Ivizia Eye Drops
1ef73cb1-da48-a713-e063-6294a90afa9d
34390-5
HUMAN OTC DRUG LABEL
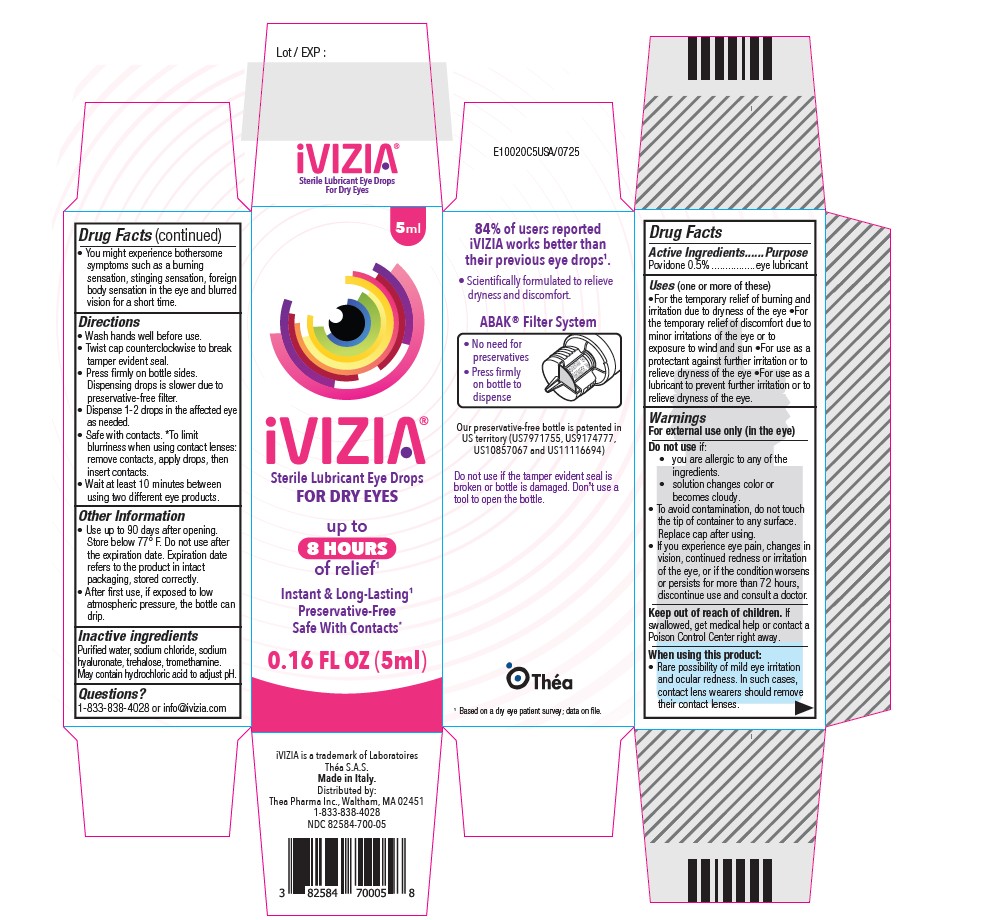
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Povidone 0.5%
Purpose
Eye lubricant
Medication Information
Purpose
Eye lubricant
Description
Povidone 0.5%
Uses
(one or more of these)
For the temporary relief of burning and irritation due to dryness of the eye
For the temporary relief of discomfort due to minor irritations of the eye or to exposure to wind and sun
For use as a protectant against further irritation or to relieve dryness of the eye
For use as a lubricant to prevent further irritation or to relieve dryness of the eye.
Warnings
For external use only(in the eye)
Do not use if:
• you are allergic to any of the ingredients.
• solution changes color or becomes cloudy.
• To avoid contamination, do not touch the tip of container to any surface.
Replace cap after using.
• If you experience eye pain, changes in vision, continued redness or irritation of the eye, or if the condition worsens or persists for more than 72 hours, discontinue use and consult a doctor.
Questions?
1-833-838-4028 or [email protected]
Directions:
Wash hands well before use.
Twist cap counterclockwise to break tamper evident seal.
Press firmly on bottle sides. Dispensing drops is slower due to preservative-free filter.
Dispense 1-2 drops in the affected eye as needed.
Safe with contacts. *To limit blurriness when using contact lenses: remove contacts, apply drops, then insert contacts.
Active Ingredient
Povidone 0.5%
Other Information
• Use up to 90 days after opening. Store below 77° F. Do not use after the expiration date on the carton. Expiration date refers to the product in intact packaging, stored correctly.
• After first use, if exposed to low atmospheric pressure, the bottle can drip.
Inactive Ingredients
Purified water, sodium chloride, sodium hyaluronate, trehalose, tromethamine. May contain hydrochloric acid to adjust pH.
Principle Display Panel
iVizia
Sterile Lubricant Eye Drops
For Dry Eyes
0.16 FL OZ (5ml)
When Using This Product
Rare possibility of mild eye irritation and ocular redness. In such cases, contact lens wearers should remove their contact lenses.
You might experience bothersome symptoms such as a burning sensation, stinging sensation, foreign body sensation in the eye and blurred vision for a short time.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
(one or more of these)
For the temporary relief of burning and irritation due to dryness of the eye
For the temporary relief of discomfort due to minor irritations of the eye or to exposure to wind and sun
For use as a protectant against further irritation or to relieve dryness of the eye
For use as a lubricant to prevent further irritation or to relieve dryness of the eye.
Purpose
Eye lubricant
Warnings
For external use only(in the eye)
Do not use if:
• you are allergic to any of the ingredients.
• solution changes color or becomes cloudy.
• To avoid contamination, do not touch the tip of container to any surface.
Replace cap after using.
• If you experience eye pain, changes in vision, continued redness or irritation of the eye, or if the condition worsens or persists for more than 72 hours, discontinue use and consult a doctor.
Questions?
1-833-838-4028 or [email protected]
Directions:
Wash hands well before use.
Twist cap counterclockwise to break tamper evident seal.
Press firmly on bottle sides. Dispensing drops is slower due to preservative-free filter.
Dispense 1-2 drops in the affected eye as needed.
Safe with contacts. *To limit blurriness when using contact lenses: remove contacts, apply drops, then insert contacts.
Active Ingredient
Povidone 0.5%
Other Information
• Use up to 90 days after opening. Store below 77° F. Do not use after the expiration date on the carton. Expiration date refers to the product in intact packaging, stored correctly.
• After first use, if exposed to low atmospheric pressure, the bottle can drip.
Inactive Ingredients
Purified water, sodium chloride, sodium hyaluronate, trehalose, tromethamine. May contain hydrochloric acid to adjust pH.
Principle Display Panel
iVizia
Sterile Lubricant Eye Drops
For Dry Eyes
0.16 FL OZ (5ml)
When Using This Product (When using this product)
Rare possibility of mild eye irritation and ocular redness. In such cases, contact lens wearers should remove their contact lenses.
You might experience bothersome symptoms such as a burning sensation, stinging sensation, foreign body sensation in the eye and blurred vision for a short time.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:34.372156 · Updated: 2026-03-14T23:13:07.898715