2105d1c1-54ef-4d1a-a63c-b1619e8b50db
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Magnesium Chloride Injection is a sterile solution of Magnesium Chloride Hexahydrate in Water for Injection q.s. Each mL contains Magnesium Chloride Hexahydrate 200 mg, Sodium Chloride 9 mg, Benzyl Alcohol 1% as a preservative, Water for Injection, q.s. pH adjusted with Hydrochloric Acid and/or Sodium Hydroxide. Total osmolarity equivalent to 2.951 mOsm/mL. Contains 1.97 mEq of Mg ++ and Cl - per mL. The structural formula is MgCl 2 •6H 2 O
Dosage and Administration
For intravenous infusion: 4 grams in 250 mL of 5% Dextrose Injection, at a rate not exceeding 3 mL per minute. Serum magnesium levels should serve as a guide to continued dosage.
Contraindications
Magnesium Chloride Injection should not be administered if there is renal impairment, marked myocardial disease or to comatose patients.
Adverse Reactions
Flushing, sweating, sharply lowered blood pressure, hypothermia, stupor and ultimately respiratory depression.
How Supplied
Magnesium Chloride Injection 200 mg/mL (20% w/v). NDC 67457-134-50 50 mL Multiple-Dose Vial. Individually boxed. Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Manufactured for: Mylan Institutional LLC Morgantown, WV 26505 Manufactured by: Mylan Institutional Galway, Ireland 1084L101 Revised: 10/2021 MI:MAGNIJ:R4
Medication Information
Dosage and Administration
For intravenous infusion: 4 grams in 250 mL of 5% Dextrose Injection, at a rate not exceeding 3 mL per minute. Serum magnesium levels should serve as a guide to continued dosage.
Contraindications
Magnesium Chloride Injection should not be administered if there is renal impairment, marked myocardial disease or to comatose patients.
Adverse Reactions
Flushing, sweating, sharply lowered blood pressure, hypothermia, stupor and ultimately respiratory depression.
How Supplied
Magnesium Chloride Injection 200 mg/mL (20% w/v).
NDC 67457-134-50
50 mL Multiple-Dose Vial. Individually boxed.
Store at 20° to 25°C (68° to 77°F). [See USP Controlled
Room Temperature.]
Manufactured for:
Mylan Institutional LLC
Morgantown, WV 26505
Manufactured by:
Mylan Institutional
Galway, Ireland
1084L101
Revised: 10/2021
MI:MAGNIJ:R4
Description
Magnesium Chloride Injection is a sterile solution of Magnesium Chloride Hexahydrate in Water for Injection q.s. Each mL contains Magnesium Chloride Hexahydrate 200 mg, Sodium Chloride 9 mg, Benzyl Alcohol 1% as a preservative, Water for Injection, q.s. pH adjusted with Hydrochloric Acid and/or Sodium Hydroxide. Total osmolarity equivalent to 2.951 mOsm/mL.
Contains 1.97 mEq of Mg++ and Cl- per mL.
The structural formula is MgCl2•6H2O
Actions
Magnesium is the second most plentiful cation within cellular fluids. It is an important activator of many enzyme systems and deficits are accompanied by a variety of functional disturbances.
Warning
Do not use if a precipitate is present.
Pregnancy
Animal reproduction studies have not been conducted with magnesium chloride. It is also not known whether magnesium chloride can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Magnesium Chloride should be given to a pregnant woman only if clearly needed.
Indications
As an electrolyte replenisher in magnesium deficiencies.
Precautions
The usual precautions for parenteral administration should be observed. Administer with caution if flushing and sweating occurs. A preparation of a calcium salt should be readily available for intravenous injection to counteract potential serious signs of magnesium intoxication. As long as deep tendon reflexes are active it is probable that the patient will not develop respiratory paralysis. Respiration and blood pressure should be carefully observed during and after administration of Magnesium Chloride Injection.
Usual Dosage Range
1 to 40 grams daily.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
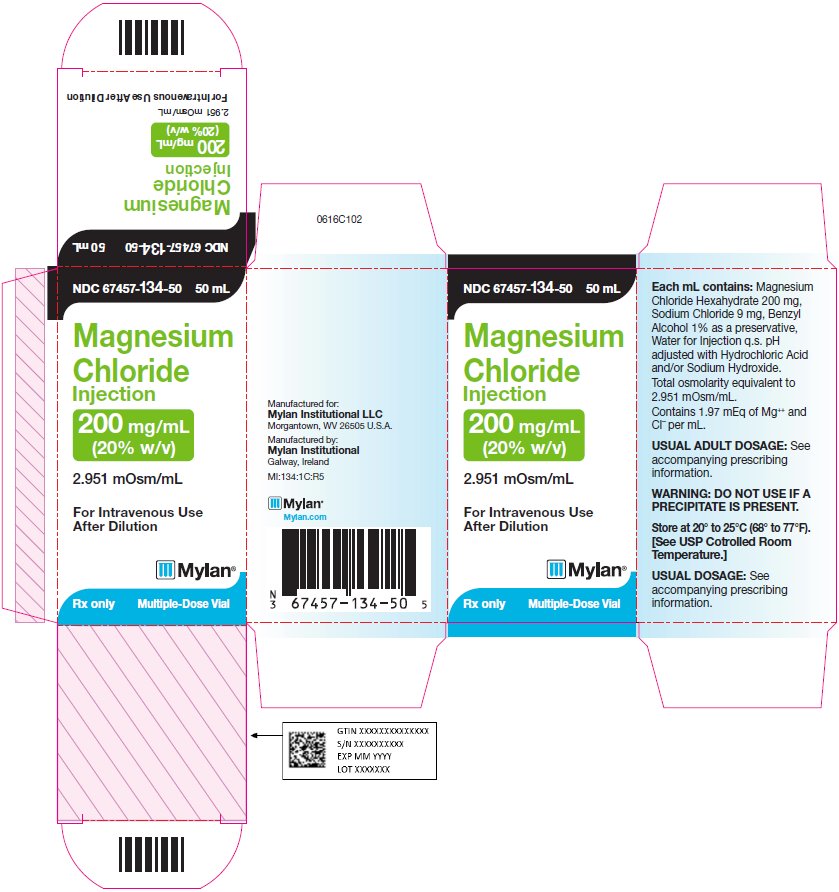
Principal Display Panel 200 Mg/ml
NDC 67457-134-50 50 mL
Magnesium
Chloride
Injection
200 mg/mL
(20% w/v)
2.951 mOsm/mL
For Intravenous Use After
Dilution
Rx only Multiple-Dose Vial
Each mL contains: Magnesium
Chloride Hexahydrate 200 mg,
Sodium Chloride 9 mg, Benzyl
Alcohol 1% as a preservative,
Water for Injection q.s. pH adjusted
with Hydrochloric Acid and/or
Sodium Hydroxide.
Total osmolarity equivalent to
2.951 mOsm/mL.
Contains 1.97 mEq of Mg++ and CI¯
per mL.
WARNING: DO NOT USE IF A
PRECIPITATE IS PRESENT.
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]
Usual Dosage: See accompanying
prescribing information.
Manufactured for:
Mylan Institutional LLC
Morgantown, WV 26505 U.S.A.
Manufactured by:
Mylan Institutional
Galway, Ireland
MI:134:1C:R5
Mylan.com
Structured Label Content
Actions (ACTIONS)
Magnesium is the second most plentiful cation within cellular fluids. It is an important activator of many enzyme systems and deficits are accompanied by a variety of functional disturbances.
Warning (WARNING)
Do not use if a precipitate is present.
Pregnancy
Animal reproduction studies have not been conducted with magnesium chloride. It is also not known whether magnesium chloride can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Magnesium Chloride should be given to a pregnant woman only if clearly needed.
Description (DESCRIPTION)
Magnesium Chloride Injection is a sterile solution of Magnesium Chloride Hexahydrate in Water for Injection q.s. Each mL contains Magnesium Chloride Hexahydrate 200 mg, Sodium Chloride 9 mg, Benzyl Alcohol 1% as a preservative, Water for Injection, q.s. pH adjusted with Hydrochloric Acid and/or Sodium Hydroxide. Total osmolarity equivalent to 2.951 mOsm/mL.
Contains 1.97 mEq of Mg++ and Cl- per mL.
The structural formula is MgCl2•6H2O
Indications (INDICATIONS)
As an electrolyte replenisher in magnesium deficiencies.
Precautions (PRECAUTIONS)
The usual precautions for parenteral administration should be observed. Administer with caution if flushing and sweating occurs. A preparation of a calcium salt should be readily available for intravenous injection to counteract potential serious signs of magnesium intoxication. As long as deep tendon reflexes are active it is probable that the patient will not develop respiratory paralysis. Respiration and blood pressure should be carefully observed during and after administration of Magnesium Chloride Injection.
How Supplied (HOW SUPPLIED)
Magnesium Chloride Injection 200 mg/mL (20% w/v).
NDC 67457-134-50
50 mL Multiple-Dose Vial. Individually boxed.
Store at 20° to 25°C (68° to 77°F). [See USP Controlled
Room Temperature.]
Manufactured for:
Mylan Institutional LLC
Morgantown, WV 26505
Manufactured by:
Mylan Institutional
Galway, Ireland
1084L101
Revised: 10/2021
MI:MAGNIJ:R4
Adverse Reactions (ADVERSE REACTIONS)
Flushing, sweating, sharply lowered blood pressure, hypothermia, stupor and ultimately respiratory depression.
Contraindications (CONTRAINDICATIONS)
Magnesium Chloride Injection should not be administered if there is renal impairment, marked myocardial disease or to comatose patients.
Usual Dosage Range (USUAL DOSAGE RANGE)
1 to 40 grams daily.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
For intravenous infusion: 4 grams in 250 mL of 5% Dextrose Injection, at a rate not exceeding 3 mL per minute. Serum magnesium levels should serve as a guide to continued dosage.
Principal Display Panel 200 Mg/ml (PRINCIPAL DISPLAY PANEL - 200 mg/mL)
NDC 67457-134-50 50 mL
Magnesium
Chloride
Injection
200 mg/mL
(20% w/v)
2.951 mOsm/mL
For Intravenous Use After
Dilution
Rx only Multiple-Dose Vial
Each mL contains: Magnesium
Chloride Hexahydrate 200 mg,
Sodium Chloride 9 mg, Benzyl
Alcohol 1% as a preservative,
Water for Injection q.s. pH adjusted
with Hydrochloric Acid and/or
Sodium Hydroxide.
Total osmolarity equivalent to
2.951 mOsm/mL.
Contains 1.97 mEq of Mg++ and CI¯
per mL.
WARNING: DO NOT USE IF A
PRECIPITATE IS PRESENT.
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]
Usual Dosage: See accompanying
prescribing information.
Manufactured for:
Mylan Institutional LLC
Morgantown, WV 26505 U.S.A.
Manufactured by:
Mylan Institutional
Galway, Ireland
MI:134:1C:R5
Mylan.com
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:36:21.861240 · Updated: 2026-03-14T21:46:05.265157