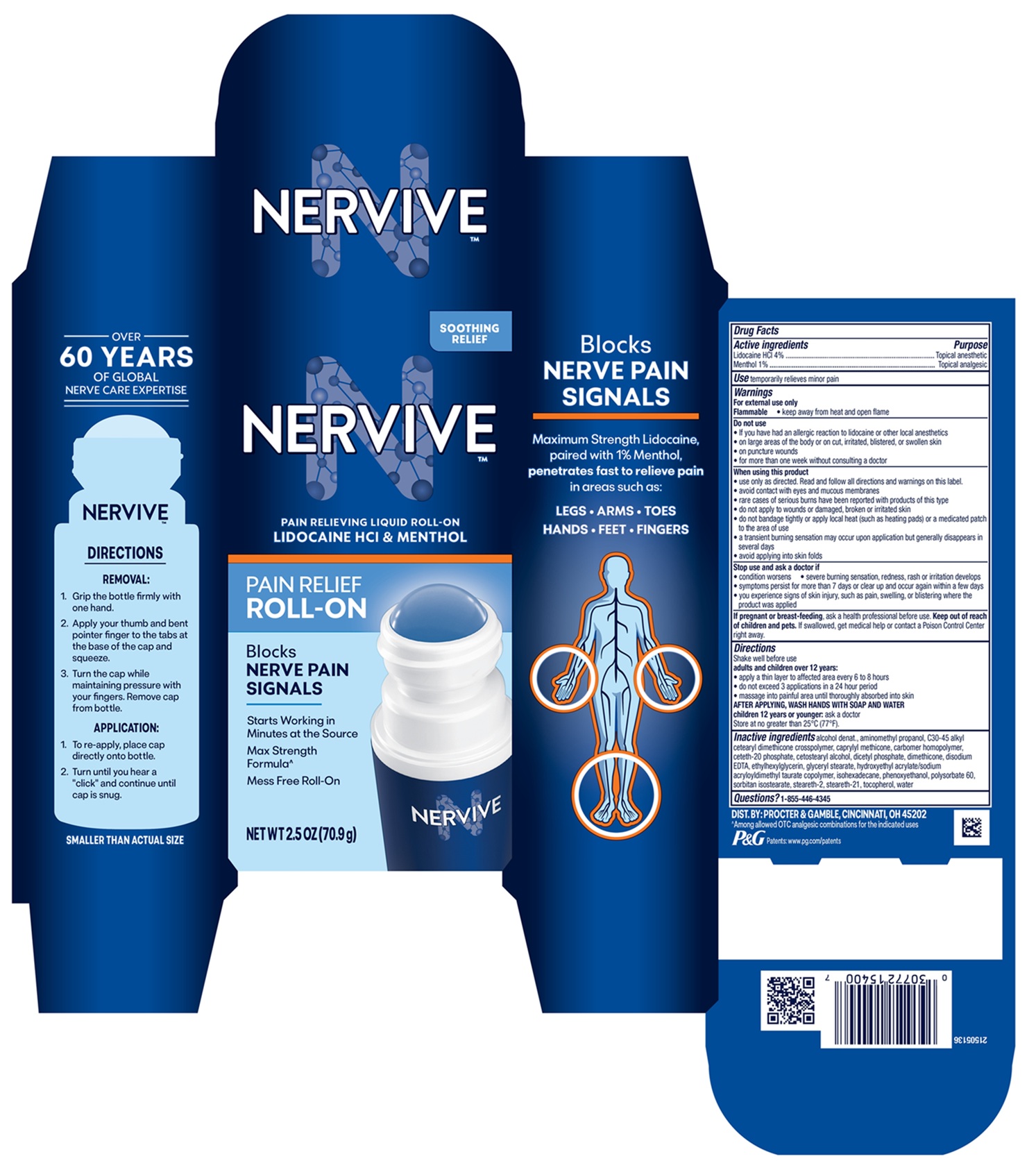
Nervive™ Pain Relieving Liquid Roll-on
1e8aeccc-8d00-9886-e063-6394a90ad7a6
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Topical anesthetic Topical analgesic
Medication Information
Purpose
Topical anesthetic Topical analgesic
Description
Drug Facts
Use
temporarily relieves minor pain
Section 34088-5
If swallowed, get medical help or contact a Poison Control Center right away.
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children and pets.
Warnings
For external use only
Flammable
- keep away from heat and open flame
Directions
Shake well before use
adults and children over 12 years:
- apply a thin layer to affected area every 6 to 8 hours
- do not exceed 3 applications in a 24 hour period
- massage into painful area until thoroughly absorbed into skin
AFTER APPLYING, WASH HANDS WITH SOAP AND WAT
ER
children 12 years or younger: ask a doctor
Store at no greater than 25°C (77°F).
Do Not Use
• If you have had an allergic reaction to lidocaine or other local anesthetics
• on large areas of the body or on cut, irritated, blistered, or swollen skin
• on puncture wounds
• for more than one week without consulting a doctor
Questions?
1-855-446-4345
Active Ingredients
Lidocaine HCl 4%
Menthol 1%
Inactive Ingredients
alcohol, aminomethyl propanol, C30-45 alkyl cetearyl dimethicone crosspolymer, caprylyl methicone, carbomer homopolymer, ceteth-20 phosphate, cetearyl alcohol, dicetyl phosphate, dimethicone, disodium EDTA, ethylhexyl glycerin, glyceryl stearate, hydroxyethyl acrylate/sodium acryloyldimethyl taurate copolymer, isohexadecane, phenoxyethanol, polysorbate 60, sorbitan isostearate, steareth-2, steareth-21, tocopherol, water
When Using This Product
• use only as directed. Read and follow all directions and warnings on this carton.
• avoid contact with eyes and mucous membranes
• rare cases of serious burns have been reported with products of this type
• do not apply to wounds or damaged, broken or irritated skin
• do not bandage tightly or apply local heat (such as heating pads) or a medicated patch to the area of use
• a transient burning sensation may occur upon application but generally disappears in several days
• avoid applying into skin folds
Active Ingredients Purpose
Topical anesthetic
Topical analgesic
Stop Use and Ask A Doctor If
• condition worsens
• severe burning sensation, redness, rash or irritation develops
• symptoms persist for more than 7 days or clear up and occur again within a few days
• you experience signs of skin injury, such as pain, swelling, or blistering where the product was applied
If Pregnant Or Breast Feeding,
ask a health professional before use.
Principal Display Panel 2.5 Oz
NERVIVE™
PAIN RELIEVING
LIQUID ROLL-ON
LIDOCAINE HCl & MENTHOL
TARGETS MULTIPLE
NERVE PAIN RECEPTORS
MASSAGING ROLL-ON
NET WT 2.5 OZ (70.9 g)
Structured Label Content
Use
temporarily relieves minor pain
Section 34088-5 (34088-5)
If swallowed, get medical help or contact a Poison Control Center right away.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children and pets.
Warnings
For external use only
Flammable
- keep away from heat and open flame
Directions
Shake well before use
adults and children over 12 years:
- apply a thin layer to affected area every 6 to 8 hours
- do not exceed 3 applications in a 24 hour period
- massage into painful area until thoroughly absorbed into skin
AFTER APPLYING, WASH HANDS WITH SOAP AND WAT
ER
children 12 years or younger: ask a doctor
Store at no greater than 25°C (77°F).
Do Not Use (Do not use)
• If you have had an allergic reaction to lidocaine or other local anesthetics
• on large areas of the body or on cut, irritated, blistered, or swollen skin
• on puncture wounds
• for more than one week without consulting a doctor
Questions?
1-855-446-4345
Active Ingredients (Active ingredients)
Lidocaine HCl 4%
Menthol 1%
Inactive Ingredients (Inactive ingredients)
alcohol, aminomethyl propanol, C30-45 alkyl cetearyl dimethicone crosspolymer, caprylyl methicone, carbomer homopolymer, ceteth-20 phosphate, cetearyl alcohol, dicetyl phosphate, dimethicone, disodium EDTA, ethylhexyl glycerin, glyceryl stearate, hydroxyethyl acrylate/sodium acryloyldimethyl taurate copolymer, isohexadecane, phenoxyethanol, polysorbate 60, sorbitan isostearate, steareth-2, steareth-21, tocopherol, water
When Using This Product (When using this product)
• use only as directed. Read and follow all directions and warnings on this carton.
• avoid contact with eyes and mucous membranes
• rare cases of serious burns have been reported with products of this type
• do not apply to wounds or damaged, broken or irritated skin
• do not bandage tightly or apply local heat (such as heating pads) or a medicated patch to the area of use
• a transient burning sensation may occur upon application but generally disappears in several days
• avoid applying into skin folds
Active Ingredients Purpose (Active ingredients Purpose)
Topical anesthetic
Topical analgesic
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
• condition worsens
• severe burning sensation, redness, rash or irritation develops
• symptoms persist for more than 7 days or clear up and occur again within a few days
• you experience signs of skin injury, such as pain, swelling, or blistering where the product was applied
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Principal Display Panel 2.5 Oz (PRINCIPAL DISPLAY PANEL - 2.5 OZ)
NERVIVE™
PAIN RELIEVING
LIQUID ROLL-ON
LIDOCAINE HCl & MENTHOL
TARGETS MULTIPLE
NERVE PAIN RECEPTORS
MASSAGING ROLL-ON
NET WT 2.5 OZ (70.9 g)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:11.807930 · Updated: 2026-03-14T23:10:24.980101