with the concurrent use of pioglitazone and TOPAMAX in a clinical trial. The clinical relevance of these observations is unknown; however, when TOPAMAX is added to pioglitazone therapy or pioglitazone is added to TOPAMAX therapy, careful attention should be given to the routine monitoring of patients for adequate control of their diabetic disease state [see Clinical Pharmacology (12.3)] .
7.8 Amitriptyline
Some patients may experience a large increase in amitriptyline concentration in the presence of TOPAMAX and any adjustments in amitriptyline dose should be made according to the patient's clinical response and not on the basis of plasma levels [see Clinical Pharmacology (12.3)] .
8.5 Geriatric Use
In clinical trials, 3% of patients were over age 60. No age-related differences in effectiveness or adverse effects were evident. However, clinical studies of topiramate did not include sufficient numbers of subjects age 65 and over to determine whether they respond differently than younger subjects. Dosage adjustment may be necessary for elderly with age-related renal impairment (creatinine clearance rate <70 mL/min/1.73 m 2) resulting in reduced clearance [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)] .
5.13 Kidney Stones
TOPAMAX increases the risk of kidney stones. During adjunctive epilepsy trials, the risk for kidney stones in TOPAMAX-treated adults was 1.5%, an incidence about 2 to 4 times greater than expected in a similar, untreated population. As in the general population, the incidence of stone formation among TOPAMAX-treated patients was higher in men. Kidney stones have also been reported in pediatric patients taking TOPAMAX for epilepsy or migraine. During long-term (up to 1 year) TOPAMAX treatment in an open-label extension study of 284 pediatric patients 1–24 months old with epilepsy, 7% developed kidney or bladder stones. TOPAMAX is not approved for treatment of epilepsy in pediatric patients less than 2 years old [see Use in Specific Populations (8.4)].
TOPAMAX is a carbonic anhydrase inhibitor. Carbonic anhydrase inhibitors can promote stone formation by reducing urinary citrate excretion and by increasing urinary pH [see Warnings and Precautions (5.4)] . The concomitant use of TOPAMAX with any other drug producing metabolic acidosis, or potentially in patients on a ketogenic diet, may create a physiological environment that increases the risk of kidney stone formation, and should therefore be avoided.
Increased fluid intake increases the urinary output, lowering the concentration of substances involved in stone formation. Hydration is recommended to reduce new stone formation.
An increase in urinary calcium and a marked decrease in urinary citrate was observed in TOPAMAX-treated pediatric patients in a one-year active-controlled study [see Use in Specific Populations (8.4)] . This increased ratio of urinary calcium/citrate increases the risk of kidney stones and/or nephrocalcinosis.
5.7 Fetal Toxicity
TOPAMAX can cause fetal harm when administered to a pregnant woman. Data from pregnancy registries indicate that infants exposed to topiramate in uterohave an increased risk of major congenital malformations, including but not limited to cleft lip and/or cleft palate (oral clefts), and of being small for gestational age (SGA). When multiple species of pregnant animals received topiramate at clinically relevant doses, structural malformations, including craniofacial defects, and reduced fetal weights occurred in offspring [see Use in Specific Populations (8.1)] .
Consider the benefits and the risks of TOPAMAX when administering this drug in women of childbearing potential, particularly when TOPAMAX is considered for a condition not usually associated with permanent injury or death [see Use in Specific Populations (8.1), Patient Counseling Information (17)] . TOPAMAX should be used during pregnancy only if the potential benefit outweighs the potential risk. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to a fetus [see Use in Specific Populations (8.1)] .
7.4 Contraceptives
The possibility of decreased contraceptive efficacy and increased breakthrough bleeding may occur in patients taking contraceptive products with TOPAMAX. Patients taking estrogen-containing or progestin-only contraceptives should be asked to report any change in their bleeding patterns. Contraceptive efficacy can be decreased even in the absence of breakthrough bleeding [see Clinical Pharmacology (12.3)].
14 Clinical Studies (14 CLINICAL STUDIES)
The studies described in the following sections were conducted using TOPAMAX (topiramate) Tablets.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in more detail in other sections of the labeling:
- Acute Myopia and Secondary Angle Closure Glaucoma [see Warnings and Precautions (5.1)]
- Visual Field Defects [see Warnings and Precautions (5.2)]
- Oligohidrosis and Hyperthermia [see Warnings and Precautions (5.3)]
- Metabolic Acidosis [see Warnings and Precautions (5.4)]
- Suicidal Behavior and Ideation [see Warnings and Precautions (5.5)]
- Cognitive/Neuropsychiatric Adverse Reactions [see Warnings and Precautions (5.6)]
- Decrease of Bone Mineral Density [see Warnings and Precautions (5.9)]
- Negative Effects on Growth (Height and Weight) [see Warnings and Precautions (5.10)]
- Serious Skin Reactions [see Warnings and Precautions (5.11)]
- Hyperammonemia and Encephalopathy (Without and With Concomitant Valproic Acid [VPA] Use) [see Warnings and Precautions (5.12)]
- Kidney Stones [see Warnings and Precautions (5.13)]
- Hypothermia with Concomitant Valproic Acid (VPA) Use [see Warnings and Precautions (5.14)]
The data described in the following sections were obtained using TOPAMAX Tablets.
7 Drug Interactions (7 DRUG INTERACTIONS)
7.3 Cns Depressants (7.3 CNS Depressants)
Concomitant administration of TOPAMAX and alcohol or other CNS depressant drugs has not been evaluated in clinical studies. Because of the potential of topiramate to cause CNS depression, as well as other cognitive and/or neuropsychiatric adverse reactions, TOPAMAX should be used with extreme caution if used in combination with alcohol and other CNS depressants.
8.6 Renal Impairment
The clearance of topiramate is reduced in patients with moderate (creatinine clearance 30 to 69 mL/min/1.73 m 2) and severe (creatinine clearance <30 mL/min/1.73 m 2) renal impairment. A dosage adjustment is recommended in patients with moderate or severe renal impairment [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)] .
12.2 Pharmacodynamics
Topiramate has anticonvulsant activity in rat and mouse maximal electroshock seizure (MES) tests. Topiramate is only weakly effective in blocking clonic seizures induced by the GABA Areceptor antagonist, pentylenetetrazole. Topiramate is also effective in rodent models of epilepsy, which include tonic and absence-like seizures in the spontaneous epileptic rat (SER) and tonic and clonic seizures induced in rats by kindling of the amygdala or by global ischemia.
Changes (increases and decreases) from baseline in vital signs (systolic blood pressure-SBP, diastolic blood pressure-DBP, pulse) occurred more frequently in pediatric patients (6 to 17 years) treated with various daily doses of topiramate (50 mg, 100 mg, 200 mg, 2 to 3 mg/kg) than in patients treated with placebo in controlled trials for the preventive treatment of migraine. The most notable changes were SBP <90 mm Hg, DBP <50 mm Hg, SBP or DBP increases or decreases ≥20 mm Hg, and pulse increases or decreases ≥30 beats per minute. These changes were often dose-related, and were most frequently associated with the greatest treatment difference at the 200 mg dose level. Systematic collection of orthostatic vital signs has not been conducted. The clinical significance of these various changes in vital signs has not been clearly established.
12.3 Pharmacokinetics
The sprinkle formulation is bioequivalent to the immediate-release tablet formulation and, therefore, may be substituted as a therapeutic equivalent.
Absorption of topiramate is rapid, with peak plasma concentrations occurring at approximately 2 hours following a 400 mg oral dose. The relative bioavailability of topiramate from the tablet formulation is about 80% compared to a solution. The bioavailability of topiramate is not affected by food.
The pharmacokinetics of topiramate are linear with dose proportional increases in plasma concentration over the dose range studied (200 to 800 mg/day). The mean plasma elimination half-life is 21 hours after single or multiple doses. Steady-state is thus reached in about 4 days in patients with normal renal function. Topiramate is 15% to 41% bound to human plasma proteins over the blood concentration range of 0.5 to 250 µg/mL. The fraction bound decreased as blood concentration increased.
Carbamazepine and phenytoin do not alter the binding of topiramate. Sodium valproate, at 500 µg/mL (a concentration 5 to 10 times higher than considered therapeutic for valproate) decreased the protein binding of topiramate from 23% to 13%. Topiramate does not influence the binding of sodium valproate.
5.4 Metabolic Acidosis
TOPAMAX can cause hyperchloremic, non-anion gap, metabolic acidosis (i.e., decreased serum bicarbonate below the normal reference range in the absence of chronic respiratory alkalosis). This metabolic acidosis is caused by renal bicarbonate loss due to carbonic anhydrase inhibition by TOPAMAX. TOPAMAX-induced metabolic acidosis can occur at any time during treatment. Bicarbonate decrements are usually mild-moderate (average decrease of 4 mEq/L at daily doses of 400 mg in adults and at approximately 6 mg/kg/day in pediatric patients); rarely, patients can experience severe decrements to values below 10 mEq/L. Conditions or therapies that predispose patients to acidosis (such as renal disease, severe respiratory disorders, status epilepticus, diarrhea, ketogenic diet, or specific drugs) may be additive to the bicarbonate lowering effects of TOPAMAX.
Metabolic acidosis was commonly observed in adult and pediatric patients treated with TOPAMAX in clinical trials. The incidence of decreased serum bicarbonate in pediatric trials, for adjunctive treatment of Lennox-Gastaut syndrome or refractory partial-onset seizures was as high as 67% for TOPAMAX (at approximately 6 mg/kg/day), and 10% for placebo. The incidence of a markedly abnormally low serum bicarbonate (i.e., absolute value < 17 mEq/L and ≥5 mEq/L decrease from pretreatment) in these trials was up to 11%, compared to ≤ 2% for placebo.
Manifestations of acute or chronic metabolic acidosis may include hyperventilation, nonspecific symptoms such as fatigue and anorexia, or more severe sequelae including cardiac arrhythmias or stupor. Chronic, untreated metabolic acidosis may increase the risk for nephrolithiasis or nephrocalcinosis, and may also result in osteomalacia (referred to as rickets in pediatric patients) and/or osteoporosis with an increased risk for fractures [see Warnings and Precautions (5.9, 5.13)] . A one-year, active-controlled study of pediatric patients treated with TOPAMAX demonstrated that TOPAMAX decreased lumbar spine bone mineral density and that this lumbar spine bone mineral density decrease was correlated (using change from baseline for lumbar spine Z score at final visit versus lowest post-treatment serum bicarbonate) with decreased serum bicarbonate, a reflection of metabolic acidosis [see Warnings and Precautions (5.9), Use in Specific Populations (8.4)] . Chronic metabolic acidosis in pediatric patients may also reduce growth rates, which may decrease the maximal height achieved. Long-term, open-label treatment of pediatric patients 1 to 24 months old with intractable partial epilepsy, for up to 1 year, showed reductions from baseline in length, weight, and head circumference compared to age and sex-matched normative data, although these patients with epilepsy are likely to have different growth rates than normal 1 to 24 month old pediatrics. Reductions in length and weight were correlated to the degree of acidosis [see Use in Specific Populations (8.4)] . TOPAMAX treatment that causes metabolic acidosis during pregnancy can possibly produce adverse effects on the fetus and might also cause metabolic acidosis in the neonate from possible transfer of topiramate to the fetus [see Warnings and Precautions (5.7), Use in Specific Populations (8.1)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
TOPAMAX is indicated for:
- Epilepsy: initial monotherapy for the treatment of partial-onset or primary generalized tonic-clonic seizures in patients 2 years of age and older ( 1.1); adjunctive therapy for the treatment of partial-onset seizures, primary generalized tonic-clonic seizures, or seizures associated with Lennox-Gastaut syndrome in patients 2 years of age and older ( 1.2)
- Preventive treatment of migraine in patients 12 years of age and older ( 1.3)
7.1 Antiepileptic Drugs
Concomitant administration of phenytoin or carbamazepine with TOPAMAX resulted in a clinically significant decrease in plasma concentrations of topiramate when compared to TOPAMAX given alone. A dosage adjustment may be needed [see Dosage and Administration (2.1), Clinical Pharmacology (12.3)].
Concomitant administration of valproic acid and TOPAMAX has been associated with hypothermia and hyperammonemia with and without encephalopathy. Examine blood ammonia levels in patients in whom the onset of hypothermia has been reported [see Warnings and Precautions (5.12, 5.14), Clinical Pharmacology (12.3)] .
1.1 Monotherapy Epilepsy
TOPAMAX is indicated as initial monotherapy for the treatment of partial-onset or primary generalized tonic-clonic seizures in patients 2 years of age and older.
12.1 Mechanism of Action
The precise mechanisms by which topiramate exerts its anticonvulsant and preventive migraine effects are unknown; however, preclinical studies have revealed four properties that may contribute to topiramate's efficacy for epilepsy and the preventive treatment of migraine. Electrophysiological and biochemical evidence suggests that topiramate, at pharmacologically relevant concentrations, blocks voltage-dependent sodium channels, augments the activity of the neurotransmitter gamma-aminobutyrate at some subtypes of the GABA-A receptor, antagonizes the AMPA/kainate subtype of the glutamate receptor, and inhibits the carbonic anhydrase enzyme, particularly isozymes II and IV.
5.2 Visual Field Defects
Visual field defects (independent of elevated intraocular pressure) have been reported in clinical trials and in postmarketing experience in patients receiving topiramate. In clinical trials, most of these events were reversible after topiramate discontinuation. If visual problems occur at any time during topiramate treatment, consideration should be given to discontinuing the drug.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Acute myopia and secondary angle closure glaucoma: can lead to permanent visual loss; discontinue TOPAMAX as soon as possible ( 5.1)
- Visual field defects: consider discontinuation of TOPAMAX ( 5.2)
- Oligohidrosis and hyperthermia: monitor decreased sweating and increased body temperature, especially in pediatric patients ( 5.3)
- Metabolic acidosis: baseline and periodic measurement of serum bicarbonate is recommended; consider dose reduction or discontinuation of TOPAMAX if clinically appropriate ( 5.4)
- Suicidal behavior and ideation: antiepileptic drugs increase the risk of suicidal behavior or ideation ( 5.5)
- Cognitive/neuropsychiatric adverse reactions: use caution when operating machinery including cars; depression and mood problems may occur ( 5.6)
- Fetal Toxicity: use during pregnancy can cause major congenital malformations, including but not limited to cleft lip and/or palate, and being small for gestational age ( 5.7)
- Withdrawal of AEDs: withdraw TOPAMAX gradually ( 5.8)
- Decrease in Bone Mineral Density: has been shown to decrease bone mineral density and bone mineral content in pediatric patients ( 5.9)
- Negative effects on growth (height and weight): may slow height increase and weight gain; carefully monitor children receiving prolonged therapy ( 5.10)
- Serious skin reactions: If SJS or TEN is suspected, discontinue TOPAMAX ( 5.11)
- Hyperammonemia/encephalopathy: measure ammonia if encephalopathic symptoms occur ( 5.12)
- Kidney stones: avoid use with other carbonic anhydrase inhibitors, drugs causing metabolic acidosis, or in patients on a ketogenic diet ( 5.13)
- Hypothermia has been reported with and without hyperammonemia during topiramate treatment with concomitant valproic acid use ( 5.14)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
TOPAMAX initial dose, titration, and recommended maintenance dose varies by indication and age group. See Full Prescribing Information for recommended dosage, and dosing considerations in patients with renal impairment, geriatric patients, and patients undergoing hemodialysis ( 2.1, 2.2, 2.3, 2.4, 2.5, 2.6)
5.11 Serious Skin Reactions
Serious skin reactions (Stevens-Johnson Syndrome [SJS] and Toxic Epidermal Necrolysis [TEN]) have been reported in patients receiving topiramate. TOPAMAX should be discontinued at the first sign of a rash, unless the rash is clearly not drug-related. If signs or symptoms suggest SJS/TEN, use of this drug should not be resumed and alternative therapy should be considered. Inform patients about the signs of serious skin reactions.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
TOPAMAX Tablets are available as debossed, coated, round tablets in the following strengths and colors:
25 mg cream (debossed "OMN" on one side; "25" on the other)
50 mg light-yellow (debossed "OMN" on one side; "50" on the other)
100 mg yellow (debossed "OMN" on one side; "100" on the other)
200 mg salmon (debossed "OMN" on one side; "200" on the other)
TOPAMAX Sprinkle Capsules contain small, white to off-white spheres. The gelatin capsules are white and clear.
They are marked as follows:
15 mg capsule with "TOP" and "15 mg" on the side
25 mg capsule with "TOP" and "25 mg" on the side
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of TOPAMAX. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as a Whole-General Disorders:oligohydrosis and hyperthermia [see Warnings and Precautions (5.3)] , hyperammonemia, hyperammonemic encephalopathy [see Warnings and Precautions (5.12)], hypothermia with concomitant valproic acid [see Warnings and Precautions (5.14)]
Gastrointestinal System Disorders:hepatic failure (including fatalities), hepatitis, pancreatitis
Skin and Appendage Disorders:bullous skin reactions (including erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis) [see Warnings and Precautions (5.11)] , pemphigus
Urinary System Disorders:kidney stones, nephrocalcinosis [see Warnings and Precautions (5.4, 5.13)]
Vision Disorders:acute myopia, secondary angle closure glaucoma [see Warnings and Precautions (5.1)] , maculopathy
Hematological Disorders:decrease of the International Normalized Ratio (INR) or prothrombin time when given concomitantly with vitamin K antagonist anticoagulant medications such as warfarin.
2.4 Administration Information
TOPAMAX can be taken without regard to meals.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the incidence of adverse reactions observed in the clinical trials of a drug cannot be directly compared to the incidence of adverse reactions in the clinical trials of another drug, and may not reflect the incidence of adverse reactions observed in practice.
7.5 Hydrochlorothiazide (hctz) (7.5 Hydrochlorothiazide (HCTZ))
Topiramate C maxand AUC increased when HCTZ was added to TOPAMAX. The clinical significance of this change is unknown. The addition of HCTZ to TOPAMAX may require a decrease in the TOPAMAX dose [see Clinical Pharmacology (12.3)] .
1.2 Adjunctive Therapy Epilepsy
TOPAMAX is indicated as adjunctive therapy for the treatment of partial-onset seizures, primary generalized tonic-clonic seizures, and seizures associated with Lennox-Gastaut syndrome in patients 2 years of age and older.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.3 Oligohidrosis and Hyperthermia
Oligohidrosis (decreased sweating), infrequently resulting in hospitalization, has been reported in association with TOPAMAX use. Decreased sweating and an elevation in body temperature above normal characterized these cases. Some of the cases were reported after exposure to elevated environmental temperatures.
The majority of the reports have been in pediatric patients. Patients (especially pediatric patients) treated with TOPAMAX should be monitored closely for evidence of decreased sweating and increased body temperature, especially in hot weather. Caution should be used when TOPAMAX is given with other drugs that predispose patients to heat-related disorders; these drugs include, but are not limited to, other carbonic anhydrase inhibitors and drugs with anticholinergic activity.
5.5 Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including TOPAMAX, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% CI:1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence rate of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5 to 100 years) in the clinical trials analyzed.
Table 4 shows absolute and relative risk by indication for all evaluated AEDs.
| Indication | Placebo Patients with Events per 1000 Patients | Drug Patients with Events per 1000 Patients | Relative Risk:
Incidence of Events in Drug Patients/Incidence in Placebo Patients |
Risk Difference:
Additional Drug Patients with Events per 1000 Patients |
|---|---|---|---|---|
| Epilepsy | 1.0 | 3.4 | 3.5 | 2.4 |
| Psychiatric | 5.7 | 8.5 | 1.5 | 2.9 |
| Other | 1.0 | 1.8 | 1.9 | 0.9 |
| Total | 2.4 | 4.3 | 1.8 | 1.9 |
The relative risk for suicidal thoughts or behavior was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
Anyone considering prescribing TOPAMAX or any other AED must balance the risk of suicidal thoughts or behavior with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
5.9 Decrease in Bone Mineral Density
Results of a one-year active-controlled study in pediatric patients (N=63) demonstrated negative effects of TOPAMAX monotherapy on bone mineral acquisition via statistically significant decreases in bone mineral density (BMD) measured in lumbar spine and in total body less head [see Use in Specific Populations (8.4)] . Twenty-one percent of TOPAMAX-treated patients experienced clinically important reductions in BMD (Z score change from baseline of –0.5 or greater) compared to 0 patients in the control group. Although decreases in BMD occurred across all pediatric age subgroups, patients 6 to 9 years of age were most commonly affected. The sample size and study duration were too small to determine if fracture risk is increased. Decreased BMD in the lumbar spine was correlated with decreased serum bicarbonate, which commonly occurs with TOPAMAX treatment and reflects metabolic acidosis, a known cause of increased bone resorption [see Warnings and Precautions (5.4)] . Although small decreases in some markers of bone metabolism (e.g., serum alkaline phosphatase, calcium, phosphorus, and 1,25-dihydroxyvitamin D) occurred in TOPAMAX-treated patients, more significant decreases in serum parathyroid hormone and 25-hydroxyvitamin D, hormones involved in bone metabolism, were observed, along with an increased excretion of urinary calcium .
8.7 Patients Undergoing Hemodialysis
Topiramate is cleared by hemodialysis at a rate that is 4 to 6 times greater than in a normal individual. A dosage adjustment may be required [see Dosage and Administration (2.6), Clinical Pharmacology (12.3)].
5.8 Withdrawal of Antiepileptic Drugs
In patients with or without a history of seizures or epilepsy, antiepileptic drugs, including TOPAMAX, should be gradually withdrawn to minimize the potential for seizures or increased seizure frequency [see Clinical Studies (14)] . In situations where rapid withdrawal of TOPAMAX is medically required, appropriate monitoring is recommended.
7.2 Other Carbonic Anhydrase Inhibitors
Concomitant use of topiramate, a carbonic anhydrase inhibitor, with any other carbonic anhydrase inhibitor (e.g., zonisamide or acetazolamide) may increase the severity of metabolic acidosis and may also increase the risk of kidney stone formation. Therefore, patients given TOPAMAX concomitantly with another carbonic anhydrase inhibitor should be monitored particularly closely for the appearance or worsening of metabolic acidosis [see Clinical Pharmacology (12.3)] .
2.5 Dosing in Patients With Renal Impairment (2.5 Dosing in Patients with Renal Impairment)
In patients with renal impairment (creatinine clearance less than 70 mL/min/1.73 m 2), one-half of the usual adult dose of TOPAMAX is recommended [see Use in Specific Populations (8.5, 8.6), Clinical Pharmacology (12.3)] .
2.6 Dosing in Patients Undergoing Hemodialysis
To avoid rapid drops in topiramate plasma concentration during hemodialysis, a supplemental dose of TOPAMAX may be required. The actual adjustment should take into account 1) the duration of dialysis period, 2) the clearance rate of the dialysis system being used, and 3) the effective renal clearance of topiramate in the patient being dialyzed [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)] .
5.6 Cognitive/neuropsychiatric Adverse Reactions (5.6 Cognitive/Neuropsychiatric Adverse Reactions)
TOPAMAX can cause cognitive/neuropsychiatric adverse reactions. The most frequent of these can be classified into three general categories: 1) Cognitive-related dysfunction (e.g., confusion, psychomotor slowing, difficulty with concentration/attention, difficulty with memory, speech or language problems, particularly word-finding difficulties); 2) Psychiatric/behavioral disturbances (e.g., depression or mood problems); and 3) Somnolence or fatigue.
2.3 Dosing for the Preventive Treatment of Migraine
The recommended total daily dose of TOPAMAX as treatment for patients 12 years of age and older for the preventive treatment of migraine is 100 mg/day administered in two divided doses (Table 3). The recommended titration rate for TOPAMAX for the preventive treatment of migraine is as follows:
| Morning Dose | Evening Dose | |
|---|---|---|
| Week 1 | None | 25 mg |
| Week 2 | 25 mg | 25 mg |
| Week 3 | 25 mg | 50 mg |
| Week 4 | 50 mg | 50 mg |
Dose and titration rate should be guided by clinical outcome. If required, longer intervals between dose adjustments can be used.
5.10 Negative Effects On Growth (height and Weight) (5.10 Negative Effects on Growth (Height and Weight))
Results of a one-year active-controlled study of pediatric patients (N=63) demonstrated negative effects of TOPAMAX monotherapy on growth (i.e., height and weight) [see Use in Specific Populations (8.4)] . Although continued growth was observed in both treatment groups, the TOPAMAX group showed statistically significant reductions in mean annual change from baseline in body weight compared to the control group. A similar trend of attenuation in height velocity and height change from baseline was also observed in the TOPAMAX group compared to the control group. Negative effects on weight and height were seen across all TOPAMAX age subgroups. Growth (height and weight) of children receiving prolonged TOPAMAX therapy should be carefully monitored.
5.14 Hypothermia With Concomitant Valproic Acid Use (5.14 Hypothermia with Concomitant Valproic Acid Use)
Hypothermia, defined as a drop in body core temperature to <35 °C (95 °F), has been reported in association with topiramate use with concomitant valproic acid both in conjunction with hyperammonemia and in the absence of hyperammonemia. This adverse reaction in patients using concomitant topiramate and valproate can occur after starting topiramate treatment or after increasing the daily dose of topiramate [see Drug Interactions (7.1)] . Consideration should be given to stopping TOPAMAX or valproate in patients who develop hypothermia, which may be manifested by a variety of clinical abnormalities including lethargy, confusion, coma, and significant alterations in other major organ systems such as the cardiovascular and respiratory systems. Clinical management and assessment should include examination of blood ammonia levels.
Principal Display Panel 25 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Label)
NDC 50458-639-65
TopAMAX®
(topiramate) Tablets
25 mg
CAUTION: Verify Prescription Before Dispensing
Attention: Dispense the enclosed Medication
Guide to each patient.
Rx only
60 tablets
Johnson
&Johnson
Principal Display Panel 50 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 50 mg Tablet Bottle Label)
NDC 50458-640-65
TopAMAX®
(topiramate) Tablets
50 mg
CAUTION: Verify Prescription Before Dispensing
Attention: Dispense the enclosed Medication
Guide to each patient.
Rx only
60 tablets
Johnson
&Johnson
Principal Display Panel 100 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Label)
NDC 50458-641-65
TopAMAX®
(topiramate) Tablets
100 mg
CAUTION: Verify Prescription Before Dispensing
Attention: Dispense the enclosed Medication
Guide to each patient.
Rx only
60 tablets
Johnson
&Johnson
Principal Display Panel 15 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 15 mg Capsule Bottle Label)
NDC 50458-647-65
Rx only
TopAMAX®
(topiramate)
Sprinkle Capsules
15 mg
Attention: Dispense the enclosed Medication
Guide to each patient.
60 capsules
Johnson
&Johnson
Principal Display Panel 200 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 200 mg Tablet Bottle Label)
NDC 50458-642-65
TopAMAX®
(topiramate) Tablets
200 mg
CAUTION: Verify Prescription Before Dispensing
Attention: Dispense the enclosed Medication
Guide to each patient.
Rx only
60 tablets
Johnson
&Johnson
Principal Display Panel 25 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 25 mg Capsule Bottle Label)
NDC 50458-645-65
Rx only
TopAMAX®
(topiramate)
Sprinkle Capsules
25 mg
Attention: Dispense the enclosed Medication
Guide to each patient.
60 capsules
Johnson
&Johnson
5.1 Acute Myopia and Secondary Angle Closure Glaucoma Syndrome
A syndrome consisting of acute myopia associated with secondary angle closure glaucoma has been reported in patients receiving TOPAMAX. Symptoms include acute onset of decreased visual acuity and/or ocular pain. Ophthalmologic findings can include some or all of the following: myopia, mydriasis, anterior chamber shallowing, ocular hyperemia (redness), choroidal detachments, retinal pigment epithelial detachments, macular striae, and increased intraocular pressure. This syndrome may be associated with supraciliary effusion resulting in anterior displacement of the lens and iris, with secondary angle closure glaucoma. Symptoms typically occur within 1 month of initiating TOPAMAX therapy. In contrast to primary narrow angle glaucoma, which is rare under 40 years of age, secondary angle closure glaucoma associated with topiramate has been reported in pediatric patients as well as adults. The primary treatment to reverse symptoms is discontinuation of TOPAMAX as rapidly as possible, according to the judgment of the treating physician. Other measures, in conjunction with discontinuation of TOPAMAX, may be helpful.
Elevated intraocular pressure of any etiology, if left untreated, can lead to serious sequelae including permanent vision loss.
5.12 Hyperammonemia and Encephalopathy (without and With Concomitant Valproic Acid Use) (5.12 Hyperammonemia and Encephalopathy (Without and With Concomitant Valproic Acid Use))
Topiramate treatment can cause hyperammonemia with or without encephalopathy [see Adverse Reactions (6.2)] . The risk for hyperammonemia with topiramate appears dose-related. Hyperammonemia has been reported more frequently when topiramate is used concomitantly with valproic acid. Postmarketing cases of hyperammonemia with or without encephalopathy have been reported with topiramate and valproic acid in patients who previously tolerated either drug alone [see Drug Interactions (7.1)] .
Clinical symptoms of hyperammonemic encephalopathy often include acute alterations in level of consciousness and/or cognitive function with lethargy and/or vomiting. In most cases, hyperammonemic encephalopathy abated with discontinuation of treatment.
The incidence of hyperammonemia in pediatric patients 12 to 17 years of age in the preventive treatment of migraine trials was 26% in patients taking TOPAMAX monotherapy at 100 mg/day, and 14% in patients taking TOPAMAX at 50 mg/day, compared to 9% in patients taking placebo. There was also an increased incidence of markedly increased hyperammonemia at the 100 mg dose.
Dose-related hyperammonemia was also seen in pediatric patients 1 to 24 months of age treated with TOPAMAX and concomitant valproic acid for partial-onset epilepsy and this was not due to a pharmacokinetic interaction.
In some patients, hyperammonemia can be asymptomatic.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:17.403122 · Updated: 2026-03-14T22:50:26.086884