These Highlights Do Not Include All The Information Needed To Use Sufentanil Citrate Injection, Safely And Effectively. See Full Prescribing Information For Sufentanil Citrate Injection.
20293943-46ff-4345-1aa4-929b4e017a25
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Boxed Warning 12/2023 Warnings and Precautions ( 5.7 ) 12/2023
Indications and Usage
Sufentanil Citrate Injection is indicated for intravenous administration in adults and pediatric patients: • as an analgesic adjunct in the maintenance of balanced general anesthesia in patients who are intubated and ventilated. • as a primary anesthetic agent for the induction and maintenance of anesthesia with 100% oxygen in patients undergoing major surgical procedures, in patients who are intubated and ventilated, such as cardiovascular surgery or neurosurgical procedures in the sitting position, to provide favorable myocardial and cerebral oxygen balance or when extended postoperative ventilation is anticipated. Sufentanil Citrate Injection is indicated for epidural administration: • as an analgesic combined with low dose (usually 12.5 mg per administration) bupivacaine usually during labor and vaginal delivery.
Dosage and Administration
• Sufentanil Citrate Injection should be administered only by persons specifically trained in the use of intravenous anesthetics and management of the respiratory effects of potent opioids. • Ensure that an opioid antagonist, resuscitative and intubation equipment, and oxygen are readily available. ( 2.1 ) • Individualize dosing based on factors such as age, body weight, physical status, underlying pathological condition, use of other drugs, type of anesthesia to be used, and the surgical procedure involved. ( 2.1 ) • Initiate analgesic treatment with 1 to 2 mcg/kg intravenously. ( 2.2 ) • Initiate epidural injection for labor and delivery at 10 to 15 mcg of Sufentanil administered with 10 mL bupivacaine 0.125% with or without epinephrine. ( 2.3 )
Warnings and Precautions
• Life-Threatening Respiratory Depression in Patients with Chronic Pulmonary Disease or in Elderly, Cachectic, or Debilitated Patients : Monitor closely, particularly during initiation and titration. ( 5.2 ) • Risks of Skeletal Muscle Rigidity and Skeletal Muscle Movement : Manage with neuromuscular blocking agent. See full prescribing information for more detail on managing these risks. ( 5.5 ) • Severe Cardiovascular Depression : Monitor during dosage initiation and titration. ( 5.6 ) • Opioid-Induced Hyperalgesia and Allodynia : Opioid-Induced Hyperalgesia (OIH) occurs when an opioid analgesic paradoxically causes an increase in pain, or an increase in sensitivity to pain. If OIH is suspected, carefully consider appropriately decreasing the dose of the current opioid analgesic, or opioid rotation. ( 5.7 ) • Serotonin Syndrome : Potentially life-threatening condition could result from concomitant serotonergic drug administration. Discontinue Sufentanil Citrate Injection if serotonin syndrome is suspected. ( 5.8 ) • Risks of Use in Patients with Increased Intracranial Pressure, Brain Tumors, or Head Injury : Monitor for sedation and respiratory depression. ( 5.10 )
Contraindications
Sufentanil Citrate Injection is contraindicated in patients with: • Hypersensitivity to sufentanil (e.g., anaphylaxis) [see Adverse Reactions (6.2) ]
Adverse Reactions
The following serious adverse reactions are described, or described in greater detail, in other sections: • Addiction, Abuse, and Misuse [see Warnings and Precautions (5.1) ] • Life-Threatening Respiratory Depression [see Warnings and Precautions (5.2) ] • Skeletal Muscle Rigidity and Skeletal Muscle Movement [see Warnings and Precautions (5.5) ] • Interactions with Benzodiazepines or Other CNS Depressants [see Warnings and Precautions (5.3) ] • Severe Cardiovascular Depression [see Warnings and Precautions (5.6) ] • Opioid-Induced Hyperalgesia and Allodynia [see Warnings and Precautions (5.7) ] • Serotonin Syndrome [see Warnings and Precautions (5.8) ] • Gastrointestinal Adverse Reactions [see Warnings and Precautions (5.11) ] • Seizures [see Warnings and Precautions (5.12) ]
Drug Interactions
Table 3 includes clinically significant drug interactions with Sufentanil Citrate Injection. Table 3: Clinically Significant Drug Interactions with Sufentanil Citrate Injection Inhibitors of CYP3A4 Clinical Impact: The concomitant use of Sufentanil Citrate Injection and CYP3A4 inhibitors can increase the plasma concentration of sufentanil, resulting in increased or prolonged opioid effects, particularly when an inhibitor is added after a stable dose of Sufentanil Citrate Injection is achieved [see Warnings and Precautions (5.4) ] . After stopping a CYP3A4 inhibitor, as the effects of the inhibitor decline, the sufentanil plasma concentration will decrease [see Clinical Pharmacology (12.3) ] , resulting in decreased opioid efficacy or a withdrawal syndrome in patients who had developed physical dependence to sufentanil. Intervention: If concomitant use is necessary, consider dosage reduction of Sufentanil Citrate Injection until stable drug effects are achieved. Monitor patients at frequent intervals for respiratory depression and sedation. If a CYP3A4 inhibitor is discontinued, consider increasing the Sufentanil Citrate Injection dosage until stable drug effects are achieved. Monitor for signs of opioid withdrawal. Examples: Macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g., ketoconazole), protease inhibitors (e.g., ritonavir), grapefruit juice. CYP3A4 Inducers Clinical Impact: The concomitant use of Sufentanil Citrate Injection and CYP3A4 inducers can decrease the plasma concentration of sufentanil [see Clinical Pharmacology (12.3) ] , resulting in decreased efficacy or onset of a withdrawal syndrome in patients who have developed physical dependence to sufentanil [see Warnings and Precautions (5.4) ] . After stopping a CYP3A4 inducer, as the effects of the inducer decline, the sufentanil plasma concentration will increase [see Clinical Pharmacology (12.3) ] , which could increase or prolong both the therapeutic effects and adverse reactions and may cause serious respiratory depression. Intervention: If concomitant use is necessary, consider increasing the Sufentanil Citrate Injection dosage until stable drug effects are achieved. Monitor for signs of opioid withdrawal. If a CYP3A4 inducer is discontinued, consider Sufentanil Citrate Injection dosage reduction and monitor for signs of respiratory depression. Examples: Rifampin, carbamazepine, phenytoin Benzodiazepines and Other Central Nervous System (CNS) Depressants Clinical Impact: The concomitant use of Sufentanil Citrate Injection with CNS depressants my result in decreased pulmonary artery pressure and may cause hypotension. Even small dosages of diazepam may cause cardiovascular depression when added to high dose or anesthetic dosages of Sufentanil Citrate Injection. As postoperative analgesia, concomitant use of Sufentanil Citrate Injection can increase the risk of hypotension, respiratory depression, profound sedation, coma, and death [see Warnings and Precautions (5.2 , 5.3 )] . Intervention: As postoperative analgesia, start with a lower dose of Sufentanil Citrate Injection and monitor patients for signs of respiratory depression, sedation, and hypotension. Fluids or other measures to counter hypotension should be available [see Warnings and Precautions (5.3) ] . Examples: Benzodiazepines and other sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, other opioids, alcohol. Serotonergic Drugs Clinical Impact: The concomitant use of opioids with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome [see Warnings and Precautions (5.8) ]. Intervention: If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dose adjustment. Discontinue Sufentanil Citrate Injection if serotonin syndrome is suspected. Examples: Selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5‑HT3 receptor antagonists, drugs that effect the serotonin neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), monoamine oxidase inhibitors (those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue). Monoamine Oxidase Inhibitors (MAOIs) Clinical Impact: MAOI interactions with opioids may manifest as serotonin syndrome or opioid toxicity (e.g., respiratory depression, coma) [see Warnings and Precautions (5.2) ]. Intervention: The use of Sufentanil Citrate Injection is not recommended for patients taking MAOIs or within 14 days of stopping such treatment. Examples: phenelzine, tranylcypromine, linezolid Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics Clinical Impact: May reduce the analgesic effect of Sufentanil Citrate Injection and/or precipitate withdrawal symptoms. Intervention: Avoid concomitant use. Examples: butorphanol, nalbuphine, pentazocine, buprenorphine Muscle Relaxants Clinical Impact: Sufentanil may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression. Intervention: Monitor patients for signs of respiratory depression that may be greater than otherwise expected and decrease the dosage of Sufentanil Citrate Injection and/or the muscle relaxant as necessary. Diuretics Clinical Impact: Opioids can reduce the efficacy of diuretics by inducing the release of antidiuretic hormone. Intervention: Monitor patients for signs of diminished diuresis and/or effects on blood pressure and increase the dosage of the diuretic as needed. Anticholinergic Drugs Clinical Impact: The concomitant use of anticholinergic drugs may increase risk of urinary retention and/or severe constipation, which may lead to paralytic ileus. Intervention: Monitor patients for signs of urinary retention or reduced gastric motility when Sufentanil Citrate Injection is used concomitantly with anticholinergic drugs. Nitrous oxide Clinical Impact: Nitrous oxide has been reported to produce cardiovascular depression when given with higher doses of Sufentanil Citrate Injection. Intervention: Monitor patients for signs of cardiovascular depression that may be greater than otherwise expected.
Storage and Handling
Sufentanil Citrate Injection, USP equivalent to 50 mcg/mL sufentanil is supplied in the following single-dose containers: Unit of Sale Concentration NDC 0409-3382-21 Carton of 10 Single-dose Fliptop Vials 50 mcg/mL NDC 0409-3382-22 Carton of 10 Single-dose Fliptop Vials 100 mcg/2 mL (50 mcg/mL) NDC 0409-3382-25 Carton of 10 Single-dose Fliptop Vials 250 mcg/5 mL (50 mcg/mL)
How Supplied
Sufentanil Citrate Injection, USP equivalent to 50 mcg/mL sufentanil is supplied in the following single-dose containers: Unit of Sale Concentration NDC 0409-3382-21 Carton of 10 Single-dose Fliptop Vials 50 mcg/mL NDC 0409-3382-22 Carton of 10 Single-dose Fliptop Vials 100 mcg/2 mL (50 mcg/mL) NDC 0409-3382-25 Carton of 10 Single-dose Fliptop Vials 250 mcg/5 mL (50 mcg/mL)
Medication Information
Warnings and Precautions
• Life-Threatening Respiratory Depression in Patients with Chronic Pulmonary Disease or in Elderly, Cachectic, or Debilitated Patients : Monitor closely, particularly during initiation and titration. ( 5.2 ) • Risks of Skeletal Muscle Rigidity and Skeletal Muscle Movement : Manage with neuromuscular blocking agent. See full prescribing information for more detail on managing these risks. ( 5.5 ) • Severe Cardiovascular Depression : Monitor during dosage initiation and titration. ( 5.6 ) • Opioid-Induced Hyperalgesia and Allodynia : Opioid-Induced Hyperalgesia (OIH) occurs when an opioid analgesic paradoxically causes an increase in pain, or an increase in sensitivity to pain. If OIH is suspected, carefully consider appropriately decreasing the dose of the current opioid analgesic, or opioid rotation. ( 5.7 ) • Serotonin Syndrome : Potentially life-threatening condition could result from concomitant serotonergic drug administration. Discontinue Sufentanil Citrate Injection if serotonin syndrome is suspected. ( 5.8 ) • Risks of Use in Patients with Increased Intracranial Pressure, Brain Tumors, or Head Injury : Monitor for sedation and respiratory depression. ( 5.10 )
Indications and Usage
Sufentanil Citrate Injection is indicated for intravenous administration in adults and pediatric patients: • as an analgesic adjunct in the maintenance of balanced general anesthesia in patients who are intubated and ventilated. • as a primary anesthetic agent for the induction and maintenance of anesthesia with 100% oxygen in patients undergoing major surgical procedures, in patients who are intubated and ventilated, such as cardiovascular surgery or neurosurgical procedures in the sitting position, to provide favorable myocardial and cerebral oxygen balance or when extended postoperative ventilation is anticipated. Sufentanil Citrate Injection is indicated for epidural administration: • as an analgesic combined with low dose (usually 12.5 mg per administration) bupivacaine usually during labor and vaginal delivery.
Dosage and Administration
• Sufentanil Citrate Injection should be administered only by persons specifically trained in the use of intravenous anesthetics and management of the respiratory effects of potent opioids. • Ensure that an opioid antagonist, resuscitative and intubation equipment, and oxygen are readily available. ( 2.1 ) • Individualize dosing based on factors such as age, body weight, physical status, underlying pathological condition, use of other drugs, type of anesthesia to be used, and the surgical procedure involved. ( 2.1 ) • Initiate analgesic treatment with 1 to 2 mcg/kg intravenously. ( 2.2 ) • Initiate epidural injection for labor and delivery at 10 to 15 mcg of Sufentanil administered with 10 mL bupivacaine 0.125% with or without epinephrine. ( 2.3 )
Contraindications
Sufentanil Citrate Injection is contraindicated in patients with: • Hypersensitivity to sufentanil (e.g., anaphylaxis) [see Adverse Reactions (6.2) ]
Adverse Reactions
The following serious adverse reactions are described, or described in greater detail, in other sections: • Addiction, Abuse, and Misuse [see Warnings and Precautions (5.1) ] • Life-Threatening Respiratory Depression [see Warnings and Precautions (5.2) ] • Skeletal Muscle Rigidity and Skeletal Muscle Movement [see Warnings and Precautions (5.5) ] • Interactions with Benzodiazepines or Other CNS Depressants [see Warnings and Precautions (5.3) ] • Severe Cardiovascular Depression [see Warnings and Precautions (5.6) ] • Opioid-Induced Hyperalgesia and Allodynia [see Warnings and Precautions (5.7) ] • Serotonin Syndrome [see Warnings and Precautions (5.8) ] • Gastrointestinal Adverse Reactions [see Warnings and Precautions (5.11) ] • Seizures [see Warnings and Precautions (5.12) ]
Drug Interactions
Table 3 includes clinically significant drug interactions with Sufentanil Citrate Injection. Table 3: Clinically Significant Drug Interactions with Sufentanil Citrate Injection Inhibitors of CYP3A4 Clinical Impact: The concomitant use of Sufentanil Citrate Injection and CYP3A4 inhibitors can increase the plasma concentration of sufentanil, resulting in increased or prolonged opioid effects, particularly when an inhibitor is added after a stable dose of Sufentanil Citrate Injection is achieved [see Warnings and Precautions (5.4) ] . After stopping a CYP3A4 inhibitor, as the effects of the inhibitor decline, the sufentanil plasma concentration will decrease [see Clinical Pharmacology (12.3) ] , resulting in decreased opioid efficacy or a withdrawal syndrome in patients who had developed physical dependence to sufentanil. Intervention: If concomitant use is necessary, consider dosage reduction of Sufentanil Citrate Injection until stable drug effects are achieved. Monitor patients at frequent intervals for respiratory depression and sedation. If a CYP3A4 inhibitor is discontinued, consider increasing the Sufentanil Citrate Injection dosage until stable drug effects are achieved. Monitor for signs of opioid withdrawal. Examples: Macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g., ketoconazole), protease inhibitors (e.g., ritonavir), grapefruit juice. CYP3A4 Inducers Clinical Impact: The concomitant use of Sufentanil Citrate Injection and CYP3A4 inducers can decrease the plasma concentration of sufentanil [see Clinical Pharmacology (12.3) ] , resulting in decreased efficacy or onset of a withdrawal syndrome in patients who have developed physical dependence to sufentanil [see Warnings and Precautions (5.4) ] . After stopping a CYP3A4 inducer, as the effects of the inducer decline, the sufentanil plasma concentration will increase [see Clinical Pharmacology (12.3) ] , which could increase or prolong both the therapeutic effects and adverse reactions and may cause serious respiratory depression. Intervention: If concomitant use is necessary, consider increasing the Sufentanil Citrate Injection dosage until stable drug effects are achieved. Monitor for signs of opioid withdrawal. If a CYP3A4 inducer is discontinued, consider Sufentanil Citrate Injection dosage reduction and monitor for signs of respiratory depression. Examples: Rifampin, carbamazepine, phenytoin Benzodiazepines and Other Central Nervous System (CNS) Depressants Clinical Impact: The concomitant use of Sufentanil Citrate Injection with CNS depressants my result in decreased pulmonary artery pressure and may cause hypotension. Even small dosages of diazepam may cause cardiovascular depression when added to high dose or anesthetic dosages of Sufentanil Citrate Injection. As postoperative analgesia, concomitant use of Sufentanil Citrate Injection can increase the risk of hypotension, respiratory depression, profound sedation, coma, and death [see Warnings and Precautions (5.2 , 5.3 )] . Intervention: As postoperative analgesia, start with a lower dose of Sufentanil Citrate Injection and monitor patients for signs of respiratory depression, sedation, and hypotension. Fluids or other measures to counter hypotension should be available [see Warnings and Precautions (5.3) ] . Examples: Benzodiazepines and other sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, other opioids, alcohol. Serotonergic Drugs Clinical Impact: The concomitant use of opioids with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome [see Warnings and Precautions (5.8) ]. Intervention: If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dose adjustment. Discontinue Sufentanil Citrate Injection if serotonin syndrome is suspected. Examples: Selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5‑HT3 receptor antagonists, drugs that effect the serotonin neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), monoamine oxidase inhibitors (those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue). Monoamine Oxidase Inhibitors (MAOIs) Clinical Impact: MAOI interactions with opioids may manifest as serotonin syndrome or opioid toxicity (e.g., respiratory depression, coma) [see Warnings and Precautions (5.2) ]. Intervention: The use of Sufentanil Citrate Injection is not recommended for patients taking MAOIs or within 14 days of stopping such treatment. Examples: phenelzine, tranylcypromine, linezolid Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics Clinical Impact: May reduce the analgesic effect of Sufentanil Citrate Injection and/or precipitate withdrawal symptoms. Intervention: Avoid concomitant use. Examples: butorphanol, nalbuphine, pentazocine, buprenorphine Muscle Relaxants Clinical Impact: Sufentanil may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression. Intervention: Monitor patients for signs of respiratory depression that may be greater than otherwise expected and decrease the dosage of Sufentanil Citrate Injection and/or the muscle relaxant as necessary. Diuretics Clinical Impact: Opioids can reduce the efficacy of diuretics by inducing the release of antidiuretic hormone. Intervention: Monitor patients for signs of diminished diuresis and/or effects on blood pressure and increase the dosage of the diuretic as needed. Anticholinergic Drugs Clinical Impact: The concomitant use of anticholinergic drugs may increase risk of urinary retention and/or severe constipation, which may lead to paralytic ileus. Intervention: Monitor patients for signs of urinary retention or reduced gastric motility when Sufentanil Citrate Injection is used concomitantly with anticholinergic drugs. Nitrous oxide Clinical Impact: Nitrous oxide has been reported to produce cardiovascular depression when given with higher doses of Sufentanil Citrate Injection. Intervention: Monitor patients for signs of cardiovascular depression that may be greater than otherwise expected.
Storage and Handling
Sufentanil Citrate Injection, USP equivalent to 50 mcg/mL sufentanil is supplied in the following single-dose containers: Unit of Sale Concentration NDC 0409-3382-21 Carton of 10 Single-dose Fliptop Vials 50 mcg/mL NDC 0409-3382-22 Carton of 10 Single-dose Fliptop Vials 100 mcg/2 mL (50 mcg/mL) NDC 0409-3382-25 Carton of 10 Single-dose Fliptop Vials 250 mcg/5 mL (50 mcg/mL)
How Supplied
Sufentanil Citrate Injection, USP equivalent to 50 mcg/mL sufentanil is supplied in the following single-dose containers: Unit of Sale Concentration NDC 0409-3382-21 Carton of 10 Single-dose Fliptop Vials 50 mcg/mL NDC 0409-3382-22 Carton of 10 Single-dose Fliptop Vials 100 mcg/2 mL (50 mcg/mL) NDC 0409-3382-25 Carton of 10 Single-dose Fliptop Vials 250 mcg/5 mL (50 mcg/mL)
Description
Boxed Warning 12/2023 Warnings and Precautions ( 5.7 ) 12/2023
Section 42229-5
Adjunct to general anesthesia:
-
•Doses of up to 8 mcg/kg (see Table 1)
-
•Total Dosage Requirements of 1 mcg/kg/hr or less are recommended
-
•Dosage should be individualized and adjusted to remaining operative time anticipated.
| Total dosage | Maintenance dosage |
|---|---|
|
Duration of anesthesia 1 to 2 hours |
|
|
Incremental or Infusion: 1 to 2 mcg/kg
|
Incremental: 10 to 25 mcg (0.2 to 0.5 mL) may be administered in increments as needed when movement and/or changes in vital signs indicate surgical stress or lightening of analgesia. Supplemental dosages should be individualized and adjusted to remaining operative time anticipated. |
|
Duration of anesthesia 2 to 8 hours |
|
|
Incremental or Infusion: 2 to 8 mcg/kg
|
Incremental: 10 to 50 mcg (0.2 to 1 mL) may be administered in increments as needed when movement and/or changes in vital signs indicate surgical stress or lightening of analgesia. Supplemental dosages should be individualized and adjusted to the remaining operative time anticipated. |
Section 43683-2
|
Boxed Warning |
12/2023 |
|
Warnings and Precautions (5.7) |
12/2023 |
Section 44425-7
Protect from light. Retain in carton until time of use.
Store at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature.]
9.2 Abuse
Sufentanil Citrate Injection contains sufentanil, a substance with high potential for misuse and abuse, which can lead to the development of substance use disorder, including addiction [see Warnings and Precautions (5.1)].
Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a healthcare provider or for whom it was not prescribed.
Abuse is the intentional, non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects.
Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence.
Misuse and abuse of Sufentanil Citrate Injection increases risk of overdose, which may lead to central nervous system and respiratory depression, hypotension, seizures, and death. The risk is increased with concurrent abuse of Sufentanil Citrate Injection with alcohol and/or other CNS depressants. Abuse of and addiction to opioids in some individuals may not be accompanied by concurrent tolerance and symptoms of physical dependence. In addition, abuse of opioids can occur in the absence of addiction.
All patients treated with opioids require careful and frequent reevaluation for signs of misuse, abuse, and addiction, because use of opioid analgesic products carries the risk of addiction even under appropriate medical use. Patients at high risk of Sufentanil Citrate Injection abuse include those with a history of prolonged use of any opioid, including products containing sufentanil, those with a history of drug or alcohol abuse, or those who use Sufentanil Citrate Injection in combination with other abused drugs.
“Drug-seeking” behavior is very common in persons with substance use disorders. Drug-seeking tactics include emergency calls or visits near the end of office hours, refusal to undergo appropriate examination, testing, or referral, repeated “loss” of prescriptions, tampering with prescriptions, and reluctance to provide prior medical records or contact information for other treating healthcare provider(s). “Doctor shopping” (visiting multiple prescribers to obtain additional prescriptions) is common among people who abuse drugs and people with substance use disorder. Preoccupation with achieving adequate pain relief can be appropriate behavior in a patient with inadequate pain control.
Sufentanil Citrate Injection, like other opioids, can be diverted for nonmedical use into illicit channels of distribution. Careful record-keeping of prescribing information, including quantity, frequency, and renewal requests, as required by state and federal law, is strongly advised.
Proper assessment of the patient, proper prescribing practices, periodic reevaluation of therapy, and proper dispensing and storage are appropriate measures that help to limit abuse of opioid drugs.
11 Description
Sufentanil Citrate Injection, USP is a sterile, nonpyrogenic solution of sufentanil citrate in water for injection. Sufentanil Citrate is a potent opioid analgesic which is administered either epidurally or by intravenous injection.
Each mL contains sufentanil citrate equivalent to 50 mcg of sufentanil. May contain sodium hydroxide and/or hydrochloric acid for pH adjustment. pH 4.2 (3.5 to 6.0).
The solution contains no bacteriostat, antimicrobial agent or added buffer and is intended for use only as a single-use injection. When smaller doses are required, the unused portion should be discarded in an appropriate manner.
Sufentanil Citrate, USP, occurs as a white crystalline powder and is chemically designated as N-[-4-(methyoxymethyl)-1-[2-(2-thienyl)ethyl]-4-piperidinyl]-N-phenylpropanamide 2-hydroxy-1,2,3-propanetricarboxylate (1:1).
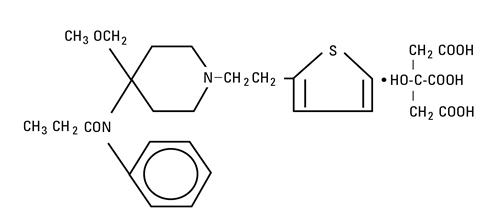
The molecular formula of sufentanil citrate is C22H30N2O2S∙C6H8O7 and the molecular weight is 578.69. Sufentanil Citrate has the following structural formula:
9.3 Dependence
Both tolerance and physical dependence can develop during use of opioid therapy.
Tolerance is a physiological state characterized by a reduced response to a drug after repeated administration (i.e., a higher dose of a drug is required to produce the same effect that was once obtained at a lower dose).
Physical dependence is a state that develops as a result of a physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug.
Withdrawal may be precipitated through the administration of drugs with opioid antagonist activity (e.g., naloxone), mixed agonist/antagonist analgesics (e.g., pentazocine, butorphanol, nalbuphine), or partial agonists (e.g., buprenorphine). Physical dependence may not occur to a clinically significant degree until after several days to weeks of continued use.
Sufentanil Citrate Injection should not be abruptly discontinued in a physically-dependent patient. If Sufentanil Citrate Injection is abruptly discontinued in a physically-dependent patient, a withdrawal syndrome may occur, typically characterized by restlessness, lacrimation, rhinorrhea, perspiration, chills, myalgia, and mydriasis. Other signs and symptoms also may develop, including irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory rate, or heart rate.
Infants born to mothers physically-dependent on opioids will also be physically-dependent and may exhibit respiratory difficulties and withdrawal signs [see Use in Specific Populations (8.1)].
8.4 Pediatric Use
The safety and efficacy of intravenous sufentanil in pediatric patients as young as 1 day old undergoing cardiovascular surgery have been documented in a limited number of cases. The clearance of sufentanil in healthy neonates is approximately one-half that in adults and children. The clearance rate of sufentanil can be further reduced by up to a third in neonates with cardiovascular disease, resulting in an increase in the elimination half-life of the drug.
8.5 Geriatric Use
Elderly patients (aged 65 years or older) may have increased sensitivity to sufentanil. In general, use caution when selecting a dosage for an elderly patient, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
Respiratory depression is the chief risk for elderly patients treated with opioids, and has occurred after large initial doses were administered to patients who were not opioid-tolerant or when opioids were co-administered with other agents that depress respiration. Titrate the dosage of Sufentanil Citrate Injection slowly in geriatric patients and monitor closely for signs of central nervous system and respiratory depression [see Warnings and Precautions (5.2) ].
2.2 Intravenous Use
Sufentanil Citrate may be administered intravenously by slow injection or infusion.
4 Contraindications
Sufentanil Citrate Injection is contraindicated in patients with:
-
•Hypersensitivity to sufentanil (e.g., anaphylaxis) [see Adverse Reactions (6.2)]
6 Adverse Reactions
The following serious adverse reactions are described, or described in greater detail, in other sections:
-
•Addiction, Abuse, and Misuse [see Warnings and Precautions (5.1)]
-
•Life-Threatening Respiratory Depression [see Warnings and Precautions (5.2)]
-
•Skeletal Muscle Rigidity and Skeletal Muscle Movement [see Warnings and Precautions (5.5) ]
-
•Interactions with Benzodiazepines or Other CNS Depressants [see Warnings and Precautions (5.3)]
-
•Severe Cardiovascular Depression [see Warnings and Precautions (5.6)]
-
•Opioid-Induced Hyperalgesia and Allodynia [see Warnings and Precautions (5.7)]
-
•Serotonin Syndrome [see Warnings and Precautions (5.8) ]
-
•Gastrointestinal Adverse Reactions [see Warnings and Precautions (5.11) ]
-
• Seizures [see Warnings and Precautions (5.12) ]
7 Drug Interactions
Table 3 includes clinically significant drug interactions with Sufentanil Citrate Injection.
|
Inhibitors of CYP3A4 |
|
|
Clinical Impact: |
The concomitant use of Sufentanil Citrate Injection and CYP3A4 inhibitors can increase the plasma concentration of sufentanil, resulting in increased or prolonged opioid effects, particularly when an inhibitor is added after a stable dose of Sufentanil Citrate Injection is achieved [see Warnings and Precautions (5.4)]. |
|
Intervention: |
If concomitant use is necessary, consider dosage reduction of Sufentanil Citrate Injection until stable drug effects are achieved. Monitor patients at frequent intervals for respiratory depression and sedation. |
|
Examples: |
Macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g., ketoconazole), protease inhibitors (e.g., ritonavir), grapefruit juice. |
|
CYP3A4 Inducers |
|
|
Clinical Impact: |
The concomitant use of Sufentanil Citrate Injection and CYP3A4 inducers can decrease the plasma concentration of sufentanil [see Clinical Pharmacology (12.3)], resulting in decreased efficacy or onset of a withdrawal syndrome in patients who have developed physical dependence to sufentanil [see Warnings and Precautions (5.4)]. |
|
Intervention: |
If concomitant use is necessary, consider increasing the Sufentanil Citrate Injection dosage until stable drug effects are achieved. Monitor for signs of opioid withdrawal. If a CYP3A4 inducer is discontinued, consider Sufentanil Citrate Injection dosage reduction and monitor for signs of respiratory depression. |
|
Examples: |
Rifampin, carbamazepine, phenytoin |
|
Benzodiazepines and Other Central Nervous System (CNS) Depressants |
|
|
Clinical Impact: |
The concomitant use of Sufentanil Citrate Injection with CNS depressants my result in decreased pulmonary artery pressure and may cause hypotension. Even small dosages of diazepam may cause cardiovascular depression when added to high dose or anesthetic dosages of Sufentanil Citrate Injection. As postoperative analgesia, concomitant use of Sufentanil Citrate Injection can increase the risk of hypotension, respiratory depression, profound sedation, coma, and death [see Warnings and Precautions (5.2, 5.3)]. |
|
Intervention: |
As postoperative analgesia, start with a lower dose of Sufentanil Citrate Injection and monitor patients for signs of respiratory depression, sedation, and hypotension. Fluids or other measures to counter hypotension should be available [see Warnings and Precautions (5.3) ]. |
|
Examples: |
Benzodiazepines and other sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, other opioids, alcohol. |
|
Serotonergic Drugs |
|
|
Clinical Impact: |
The concomitant use of opioids with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome [see Warnings and Precautions (5.8) ]. |
|
Intervention: |
If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dose adjustment. Discontinue Sufentanil Citrate Injection if serotonin syndrome is suspected. |
|
Examples: |
Selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5‑HT3 receptor antagonists, drugs that effect the serotonin neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), monoamine oxidase inhibitors (those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue). |
|
Monoamine Oxidase Inhibitors (MAOIs) |
|
|
Clinical Impact: |
MAOI interactions with opioids may manifest as serotonin syndrome or opioid toxicity (e.g., respiratory depression, coma) [see Warnings and Precautions (5.2)]. |
|
Intervention: |
The use of Sufentanil Citrate Injection is not recommended for patients taking MAOIs or within 14 days of stopping such treatment. |
|
Examples: |
phenelzine, tranylcypromine, linezolid |
|
Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics |
|
|
Clinical Impact: |
May reduce the analgesic effect of Sufentanil Citrate Injection and/or precipitate withdrawal symptoms. |
|
Intervention: |
Avoid concomitant use. |
|
Examples: |
butorphanol, nalbuphine, pentazocine, buprenorphine |
|
Muscle Relaxants |
|
|
Clinical Impact: |
Sufentanil may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression. |
|
Intervention: |
Monitor patients for signs of respiratory depression that may be greater than otherwise expected and decrease the dosage of Sufentanil Citrate Injection and/or the muscle relaxant as necessary. |
|
Diuretics |
|
|
Clinical Impact: |
Opioids can reduce the efficacy of diuretics by inducing the release of antidiuretic hormone. |
|
Intervention: |
Monitor patients for signs of diminished diuresis and/or effects on blood pressure and increase the dosage of the diuretic as needed. |
|
Anticholinergic Drugs |
|
|
Clinical Impact: |
The concomitant use of anticholinergic drugs may increase risk of urinary retention and/or severe constipation, which may lead to paralytic ileus. |
|
Intervention: |
Monitor patients for signs of urinary retention or reduced gastric motility when Sufentanil Citrate Injection is used concomitantly with anticholinergic drugs. |
|
Nitrous oxide |
|
|
Clinical Impact: |
Nitrous oxide has been reported to produce cardiovascular depression when given with higher doses of Sufentanil Citrate Injection. |
|
Intervention: |
Monitor patients for signs of cardiovascular depression that may be greater than otherwise expected. |
8.7 Renal Impairment
Sufentanil Citrate Injection should be administered with caution to patients with kidney dysfunction because of the renal excretion of sufentanil citrate and its metabolites. Reduce the dosage as needed and monitor for signs of respiratory depression, sedation, and hypotension.
12.3 Pharmacokinetics
Sufentanil Citrate Injection is administered by the intravenous or epidural route. The pharmacokinetics of intravenous sufentanil can be described as a three-compartment model.
8.6 Hepatic Impairment
Sufentanil Citrate Injection should be administered with caution to patients with liver dysfunction because of the extensive hepatic metabolism. Reduce the dosage as needed and monitor closely for signs of respiratory depression, sedation, and hypotension.
1 Indications and Usage
Sufentanil Citrate Injection is indicated for intravenous administration in adults and pediatric patients:
-
•as an analgesic adjunct in the maintenance of balanced general anesthesia in patients who are intubated and ventilated.
-
•as a primary anesthetic agent for the induction and maintenance of anesthesia with 100% oxygen in patients undergoing major surgical procedures, in patients who are intubated and ventilated, such as cardiovascular surgery or neurosurgical procedures in the sitting position, to provide favorable myocardial and cerebral oxygen balance or when extended postoperative ventilation is anticipated.
Sufentanil Citrate Injection is indicated for epidural administration:
-
•as an analgesic combined with low dose (usually 12.5 mg per administration) bupivacaine usually during labor and vaginal delivery.
12.1 Mechanism of Action
Sufentanil is an opioid agonist. When used in balanced general anesthesia, sufentanil has been reported to be as much as 10 times as potent as fentanyl. When administered intravenously as a primary anesthetic agent with 100% oxygen, sufentanil is approximately 5 to 7 times as potent as fentanyl.
9.1 Controlled Substance
Sufentanil Citrate Injection contains sufentanil, a Schedule II controlled substance.
5 Warnings and Precautions
-
•Life-Threatening Respiratory Depression in Patients with Chronic Pulmonary Disease or in Elderly, Cachectic, or
Debilitated Patients: Monitor closely, particularly during initiation and titration. (5.2) -
•Risks of Skeletal Muscle Rigidity and Skeletal Muscle Movement: Manage with neuromuscular blocking agent. See full prescribing information for more detail on managing these risks. (5.5)
-
•Severe Cardiovascular Depression: Monitor during dosage initiation and titration. (5.6)
-
•Opioid-Induced Hyperalgesia and Allodynia: Opioid-Induced Hyperalgesia (OIH) occurs when an opioid analgesic paradoxically causes an increase in pain, or an increase in sensitivity to pain. If OIH is suspected, carefully consider appropriately decreasing the dose of the current opioid analgesic, or opioid rotation. (5.7)
-
•Serotonin Syndrome: Potentially life-threatening condition could result from concomitant serotonergic drug administration. Discontinue Sufentanil Citrate Injection if serotonin syndrome is suspected. (5.8)
-
•Risks of Use in Patients with Increased Intracranial Pressure, Brain Tumors, or Head Injury: Monitor for sedation and respiratory depression. (5.10)
2 Dosage and Administration
-
•Sufentanil Citrate Injection should be administered only by persons specifically trained in the use of intravenous anesthetics and management of the respiratory effects of potent opioids.
-
•Ensure that an opioid antagonist, resuscitative and intubation equipment, and oxygen are readily available. (2.1)
-
•Individualize dosing based on factors such as age, body weight, physical status, underlying pathological condition, use of other drugs, type of anesthesia to be used, and the surgical procedure involved. (2.1)
-
•Initiate analgesic treatment with 1 to 2 mcg/kg intravenously. (2.2)
-
•Initiate epidural injection for labor and delivery at 10 to 15 mcg of Sufentanil administered with 10 mL bupivacaine 0.125% with or without epinephrine. (2.3)
3 Dosage Forms and Strengths
Sufentanil Citrate Injection, USP 50 mcg/mL (equivalent to 50 mcg/mL sufentanil base).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of sufentanil. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.1 Addiction, Abuse, and Misuse
Sufentanil Citrate Injection contains sufentanil, a Schedule II controlled substance. As an opioid, Sufentanil Citrate Injection exposes users to the risks of addiction, abuse, and misuse [see Drug Abuse and Dependence (9)].
Opioids are sought for nonmedical use and are subject to diversion from legitimate prescribed use. Consider these risks when handling Sufentanil Citrate Injection. Strategies to reduce these risks include proper product storage and control practices for a C-II drug. Contact local state professional licensing board or state-controlled substances authority for information on how to prevent and detect abuse or diversion of this product.
17 Patient Counseling Information
Addiction, Abuse, and Misuse
Inform patients that the use of Sufentanil Citrate Injection, even when taken as recommended, can result in addiction, abuse, and misuse, which can lead to overdose and death [see Warnings and Precautions (5.1)].
Life-Threatening Respiratory Depression
Inform patients of the risk of life-threatening respiratory depression, including information that the risk is greatest when starting Sufentanil Citrate Injection or when the dosage is increased, and that it can occur even at recommended dosages.
Hyperalgesia and Allodynia
Advise patients to inform their healthcare provider if they experience symptoms of hyperalgesia, including worsening pain, increased sensitivity to pain, or new pain [see Warnings and Precautions (5.7), Adverse Reactions (6)].
Serotonin Syndrome
Inform patients that opioids could cause a rare but potentially life-threatening condition called serotonin syndrome resulting from concomitant administration of serotonergic drugs. Warn patients of the symptoms of serotonin syndrome and to seek medical attention right away if symptoms develop after discharge from the hospital. Instruct patients to inform their healthcare provider if they are taking, or plan to take serotonergic medications [see Warnings and Precautions (5.8), Drug Interactions (7)].
Constipation
Advise patients of the potential for severe constipation, including management instructions and when to seek medical attention [see Adverse Reactions (6), Clinical Pharmacology (12.2)].
16 How Supplied/storage and Handling
Sufentanil Citrate Injection, USP equivalent to 50 mcg/mL sufentanil is supplied in the following single-dose containers:
|
Unit of Sale |
Concentration |
|
NDC 0409-3382-21 Carton of 10 Single-dose Fliptop Vials |
50 mcg/mL |
|
NDC 0409-3382-22 Carton of 10 Single-dose Fliptop Vials |
100 mcg/2 mL (50 mcg/mL) |
|
NDC 0409-3382-25 Carton of 10 Single-dose Fliptop Vials |
250 mcg/5 mL (50 mcg/mL) |
5.6 Severe Cardiovascular Depression
Sufentanil Citrate Injection may cause severe bradycardia, severe hypotension including orthostatic hypotension, and syncope. There is increased risk in patients whose ability to maintain blood pressure has already been compromised by a reduced blood volume or concurrent administration of certain CNS depressant drugs (e.g., phenothiazines or general anesthetics) [see Drug Interactions (7)]. In patients with circulatory shock, Sufentanil Citrate Injection may cause vasodilation that can further reduce cardiac output and blood pressure. Monitor these patients for signs of hypotension after initiating or titrating the dosage of Sufentanil Citrate Injection.
2.3 Epidural Use in Labor and Delivery
Proper placement of the needle or catheter in the epidural space should be verified before sufentanil citrate is injected to assure that unintentional intravascular or intrathecal administration does not occur. Unintentional intravascular injection of sufentanil could result in a potentially serious overdose, including acute truncal muscular rigidity and apnea. Unintentional intrathecal injection of the full sufentanil, bupivacaine epidural doses and volume could produce effects of high spinal anesthesia including prolonged paralysis and delayed recovery.
-
•Sufentanil should be administered by slow injection. Respiration should be closely monitored following each administration of an epidural injection of sufentanil.
-
•If analgesia is inadequate, the placement and integrity of the catheter should be verified prior to the administration of any additional epidural medications.
Principal Display Panel 1 Ml Vial Label
1 mL Single-dose
NDC 0409-3382-11
Preservative-Free
Rx only
SUFENTANIL CITRATE
Injection, USP
50 mcg/mL
CII
For Intravenous and Epidural Use.
Distributed by Hospira, Inc.,
Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 2 Ml Vial Label
2 mL Single-dose
NDC 0409-3382-12
Preservative-Free
Rx only
SUFENTANIL
CITRATE Inj., USP
CII
100 mcg/2 mL (50 mcg/mL)
For Intravenous and Epidural Use.
Distributed by Hospira, Inc.,
Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 5 Ml Vial Label
5 mL Single-dose
Preservative-Free
NDC 0409-3382-15
SUFENTANIL CITRATE
Injection, USP
CII
250 mcg/5 mL
(50 mcg/mL)
Rx only
For Intravenous
and Epidural
Use. Protect
from light.
Retain in
carton until
time of use.
Distributed by
Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 1 Ml Vial Carton
1 mL
10 Single-dose Fliptop Vials
Preservative-Free
Rx only
NDC 0409-3382-21
Contains 10 of NDC 0409-3382-11
Protect from light.
Retain in carton
until time of use.
CII
SUFENTANIL CITRATE Inj., USP
50 mcg/mL*
For Intravenous and Epidural Use.
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 2 Ml Vial Carton
2 mL
10 Single-dose
Fliptop Vials
Preservative-Free
Rx only
NDC 0409-3382-22
Contains 10 of NDC 0409-3382-12
Protect from light.
Retain in carton
until time of use.
CII
SUFENTANIL CITRATE Inj., USP
100 mcg/2 mL (50 mcg/mL)*
For Intravenous and Epidural Use.
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 5 Ml Vial Carton
5 mL
10 Single-dose Fliptop Vials
Preservative-Free
Rx only
NDC 0409-3382-25
Contains 10 of NDC 0409-3382-15
SUFENTANIL CITRATE Inj., USP
CII
250 mcg/5 mL (50 mcg/mL)*
Protect from light. Retain in carton until time of use.
For Intravenous and Epidural Use.
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
5.2 Life Threatening Respiratory Depression
Serious, life-threatening, or fatal respiratory depression has been reported with the use of opioids, even when used as recommended. Respiratory depression, if not immediately recognized and treated, may lead to respiratory arrest and death. Sufentanil Citrate Injection should be administered only by persons specifically trained in the use of anesthetic drugs and the management of the respiratory effects of potent opioids, including respiration and cardiac resuscitation of patients in the age group being treated. Such training must include the establishment and maintenance of a patent airway and assisted ventilation. Adequate facilities should be available for postoperative monitoring and ventilation of patients administered anesthetic doses of Sufentanil Citrate Injection. It is essential that these facilities be fully equipped to handle all degrees of respiratory depression. Management of respiratory depression may include close observation, supportive measures, and use of opioid antagonists, depending on the patient's clinical status [see Overdosage (10)]. Carbon dioxide (CO2) retention from opioid-induced respiratory depression can exacerbate the sedating effects of opioids.
As with other potent opioids, the respiratory depressant effect of Sufentanil Citrate Injection may persist longer than the measured analgesic effect. The total dose of all opioid agonists administered should be considered by the practitioner before ordering opioid analgesics during recovery from anesthesia.
Certain forms of conduction anesthesia, such as spinal anesthesia and some epidural anesthetics, can alter respiration by blocking intercostal nerves [see Clinical Pharmacology (12.2)]. Sufentanil Citrate Injection can also alter respiration. Therefore, when Sufentanil Citrate Injection is used to supplement these forms of anesthesia, the anesthetist should be familiar with the physiological alterations involved, and be prepared to manage them in the patients selected for these forms of anesthesia.
Patients with significant chronic obstructive pulmonary disease or cor pulmonale, and those with a substantially decreased respiratory reserve, hypoxia, hypercapnia, or pre-existing respiratory depression are at increased risk of decreased respiratory drive including apnea, even at recommended dosages of Sufentanil Citrate Injection. Elderly, cachectic, or debilitated patients may have altered pharmacokinetics or altered clearance compared to younger, healthier patients resulting in greater risk for respiratory depression.
Monitor such patients closely including vital signs, particularly when initiating and titrating Sufentanil Citrate Injection and when Sufentanil Citrate Injection is given concomitantly with other drugs that depress respiration. To reduce the risk of respiratory depression, proper dosing and titration of Sufentanil Citrate Injection are essential [see Dosage and Administration (2.1)].
Opioids can cause sleep-related breathing disorders including central sleep apnea (CSA) and sleep-related hypoxemia. Opioid use increases the risk of CSA in a dose-dependent fashion. In patients who present with CSA, consider decreasing the opioid dosage using best practices for opioid taper [see Dosage and Administration (2.1)].
5.9 Risks Due to Improper Epidural Injection
Proper placement of the needle or catheter in the epidural space should be verified before sufentanil is injected to assure that unintentional intravascular or intrathecal administration does not occur. Unintentional intravascular injection of sufentanil could result in a potentially serious overdose, including acute truncal muscular rigidity and apnea. Unintentional intrathecal injection of the full sufentanil/bupivacaine epidural doses and volume could produce effects of high spinal anesthesia including prolonged paralysis and delayed recovery. If analgesia is inadequate, the placement and integrity of the catheter should be verified prior to the administration of any additional epidural medications. Sufentanil should be administered epidurally by slow injection.
5.13 Risks of Driving and Operating Machinery
Sufentanil Citrate Injection may impair the mental or physical abilities needed to perform potentially hazardous activities such as driving a car or operating machinery. Warn patients not to drive or operate dangerous machinery unless they are tolerant to the effects of Sufentanil Citrate Injection and know how they will react to the medication.
5.7 Opioid Induced Hyperalgesia and Allodynia
Opioid-Induced Hyperalgesia (OIH) occurs when an opioid analgesic paradoxically causes an increase in pain, or an increase in sensitivity to pain. This condition differs from tolerance, which is the need for increasing doses of opioids to maintain a defined effect [see Dependence (9.3)]. Symptoms of OIH include (but may not be limited to) increased levels of pain upon opioid dosage increase, decreased levels of pain upon opioid dosage decrease, or pain from ordinarily non-painful stimuli (allodynia). These symptoms may suggest OIH only if there is no evidence of underlying disease progression, opioid tolerance, opioid withdrawal, or addictive behavior.
Cases of OIH have been reported, both with short-term and longer-term use of opioid analgesics. Though the mechanism of OIH is not fully understood, multiple biochemical pathways have been implicated. Medical literature suggests a strong biologic plausibility between opioid analgesics and OIH and allodynia. If a patient is suspected to be experiencing OIH, carefully consider appropriately decreasing the dose of the current opioid analgesic or opioid rotation (safely switching the patient to a different opioid moiety) [see Dosage and Administration (2), Warnings and Precautions (5.2)].
2.1 Important Dosage and Administration Instructions
Sufentanil Citrate Injection should be administered only by persons specifically trained in the use of intravenous or epidural anesthetics and management of the respiratory effects of potent opioids.
In patients administered high doses of Sufentanil Citrate Injection, it is essential that qualified personnel and adequate facilities are available for the management of postoperative respiratory depression.
For purposes of administering small volumes of Sufentanil Citrate Injection accurately, the use of a tuberculin syringe or equivalent is recommended.
-
•Ensure that an opioid antagonist, resuscitative and intubation equipment, and oxygen are readily available.
-
•Individualize dosage based on factors such as age, body weight, physical status, underlying pathological condition, use of other drugs, type of anesthesia to be used, and the surgical procedure involved.
-
•Monitor vital signs regularly.
-
•The selection of preanesthetic medications should be based upon the needs of the individual patient.
-
•The neuromuscular blocking agent selected should be compatible with the patient's condition, taking into account the hemodynamic effects of a particular muscle relaxant and the degree of skeletal muscle relaxation required.
As with other potent opioids, the respiratory depressant effect of sufentanil may persist longer than the measured analgesic effect. The total dose of all opioid agonists administered should be considered by the practitioner before ordering opioid analgesics during recovery from anesthesia.
If Sufentanil Citrate Injection is administered with a CNS depressant, become familiar with the properties of each drug, particularly each product's duration of action. In addition, when such a combination is used, fluids and other countermeasures to manage hypotension should be available [see Warnings and Precautions (5.3) ].
Inspect parenteral drug products visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
5.5 Risks of Muscle Rigidity and Skeletal Muscle Movement
Intravenous administration or unintentional intravascular injection during epidural administration of Sufentanil Citrate Injection may cause muscle rigidity, particularly involving the muscles of respiration. The incidence and severity of muscle rigidity is dose related. These effects are related to the dose and speed of injection. Administration of sufentanil may produce muscular rigidity with a more rapid onset of action than that seen with fentanyl. Skeletal muscle rigidity also has been reported to occur or recur infrequently in the extended postoperative period usually following high dose administration. In addition, skeletal muscle movements of various groups in the extremities, neck, and external eye have been reported during induction of anesthesia with Sufentanil Citrate Injection; these reported movements have, on rare occasions, been strong enough to pose patient management problems.
The incidence of skeletal muscle rigidity can be reduced by: 1) administration of up to 1/4 of the full paralyzing dose of a non-depolarizing neuromuscular blocking agent just prior to administration of sufentanil at dosages of up to 8 mcg/kg, 2) administration of a full paralyzing dose of a neuromuscular blocking agent following loss of consciousness when sufentanil is used in anesthetic dosages (above 8 mcg/kg) titrated by slow intravenous infusion, or, 3) simultaneous administration of sufentanil and a full paralyzing dose of a neuromuscular blocking agent when sufentanil is used in rapidly administered anesthetic dosages (above 8 mcg/kg).
The neuromuscular blocking agents used should be compatible with the patient's cardiovascular status. The hemodynamic effects and degree of skeletal muscle relaxation required should be considered in the selection of a neuromuscular blocking agent. High doses of pancuronium may produce increases in heart rate during sufentanil-oxygen anesthesia. Bradycardia and hypotension have been reported with other muscle relaxants during sufentanil-oxygen anesthesia; this effect may be more pronounced in the presence of calcium channel and/or beta-blockers. Muscle relaxants with no clinically significant effect on heart rate (at recommended doses) would not counteract the vagotonic effect of sufentanil, therefore a lower heart rate would be expected. Rare reports of bradycardia associated with the concomitant use of succinylcholine and sufentanil have been reported.
5.11 Risks of Use in Patients With Gastrointestinal Conditions
Sufentanil may cause spasm of the sphincter of Oddi. Opioids may cause increases in serum amylase. Monitor patients with biliary tract disease, including acute pancreatitis for worsening symptoms.
5.8 Serotonin Syndrome With Concomitant Use of Serotonergic Drugs
Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of sufentanil with serotonergic drugs. Serotonergic drugs include selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that affect the serotonergic neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), and drugs that impair metabolism of serotonin (including MAO inhibitors, both those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue) [see Drug Interactions (7)]. This may occur within the recommended dosage range.
Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination, rigidity), and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea) and can be fatal. The onset of symptoms generally occurs within several hours to a few days of concomitant use, but may occur later than that. Discontinue Sufentanil Citrate Injection if serotonin syndrome is suspected.
5.12 Increased Risk of Seizures in Patients With Seizure Disorders
Sufentanil may increase the frequency of seizures in patients with seizure disorders, and may increase the risk of seizures occurring in other clinical settings associated with seizures. Monitor patients with a history of seizure disorders for worsened seizure control during Sufentanil Citrate Injection therapy.
5.3 Risks From Concomitant Use With Benzodiazepines Or Other Cns Depressants
When benzodiazepines or other CNS depressants are used with Sufentanil Citrate Injection, pulmonary arterial pressure may be decreased. This fact should be considered by those who conduct diagnostic and surgical procedures where interpretation of pulmonary arterial pressure measurements might determine final management of the patient. When high dose or anesthetic dosages of Sufentanil Citrate Injection are employed, even relatively small dosages of diazepam may cause cardiovascular depression.
When Sufentanil Citrate Injection is used with CNS depressants, hypotension can occur. If it occurs, consider the possibility of hypovolemia and manage with appropriate parenteral fluid therapy. When operative conditions permit, consider repositioning the patient to improve venous return to the heart. Exercise care in moving and repositioning of patients because of the possibility of orthostatic hypotension. If volume expansion with fluids plus other countermeasures do not correct hypotension, consider administration of pressor agents other than epinephrine. Epinephrine may paradoxically decrease blood pressure in patients treated with a neuroleptic that blocks alpha adrenergic activity.
Profound sedation, respiratory depression, coma, and death may result from the concomitant use of Sufentanil Citrate Injection with benzodiazepines and/or other CNS depressants including alcohol (e.g., nonbenzodiazepine sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, other opioids).
Observational studies have demonstrated that concomitant use of opioid analgesics and benzodiazepines increases the risk of drug-related mortality compared to use of opioid analgesics alone. Because of similar pharmacological properties, it is reasonable to expect similar risk with the concomitant use of other CNS depressant drugs with opioid analgesics [see Drug Interactions (7)].
If the decision is made to manage postoperative pain with Sufentanil Citrate Injection concomitantly with a benzodiazepine or other CNS depressant, start dosing with the lowest effective dosage and titrate based on clinical response. Monitor patients closely for signs and symptoms of respiratory depression, sedation, and hypotension. Fluids or other measures to counter hypotension should be available [see Drug Interactions (7)].
Warning: serious and Life Threatening Risks From Use of Sufentanil Citrate injection
Addiction, Abuse, and Misuse
Because the use of Sufentanil Citrate Injection exposes patients and other users to the risks of opioid addiction, abuse, and misuse, which can lead to overdose and death, assess each patient's risk prior to prescribing and reassess all patients regularly for the development of these behaviors and conditions [see Warnings and Precautions (5.1)] .
Life-Threatening Respiratory Depression
Serious, life-threatening, or fatal respiratory depression may occur with use of Sufentanil Citrate Injection, especially during initiation or following a dosage increase. To reduce the risk of respiratory depression, proper dosing and titration of Sufentanil Citrate Injection are essential [see Warnings and Precautions (5.2)].
Risks from Concomitant Use with Benzodiazepines or Other CNS Depressants
Concomitant use of opioids with benzodiazepines or other central nervous system (CNS) depressants, including alcohol, may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing of Sufentanil Citrate Injection and benzodiazepines or other CNS depressants for use in patients for whom alternative treatment options are inadequate [see Warnings and Precautions (5.3), Drug Interactions (7) ].
Cytochrome P450 3A4 Interaction
The concomitant use of Sufentanil Citrate Injection with all cytochrome P450 3A4 inhibitors may result in an increase in sufentanil plasma concentrations, which could increase or prolong adverse reactions and may cause potentially fatal respiratory depression. In addition, discontinuation of a concomitantly used cytochrome P450 3A4 inducer may result in an increase in sufentanil plasma concentration. Monitor patients receiving Sufentanil Citrate Injection and any CYP3A4 inhibitor or inducer [see Warnings and Precautions (5.4), Drug Interactions (7), Clinical Pharmacology (12.3)] .
5.4 Risks of Concomitant Use Or Discontinuation of Cytochrome P450 3a4 Inhibitors and Inducers
Concomitant use of Sufentanil Citrate Injection with a CYP3A4 inhibitor, such as macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g., ketoconazole), and protease inhibitors (e.g., ritonavir), may increase plasma concentrations of sufentanil and prolong opioid adverse reactions, which may exacerbate fatal respiratory depression [see Warnings and Precautions (5.2)], particularly when an inhibitor is added after a stable dose of Sufentanil Citrate Injection is achieved. Similarly, discontinuation of a CYP3A4 inducer, such as rifampin, carbamazepine, and phenytoin, in Sufentanil Citrate Injection-treated patients may increase sufentanil plasma concentrations and prolong opioid adverse reactions. When using Sufentanil Citrate Injection with CYP3A4 inhibitors or discontinuing CYP3A4 inducers in Sufentanil Citrate Injection-treated patients, monitor patients closely at frequent intervals and consider dosage reduction of Sufentanil Citrate Injection [see Dosage and Administration (2.1), Drug Interactions (7)].
Concomitant use of Sufentanil Citrate Injection with CYP3A4 inducers or discontinuation of an CYP3A4 inhibitor could result in lower than expected sufentanil plasma concentrations, decreased efficacy, or, possibly, lead to a withdrawal syndrome in a patient who had developed physical dependence to sufentanil. When using Sufentanil Citrate Injection with CYP3A4 inducers or discontinuing CYP3A4 inhibitors, monitor patients closely at frequent intervals and consider increasing the Sufentanil Citrate Injection dosage [see Dosage and Administration (2.1), Drug Interactions (7)].
5.10 Risks of Use in Patients With Increased Intracranial Pressure, Brain Tumors, Or Head Injury
In patients who may be susceptible to the intracranial effects of CO2 retention (e.g., those with evidence of increased intracranial pressure or brain tumors), Sufentanil Citrate Injection may reduce respiratory drive, and the resultant CO2 retention can further increase intracranial pressure. Monitor such patients for signs of sedation and respiratory depression, particularly when initiating therapy with Sufentanil Citrate Injection. Opioids may also obscure the clinical course in a patient with a head injury.
Structured Label Content
Section 42229-5 (42229-5)
Adjunct to general anesthesia:
-
•Doses of up to 8 mcg/kg (see Table 1)
-
•Total Dosage Requirements of 1 mcg/kg/hr or less are recommended
-
•Dosage should be individualized and adjusted to remaining operative time anticipated.
| Total dosage | Maintenance dosage |
|---|---|
|
Duration of anesthesia 1 to 2 hours |
|
|
Incremental or Infusion: 1 to 2 mcg/kg
|
Incremental: 10 to 25 mcg (0.2 to 0.5 mL) may be administered in increments as needed when movement and/or changes in vital signs indicate surgical stress or lightening of analgesia. Supplemental dosages should be individualized and adjusted to remaining operative time anticipated. |
|
Duration of anesthesia 2 to 8 hours |
|
|
Incremental or Infusion: 2 to 8 mcg/kg
|
Incremental: 10 to 50 mcg (0.2 to 1 mL) may be administered in increments as needed when movement and/or changes in vital signs indicate surgical stress or lightening of analgesia. Supplemental dosages should be individualized and adjusted to the remaining operative time anticipated. |
Section 43683-2 (43683-2)
|
Boxed Warning |
12/2023 |
|
Warnings and Precautions (5.7) |
12/2023 |
Section 44425-7 (44425-7)
Protect from light. Retain in carton until time of use.
Store at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature.]
9.2 Abuse
Sufentanil Citrate Injection contains sufentanil, a substance with high potential for misuse and abuse, which can lead to the development of substance use disorder, including addiction [see Warnings and Precautions (5.1)].
Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a healthcare provider or for whom it was not prescribed.
Abuse is the intentional, non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects.
Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence.
Misuse and abuse of Sufentanil Citrate Injection increases risk of overdose, which may lead to central nervous system and respiratory depression, hypotension, seizures, and death. The risk is increased with concurrent abuse of Sufentanil Citrate Injection with alcohol and/or other CNS depressants. Abuse of and addiction to opioids in some individuals may not be accompanied by concurrent tolerance and symptoms of physical dependence. In addition, abuse of opioids can occur in the absence of addiction.
All patients treated with opioids require careful and frequent reevaluation for signs of misuse, abuse, and addiction, because use of opioid analgesic products carries the risk of addiction even under appropriate medical use. Patients at high risk of Sufentanil Citrate Injection abuse include those with a history of prolonged use of any opioid, including products containing sufentanil, those with a history of drug or alcohol abuse, or those who use Sufentanil Citrate Injection in combination with other abused drugs.
“Drug-seeking” behavior is very common in persons with substance use disorders. Drug-seeking tactics include emergency calls or visits near the end of office hours, refusal to undergo appropriate examination, testing, or referral, repeated “loss” of prescriptions, tampering with prescriptions, and reluctance to provide prior medical records or contact information for other treating healthcare provider(s). “Doctor shopping” (visiting multiple prescribers to obtain additional prescriptions) is common among people who abuse drugs and people with substance use disorder. Preoccupation with achieving adequate pain relief can be appropriate behavior in a patient with inadequate pain control.
Sufentanil Citrate Injection, like other opioids, can be diverted for nonmedical use into illicit channels of distribution. Careful record-keeping of prescribing information, including quantity, frequency, and renewal requests, as required by state and federal law, is strongly advised.
Proper assessment of the patient, proper prescribing practices, periodic reevaluation of therapy, and proper dispensing and storage are appropriate measures that help to limit abuse of opioid drugs.
11 Description (11 DESCRIPTION)
Sufentanil Citrate Injection, USP is a sterile, nonpyrogenic solution of sufentanil citrate in water for injection. Sufentanil Citrate is a potent opioid analgesic which is administered either epidurally or by intravenous injection.
Each mL contains sufentanil citrate equivalent to 50 mcg of sufentanil. May contain sodium hydroxide and/or hydrochloric acid for pH adjustment. pH 4.2 (3.5 to 6.0).
The solution contains no bacteriostat, antimicrobial agent or added buffer and is intended for use only as a single-use injection. When smaller doses are required, the unused portion should be discarded in an appropriate manner.
Sufentanil Citrate, USP, occurs as a white crystalline powder and is chemically designated as N-[-4-(methyoxymethyl)-1-[2-(2-thienyl)ethyl]-4-piperidinyl]-N-phenylpropanamide 2-hydroxy-1,2,3-propanetricarboxylate (1:1).
The molecular formula of sufentanil citrate is C22H30N2O2S∙C6H8O7 and the molecular weight is 578.69. Sufentanil Citrate has the following structural formula:
9.3 Dependence
Both tolerance and physical dependence can develop during use of opioid therapy.
Tolerance is a physiological state characterized by a reduced response to a drug after repeated administration (i.e., a higher dose of a drug is required to produce the same effect that was once obtained at a lower dose).
Physical dependence is a state that develops as a result of a physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug.
Withdrawal may be precipitated through the administration of drugs with opioid antagonist activity (e.g., naloxone), mixed agonist/antagonist analgesics (e.g., pentazocine, butorphanol, nalbuphine), or partial agonists (e.g., buprenorphine). Physical dependence may not occur to a clinically significant degree until after several days to weeks of continued use.
Sufentanil Citrate Injection should not be abruptly discontinued in a physically-dependent patient. If Sufentanil Citrate Injection is abruptly discontinued in a physically-dependent patient, a withdrawal syndrome may occur, typically characterized by restlessness, lacrimation, rhinorrhea, perspiration, chills, myalgia, and mydriasis. Other signs and symptoms also may develop, including irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory rate, or heart rate.
Infants born to mothers physically-dependent on opioids will also be physically-dependent and may exhibit respiratory difficulties and withdrawal signs [see Use in Specific Populations (8.1)].
8.4 Pediatric Use
The safety and efficacy of intravenous sufentanil in pediatric patients as young as 1 day old undergoing cardiovascular surgery have been documented in a limited number of cases. The clearance of sufentanil in healthy neonates is approximately one-half that in adults and children. The clearance rate of sufentanil can be further reduced by up to a third in neonates with cardiovascular disease, resulting in an increase in the elimination half-life of the drug.
8.5 Geriatric Use
Elderly patients (aged 65 years or older) may have increased sensitivity to sufentanil. In general, use caution when selecting a dosage for an elderly patient, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
Respiratory depression is the chief risk for elderly patients treated with opioids, and has occurred after large initial doses were administered to patients who were not opioid-tolerant or when opioids were co-administered with other agents that depress respiration. Titrate the dosage of Sufentanil Citrate Injection slowly in geriatric patients and monitor closely for signs of central nervous system and respiratory depression [see Warnings and Precautions (5.2) ].
2.2 Intravenous Use (2.2 Intravenous use)
Sufentanil Citrate may be administered intravenously by slow injection or infusion.
4 Contraindications (4 CONTRAINDICATIONS)
Sufentanil Citrate Injection is contraindicated in patients with:
-
•Hypersensitivity to sufentanil (e.g., anaphylaxis) [see Adverse Reactions (6.2)]
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described, or described in greater detail, in other sections:
-
•Addiction, Abuse, and Misuse [see Warnings and Precautions (5.1)]
-
•Life-Threatening Respiratory Depression [see Warnings and Precautions (5.2)]
-
•Skeletal Muscle Rigidity and Skeletal Muscle Movement [see Warnings and Precautions (5.5) ]
-
•Interactions with Benzodiazepines or Other CNS Depressants [see Warnings and Precautions (5.3)]
-
•Severe Cardiovascular Depression [see Warnings and Precautions (5.6)]
-
•Opioid-Induced Hyperalgesia and Allodynia [see Warnings and Precautions (5.7)]
-
•Serotonin Syndrome [see Warnings and Precautions (5.8) ]
-
•Gastrointestinal Adverse Reactions [see Warnings and Precautions (5.11) ]
-
• Seizures [see Warnings and Precautions (5.12) ]
7 Drug Interactions (7 DRUG INTERACTIONS)
Table 3 includes clinically significant drug interactions with Sufentanil Citrate Injection.
|
Inhibitors of CYP3A4 |
|
|
Clinical Impact: |
The concomitant use of Sufentanil Citrate Injection and CYP3A4 inhibitors can increase the plasma concentration of sufentanil, resulting in increased or prolonged opioid effects, particularly when an inhibitor is added after a stable dose of Sufentanil Citrate Injection is achieved [see Warnings and Precautions (5.4)]. |
|
Intervention: |
If concomitant use is necessary, consider dosage reduction of Sufentanil Citrate Injection until stable drug effects are achieved. Monitor patients at frequent intervals for respiratory depression and sedation. |
|
Examples: |
Macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g., ketoconazole), protease inhibitors (e.g., ritonavir), grapefruit juice. |
|
CYP3A4 Inducers |
|
|
Clinical Impact: |
The concomitant use of Sufentanil Citrate Injection and CYP3A4 inducers can decrease the plasma concentration of sufentanil [see Clinical Pharmacology (12.3)], resulting in decreased efficacy or onset of a withdrawal syndrome in patients who have developed physical dependence to sufentanil [see Warnings and Precautions (5.4)]. |
|
Intervention: |
If concomitant use is necessary, consider increasing the Sufentanil Citrate Injection dosage until stable drug effects are achieved. Monitor for signs of opioid withdrawal. If a CYP3A4 inducer is discontinued, consider Sufentanil Citrate Injection dosage reduction and monitor for signs of respiratory depression. |
|
Examples: |
Rifampin, carbamazepine, phenytoin |
|
Benzodiazepines and Other Central Nervous System (CNS) Depressants |
|
|
Clinical Impact: |
The concomitant use of Sufentanil Citrate Injection with CNS depressants my result in decreased pulmonary artery pressure and may cause hypotension. Even small dosages of diazepam may cause cardiovascular depression when added to high dose or anesthetic dosages of Sufentanil Citrate Injection. As postoperative analgesia, concomitant use of Sufentanil Citrate Injection can increase the risk of hypotension, respiratory depression, profound sedation, coma, and death [see Warnings and Precautions (5.2, 5.3)]. |
|
Intervention: |
As postoperative analgesia, start with a lower dose of Sufentanil Citrate Injection and monitor patients for signs of respiratory depression, sedation, and hypotension. Fluids or other measures to counter hypotension should be available [see Warnings and Precautions (5.3) ]. |
|
Examples: |
Benzodiazepines and other sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, other opioids, alcohol. |
|
Serotonergic Drugs |
|
|
Clinical Impact: |
The concomitant use of opioids with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome [see Warnings and Precautions (5.8) ]. |
|
Intervention: |
If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dose adjustment. Discontinue Sufentanil Citrate Injection if serotonin syndrome is suspected. |
|
Examples: |
Selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5‑HT3 receptor antagonists, drugs that effect the serotonin neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), monoamine oxidase inhibitors (those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue). |
|
Monoamine Oxidase Inhibitors (MAOIs) |
|
|
Clinical Impact: |
MAOI interactions with opioids may manifest as serotonin syndrome or opioid toxicity (e.g., respiratory depression, coma) [see Warnings and Precautions (5.2)]. |
|
Intervention: |
The use of Sufentanil Citrate Injection is not recommended for patients taking MAOIs or within 14 days of stopping such treatment. |
|
Examples: |
phenelzine, tranylcypromine, linezolid |
|
Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics |
|
|
Clinical Impact: |
May reduce the analgesic effect of Sufentanil Citrate Injection and/or precipitate withdrawal symptoms. |
|
Intervention: |
Avoid concomitant use. |
|
Examples: |
butorphanol, nalbuphine, pentazocine, buprenorphine |
|
Muscle Relaxants |
|
|
Clinical Impact: |
Sufentanil may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression. |
|
Intervention: |
Monitor patients for signs of respiratory depression that may be greater than otherwise expected and decrease the dosage of Sufentanil Citrate Injection and/or the muscle relaxant as necessary. |
|
Diuretics |
|
|
Clinical Impact: |
Opioids can reduce the efficacy of diuretics by inducing the release of antidiuretic hormone. |
|
Intervention: |
Monitor patients for signs of diminished diuresis and/or effects on blood pressure and increase the dosage of the diuretic as needed. |
|
Anticholinergic Drugs |
|
|
Clinical Impact: |
The concomitant use of anticholinergic drugs may increase risk of urinary retention and/or severe constipation, which may lead to paralytic ileus. |
|
Intervention: |
Monitor patients for signs of urinary retention or reduced gastric motility when Sufentanil Citrate Injection is used concomitantly with anticholinergic drugs. |
|
Nitrous oxide |
|
|
Clinical Impact: |
Nitrous oxide has been reported to produce cardiovascular depression when given with higher doses of Sufentanil Citrate Injection. |
|
Intervention: |
Monitor patients for signs of cardiovascular depression that may be greater than otherwise expected. |
8.7 Renal Impairment
Sufentanil Citrate Injection should be administered with caution to patients with kidney dysfunction because of the renal excretion of sufentanil citrate and its metabolites. Reduce the dosage as needed and monitor for signs of respiratory depression, sedation, and hypotension.
12.3 Pharmacokinetics
Sufentanil Citrate Injection is administered by the intravenous or epidural route. The pharmacokinetics of intravenous sufentanil can be described as a three-compartment model.
8.6 Hepatic Impairment
Sufentanil Citrate Injection should be administered with caution to patients with liver dysfunction because of the extensive hepatic metabolism. Reduce the dosage as needed and monitor closely for signs of respiratory depression, sedation, and hypotension.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Sufentanil Citrate Injection is indicated for intravenous administration in adults and pediatric patients:
-
•as an analgesic adjunct in the maintenance of balanced general anesthesia in patients who are intubated and ventilated.
-
•as a primary anesthetic agent for the induction and maintenance of anesthesia with 100% oxygen in patients undergoing major surgical procedures, in patients who are intubated and ventilated, such as cardiovascular surgery or neurosurgical procedures in the sitting position, to provide favorable myocardial and cerebral oxygen balance or when extended postoperative ventilation is anticipated.
Sufentanil Citrate Injection is indicated for epidural administration:
-
•as an analgesic combined with low dose (usually 12.5 mg per administration) bupivacaine usually during labor and vaginal delivery.
12.1 Mechanism of Action
Sufentanil is an opioid agonist. When used in balanced general anesthesia, sufentanil has been reported to be as much as 10 times as potent as fentanyl. When administered intravenously as a primary anesthetic agent with 100% oxygen, sufentanil is approximately 5 to 7 times as potent as fentanyl.
9.1 Controlled Substance
Sufentanil Citrate Injection contains sufentanil, a Schedule II controlled substance.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Life-Threatening Respiratory Depression in Patients with Chronic Pulmonary Disease or in Elderly, Cachectic, or
Debilitated Patients: Monitor closely, particularly during initiation and titration. (5.2) -
•Risks of Skeletal Muscle Rigidity and Skeletal Muscle Movement: Manage with neuromuscular blocking agent. See full prescribing information for more detail on managing these risks. (5.5)
-
•Severe Cardiovascular Depression: Monitor during dosage initiation and titration. (5.6)
-
•Opioid-Induced Hyperalgesia and Allodynia: Opioid-Induced Hyperalgesia (OIH) occurs when an opioid analgesic paradoxically causes an increase in pain, or an increase in sensitivity to pain. If OIH is suspected, carefully consider appropriately decreasing the dose of the current opioid analgesic, or opioid rotation. (5.7)
-
•Serotonin Syndrome: Potentially life-threatening condition could result from concomitant serotonergic drug administration. Discontinue Sufentanil Citrate Injection if serotonin syndrome is suspected. (5.8)
-
•Risks of Use in Patients with Increased Intracranial Pressure, Brain Tumors, or Head Injury: Monitor for sedation and respiratory depression. (5.10)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Sufentanil Citrate Injection should be administered only by persons specifically trained in the use of intravenous anesthetics and management of the respiratory effects of potent opioids.
-
•Ensure that an opioid antagonist, resuscitative and intubation equipment, and oxygen are readily available. (2.1)
-
•Individualize dosing based on factors such as age, body weight, physical status, underlying pathological condition, use of other drugs, type of anesthesia to be used, and the surgical procedure involved. (2.1)
-
•Initiate analgesic treatment with 1 to 2 mcg/kg intravenously. (2.2)
-
•Initiate epidural injection for labor and delivery at 10 to 15 mcg of Sufentanil administered with 10 mL bupivacaine 0.125% with or without epinephrine. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Sufentanil Citrate Injection, USP 50 mcg/mL (equivalent to 50 mcg/mL sufentanil base).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of sufentanil. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.1 Addiction, Abuse, and Misuse
Sufentanil Citrate Injection contains sufentanil, a Schedule II controlled substance. As an opioid, Sufentanil Citrate Injection exposes users to the risks of addiction, abuse, and misuse [see Drug Abuse and Dependence (9)].
Opioids are sought for nonmedical use and are subject to diversion from legitimate prescribed use. Consider these risks when handling Sufentanil Citrate Injection. Strategies to reduce these risks include proper product storage and control practices for a C-II drug. Contact local state professional licensing board or state-controlled substances authority for information on how to prevent and detect abuse or diversion of this product.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Addiction, Abuse, and Misuse
Inform patients that the use of Sufentanil Citrate Injection, even when taken as recommended, can result in addiction, abuse, and misuse, which can lead to overdose and death [see Warnings and Precautions (5.1)].
Life-Threatening Respiratory Depression
Inform patients of the risk of life-threatening respiratory depression, including information that the risk is greatest when starting Sufentanil Citrate Injection or when the dosage is increased, and that it can occur even at recommended dosages.
Hyperalgesia and Allodynia
Advise patients to inform their healthcare provider if they experience symptoms of hyperalgesia, including worsening pain, increased sensitivity to pain, or new pain [see Warnings and Precautions (5.7), Adverse Reactions (6)].
Serotonin Syndrome
Inform patients that opioids could cause a rare but potentially life-threatening condition called serotonin syndrome resulting from concomitant administration of serotonergic drugs. Warn patients of the symptoms of serotonin syndrome and to seek medical attention right away if symptoms develop after discharge from the hospital. Instruct patients to inform their healthcare provider if they are taking, or plan to take serotonergic medications [see Warnings and Precautions (5.8), Drug Interactions (7)].
Constipation
Advise patients of the potential for severe constipation, including management instructions and when to seek medical attention [see Adverse Reactions (6), Clinical Pharmacology (12.2)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Sufentanil Citrate Injection, USP equivalent to 50 mcg/mL sufentanil is supplied in the following single-dose containers:
|
Unit of Sale |
Concentration |
|
NDC 0409-3382-21 Carton of 10 Single-dose Fliptop Vials |
50 mcg/mL |
|
NDC 0409-3382-22 Carton of 10 Single-dose Fliptop Vials |
100 mcg/2 mL (50 mcg/mL) |
|
NDC 0409-3382-25 Carton of 10 Single-dose Fliptop Vials |
250 mcg/5 mL (50 mcg/mL) |
5.6 Severe Cardiovascular Depression
Sufentanil Citrate Injection may cause severe bradycardia, severe hypotension including orthostatic hypotension, and syncope. There is increased risk in patients whose ability to maintain blood pressure has already been compromised by a reduced blood volume or concurrent administration of certain CNS depressant drugs (e.g., phenothiazines or general anesthetics) [see Drug Interactions (7)]. In patients with circulatory shock, Sufentanil Citrate Injection may cause vasodilation that can further reduce cardiac output and blood pressure. Monitor these patients for signs of hypotension after initiating or titrating the dosage of Sufentanil Citrate Injection.
2.3 Epidural Use in Labor and Delivery
Proper placement of the needle or catheter in the epidural space should be verified before sufentanil citrate is injected to assure that unintentional intravascular or intrathecal administration does not occur. Unintentional intravascular injection of sufentanil could result in a potentially serious overdose, including acute truncal muscular rigidity and apnea. Unintentional intrathecal injection of the full sufentanil, bupivacaine epidural doses and volume could produce effects of high spinal anesthesia including prolonged paralysis and delayed recovery.
-
•Sufentanil should be administered by slow injection. Respiration should be closely monitored following each administration of an epidural injection of sufentanil.
-
•If analgesia is inadequate, the placement and integrity of the catheter should be verified prior to the administration of any additional epidural medications.
Principal Display Panel 1 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 1 mL Vial Label)
1 mL Single-dose
NDC 0409-3382-11
Preservative-Free
Rx only
SUFENTANIL CITRATE
Injection, USP
50 mcg/mL
CII
For Intravenous and Epidural Use.
Distributed by Hospira, Inc.,
Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 2 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 2 mL Vial Label)
2 mL Single-dose
NDC 0409-3382-12
Preservative-Free
Rx only
SUFENTANIL
CITRATE Inj., USP
CII
100 mcg/2 mL (50 mcg/mL)
For Intravenous and Epidural Use.
Distributed by Hospira, Inc.,
Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 5 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 5 mL Vial Label)
5 mL Single-dose
Preservative-Free
NDC 0409-3382-15
SUFENTANIL CITRATE
Injection, USP
CII
250 mcg/5 mL
(50 mcg/mL)
Rx only
For Intravenous
and Epidural
Use. Protect
from light.
Retain in
carton until
time of use.
Distributed by
Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 1 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 1 mL Vial Carton)
1 mL
10 Single-dose Fliptop Vials
Preservative-Free
Rx only
NDC 0409-3382-21
Contains 10 of NDC 0409-3382-11
Protect from light.
Retain in carton
until time of use.
CII
SUFENTANIL CITRATE Inj., USP
50 mcg/mL*
For Intravenous and Epidural Use.
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 2 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 2 mL Vial Carton)
2 mL
10 Single-dose
Fliptop Vials
Preservative-Free
Rx only
NDC 0409-3382-22
Contains 10 of NDC 0409-3382-12
Protect from light.
Retain in carton
until time of use.
CII
SUFENTANIL CITRATE Inj., USP
100 mcg/2 mL (50 mcg/mL)*
For Intravenous and Epidural Use.
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 5 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 5 mL Vial Carton)
5 mL
10 Single-dose Fliptop Vials
Preservative-Free
Rx only
NDC 0409-3382-25
Contains 10 of NDC 0409-3382-15
SUFENTANIL CITRATE Inj., USP
CII
250 mcg/5 mL (50 mcg/mL)*
Protect from light. Retain in carton until time of use.
For Intravenous and Epidural Use.
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
5.2 Life Threatening Respiratory Depression (5.2 Life-Threatening Respiratory Depression)
Serious, life-threatening, or fatal respiratory depression has been reported with the use of opioids, even when used as recommended. Respiratory depression, if not immediately recognized and treated, may lead to respiratory arrest and death. Sufentanil Citrate Injection should be administered only by persons specifically trained in the use of anesthetic drugs and the management of the respiratory effects of potent opioids, including respiration and cardiac resuscitation of patients in the age group being treated. Such training must include the establishment and maintenance of a patent airway and assisted ventilation. Adequate facilities should be available for postoperative monitoring and ventilation of patients administered anesthetic doses of Sufentanil Citrate Injection. It is essential that these facilities be fully equipped to handle all degrees of respiratory depression. Management of respiratory depression may include close observation, supportive measures, and use of opioid antagonists, depending on the patient's clinical status [see Overdosage (10)]. Carbon dioxide (CO2) retention from opioid-induced respiratory depression can exacerbate the sedating effects of opioids.
As with other potent opioids, the respiratory depressant effect of Sufentanil Citrate Injection may persist longer than the measured analgesic effect. The total dose of all opioid agonists administered should be considered by the practitioner before ordering opioid analgesics during recovery from anesthesia.
Certain forms of conduction anesthesia, such as spinal anesthesia and some epidural anesthetics, can alter respiration by blocking intercostal nerves [see Clinical Pharmacology (12.2)]. Sufentanil Citrate Injection can also alter respiration. Therefore, when Sufentanil Citrate Injection is used to supplement these forms of anesthesia, the anesthetist should be familiar with the physiological alterations involved, and be prepared to manage them in the patients selected for these forms of anesthesia.
Patients with significant chronic obstructive pulmonary disease or cor pulmonale, and those with a substantially decreased respiratory reserve, hypoxia, hypercapnia, or pre-existing respiratory depression are at increased risk of decreased respiratory drive including apnea, even at recommended dosages of Sufentanil Citrate Injection. Elderly, cachectic, or debilitated patients may have altered pharmacokinetics or altered clearance compared to younger, healthier patients resulting in greater risk for respiratory depression.
Monitor such patients closely including vital signs, particularly when initiating and titrating Sufentanil Citrate Injection and when Sufentanil Citrate Injection is given concomitantly with other drugs that depress respiration. To reduce the risk of respiratory depression, proper dosing and titration of Sufentanil Citrate Injection are essential [see Dosage and Administration (2.1)].
Opioids can cause sleep-related breathing disorders including central sleep apnea (CSA) and sleep-related hypoxemia. Opioid use increases the risk of CSA in a dose-dependent fashion. In patients who present with CSA, consider decreasing the opioid dosage using best practices for opioid taper [see Dosage and Administration (2.1)].
5.9 Risks Due to Improper Epidural Injection (5.9 Risks due to Improper Epidural Injection)
Proper placement of the needle or catheter in the epidural space should be verified before sufentanil is injected to assure that unintentional intravascular or intrathecal administration does not occur. Unintentional intravascular injection of sufentanil could result in a potentially serious overdose, including acute truncal muscular rigidity and apnea. Unintentional intrathecal injection of the full sufentanil/bupivacaine epidural doses and volume could produce effects of high spinal anesthesia including prolonged paralysis and delayed recovery. If analgesia is inadequate, the placement and integrity of the catheter should be verified prior to the administration of any additional epidural medications. Sufentanil should be administered epidurally by slow injection.
5.13 Risks of Driving and Operating Machinery
Sufentanil Citrate Injection may impair the mental or physical abilities needed to perform potentially hazardous activities such as driving a car or operating machinery. Warn patients not to drive or operate dangerous machinery unless they are tolerant to the effects of Sufentanil Citrate Injection and know how they will react to the medication.
5.7 Opioid Induced Hyperalgesia and Allodynia (5.7 Opioid-Induced Hyperalgesia and Allodynia)
Opioid-Induced Hyperalgesia (OIH) occurs when an opioid analgesic paradoxically causes an increase in pain, or an increase in sensitivity to pain. This condition differs from tolerance, which is the need for increasing doses of opioids to maintain a defined effect [see Dependence (9.3)]. Symptoms of OIH include (but may not be limited to) increased levels of pain upon opioid dosage increase, decreased levels of pain upon opioid dosage decrease, or pain from ordinarily non-painful stimuli (allodynia). These symptoms may suggest OIH only if there is no evidence of underlying disease progression, opioid tolerance, opioid withdrawal, or addictive behavior.
Cases of OIH have been reported, both with short-term and longer-term use of opioid analgesics. Though the mechanism of OIH is not fully understood, multiple biochemical pathways have been implicated. Medical literature suggests a strong biologic plausibility between opioid analgesics and OIH and allodynia. If a patient is suspected to be experiencing OIH, carefully consider appropriately decreasing the dose of the current opioid analgesic or opioid rotation (safely switching the patient to a different opioid moiety) [see Dosage and Administration (2), Warnings and Precautions (5.2)].
2.1 Important Dosage and Administration Instructions
Sufentanil Citrate Injection should be administered only by persons specifically trained in the use of intravenous or epidural anesthetics and management of the respiratory effects of potent opioids.
In patients administered high doses of Sufentanil Citrate Injection, it is essential that qualified personnel and adequate facilities are available for the management of postoperative respiratory depression.
For purposes of administering small volumes of Sufentanil Citrate Injection accurately, the use of a tuberculin syringe or equivalent is recommended.
-
•Ensure that an opioid antagonist, resuscitative and intubation equipment, and oxygen are readily available.
-
•Individualize dosage based on factors such as age, body weight, physical status, underlying pathological condition, use of other drugs, type of anesthesia to be used, and the surgical procedure involved.
-
•Monitor vital signs regularly.
-
•The selection of preanesthetic medications should be based upon the needs of the individual patient.
-
•The neuromuscular blocking agent selected should be compatible with the patient's condition, taking into account the hemodynamic effects of a particular muscle relaxant and the degree of skeletal muscle relaxation required.
As with other potent opioids, the respiratory depressant effect of sufentanil may persist longer than the measured analgesic effect. The total dose of all opioid agonists administered should be considered by the practitioner before ordering opioid analgesics during recovery from anesthesia.
If Sufentanil Citrate Injection is administered with a CNS depressant, become familiar with the properties of each drug, particularly each product's duration of action. In addition, when such a combination is used, fluids and other countermeasures to manage hypotension should be available [see Warnings and Precautions (5.3) ].
Inspect parenteral drug products visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
5.5 Risks of Muscle Rigidity and Skeletal Muscle Movement
Intravenous administration or unintentional intravascular injection during epidural administration of Sufentanil Citrate Injection may cause muscle rigidity, particularly involving the muscles of respiration. The incidence and severity of muscle rigidity is dose related. These effects are related to the dose and speed of injection. Administration of sufentanil may produce muscular rigidity with a more rapid onset of action than that seen with fentanyl. Skeletal muscle rigidity also has been reported to occur or recur infrequently in the extended postoperative period usually following high dose administration. In addition, skeletal muscle movements of various groups in the extremities, neck, and external eye have been reported during induction of anesthesia with Sufentanil Citrate Injection; these reported movements have, on rare occasions, been strong enough to pose patient management problems.
The incidence of skeletal muscle rigidity can be reduced by: 1) administration of up to 1/4 of the full paralyzing dose of a non-depolarizing neuromuscular blocking agent just prior to administration of sufentanil at dosages of up to 8 mcg/kg, 2) administration of a full paralyzing dose of a neuromuscular blocking agent following loss of consciousness when sufentanil is used in anesthetic dosages (above 8 mcg/kg) titrated by slow intravenous infusion, or, 3) simultaneous administration of sufentanil and a full paralyzing dose of a neuromuscular blocking agent when sufentanil is used in rapidly administered anesthetic dosages (above 8 mcg/kg).
The neuromuscular blocking agents used should be compatible with the patient's cardiovascular status. The hemodynamic effects and degree of skeletal muscle relaxation required should be considered in the selection of a neuromuscular blocking agent. High doses of pancuronium may produce increases in heart rate during sufentanil-oxygen anesthesia. Bradycardia and hypotension have been reported with other muscle relaxants during sufentanil-oxygen anesthesia; this effect may be more pronounced in the presence of calcium channel and/or beta-blockers. Muscle relaxants with no clinically significant effect on heart rate (at recommended doses) would not counteract the vagotonic effect of sufentanil, therefore a lower heart rate would be expected. Rare reports of bradycardia associated with the concomitant use of succinylcholine and sufentanil have been reported.
5.11 Risks of Use in Patients With Gastrointestinal Conditions (5.11 Risks of Use in Patients with Gastrointestinal Conditions)
Sufentanil may cause spasm of the sphincter of Oddi. Opioids may cause increases in serum amylase. Monitor patients with biliary tract disease, including acute pancreatitis for worsening symptoms.
5.8 Serotonin Syndrome With Concomitant Use of Serotonergic Drugs (5.8 Serotonin Syndrome with Concomitant Use of Serotonergic Drugs)
Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of sufentanil with serotonergic drugs. Serotonergic drugs include selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that affect the serotonergic neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), and drugs that impair metabolism of serotonin (including MAO inhibitors, both those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue) [see Drug Interactions (7)]. This may occur within the recommended dosage range.
Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination, rigidity), and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea) and can be fatal. The onset of symptoms generally occurs within several hours to a few days of concomitant use, but may occur later than that. Discontinue Sufentanil Citrate Injection if serotonin syndrome is suspected.
5.12 Increased Risk of Seizures in Patients With Seizure Disorders (5.12 Increased Risk of Seizures in Patients with Seizure Disorders)
Sufentanil may increase the frequency of seizures in patients with seizure disorders, and may increase the risk of seizures occurring in other clinical settings associated with seizures. Monitor patients with a history of seizure disorders for worsened seizure control during Sufentanil Citrate Injection therapy.
5.3 Risks From Concomitant Use With Benzodiazepines Or Other Cns Depressants (5.3 Risks from Concomitant Use with Benzodiazepines or Other CNS Depressants)
When benzodiazepines or other CNS depressants are used with Sufentanil Citrate Injection, pulmonary arterial pressure may be decreased. This fact should be considered by those who conduct diagnostic and surgical procedures where interpretation of pulmonary arterial pressure measurements might determine final management of the patient. When high dose or anesthetic dosages of Sufentanil Citrate Injection are employed, even relatively small dosages of diazepam may cause cardiovascular depression.
When Sufentanil Citrate Injection is used with CNS depressants, hypotension can occur. If it occurs, consider the possibility of hypovolemia and manage with appropriate parenteral fluid therapy. When operative conditions permit, consider repositioning the patient to improve venous return to the heart. Exercise care in moving and repositioning of patients because of the possibility of orthostatic hypotension. If volume expansion with fluids plus other countermeasures do not correct hypotension, consider administration of pressor agents other than epinephrine. Epinephrine may paradoxically decrease blood pressure in patients treated with a neuroleptic that blocks alpha adrenergic activity.
Profound sedation, respiratory depression, coma, and death may result from the concomitant use of Sufentanil Citrate Injection with benzodiazepines and/or other CNS depressants including alcohol (e.g., nonbenzodiazepine sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, other opioids).
Observational studies have demonstrated that concomitant use of opioid analgesics and benzodiazepines increases the risk of drug-related mortality compared to use of opioid analgesics alone. Because of similar pharmacological properties, it is reasonable to expect similar risk with the concomitant use of other CNS depressant drugs with opioid analgesics [see Drug Interactions (7)].
If the decision is made to manage postoperative pain with Sufentanil Citrate Injection concomitantly with a benzodiazepine or other CNS depressant, start dosing with the lowest effective dosage and titrate based on clinical response. Monitor patients closely for signs and symptoms of respiratory depression, sedation, and hypotension. Fluids or other measures to counter hypotension should be available [see Drug Interactions (7)].
Warning: serious and Life Threatening Risks From Use of Sufentanil Citrate injection (WARNING: SERIOUS AND LIFE-THREATENING RISKS FROM USE OF SUFENTANIL CITRATE INJECTION)
Addiction, Abuse, and Misuse
Because the use of Sufentanil Citrate Injection exposes patients and other users to the risks of opioid addiction, abuse, and misuse, which can lead to overdose and death, assess each patient's risk prior to prescribing and reassess all patients regularly for the development of these behaviors and conditions [see Warnings and Precautions (5.1)] .
Life-Threatening Respiratory Depression
Serious, life-threatening, or fatal respiratory depression may occur with use of Sufentanil Citrate Injection, especially during initiation or following a dosage increase. To reduce the risk of respiratory depression, proper dosing and titration of Sufentanil Citrate Injection are essential [see Warnings and Precautions (5.2)].
Risks from Concomitant Use with Benzodiazepines or Other CNS Depressants
Concomitant use of opioids with benzodiazepines or other central nervous system (CNS) depressants, including alcohol, may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing of Sufentanil Citrate Injection and benzodiazepines or other CNS depressants for use in patients for whom alternative treatment options are inadequate [see Warnings and Precautions (5.3), Drug Interactions (7) ].
Cytochrome P450 3A4 Interaction
The concomitant use of Sufentanil Citrate Injection with all cytochrome P450 3A4 inhibitors may result in an increase in sufentanil plasma concentrations, which could increase or prolong adverse reactions and may cause potentially fatal respiratory depression. In addition, discontinuation of a concomitantly used cytochrome P450 3A4 inducer may result in an increase in sufentanil plasma concentration. Monitor patients receiving Sufentanil Citrate Injection and any CYP3A4 inhibitor or inducer [see Warnings and Precautions (5.4), Drug Interactions (7), Clinical Pharmacology (12.3)] .
5.4 Risks of Concomitant Use Or Discontinuation of Cytochrome P450 3a4 Inhibitors and Inducers (5.4 Risks of Concomitant Use or Discontinuation of Cytochrome P450 3A4 Inhibitors and Inducers)
Concomitant use of Sufentanil Citrate Injection with a CYP3A4 inhibitor, such as macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g., ketoconazole), and protease inhibitors (e.g., ritonavir), may increase plasma concentrations of sufentanil and prolong opioid adverse reactions, which may exacerbate fatal respiratory depression [see Warnings and Precautions (5.2)], particularly when an inhibitor is added after a stable dose of Sufentanil Citrate Injection is achieved. Similarly, discontinuation of a CYP3A4 inducer, such as rifampin, carbamazepine, and phenytoin, in Sufentanil Citrate Injection-treated patients may increase sufentanil plasma concentrations and prolong opioid adverse reactions. When using Sufentanil Citrate Injection with CYP3A4 inhibitors or discontinuing CYP3A4 inducers in Sufentanil Citrate Injection-treated patients, monitor patients closely at frequent intervals and consider dosage reduction of Sufentanil Citrate Injection [see Dosage and Administration (2.1), Drug Interactions (7)].
Concomitant use of Sufentanil Citrate Injection with CYP3A4 inducers or discontinuation of an CYP3A4 inhibitor could result in lower than expected sufentanil plasma concentrations, decreased efficacy, or, possibly, lead to a withdrawal syndrome in a patient who had developed physical dependence to sufentanil. When using Sufentanil Citrate Injection with CYP3A4 inducers or discontinuing CYP3A4 inhibitors, monitor patients closely at frequent intervals and consider increasing the Sufentanil Citrate Injection dosage [see Dosage and Administration (2.1), Drug Interactions (7)].
5.10 Risks of Use in Patients With Increased Intracranial Pressure, Brain Tumors, Or Head Injury (5.10 Risks of Use in Patients with Increased Intracranial Pressure, Brain Tumors, or Head Injury)
In patients who may be susceptible to the intracranial effects of CO2 retention (e.g., those with evidence of increased intracranial pressure or brain tumors), Sufentanil Citrate Injection may reduce respiratory drive, and the resultant CO2 retention can further increase intracranial pressure. Monitor such patients for signs of sedation and respiratory depression, particularly when initiating therapy with Sufentanil Citrate Injection. Opioids may also obscure the clinical course in a patient with a head injury.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:44:45.540516 · Updated: 2026-03-14T22:21:06.327897