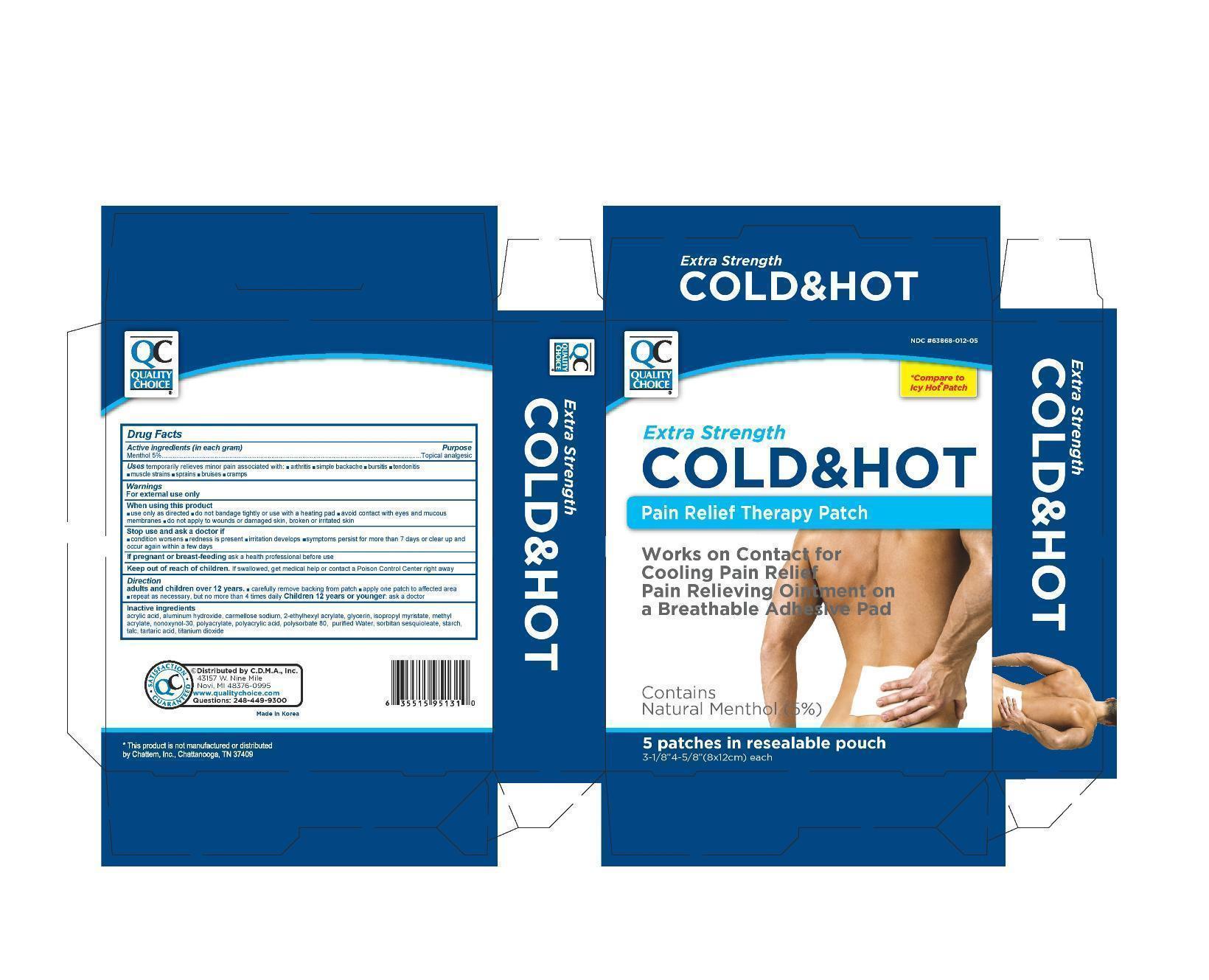
Drug Facts
1ff58c3a-95c1-4ca0-81d8-ce580f08481d
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Menthol 5.0%
Purpose
Topical Analgesic
Medication Information
Purpose
Topical Analgesic
Description
Menthol 5.0%
Uses
temporarily relieves minor pain associated with
- arthritis
- simple backache
- bursitis
- tendonitis
- muscle strains
- sprains
- bruises
- cramps
Warnings
For external use only
Directions
Adults and children over 12 years:
- carefully remove backing from patch
- apply one patch to affected area
- repeat as necessary, but no more than 4 times daily
Children 12 years or younger: ask a doctor
Package Label
QC Cold and Hot Patch
Active Ingredient
Inactive Ingredients
Acrylic Acid, Aluminum Hydroxide, Carmellose sodium, 2-Ethylhexyl Acrylate, Glycerin, Isopropyl Myristate, Methyl Acrylate, Nonoxymol-30, Sodium Polyacrylate, Polyacrylic Acid, Polysorbate 80, Purified Water, Sorbitan Sesquioleate, Starch, Talc, Tartaric acid, Titanium Dioxide
When Using This Product
- use only as directed
- avoid contact with eyes and mucous membranes
- do not bandage tightly or use with a heating pad
- do not apply to wounds or damaged skin, broken or irritated skin
Stop Use and Ask A Doctor If
- condition worsens
- redness is present
- irritation develops
- symptoms persist for more than 7 days or clear up and occur again within a few days
If Pregnant Or Breast Feeding,
ask a health professional before use
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
temporarily relieves minor pain associated with
- arthritis
- simple backache
- bursitis
- tendonitis
- muscle strains
- sprains
- bruises
- cramps
Purpose
Topical Analgesic
Warnings
For external use only
Directions
Adults and children over 12 years:
- carefully remove backing from patch
- apply one patch to affected area
- repeat as necessary, but no more than 4 times daily
Children 12 years or younger: ask a doctor
Package Label (package label)
QC Cold and Hot Patch
Active Ingredient
Inactive Ingredients (Inactive ingredients)
Acrylic Acid, Aluminum Hydroxide, Carmellose sodium, 2-Ethylhexyl Acrylate, Glycerin, Isopropyl Myristate, Methyl Acrylate, Nonoxymol-30, Sodium Polyacrylate, Polyacrylic Acid, Polysorbate 80, Purified Water, Sorbitan Sesquioleate, Starch, Talc, Tartaric acid, Titanium Dioxide
When Using This Product (When using this product)
- use only as directed
- avoid contact with eyes and mucous membranes
- do not bandage tightly or use with a heating pad
- do not apply to wounds or damaged skin, broken or irritated skin
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens
- redness is present
- irritation develops
- symptoms persist for more than 7 days or clear up and occur again within a few days
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:33.690831 · Updated: 2026-03-14T22:53:20.556394