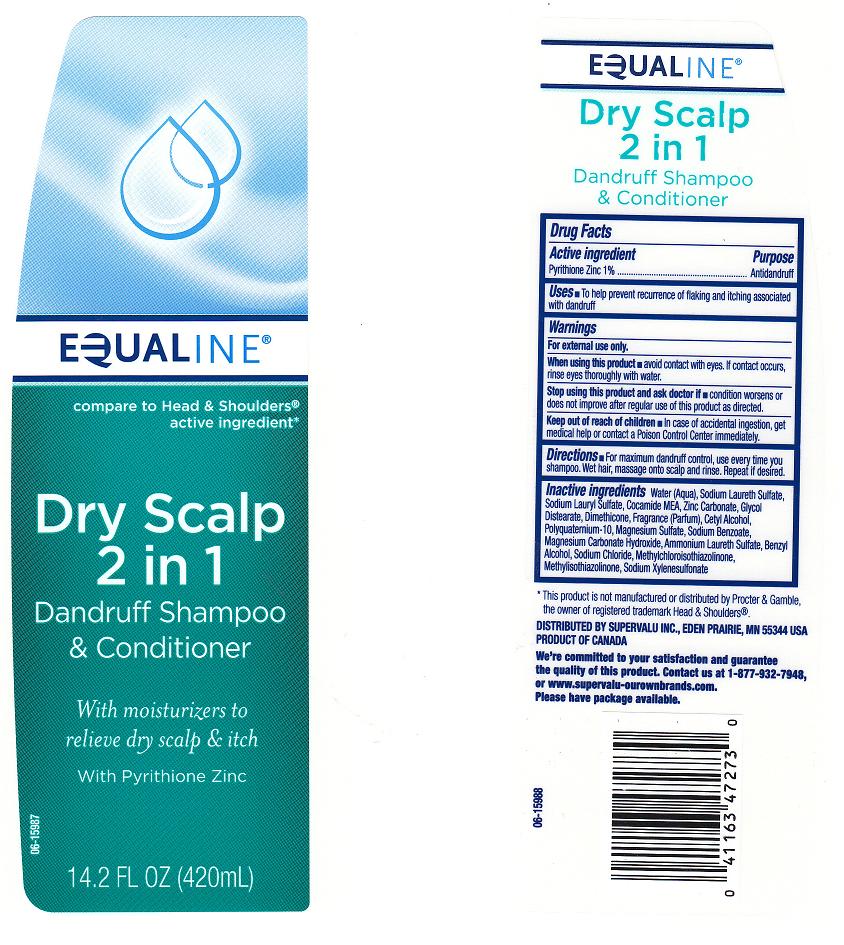
Back Label - Drug Facts Box
20e034ee-9540-4488-8293-5bbea7c4d2c7
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
PYRITHIONE ZINC 1%
Purpose
ANTI-DANDRUFF
Medication Information
Purpose
Description
PYRITHIONE ZINC 1%
Uses
Warnings
FOR EXTERNAL USE ONLY
Directions
Active Ingredient
PYRITHIONE ZINC 1%
Inactive Ingredients
Water (Aqua), Sodium Laureth Sulfate, Sodium Lauryl Sulfate, Cocamide MEA, Zinc Carbonate, Glycol Distearate, Dimethicone, Fragrance (Parfum), Cetyl Alcohol, Polyquaternium-10, Magnesium Sulfate, Sodium Benzoate, Magnesium Carbonate Hydroxide, Ammonium Laureth Sulfate, Benzyl Alcohol, Sodium Chloride, Methylchloroisothiazolinone, Methylisothiazolinone, Sodium Xylenesulfonate
When Using This Product
AVOID CONTACT WITH EYES. IF CONTACT OCCURS, RINSE EYES THROUGHLY WITH WATER.
Stop Use and Ask A Doctor If
CONDITION WORSENS OR DOES NOT IMPROVE AFTER REGULAR USE OF THIS PRODUCT AS DIRECTED
Keep Out of Reach of Children
IN CASE OF ACCIDENTAL INGESTION, GET MEDICAL HELP OR CONTACT POISON CONTROL CENTER IMMEDIATELY.
Package Front and Back Labels
14OZ Front and Back Labels: eql14.jpg
Structured Label Content
Uses
Purpose (PURPOSE)
Warnings (WARNINGS)
FOR EXTERNAL USE ONLY
Directions
Active Ingredient (ACTIVE INGREDIENT)
PYRITHIONE ZINC 1%
Inactive Ingredients
Water (Aqua), Sodium Laureth Sulfate, Sodium Lauryl Sulfate, Cocamide MEA, Zinc Carbonate, Glycol Distearate, Dimethicone, Fragrance (Parfum), Cetyl Alcohol, Polyquaternium-10, Magnesium Sulfate, Sodium Benzoate, Magnesium Carbonate Hydroxide, Ammonium Laureth Sulfate, Benzyl Alcohol, Sodium Chloride, Methylchloroisothiazolinone, Methylisothiazolinone, Sodium Xylenesulfonate
When Using This Product (WHEN USING THIS PRODUCT)
AVOID CONTACT WITH EYES. IF CONTACT OCCURS, RINSE EYES THROUGHLY WITH WATER.
Stop Use and Ask A Doctor If (STOP USE AND ASK A DOCTOR IF)
CONDITION WORSENS OR DOES NOT IMPROVE AFTER REGULAR USE OF THIS PRODUCT AS DIRECTED
Keep Out of Reach of Children (KEEP OUT OF REACH OF CHILDREN)
IN CASE OF ACCIDENTAL INGESTION, GET MEDICAL HELP OR CONTACT POISON CONTROL CENTER IMMEDIATELY.
Package Front and Back Labels (PACKAGE FRONT AND BACK LABELS)
14OZ Front and Back Labels: eql14.jpg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:07.924404 · Updated: 2026-03-14T22:52:28.188101