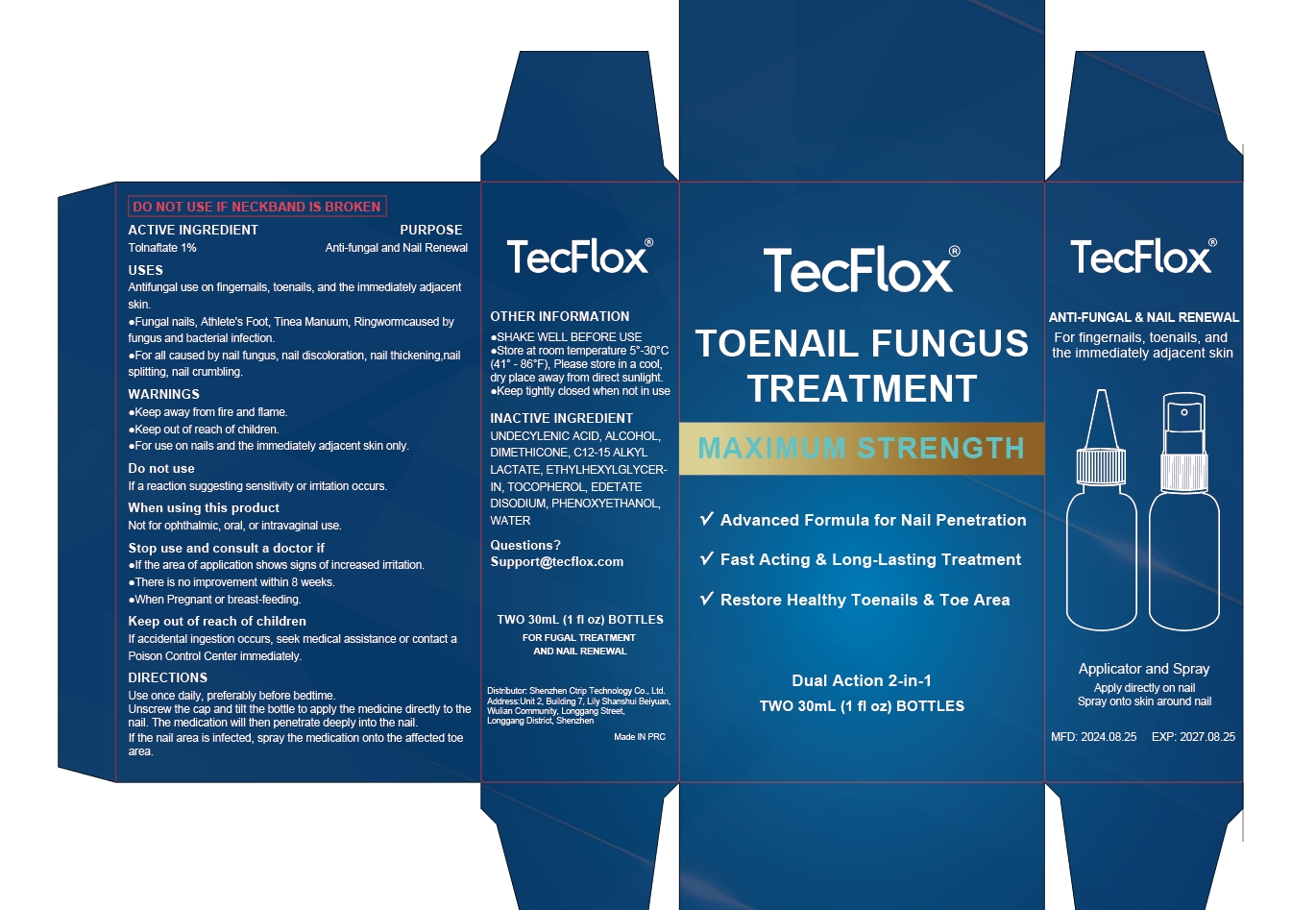
84614-002 Toenail Fungus Treatment Maximum Strength Liquid
1fb03184-5f21-c82a-e063-6294a90a9fa4
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
TOENAIL FUNGUS TREATMENT MAXIMUM STRENGTH Liquid
Medication Information
Warnings and Precautions
Keep away from fire and flame.
Keep out of reach of children.
For use on nails and the immediately adjacent skin only.
Indications and Usage
Antifungal use on fingemails, toenails, and the immediately adjacent skin.
Fungal nails, Athlete's Foot, Tinea Manuum, Ringwomcaused byfungus and bacterial infection.
For all caused by nail fungus, nail discoloration, nail thickening,nailsplitting, nail crumbling
Dosage and Administration
Use once daily, preferably before bedtimeUnscrew the cap and tilt the bottle to apply the medicine directly to thenail. The medication will then penetrate deeply into the nail.If the nail area is infected, spray the medication onto the affected toearea.
Description
TOENAIL FUNGUS TREATMENT MAXIMUM STRENGTH Liquid
Section 42229-5
TOENAIL FUNGUS TREATMENT MAXIMUM STRENGTH Liquid
Section 44425-7
SHAKE WELL BEFORE USE
Store at room temperature 5--30°C(41°-86°F), Please store in a cool,dry place away from direct sunlight.
Keep tightly closed when not in use
Section 50565-1
If accidental ingestion occurs, seek medical assistance or contact a Poison Control Center immediately.
Section 50566-9
lf the area of application shows signs of increased iritation.
There is no improvement within 8 weeks.
When Pregnant or breast-feeding
Section 50567-7
Not for ophthalmic, oral, or intravaginal use
Section 50570-1
If a reaction suggesting sensitivity or iritation occurs.
Section 51727-6
UNDECYLENIC ACID
ALCOHOL
DIMETHICONE
C12-15 ALKYL LACTATE
ETHYLHEXYLGLYCERIN
TOCOPHEROL
EDETATE DISODIUM
PHENOXYETHANOL
WATER
Section 51945-4
Section 55105-1
Anti-fungal and Nail Renewal
Section 55106-9
Tolnaftate 1%
Structured Label Content
Indications and Usage (34067-9)
Antifungal use on fingemails, toenails, and the immediately adjacent skin.
Fungal nails, Athlete's Foot, Tinea Manuum, Ringwomcaused byfungus and bacterial infection.
For all caused by nail fungus, nail discoloration, nail thickening,nailsplitting, nail crumbling
Dosage and Administration (34068-7)
Use once daily, preferably before bedtimeUnscrew the cap and tilt the bottle to apply the medicine directly to thenail. The medication will then penetrate deeply into the nail.If the nail area is infected, spray the medication onto the affected toearea.
Warnings and Precautions (34071-1)
Keep away from fire and flame.
Keep out of reach of children.
For use on nails and the immediately adjacent skin only.
Section 42229-5 (42229-5)
TOENAIL FUNGUS TREATMENT MAXIMUM STRENGTH Liquid
Section 44425-7 (44425-7)
SHAKE WELL BEFORE USE
Store at room temperature 5--30°C(41°-86°F), Please store in a cool,dry place away from direct sunlight.
Keep tightly closed when not in use
Section 50565-1 (50565-1)
If accidental ingestion occurs, seek medical assistance or contact a Poison Control Center immediately.
Section 50566-9 (50566-9)
lf the area of application shows signs of increased iritation.
There is no improvement within 8 weeks.
When Pregnant or breast-feeding
Section 50567-7 (50567-7)
Not for ophthalmic, oral, or intravaginal use
Section 50570-1 (50570-1)
If a reaction suggesting sensitivity or iritation occurs.
Section 51727-6 (51727-6)
UNDECYLENIC ACID
ALCOHOL
DIMETHICONE
C12-15 ALKYL LACTATE
ETHYLHEXYLGLYCERIN
TOCOPHEROL
EDETATE DISODIUM
PHENOXYETHANOL
WATER
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Anti-fungal and Nail Renewal
Section 55106-9 (55106-9)
Tolnaftate 1%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:16.633009 · Updated: 2026-03-14T23:12:28.058882