papaverine hydrochloride injection, usp
1e76839f-82eb-42a8-abc2-e7fbc533082d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Papaverine is recommended in various conditions accompanied by spasm of smooth muscle, such as vascular spasm associated with acute myocardial infarction (coronary occlusion), angina pectoris, peripheral and pulmonary embolism, peripheral vascular disease in which there is a vasospastic element, or certain cerebral angiospastic states; and visceral spasm, as in ureteral, biliary, or gastrointestinal colic.
Dosage and Administration
Papaverine Hydrochloride may be administered intravenously or intramuscularly. The intravenous route is recommended when an immediate effect is desired, but the drug must be injected slowly over the course of 1 or 2 minutes to avoid uncomfortable or alarming side effects. Parenteral administration of papaverine hydrochloride in doses of 1 to 4 mL is repeated every 3 hours as indicated. In the treatment of cardiac extrasystoles, 2 doses may be given 10 minutes apart.
Contraindications
Intravenous injection of papaverine is contraindicated in the presence of complete atrioventricular heart block. When conduction is depressed, the drug may produce transient ectopic rhythms of ventricular origin, either premature beats or paroxysmal tachycardia. Papaverine Hydrochloride is not indicated for the treatment of impotence by intracorporeal injection. The intracorporeal injection of papaverine hydrochloride has been reported to have resulted in persistent priapism requiring medical and surgical intervention.
Adverse Reactions
The following side effects have been reported: general discomfort, nausea, abdominal discomfort, anorexia, constipation or diarrhea, skin rash, malaise, vertigo, headache, intensive flushing of the face, perspiration, increase in the depth of respiration, increase in heart rate, a slight rise in blood pressure, and excessive sedation. Hepatitis, probably related to an immune mechanism, has been reported infrequently. Rarely, this has progressed to cirrhosis.
How Supplied
Papaverine Hydrochloride Injection, USP, 30 mg/mL NDC: 14789-121-07 2 mL Single-dose Vial NDC: 14789-121-05 10 pack carton of 2 mL Single-dose Vials Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) (See USP Controlled Room Temperature). PROTECT FROM LIGHT. RETAIN IN CARTON UNTIL TIME OF USE. Container closer was not made with natural rubber latex. Manufactured in Italy for: Nexus Pharmaceuticals, LLC Lincolnshire, IL 60069 PAPPI01ITR04 Revised 08/2024 NEXUS PHARMACEUTICALS
Description
Rx Only This product is to be used by or under the direction of a physician. Each vial contains a sufficient amount to permit withdrawal and administration of the volume specified on the label.
Medication Information
Indications and Usage
Papaverine is recommended in various conditions accompanied by spasm of smooth muscle, such as vascular spasmassociated with acute myocardial infarction (coronary occlusion), angina pectoris, peripheral and pulmonary embolism, peripheral vascular disease in which there is a vasospastic element, or certain cerebral angiospastic states; and visceral spasm,as in ureteral, biliary, or gastrointestinal colic.
Dosage and Administration
Papaverine Hydrochloride may be administered intravenously or intramuscularly. The intravenous route is recommended when an immediate effect is desired, but the drug mustbe injected slowlyover the course of 1 or 2 minutes to avoid uncomfortable or alarming side effects.
Parenteral administration of papaverine hydrochloride in doses of 1 to 4 mL is repeated every 3 hours as indicated. In the treatment of cardiac extrasystoles, 2 doses may be given 10 minutes apart.
Contraindications
Intravenous injection of papaverine is contraindicated in the presence of complete atrioventricular heart block. When conduction is depressed, the drug may produce transient ectopic rhythms of ventricular origin, either premature beats or paroxysmal tachycardia.
Papaverine Hydrochloride is not indicated for the treatment of impotence by intracorporeal injection. The intracorporeal injection of papaverine hydrochloride has been reported to have resulted in persistent priapism requiring medical and surgical intervention.
Adverse Reactions
The following side effects have been reported: general discomfort, nausea, abdominal discomfort, anorexia, constipation or diarrhea, skin rash, malaise, vertigo, headache, intensive flushing of the face, perspiration, increase in the depth of respiration, increase in heart rate, a slight rise in blood pressure, and excessive sedation.
Hepatitis, probably related to an immune mechanism, has been reported infrequently. Rarely, this has progressed to cirrhosis.
How Supplied
Papaverine Hydrochloride Injection, USP, 30 mg/mL NDC: 14789-121-07 2 mL Single-dose Vial NDC: 14789-121-05 10 pack carton of 2 mL Single-dose Vials Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) (See USP Controlled Room Temperature). PROTECT FROM LIGHT. RETAIN IN CARTON UNTIL TIME OF USE. Container closer was not made with natural rubber latex. Manufactured in Italy for: Nexus Pharmaceuticals, LLC Lincolnshire, IL 60069 PAPPI01ITR04 Revised 08/2024 NEXUS PHARMACEUTICALS
Description
Papaverine Hydrochloride, USP, is the hydrochloride of an alkaloid obtained from opium or prepared synthetically. It belongs to the benzylisoquinoline group of alkaloids. It does not contain a phenanthrene group as do morphine and codeine.
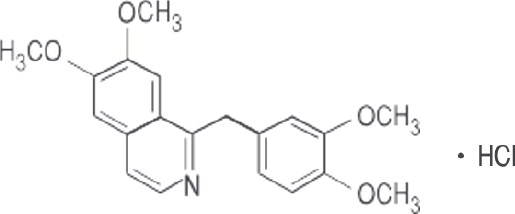
Papaverine Hydrochloride, USP, is 6,7-dimethoxy-1- veratrylisoquinoline hydrochloride and contains, on the dried basis, not less than 98.5% of C 20H 21NO 4•HCl. The molecular weight is 375.85. The structural formula is as shown.
Papaverine Hydrochloride occurs as white crystals or white crystalline powder. One gram dissolves in about 30 mL of water and in 120 mL of alcohol. It is soluble in chloroform and practically insoluble in ether.
Papaverine Hydrochloride Injection, USP, is a clear, colorless to pale-yellow solution.
Papaverine Hydrochloride, for parenteral administration, is a smooth-muscle relaxant that is available in vials containing 30 mg/mL. Each vial also contains edetate disodium 0.005%.
pH may be adjusted with sodium citrate and/or citric acid.
Section 34072-9
General - Papaverine Hydrochloride Injection, USP, should not be added to Lactated Ringer's Injection, because precipitation would result.
Papaverine Hydrochloride should be used with caution in patients with glaucoma. The medication should be discontinued if hepatic hypersensitivity with gastrointestinal symptoms, jaundice, or eosinophilia becomes evident or if liver function test values become altered.
Section 34080-2
Nursing Mothers - It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when papaverine hydrochloride is administered to a nursing woman.
Section 34081-0
Usage In Children - Safety and effectiveness in children have not been established.
Section 42228-7
Usage In Pregnancy - Pregnancy Category C - No teratogenic effects were observed in rats when papaverine hydrochloride was administered subcutaneously as a single agent. It is not known whether papaverine can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Papaverine Hydrochloride should be given to a pregnant woman only if clearly needed.
Section 42229-5
Rx Only
This product is to be used by or under the direction of a physician.
Each vial contains a sufficient amount to permit withdrawal and administration of the volume specified on the label.
How Supplied:
Papaverine Hydrochloride Injection, USP, 30 mg/mL
NDC: 14789-121-07 2 mL Single-dose Vial
NDC: 14789-121-05 10 pack carton of 2 mL Single-dose Vials
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) (See USP Controlled Room Temperature).
PROTECT FROM LIGHT.RETAIN IN CARTON UNTIL TIME OF USE.
Container closer was not made with natural rubber latex.
Manufactured in Italy for:
Nexus Pharmaceuticals, LLC
Lincolnshire, IL 60069
PAPPI01ITR04 Revised 08/2024
NEXUS
PHARMACEUTICALS
Clinical Pharmacology
The most characteristic effect of papaverine is relaxation of the tonus of all smooth muscle, especially when it has been spasmodically contracted. Papaverine Hydrochloride apparently acts directly on the muscle itself. This relaxation is noted in the vascular systemand bronchial musculatureand in the gastrointestinal, biliaryand urinary tracts.
The main actions of papaverine are exerted on cardiac and smooth muscle. Papaverine relaxes various smooth muscles, especially those of larger arteries; this relaxation may be prominent if spasm exists. The antispasmodic effect is a direct one and unrelated to muscle innervation, and the muscle still responds to drugs and other stimuli causing contraction. Papaverine has minimal actions on the central nervous system, although very large doses tend to produce some sedation and sleepiness in some patients. In certain circumstances, mild respiratory stimulation can be observed, but this is therapeutically inconsequential. Papaverine stimulates respiration by acting on carotid and aortic body chemoreceptors.
Papaverine relaxes the smooth musculature of the larger blood vessels, including the coronary, cerebral, peripheral, and pulmonary arteries. This action is particularly evident when such vessels are in spasm, induced reflexly or by drugs, and it provides the basis for the clinical use of papaverine in peripheral or pulmonary arterial embolism.
Experimentally in dogs, the alkaloid has been shown to cause fairly marked and long-lasting coronary vasodilatation and an increase in coronary blood flow. However, it also appears to have a direct inotropic effect and, when increased mechanical activity coincides with decreased systemic pressure, increases in coronary blood flow may not be sufficient to prevent brief periods of hypoxic myocardial depression.
Papaverine is effective by all routes of administration. A considerable fraction of the drug localizes in fat deposits and in the liver, with the remainder being distributed throughout the body. It is metabolized in the liver. About 90% of the drug is bound to plasma protein. Although estimates of its biologic half-life vary widely, reasonably constant plasma levels can be maintained with oral administration at 6 hour intervals. The drug is excreted in the urine in an inactive form.
Drug Abuse and Dependence
Drug dependence resulting from the abuse of many of the selective depressants, including papaverine hydrochloride, has been reported.
Principal Display Panel – 2 Ml Vial Label
NDC 14789-121-07
Rx Only
PAPAVERINE HYDROCHLORIDE
INJECTION, USP
60mg/2mL
(30 mg/mL)
Discard Unused Portion
2mL Single-dose Vials
NEXUS
PHARMACEUTICALS
Principal Display Panel – 2 Ml Carton Label
NDC 14789-121-05
Rx Only
PAPAVERINE HYDROCHLORIDE
INJECTION, USP
60mg/2mL
(30 mg/mL)
Discard Unused Portion
10 x 2mL Single-dose Vials
NEXUS
PHARMACEUTICALS
Structured Label Content
Indications and Usage (INDICATIONS AND USAGE)
Papaverine is recommended in various conditions accompanied by spasm of smooth muscle, such as vascular spasmassociated with acute myocardial infarction (coronary occlusion), angina pectoris, peripheral and pulmonary embolism, peripheral vascular disease in which there is a vasospastic element, or certain cerebral angiospastic states; and visceral spasm,as in ureteral, biliary, or gastrointestinal colic.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Papaverine Hydrochloride may be administered intravenously or intramuscularly. The intravenous route is recommended when an immediate effect is desired, but the drug mustbe injected slowlyover the course of 1 or 2 minutes to avoid uncomfortable or alarming side effects.
Parenteral administration of papaverine hydrochloride in doses of 1 to 4 mL is repeated every 3 hours as indicated. In the treatment of cardiac extrasystoles, 2 doses may be given 10 minutes apart.
Contraindications (CONTRAINDICATIONS)
Intravenous injection of papaverine is contraindicated in the presence of complete atrioventricular heart block. When conduction is depressed, the drug may produce transient ectopic rhythms of ventricular origin, either premature beats or paroxysmal tachycardia.
Papaverine Hydrochloride is not indicated for the treatment of impotence by intracorporeal injection. The intracorporeal injection of papaverine hydrochloride has been reported to have resulted in persistent priapism requiring medical and surgical intervention.
Adverse Reactions (ADVERSE REACTIONS)
The following side effects have been reported: general discomfort, nausea, abdominal discomfort, anorexia, constipation or diarrhea, skin rash, malaise, vertigo, headache, intensive flushing of the face, perspiration, increase in the depth of respiration, increase in heart rate, a slight rise in blood pressure, and excessive sedation.
Hepatitis, probably related to an immune mechanism, has been reported infrequently. Rarely, this has progressed to cirrhosis.
Description (DESCRIPTION)
Papaverine Hydrochloride, USP, is the hydrochloride of an alkaloid obtained from opium or prepared synthetically. It belongs to the benzylisoquinoline group of alkaloids. It does not contain a phenanthrene group as do morphine and codeine.
Papaverine Hydrochloride, USP, is 6,7-dimethoxy-1- veratrylisoquinoline hydrochloride and contains, on the dried basis, not less than 98.5% of C 20H 21NO 4•HCl. The molecular weight is 375.85. The structural formula is as shown.
Papaverine Hydrochloride occurs as white crystals or white crystalline powder. One gram dissolves in about 30 mL of water and in 120 mL of alcohol. It is soluble in chloroform and practically insoluble in ether.
Papaverine Hydrochloride Injection, USP, is a clear, colorless to pale-yellow solution.
Papaverine Hydrochloride, for parenteral administration, is a smooth-muscle relaxant that is available in vials containing 30 mg/mL. Each vial also contains edetate disodium 0.005%.
pH may be adjusted with sodium citrate and/or citric acid.
Section 34072-9 (34072-9)
General - Papaverine Hydrochloride Injection, USP, should not be added to Lactated Ringer's Injection, because precipitation would result.
Papaverine Hydrochloride should be used with caution in patients with glaucoma. The medication should be discontinued if hepatic hypersensitivity with gastrointestinal symptoms, jaundice, or eosinophilia becomes evident or if liver function test values become altered.
Section 34080-2 (34080-2)
Nursing Mothers - It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when papaverine hydrochloride is administered to a nursing woman.
Section 34081-0 (34081-0)
Usage In Children - Safety and effectiveness in children have not been established.
Section 42228-7 (42228-7)
Usage In Pregnancy - Pregnancy Category C - No teratogenic effects were observed in rats when papaverine hydrochloride was administered subcutaneously as a single agent. It is not known whether papaverine can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Papaverine Hydrochloride should be given to a pregnant woman only if clearly needed.
Section 42229-5 (42229-5)
Rx Only
This product is to be used by or under the direction of a physician.
Each vial contains a sufficient amount to permit withdrawal and administration of the volume specified on the label.
How Supplied: (HOW SUPPLIED:)
Papaverine Hydrochloride Injection, USP, 30 mg/mL
NDC: 14789-121-07 2 mL Single-dose Vial
NDC: 14789-121-05 10 pack carton of 2 mL Single-dose Vials
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) (See USP Controlled Room Temperature).
PROTECT FROM LIGHT.RETAIN IN CARTON UNTIL TIME OF USE.
Container closer was not made with natural rubber latex.
Manufactured in Italy for:
Nexus Pharmaceuticals, LLC
Lincolnshire, IL 60069
PAPPI01ITR04 Revised 08/2024
NEXUS
PHARMACEUTICALS
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
The most characteristic effect of papaverine is relaxation of the tonus of all smooth muscle, especially when it has been spasmodically contracted. Papaverine Hydrochloride apparently acts directly on the muscle itself. This relaxation is noted in the vascular systemand bronchial musculatureand in the gastrointestinal, biliaryand urinary tracts.
The main actions of papaverine are exerted on cardiac and smooth muscle. Papaverine relaxes various smooth muscles, especially those of larger arteries; this relaxation may be prominent if spasm exists. The antispasmodic effect is a direct one and unrelated to muscle innervation, and the muscle still responds to drugs and other stimuli causing contraction. Papaverine has minimal actions on the central nervous system, although very large doses tend to produce some sedation and sleepiness in some patients. In certain circumstances, mild respiratory stimulation can be observed, but this is therapeutically inconsequential. Papaverine stimulates respiration by acting on carotid and aortic body chemoreceptors.
Papaverine relaxes the smooth musculature of the larger blood vessels, including the coronary, cerebral, peripheral, and pulmonary arteries. This action is particularly evident when such vessels are in spasm, induced reflexly or by drugs, and it provides the basis for the clinical use of papaverine in peripheral or pulmonary arterial embolism.
Experimentally in dogs, the alkaloid has been shown to cause fairly marked and long-lasting coronary vasodilatation and an increase in coronary blood flow. However, it also appears to have a direct inotropic effect and, when increased mechanical activity coincides with decreased systemic pressure, increases in coronary blood flow may not be sufficient to prevent brief periods of hypoxic myocardial depression.
Papaverine is effective by all routes of administration. A considerable fraction of the drug localizes in fat deposits and in the liver, with the remainder being distributed throughout the body. It is metabolized in the liver. About 90% of the drug is bound to plasma protein. Although estimates of its biologic half-life vary widely, reasonably constant plasma levels can be maintained with oral administration at 6 hour intervals. The drug is excreted in the urine in an inactive form.
Drug Abuse and Dependence (DRUG ABUSE AND DEPENDENCE)
Drug dependence resulting from the abuse of many of the selective depressants, including papaverine hydrochloride, has been reported.
Principal Display Panel – 2 Ml Vial Label (Principal Display Panel – 2 mL Vial Label)
NDC 14789-121-07
Rx Only
PAPAVERINE HYDROCHLORIDE
INJECTION, USP
60mg/2mL
(30 mg/mL)
Discard Unused Portion
2mL Single-dose Vials
NEXUS
PHARMACEUTICALS
Principal Display Panel – 2 Ml Carton Label (Principal Display Panel – 2 mL Carton Label)
NDC 14789-121-05
Rx Only
PAPAVERINE HYDROCHLORIDE
INJECTION, USP
60mg/2mL
(30 mg/mL)
Discard Unused Portion
10 x 2mL Single-dose Vials
NEXUS
PHARMACEUTICALS
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:44:06.627687 · Updated: 2026-03-14T22:18:42.758225