Tremfya
1e6dc9ae-1c4c-42d9-87aa-c315ecc51b56
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
TREMFYA is an interleukin-23 antagonist indicated for the treatment of: adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with moderate-to-severe plaque psoriasis and who are candidates for systemic therapy or phototherapy. ( 1.1 ) adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with active psoriatic arthritis. ( 1.2 ) adults with moderately to severely active ulcerative colitis. ( 1.3 ) adults with moderately to severely active Crohn's disease. ( 1.4 )
Dosage and Administration
For the treatment of ulcerative colitis or Crohn’s disease: Obtain liver enzymes and bilirubin levels prior to initiating treatment with TREMFYA. ( 2.1 , 5.4 ). For the treatment of plaque psoriasis or psoriatic arthritis, if clinically indicated, evaluate liver enzymes and bilirubin at baseline prior to initiating treatment with TREMFYA ( 2.1 , 5.4 ). Complete all age-appropriate vaccinations as recommended by current immunization guidelines prior to treatment initiation. ( 2.1 ) Recommended Dosage Plaque Psoriasis Adults 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. ( 2.2 ) Pediatric Patients 6 Years of Age and Older Who Also Weigh at Least 40 kg 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. ( 2.2 ) Psoriatic Arthritis Adults 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. TREMFYA can be used alone or in combination with a conventional DMARD (e.g., methotrexate). ( 2.3 ) Pediatric Patients 6 Years of Age and Older Who Also Weigh at Least 40 kg 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. TREMFYA may be administered alone or in combination with a conventional disease-modifying antirheumatic drug (e.g., methotrexate). ( 2.3 ) Ulcerative Colitis and Crohn’s Disease Induction : 200 mg administered by intravenous infusion over at least one hour at Week 0, Week 4, and Week 8 or 400 mg administered by subcutaneous injection at Week 0, Week 4, and Week 8. ( 2.4 ) Maintenance : 100 mg administered by subcutaneous injection at Week 16, and every 8 weeks thereafter, or 200 mg administered by subcutaneous injection at Week 12, and every 4 weeks thereafter. Use the lowest effective recommended dosage to maintain therapeutic response. ( 2.4 )
Contraindications
TREMFYA is contraindicated in patients with a history of serious hypersensitivity reaction to guselkumab or to any of the excipients [see Warnings and Precautions (5.1) ] .
Warnings and Precautions
Hypersensitivity Reactions : Serious hypersensitivity reactions, including anaphylaxis, may occur. ( 5.1 ) Infections : TREMFYA may increase the risk of infections. Do not initiate treatment with TREMFYA in patients with clinically important active infection until the infection resolves or is adequately treated. If such an infection develops, discontinue TREMFYA until the infection resolves. ( 5.2 ) Tuberculosis (TB) : Evaluate for TB prior to initiating treatment with TREMFYA. Monitor patients for signs and symptoms of active TB during and after treatment with TREMFYA. ( 5.3 ) Hepatotoxicity : Drug-induced liver injury has been reported. For the treatment of ulcerative colitis or Crohn’s disease, evaluate liver enzymes and bilirubin levels at baseline, for at least 16 weeks of treatment, and periodically thereafter according to routine patient management. For the treatment of plaque psoriasis or psoriatic arthritis, if clinically indicated, evaluate liver enzymes and bilirubin at baseline, and periodically thereafter according to routine patient management. Interrupt treatment if drug-induced liver injury is suspected, until this diagnosis is excluded. ( 5.4 ) Immunizations : Avoid use of live vaccines. ( 5.5 )
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of labeling: Hypersensitivity Reactions [see Contraindications (4) and Warnings and Precautions (5.1) ] Infections [see Warnings and Precautions (5.2) ] Tuberculosis [see Warnings and Precautions (5.3) ] Hepatotoxicity [see Warnings and Precautions (5.4) ]
Description
Indications and Usage ( 1.4 ) 03/2025 Indications and Usage ( 1.1 , 1.2 ) 09/2025 Dosage and Administration ( 2.1 , 2.5 , 2.6 ) 03/2025 Dosage and Administration ( 2.2 , 2.3 , 2.5 ) 09/2025 Dosage and Administration ( 2.4 ) 09/2025 Warnings and Precautions ( 5.3 , 5.4 ) 03/2025 Warnings and Precautions ( 5.2 , 5.4 ) 09/2025
Medication Information
Warnings and Precautions
Hypersensitivity Reactions : Serious hypersensitivity reactions, including anaphylaxis, may occur. ( 5.1 ) Infections : TREMFYA may increase the risk of infections. Do not initiate treatment with TREMFYA in patients with clinically important active infection until the infection resolves or is adequately treated. If such an infection develops, discontinue TREMFYA until the infection resolves. ( 5.2 ) Tuberculosis (TB) : Evaluate for TB prior to initiating treatment with TREMFYA. Monitor patients for signs and symptoms of active TB during and after treatment with TREMFYA. ( 5.3 ) Hepatotoxicity : Drug-induced liver injury has been reported. For the treatment of ulcerative colitis or Crohn’s disease, evaluate liver enzymes and bilirubin levels at baseline, for at least 16 weeks of treatment, and periodically thereafter according to routine patient management. For the treatment of plaque psoriasis or psoriatic arthritis, if clinically indicated, evaluate liver enzymes and bilirubin at baseline, and periodically thereafter according to routine patient management. Interrupt treatment if drug-induced liver injury is suspected, until this diagnosis is excluded. ( 5.4 ) Immunizations : Avoid use of live vaccines. ( 5.5 )
Indications and Usage
TREMFYA is an interleukin-23 antagonist indicated for the treatment of: adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with moderate-to-severe plaque psoriasis and who are candidates for systemic therapy or phototherapy. ( 1.1 ) adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with active psoriatic arthritis. ( 1.2 ) adults with moderately to severely active ulcerative colitis. ( 1.3 ) adults with moderately to severely active Crohn's disease. ( 1.4 )
Dosage and Administration
For the treatment of ulcerative colitis or Crohn’s disease: Obtain liver enzymes and bilirubin levels prior to initiating treatment with TREMFYA. ( 2.1 , 5.4 ). For the treatment of plaque psoriasis or psoriatic arthritis, if clinically indicated, evaluate liver enzymes and bilirubin at baseline prior to initiating treatment with TREMFYA ( 2.1 , 5.4 ). Complete all age-appropriate vaccinations as recommended by current immunization guidelines prior to treatment initiation. ( 2.1 ) Recommended Dosage Plaque Psoriasis Adults 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. ( 2.2 ) Pediatric Patients 6 Years of Age and Older Who Also Weigh at Least 40 kg 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. ( 2.2 ) Psoriatic Arthritis Adults 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. TREMFYA can be used alone or in combination with a conventional DMARD (e.g., methotrexate). ( 2.3 ) Pediatric Patients 6 Years of Age and Older Who Also Weigh at Least 40 kg 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. TREMFYA may be administered alone or in combination with a conventional disease-modifying antirheumatic drug (e.g., methotrexate). ( 2.3 ) Ulcerative Colitis and Crohn’s Disease Induction : 200 mg administered by intravenous infusion over at least one hour at Week 0, Week 4, and Week 8 or 400 mg administered by subcutaneous injection at Week 0, Week 4, and Week 8. ( 2.4 ) Maintenance : 100 mg administered by subcutaneous injection at Week 16, and every 8 weeks thereafter, or 200 mg administered by subcutaneous injection at Week 12, and every 4 weeks thereafter. Use the lowest effective recommended dosage to maintain therapeutic response. ( 2.4 )
Contraindications
TREMFYA is contraindicated in patients with a history of serious hypersensitivity reaction to guselkumab or to any of the excipients [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of labeling: Hypersensitivity Reactions [see Contraindications (4) and Warnings and Precautions (5.1) ] Infections [see Warnings and Precautions (5.2) ] Tuberculosis [see Warnings and Precautions (5.3) ] Hepatotoxicity [see Warnings and Precautions (5.4) ]
Description
Indications and Usage ( 1.4 ) 03/2025 Indications and Usage ( 1.1 , 1.2 ) 09/2025 Dosage and Administration ( 2.1 , 2.5 , 2.6 ) 03/2025 Dosage and Administration ( 2.2 , 2.3 , 2.5 ) 09/2025 Dosage and Administration ( 2.4 ) 09/2025 Warnings and Precautions ( 5.3 , 5.4 ) 03/2025 Warnings and Precautions ( 5.2 , 5.4 ) 09/2025
Section 42229-5
Adults
Induction:
The recommended induction dosage of TREMFYA is:
- 200 mg administered by intravenous infusion over at least one hour at Week 0, Week 4, and Week 8 [see Dosage and Administration (2.6)] or
- 400 mg administered by subcutaneous injection (given as two consecutive injections of 200 mg each) at Week 0, Week 4, and Week 8.
Section 42231-1
| Medication Guide | |||
|---|---|---|---|
| TREMFYA® (trem fye´ ah)
(guselkumab) injection, for subcutaneous or intravenous use |
|||
| This Medication Guide had been approved by the U.S. Food and Drug Administration. | Revised: 09/2025 | ||
|
What is the most important information I should know about TREMFYA?
|
|||
|
|||
|
|
||
|
|||
|
|
|
|
|
|||
|
|
||
| See "What are the possible side effects of TREMFYA?" for more information about side effects. | |||
| What is TREMFYA? | |||
| TREMFYA is a prescription medicine used to treat: | |||
|
|||
| It is not known if TREMFYA is safe and effective in children under 18 years of age with ulcerative colitis or Crohn’s disease or in children under 6 years of age with plaque psoriasis or psoriatic arthritis. | |||
| Do not use TREMFYA if you have had a serious allergic reaction to guselkumab or any of the other ingredients in TREMFYA. See the end of this Medication Guide for a complete list of ingredients in TREMFYA. | |||
| Before using TREMFYA, tell your healthcare provider about all of your medical conditions, including if you: | |||
|
|||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||
| How should I use TREMFYA? | |||
| See the detailed "Instructions for Use" that comes with TREMFYA for information on how to prepare and inject a dose of TREMFYA, and how to properly throw away (dispose of) the used TREMFYA prefilled syringe, One-Press injector or prefilled pen (TREMFYA PEN). | |||
|
|||
|
How will my child receive TREMFYA? See the detailed "Instructions for Use" that comes with TREMFYA.
|
|||
|
What are the possible side effects of TREMFYA?
|
|||
| The most common side effects of TREMFYA include: | |||
|
|
|
|
| These are not all the possible side effects of TREMFYA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||
How should I store TREMFYA?
|
|||
| General information about the safe and effective use of TREMFYA. | |||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use TREMFYA for a condition for which it was not prescribed. Do not give TREMFYA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about TREMFYA that is written for health professionals. | |||
|
What are the ingredients in TREMFYA?
Active ingredient: guselkumab Inactive ingredients: Single-dose prefilled syringe, single-dose One-Press patient-controlled injector, and single-dose prefilled pen (TREMFYA PEN) for subcutaneous use: L-histidine, L-histidine monohydrochloride monohydrate, polysorbate 80, sucrose and water for injection. Single-dose vial for intravenous infusion: EDTA disodium dihydrate, L-histidine, L-histidine monohydrochloride monohydrate, L-methionine, polysorbate 80, sucrose and water for injection. |
|||
| Manufactured by: Janssen Biotech, Inc., Horsham, PA 19044, USA. U.S. License Number 1864
For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2017–2025 For more information, call 1-800-526-7736 or go to www.tremfya.com. |
Section 43683-2
| Indications and Usage (1.4) | 03/2025 |
| Indications and Usage (1.1, 1.2) | 09/2025 |
| Dosage and Administration (2.1, 2.5, 2.6) | 03/2025 |
| Dosage and Administration (2.2, 2.3, 2.5) | 09/2025 |
| Dosage and Administration (2.4) | 09/2025 |
| Warnings and Precautions (5.3, 5.4) | 03/2025 |
| Warnings and Precautions (5.2, 5.4) | 09/2025 |
Section 44425-7
Storage and Handling
TREMFYA is sterile and preservative-free. Discard any unused portion.
- Store in a refrigerator between 2 °C to 8 °C (36 °F to 46 °F).
- Store in original carton until time of use.
- Protect from light until use.
- Do not freeze.
- Do not shake.
- Not made with natural rubber latex.
11 Description
Guselkumab, an interleukin-23 antagonist, is a human immunoglobulin G1 lambda (IgG1λ) monoclonal antibody. Guselkumab is produced in a mammalian cell line using recombinant DNA technology and has an approximate molecular weight of approximately 147 kDa.
TREMFYA® (guselkumab) injection is a sterile, preservative free, clear, and colorless to light yellow solution.
5.2 Infections
TREMFYA may increase the risk of infection [see Adverse Reactions (6.1)].
In placebo-controlled clinical trials of up to 48 weeks in subjects with ulcerative colitis and Crohn’s disease, serious infections occurred in ≤ 2% of subjects who received TREMFYA. In the 16-week placebo-controlled trials in subjects with plaque psoriasis, the rate of serious infections for the TREMFYA group and the placebo group was ≤ 0.2%. A similar rate of serious infections was seen in placebo-controlled trials in subjects with psoriatic arthritis. The overall rates of infections were similar between subjects in the TREMFYA groups and subjects in the placebo groups in clinical trials for all indicated populations [see Adverse Reactions (6.1)].
Treatment with TREMFYA should not be initiated in patients with any clinically important active infection until the infection resolves or is adequately treated.
In patients with a chronic infection or a history of recurrent infection, consider the risks and benefits prior to prescribing TREMFYA. Instruct patients to seek medical help if signs or symptoms of clinically important chronic or acute infection occur. If a patient develops a clinically important or serious infection or is not responding to standard therapy, monitor the patient closely and discontinue TREMFYA until the infection resolves.
5.3 Tuberculosis
Evaluate patients for tuberculosis (TB) infection prior to initiating TREMFYA treatment. Do not administer TREMFYA to patients with active TB infection. Initiate treatment of latent TB prior to administering TREMFYA. Consider anti-TB therapy prior to initiating TREMFYA in patients with a past history of latent or active TB in whom an adequate course of treatment cannot be confirmed. Monitor all patients for signs and symptoms of active TB during and after TREMFYA treatment.
In clinical trials, 105 subjects with plaque psoriasis, 71 subjects with psoriatic arthritis, 43 subjects with ulcerative colitis, and 36 subjects with Crohn's disease with latent TB who were concurrently treated with TREMFYA, and appropriate TB prophylaxis did not develop active TB. In clinical trials of TREMFYA in subjects with Crohn's disease, active TB was reported in 2 subjects during treatment with TREMFYA [see Adverse Reactions (6.1)].
5.5 Immunizations
Avoid use of live vaccines in patients treated with TREMFYA. Medications that interact with the immune system may increase the risk of infection following administration of live vaccines. Prior to initiating therapy with TREMFYA, complete all age-appropriate vaccinations according to current immunization guidelines. No data are available on the response to live or inactive vaccines.
8.5 Geriatric Use
Of the 5723 subjects with plaque psoriasis, psoriatic arthritis, ulcerative colitis, or Crohn's disease exposed to TREMFYA, a total of 313 subjects were 65 years or older, and 32 subjects were 75 years or older. Clinical studies of TREMFYA, within each indication, did not include sufficient numbers of subjects 65 years of age and older to determine whether they respond differently from younger adult subjects.
No clinically meaningful differences in the pharmacokinetics of guselkumab were observed based on age [see Clinical Pharmacology (12.3)].
5.4 Hepatotoxicity
A serious adverse reaction of drug-induced liver injury was reported in a clinical trial subject with Crohn's disease following three doses of a higher than the recommended induction regimen. This subject had peak alanine aminotransferase (ALT) of 18x the upper limit of normal (ULN), aspartate aminotransferase (AST) of 11x ULN, and total bilirubin of 2.4x ULN. TREMFYA was subsequently discontinued, and the liver test abnormalities resolved following administration of corticosteroids [see Adverse Reactions (6.1)].
In patients with ulcerative colitis or Crohn's disease, evaluate liver enzymes and bilirubin at baseline, for at least 16 weeks of treatment, and periodically thereafter according to routine patient management.
In patients with plaque psoriasis or psoriatic arthritis, if clinically indicated, evaluate liver enzymes and bilirubin at baseline, and periodically thereafter according to routine patient management.
Consider other treatment options in patients with evidence of acute liver disease or cirrhosis. Prompt investigation of the cause of liver enzyme elevation is recommended to identify potential cases of drug-induced liver injury. Interrupt treatment if drug-induced liver injury is suspected, until this diagnosis is excluded. Instruct patients to seek immediate medical attention if they experience symptoms suggestive of hepatic dysfunction.
1.4 Crohn's Disease
TREMFYA is indicated for the treatment of adult patients with moderately to severely active Crohn's disease.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of guselkumab or of other guselkumab products.
4 Contraindications
TREMFYA is contraindicated in patients with a history of serious hypersensitivity reaction to guselkumab or to any of the excipients [see Warnings and Precautions (5.1)].
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of labeling:
- Hypersensitivity Reactions [see Contraindications (4) and Warnings and Precautions (5.1)]
- Infections [see Warnings and Precautions (5.2)]
- Tuberculosis [see Warnings and Precautions (5.3)]
- Hepatotoxicity [see Warnings and Precautions (5.4)]
1.1 Plaque Psoriasis
TREMFYA is indicated for the treatment of adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with moderate-to-severe plaque psoriasis and who are candidates for systemic therapy or phototherapy.
Instructions for Use
TREMFYA® [trem fye' ah]
(guselkumab)
injection, for subcutaneous use
200 mg/2mL (100 mg/mL) Prefilled Syringe
This Instructions for Use contains information on how to inject TREMFYA.
| SINGLE-DOSE |
Important Information You Need to Know Before Injecting TREMFYA
TREMFYA comes in a single-dose prefilled syringe containing one 200 mg dose.
| Your healthcare provider will tell you if you will need to use 1 or 2 prefilled syringes. |
If your healthcare provider decides that you or a caregiver may be able to give your injections of TREMFYA at home, you should receive training on the correct way to prepare and inject TREMFYA before using the prefilled syringe.
Read this Instructions for Use before using your TREMFYA prefilled syringe and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your healthcare provider about your medical condition or your treatment.
Each TREMFYA prefilled syringe can only be used one time. Throw the used prefilled syringe away (see Step 4) after one dose, even if there is still medicine left in it. Do not reuse your TREMFYA prefilled syringe.
The TREMFYA prefilled syringe is intended for injection under the skin, not into the muscle or vein. After injection, the needle will retract into the device and lock into place.
Storage information
Store in refrigerator between 36°F to 46°F (2°C to 8°C).
Do not freeze TREMFYA prefilled syringe.
Do not shake your TREMFYA prefilled syringe.
Keep TREMFYA prefilled syringe and all medicines out of reach of children.
Keep TREMFYA prefilled syringe in the original carton to protect from light and physical damage.
Prefilled syringe parts
1. Get ready
Check your dose to see if you will need to use 1 or 2 prefilled syringes and inspect carton(s)
Remove the carton(s) from the refrigerator.
Check the expiration ('EXP') date. Do not use your prefilled syringe if the expiration date has passed or if the seal on the carton is broken. Contact your healthcare provider or pharmacist for a new prefilled syringe.
Allow TREMFYA to come to room temperature
Let the cartons sit on a flat surface at room temperature for approximately 30 minutes before use.
Do not warm the prefilled syringe(s) any other way.
2. Prepare to inject TREMFYA
Take the prefilled syringe out of the carton.
Inspect liquid to see that it is clear and colorless to slightly yellow
Check the liquid in the viewing window. It should be clear and colorless to slightly yellow and may contain tiny white or clear particles. You may also see air bubbles. This is normal.
Do not inject if the liquid is:
- cloudy or
- discolored or
- has large particles
Do not use the prefilled syringe if it is dropped. Call your healthcare provider or pharmacist for a new prefilled syringe.
Choose injection site
Select a site from the following areas for your injection:
- Front of thighs
- Lower stomach area (lower abdomen), except for a 2-inch area right around your navel (belly-button)
- Back of upper arms (only if someone else is giving you the injection)
If you need to give 2 injections to complete your dose, choose different areas or leave at least 2 inches between injection sites.
Do not inject into skin that is tender, bruised, red, scaly, thick or hard. Avoid areas with scars or stretch marks.
Wash hands and clean injection site
Wash your hands well with soap and warm water.
Wipe your chosen injection site with an alcohol swab and allow it to dry.
Do not touch, fan, or blow on the injection site after you have cleaned it.
3. Inject TREMFYA
Remove needle cover when you are ready to inject
Hold the prefilled syringe by the body and pull needle cover straight off. It is normal to see a few drops of liquid.
Inject TREMFYA within 5 minutes of removing the needle cover.
Do not put needle cover back on, as this may damage the needle or cause a needle stick injury.
Do not touch needle or let it touch any surface.
Do not use the prefilled syringe if it is dropped. Call your healthcare provider or pharmacist for a new prefilled syringe.
Do not hold or pull the plunger at any time.
Pinch injection site and insert needle at about a 45-degree angle
It is important to pinch enough skin to inject under the skin and not into muscle.
Insert needle with a quick dart-like motion.
Slowly press plunger all the way down until it stops to inject all of the liquid
You will feel some resistance as you press the plunger, this is normal.
Release pressure from plunger to remove the needle from the skin
The needle will retract into the device and lock into place.
If your prescribed dose requires two injections, repeat Steps 2 to 4 with the second prefilled syringe.
4. After your injection
Check injection site
There may be a small amount of blood or liquid at the injection site. Gently hold pressure over the injection site with a cotton ball or gauze pad until any bleeding stops.
Do not rub the injection site.
If needed, cover the injection site with a bandage.
Dispose of your prefilled syringe
Put the used prefilled syringe in an FDA-cleared sharps disposal container right away after use.
Do not throw away (dispose of) your prefilled syringe in your household trash.
Do not recycle your used sharps disposal container.
For more information, see Disposing of TREMFYA prefilled syringe.
Disposing of TREMFYA prefilled syringe
If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- made of heavy-duty plastic
- can be closed with a tight-fitting, puncture resistant lid, without sharps being able to come out
- upright and stable during use
- leak-resistant
- properly labeled to warn of hazardous waste inside the container
When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes.
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: www.fda.gov/safesharpsdisposal. You may also consult your pharmacist.
Need help?
Call your healthcare provider to talk about any questions you may have. For additional assistance or to share your feedback, call 800-526-7736.
Manufactured by:
Janssen Biotech, Inc.
Horsham, PA 19044, USA
US License No. 1864
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Approved: August / 2025
Instructions for Use
TREMFYA®
(trem fye' ah)
(guselkumab)
Prefilled Syringe
SINGLE-DOSE
Important
TREMFYA comes as a single-dose prefilled syringe containing one 100 mg dose. Each TREMFYA prefilled syringe can only be used one time. Throw the used prefilled syringe away (See Step 3) after one dose, even if there is medicine left in it. Do not reuse your TREMFYA prefilled syringe.
If your healthcare provider decides that you or a caregiver may be able to give your injections of TREMFYA at home, you should receive training on the right way to prepare and inject TREMFYA using the prefilled syringe before attempting to inject. Do not try to inject yourself until you have been shown the right way to give the injections by your healthcare provider.
For children, TREMFYA should be administered by a healthcare provider or an adult caregiver who has been shown the right way to give the injections by your healthcare provider.
Read this Instructions for Use before using your TREMFYA prefilled syringe and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your healthcare provider about your medical condition or your treatment.
The TREMFYA prefilled syringe is intended for injection under the skin, not into the muscle or vein. After injection, the needle will retract into the body of the device and lock into place.
Storage information
Store in refrigerator at 36 °F to 46 °F (2 °C to 8 °C). Do not freeze TREMFYA prefilled syringe.
Keep TREMFYA prefilled syringe and all medicines out of reach of children.
Do not shake your TREMFYA prefilled syringe.
Keep TREMFYA prefilled syringe in the original carton to protect from light and physical damage.
Prefilled syringe parts
Before use
After use
|
You will need these supplies:
Not provided in the TREMFYA prefilled syringe carton:
|
1. Prepare for your injection
Inspect carton
Remove your TREMFYA prefilled syringe carton from the refrigerator.
Keep the prefilled syringe in the carton and let it sit on a flat surface at room temperature for at least 30 minutes before use.
Do not warm the prefilled syringe any other way.
Check the expiration date ('EXP') on the back panel of the carton.
Do not use your prefilled syringe if the expiration date has passed.
Do not inject TREMFYA if the perforations on the carton are broken. Call your healthcare provider or pharmacist for a refill.
Choose injection site
Select from the following areas for your injection:
- Front of thighs (recommended)
- Lower stomach area (lower abdomen), except for a 2-inch area right around your navel (belly-button)
- Back of upper arms (only if someone else is giving you the injection)
Do not inject into skin that is tender, bruised, red, hard, thick, scaly or affected by psoriasis.
Clean injection site
Wash your hands well with soap and warm water.
Wipe your chosen injection site with an alcohol swab and allow it to dry.
Do not touch, fan, or blow on the injection site after you have cleaned it.
Inspect liquid
Take your TREMFYA prefilled syringe out of the carton.
Check the TREMFYA prefilled syringe liquid in the viewing window. It should be clear to slightly yellow and may contain tiny white or clear particles. You may also see one or more air bubbles. This is normal.
Do not inject if the liquid is cloudy or discolored, or has large particles. Call your healthcare provider or pharmacist for a refill.
2. Inject TREMFYA using prefilled syringe
Remove needle cover
Hold your prefilled syringe by the body and pull needle cover straight off. It is normal to see a drop of liquid.
Inject TREMFYA within 5 minutes of removing the needle cover.
Do not put needle cover back on, as this may damage the needle or cause a needle stick injury.
Do not touch needle or let it touch any surface.
Do not use a TREMFYA prefilled syringe if it is dropped. Call your healthcare provider or pharmacist for a refill.
Position fingers and insert needle
Place your thumb, index and middle fingers directly under the finger flange, as shown.
Do not touch plunger or area above finger flange as this may cause the needle safety device to activate.
Use your other hand to pinch skin at the injection site. Position syringe at about a 45 degree angle to the skin.
It is important to pinch enough skin to inject under the skin and not into the muscle.
Insert needle with a quick, dart-like motion.
Release pinch and reposition hand
Use your free hand to grasp the body of the prefilled syringe.
Press plunger
Place thumb from the opposite hand on the plunger and press the plunger all the way down until it stops.
Release pressure from plunger
The safety guard will cover the needle and lock into place, removing the needle from your skin.
3. After your injection
Dispose of your prefilled syringe
Put your used TREMFYA prefilled syringe in an FDA-cleared sharps disposal container right away after use.
Do not throw away (dispose of) your TREMFYA prefilled syringe in your household trash.
Do not recycle your used sharps disposal container.
For more information, see "How should I dispose of the used prefilled syringe?"
Check injection site
There may be a small amount of blood or liquid at the injection site. Hold pressure over your skin with a cotton ball or gauze pad until any bleeding stops.
Do not rub the injection site.
If needed, cover injection site with a bandage.
Need help?
Call your healthcare provider to talk about any questions you may have. For additional assistance or to share your feedback call 1-800-526-7736.
How should I dispose of the used prefilled syringe?
If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out
- upright and stable during use
- leak-resistant
- properly labeled to warn of hazardous waste inside the container
When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes.
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: www.fda.gov/safesharpsdisposal
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by:
Janssen Biotech, Inc.
Horsham, PA 19044, USA
U.S. License No. 1864
Approved: September 2025
12.2 Pharmacodynamics
In evaluated adult subjects with moderate-to-severe plaque psoriasis, guselkumab reduced serum levels of IL-17A, IL-17F and IL-22 relative to pre-treatment levels based on exploratory analyses of the pharmacodynamic markers.
In evaluated adult subjects with active psoriatic arthritis, serum levels of acute phase proteins C-reactive protein, serum amyloid A and IL-6, and Th17 effector cytokines IL-17A, IL-17F and IL-22 were elevated at baseline. Serum levels of these proteins measured at Week 4 and Week 24 were decreased compared to baseline following guselkumab treatment at Week 0, Week 4, and every 8 weeks thereafter.
The relationship between these pharmacodynamic markers and the mechanism(s) by which guselkumab exerts its clinical effects is unknown.
12.3 Pharmacokinetics
Guselkumab exhibited linear pharmacokinetics in healthy adult subjects and adult subjects with plaque psoriasis following subcutaneous injections. In subjects with plaque psoriasis, following subcutaneous administration of 100 mg of TREMFYA at Weeks 0 and 4, and every 8 weeks thereafter, mean steady-state trough serum guselkumab concentration was approximately 1.2 mcg/mL.
The pharmacokinetics of guselkumab in adult subjects with psoriatic arthritis was similar to that in adult subjects with plaque psoriasis. Following subcutaneous administration of 100 mg of TREMFYA at Weeks 0, 4, and every 8 weeks thereafter, mean steady-state trough serum guselkumab concentration was approximately 1.2 mcg/mL.
Following subcutaneous maintenance dosing of 100 mg TREMFYA every 8 weeks or 200 mg TREMFYA every 4 weeks in adult subjects with ulcerative colitis, mean steady-state trough serum guselkumab concentrations were approximately 1.4 mcg/mL and 10.7 mcg/mL, respectively.
Following subcutaneous maintenance dosing of 100 mg TREMFYA every 8 weeks, or 200 mg TREMFYA every 4 weeks, in adult subjects with Crohn's disease mean steady-state trough serum guselkumab concentrations were approximately 1.2 mcg/mL and 10.1 mcg/mL, respectively.
7.1 Cyp450 Substrates
The formation of CYP450 enzymes can be altered by increased levels of certain cytokines (e.g., IL-1, IL-6, IL-10, TNFα, interferon) during chronic inflammation.
Results from an exploratory drug-drug interaction trial in subjects with moderate-to-severe plaque psoriasis suggested a low potential for clinically relevant drug interactions for drugs metabolized by CYP3A4, CYP2C9, CYP2C19 and CYP1A2 but the interaction potential cannot be ruled out for drugs metabolized by CYP2D6. However, the results were highly variable because of the limited number of subjects in the trial.
Upon initiation of TREMFYA in patients who are receiving concomitant CYP450 substrates, particularly those with a narrow therapeutic index, consider monitoring for therapeutic effect or drug concentration and consider dosage adjustment as needed [see Clinical Pharmacology (12.3)].
1.3 Ulcerative Colitis
TREMFYA is indicated for the treatment of adult patients with moderately to severely active ulcerative colitis.
1 Indications and Usage
TREMFYA is an interleukin-23 antagonist indicated for the treatment of:
- adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with moderate-to-severe plaque psoriasis and who are candidates for systemic therapy or phototherapy. (1.1)
- adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with active psoriatic arthritis. (1.2)
- adults with moderately to severely active ulcerative colitis. (1.3)
- adults with moderately to severely active Crohn's disease. (1.4)
1.2 Psoriatic Arthritis
TREMFYA is indicated for the treatment of adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with active psoriatic arthritis.
12.1 Mechanism of Action
Guselkumab is a human monoclonal IgG1λ antibody that selectively binds to the p19 subunit of interleukin 23 (IL-23) and inhibits its interaction with the IL-23 receptor. IL-23 is a naturally occurring cytokine that is involved in normal inflammatory and immune responses. Guselkumab inhibits the release of proinflammatory cytokines and chemokines.
5 Warnings and Precautions
- Hypersensitivity Reactions: Serious hypersensitivity reactions, including anaphylaxis, may occur. (5.1)
- Infections: TREMFYA may increase the risk of infections. Do not initiate treatment with TREMFYA in patients with clinically important active infection until the infection resolves or is adequately treated. If such an infection develops, discontinue TREMFYA until the infection resolves. (5.2)
- Tuberculosis (TB): Evaluate for TB prior to initiating treatment with TREMFYA. Monitor patients for signs and symptoms of active TB during and after treatment with TREMFYA. (5.3)
- Hepatotoxicity: Drug-induced liver injury has been reported. For the treatment of ulcerative colitis or Crohn’s disease, evaluate liver enzymes and bilirubin levels at baseline, for at least 16 weeks of treatment, and periodically thereafter according to routine patient management. For the treatment of plaque psoriasis or psoriatic arthritis, if clinically indicated, evaluate liver enzymes and bilirubin at baseline, and periodically thereafter according to routine patient management. Interrupt treatment if drug-induced liver injury is suspected, until this diagnosis is excluded. (5.4)
- Immunizations: Avoid use of live vaccines. (5.5)
2 Dosage and Administration
- For the treatment of ulcerative colitis or Crohn’s disease: Obtain liver enzymes and bilirubin levels prior to initiating treatment with TREMFYA. (2.1, 5.4).
- For the treatment of plaque psoriasis or psoriatic arthritis, if clinically indicated, evaluate liver enzymes and bilirubin at baseline prior to initiating treatment with TREMFYA (2.1, 5.4).
- Complete all age-appropriate vaccinations as recommended by current immunization guidelines prior to treatment initiation. (2.1)
Recommended Dosage
Plaque Psoriasis
Adults
- 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. (2.2)
Pediatric Patients 6 Years of Age and Older Who Also Weigh at Least 40 kg
- 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. (2.2)
Psoriatic Arthritis
Adults
- 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. TREMFYA can be used alone or in combination with a conventional DMARD (e.g., methotrexate). (2.3)
Pediatric Patients 6 Years of Age and Older Who Also Weigh at Least 40 kg
- 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. TREMFYA may be administered alone or in combination with a conventional disease-modifying antirheumatic drug (e.g., methotrexate). (2.3)
Ulcerative Colitis and Crohn’s Disease
- Induction: 200 mg administered by intravenous infusion over at least one hour at Week 0, Week 4, and Week 8 or 400 mg administered by subcutaneous injection at Week 0, Week 4, and Week 8. (2.4)
- Maintenance: 100 mg administered by subcutaneous injection at Week 16, and every 8 weeks thereafter, or 200 mg administered by subcutaneous injection at Week 12, and every 4 weeks thereafter. Use the lowest effective recommended dosage to maintain therapeutic response. (2.4)
3 Dosage Forms and Strengths
TREMFYA is a clear and colorless to light yellow solution.
6.2 Postmarketing Experience
The following adverse reactions have been reported during post-approval of TREMFYA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to TREMFYA exposure.
Immune system disorders: Hypersensitivity, including anaphylaxis [see Warnings and Precautions (5.1)]
Skin and subcutaneous tissue disorders: Rash [see Warnings and Precautions (5.1)]
5.1 Hypersensitivity Reactions
Serious hypersensitivity reactions, including anaphylaxis, have been reported with post market use of TREMFYA. Some cases required hospitalization. If a serious hypersensitivity reaction occurs, discontinue TREMFYA and initiate appropriate therapy.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patients and/or caregivers to read the FDA-approved patient labeling (Medication Guide and Instructions for Use) before starting TREMFYA therapy, and each time the prescription is renewed, as there may be new information they need to know.
Principal Display Panel 100 Mg/ml Pen Carton
NDC 57894-640-06
Tremfya® PEN
(guselkumab)
Injection
100 mg/mL
FOR SUBCUTANEOUS USE ONLY
Rx only
One single-dose prefilled pen
Discard unused portion
Attention: Dispense the enclosed
Medication Guide to each patient.
Principal Display Panel 200 Mg/2 Ml Pen Carton
NDC 57894-651-02
Tremfya® PEN
(guselkumab)
Injection
200 mg/2 mL
FOR SUBCUTANEOUS USE ONLY
Rx only
One single-dose prefilled pen
Discard unused portion
ATTENTION: Dispense the enclosed
Medication Guide to each patient.
Principal Display Panel 200 Mg/20 Ml Vial Carton
NDC 57894-650-02
Tremfya®
(guselkumab)
Injection
200 mg/20 mL
(10 mg/mL)
FOR INTRAVENOUS
INFUSION ONLY
Attention: Dispense the
enclosed Medication Guide
to each patient.
Rx only
Single-dose vial
Discard unused portion
14.4 Clinical Studies in Adults With Crohn's Disease
The efficacy and safety of TREMFYA were assessed in three randomized, double-blind, placebo-controlled trials that enrolled adult subjects with moderately to severely active Crohn's disease who had a history of inadequate response, loss of response, or intolerance to oral corticosteroids, immunomodulators (azathioprine, 6-mercaptopurine, methotrexate), and/or biologic therapy (TNF blockers or vedolizumab). Moderately to severely active Crohn's disease was defined as a Crohn's Disease Activity Index (CDAI) score of ≥220 and a Simple Endoscopic Score for Crohn's Disease (SES-CD) of ≥6 (or ≥4 for subjects with isolated ileal disease). Subjects were permitted to use stable doses of oral corticosteroids (prednisone ≤40 mg/day or equivalent), immunomodulators (azathioprine, 6-mercaptopurine, methotrexate), and/or aminosalicylates.
Principal Display Panel 200 Mg/2 Ml Syringe Carton
NDC 57894-651-22
Tremfya®
(guselkumab)
Injection
200 mg/2 mL
FOR SUBCUTANEOUS USE ONLY
Rx only
One single-dose prefilled syringe
Discard unused portion
ATTENTION: Dispense the enclosed
Medication Guide to each patient.
2.3 Recommended Dosage for Active Psoriatic Arthritis
Administer TREMFYA by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter.
TREMFYA may be administered alone or in combination with a conventional disease-modifying antirheumatic drug (e.g., methotrexate).
Adults
The recommended dose is 100 mg.
Pediatric Patients 6 Years of Age and Older Who Also Weigh at Least 40 kg
The recommended dose is 100 mg.
14.3 Clinical Studies in Adults With Ulcerative Colitis
The efficacy and safety of TREMFYA was assessed in three randomized, double-blind, placebo-controlled trials (UC1, UC2, and UC 4) that enrolled adult subjects with moderately to severely active ulcerative colitis. Disease activity was assessed by the modified Mayo score (mMS), a 3-component Mayo score (0 –9) which consists of the following subscores (0 to 3 for each subscore): stool frequency (SFS), rectal bleeding (RBS), and findings on centrally reviewed endoscopy (ES). An ES of 2 was defined by marked erythema, lack of vascular pattern, friability, and/or erosions; an ES of 3 was defined by spontaneous bleeding and ulceration. Enrolled subjects with a mMS between 5 and 9 and an ES of 2 or 3 were classified as having moderately to severely active ulcerative colitis. Subjects with inadequate response, loss of response, or intolerance to corticosteroids, immunomodulators (azathioprine, 6-mercaptopurine), biologic therapy (TNF blockers, vedolizumab), and/or Janus kinase (JAK) inhibitors were enrolled. In UC4, subjects with inadequate response, loss of response, or intolerance to sphingosine-1-phosphate receptor modulators (S1PRM) were also enrolled.
14.2 Clinical Studies in Adults With Psoriatic Arthritis
The safety and efficacy of TREMFYA were assessed in 2 randomized, double-blind, placebo-controlled trials (PsA1 [NCT03162796] and PsA2 [NCT03158285]) in 1120 adult subjects with active psoriatic arthritis (PsA) (≥3 swollen joints, ≥3 tender joints, and a C-reactive protein (CRP) level of ≥0.3 mg/dL in PsA1 and ≥5 swollen joints, ≥5 tender joints, and a CRP level of ≥0.6 mg/dL in PsA2) who had inadequate response to standard therapies (e.g., conventional DMARDs [cDMARDs]), apremilast, or nonsteroidal anti-inflammatory drugs [NSAIDs]). Subjects in these trials had a diagnosis of PsA for at least 6 months based on the Classification criteria for Psoriatic Arthritis (CASPAR) and a median duration of PsA of 4 years at baseline.
In trial PsA1, approximately 31% of subjects had been previously treated with up to 2 anti-tumor necrosis factor alpha (anti-TNFα) agents whereas in trial PsA2, all subjects were biologic naïve. Approximately 58% of subjects from both trials had concomitant methotrexate (MTX) use. Subjects with different subtypes of PsA were enrolled in both trials, including polyarticular arthritis with the absence of rheumatoid nodules (40%), spondylitis with peripheral arthritis (30%), asymmetric peripheral arthritis (23%), distal interphalangeal involvement (7%) and arthritis mutilans (1%). At baseline, over 65% and 42% of the subjects had enthesitis and dactylitis, respectively and 79% had ≥3% body surface area (BSA) psoriasis skin involvement.
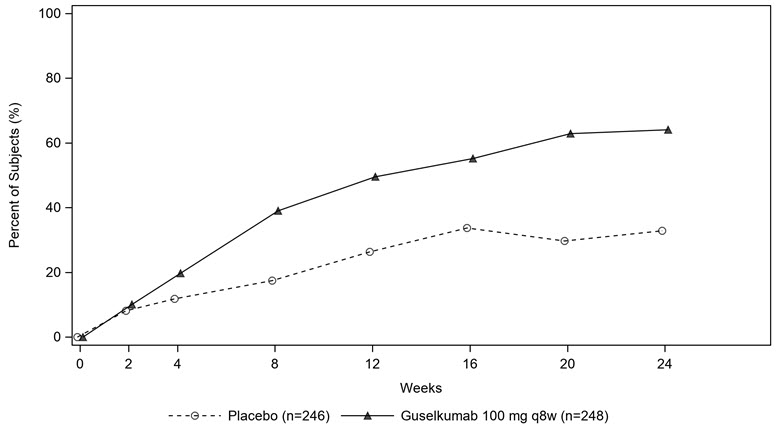
Trial PsA1 evaluated 381 subjects who were treated with placebo SC, TREMFYA 100 mg SC at Weeks 0, 4 and every 8 weeks (q8w) thereafter, or TREMFYA 100 mg SC every 4 weeks (q4w). Trial PsA2 evaluated 739 subjects who were treated with placebo SC, TREMFYA 100 mg SC at Weeks 0, 4 and q8w thereafter, or TREMFYA 100 mg SC q4w. The primary endpoint in both trials was the percentage of subjects achieving an ACR20 response at Week 24.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Animal studies have not been conducted to evaluate the carcinogenic or mutagenic potential of TREMFYA.
No effects on fertility parameters were observed after male guinea pigs were subcutaneously administered guselkumab at a dose of 25 mg/kg twice weekly (6 times the exposure (AUC) in humans administered 200 mg intravenously and 5 times the exposure (AUC) at the 400 mg subcutaneous dose).
No effects on fertility parameters were observed after female guinea pigs were subcutaneously administered guselkumab at doses up to 100 mg/kg twice weekly (12 times the exposure (AUC) in humans administered 200 mg intravenously and 10 times the exposure (AUC) at the 400 mg subcutaneous dose).
2.2 Recommended Dosage for Moderate to Severe Plaque Psoriasis
Administer TREMFYA by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter.
Adults
The recommended dose is 100 mg.
Pediatric Patients 6 Years of Age and Older Who Also Weigh at Least 40 kg
The recommended dose is 100 mg.
Principal Display Panel 200 Mg/2 Ml Pen Carton Ndc 57894 651 04
NDC 57894-651-04
Rx only
Tremfya® PEN
(guselkumab)
Injection
200 mg/2 mL
FOR SUBCUTANEOUS USE ONLY
Induction Pack for Crohn's Disease
and Ulcerative Colitis
(includes 1 induction dose)
The entire carton is to
be dispensed as a unit.
Contains: Two cartons each containing
one single-dose prefilled pen.
Discard unused portion.
ATTENTION: Dispense the enclosed
Medication Guide to each patient.
Principal Display Panel 100 Mg/ml Syringe Carton Ndc 57894 640 01
Tremfya®
(guselkumab)
Injection
100 mg/mL
FOR SUBCUTANEOUS USE ONLY
Rx only
NDC 57894-640-01
One single-dose prefilled syringe
Discard unused portion
Contains no preservative
ATTENTION: Dispense the enclosed
Medication Guide to each patient
Principal Display Panel 100 Mg/ml Syringe Carton Ndc 57894 640 11
Tremfya®
(guselkumab)
Injection
100 mg/mL
FOR SUBCUTANEOUS USE ONLY
Rx only
NDC 57894-640-11
Single-dose One-Press
patient-controlled injector
Discard unused portion
Contains no preservative
ATTENTION: Dispense the enclosed
Medication Guide to each patient
2.5 Preparation and Administration Instructions for Subcutaneous Injection
TREMFYA is available for subcutaneous use in the following presentations: prefilled pen (TREMFYA PEN), One-Press injector, and prefilled syringes [see Dosage Forms and Strengths (3) and How Supplied/Storage and Handling (16)].
- Each prefilled pen, One-Press injector, or prefilled syringe is for one time use in one patient only. Instruct patients to inject the full amount: 100 mg or 200 mg of TREMFYA (1 mL or 2 mL, respectively).
-
TREMFYA is intended for use under the guidance and supervision of a healthcare professional. After proper training in subcutaneous injection technique:
Adults- Adults may self-inject with the TREMFYA prefilled syringe, One-Press injector, and prefilled pen.
- Inject into the front of the thighs, the lower abdomen except for the 2 inches around the navel, or the back of the upper arms (healthcare professional or caregiver only).
- Pediatric self-administration is not recommended. Administration of TREMFYA to pediatric patients with the prefilled syringe, One-Press injector and prefilled pen should be performed by a healthcare provider or by a caregiver who has received training and demonstrated proper subcutaneous injection technique.
- Inject into the front of the thighs or the lower abdomen except for the 2 inches around the navel. For the prefilled syringe only, injection can also be given in the back of the upper arms.
- Before injection, remove TREMFYA from the refrigerator and allow to reach room temperature up to 25 °C (77 °F) (30 minutes) without removing the needle cap.
- Do not inject TREMFYA into areas where the skin is tender, bruised, red, hard, thick, scaly, or affected by psoriasis [see Instructions for Use].
- The TREMFYA Instructions for Use contains more detailed patient instructions on the preparation and administration of TREMFYA [see Instructions for Use].
- If a dose is missed, administer the dose as soon as possible. Thereafter, resume dosing at the regular scheduled time.
- Inspect TREMFYA visually for particulate matter and discoloration prior to administration. TREMFYA is a clear and colorless to light yellow solution that may contain small translucent particles. Do not use if the liquid contains large particles, is discolored or cloudy. TREMFYA does not contain preservatives; therefore, discard any unused product remaining in the prefilled pen, One-Press injector, or prefilled syringe.
2.1 Recommended Evaluations and Immunizations Prior to Treatment Initiation
- Evaluate patients for tuberculosis (TB) infection prior to initiating treatment with TREMFYA [see Warnings and Precautions (5.3)].
- For the treatment of ulcerative colitis or Crohn’s disease, obtain liver enzymes and bilirubin levels prior to initiating treatment with TREMFYA [see Warnings and Precautions (5.4)].
- For the treatment of plaque psoriasis or psoriatic arthritis, if clinically indicated, evaluate liver enzymes and bilirubin prior to initiating treatment with TREMFYA [see Warnings and Precautions (5.4)].
- Complete all age-appropriate vaccinations according to current immunization guidelines [see Warnings and Precautions (5.5)].
Structured Label Content
Section 42229-5 (42229-5)
Adults
Induction:
The recommended induction dosage of TREMFYA is:
- 200 mg administered by intravenous infusion over at least one hour at Week 0, Week 4, and Week 8 [see Dosage and Administration (2.6)] or
- 400 mg administered by subcutaneous injection (given as two consecutive injections of 200 mg each) at Week 0, Week 4, and Week 8.
Section 42231-1 (42231-1)
| Medication Guide | |||
|---|---|---|---|
| TREMFYA® (trem fye´ ah)
(guselkumab) injection, for subcutaneous or intravenous use |
|||
| This Medication Guide had been approved by the U.S. Food and Drug Administration. | Revised: 09/2025 | ||
|
What is the most important information I should know about TREMFYA?
|
|||
|
|||
|
|
||
|
|||
|
|
|
|
|
|||
|
|
||
| See "What are the possible side effects of TREMFYA?" for more information about side effects. | |||
| What is TREMFYA? | |||
| TREMFYA is a prescription medicine used to treat: | |||
|
|||
| It is not known if TREMFYA is safe and effective in children under 18 years of age with ulcerative colitis or Crohn’s disease or in children under 6 years of age with plaque psoriasis or psoriatic arthritis. | |||
| Do not use TREMFYA if you have had a serious allergic reaction to guselkumab or any of the other ingredients in TREMFYA. See the end of this Medication Guide for a complete list of ingredients in TREMFYA. | |||
| Before using TREMFYA, tell your healthcare provider about all of your medical conditions, including if you: | |||
|
|||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||
| How should I use TREMFYA? | |||
| See the detailed "Instructions for Use" that comes with TREMFYA for information on how to prepare and inject a dose of TREMFYA, and how to properly throw away (dispose of) the used TREMFYA prefilled syringe, One-Press injector or prefilled pen (TREMFYA PEN). | |||
|
|||
|
How will my child receive TREMFYA? See the detailed "Instructions for Use" that comes with TREMFYA.
|
|||
|
What are the possible side effects of TREMFYA?
|
|||
| The most common side effects of TREMFYA include: | |||
|
|
|
|
| These are not all the possible side effects of TREMFYA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||
How should I store TREMFYA?
|
|||
| General information about the safe and effective use of TREMFYA. | |||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use TREMFYA for a condition for which it was not prescribed. Do not give TREMFYA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about TREMFYA that is written for health professionals. | |||
|
What are the ingredients in TREMFYA?
Active ingredient: guselkumab Inactive ingredients: Single-dose prefilled syringe, single-dose One-Press patient-controlled injector, and single-dose prefilled pen (TREMFYA PEN) for subcutaneous use: L-histidine, L-histidine monohydrochloride monohydrate, polysorbate 80, sucrose and water for injection. Single-dose vial for intravenous infusion: EDTA disodium dihydrate, L-histidine, L-histidine monohydrochloride monohydrate, L-methionine, polysorbate 80, sucrose and water for injection. |
|||
| Manufactured by: Janssen Biotech, Inc., Horsham, PA 19044, USA. U.S. License Number 1864
For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2017–2025 For more information, call 1-800-526-7736 or go to www.tremfya.com. |
Section 43683-2 (43683-2)
| Indications and Usage (1.4) | 03/2025 |
| Indications and Usage (1.1, 1.2) | 09/2025 |
| Dosage and Administration (2.1, 2.5, 2.6) | 03/2025 |
| Dosage and Administration (2.2, 2.3, 2.5) | 09/2025 |
| Dosage and Administration (2.4) | 09/2025 |
| Warnings and Precautions (5.3, 5.4) | 03/2025 |
| Warnings and Precautions (5.2, 5.4) | 09/2025 |
Section 44425-7 (44425-7)
Storage and Handling
TREMFYA is sterile and preservative-free. Discard any unused portion.
- Store in a refrigerator between 2 °C to 8 °C (36 °F to 46 °F).
- Store in original carton until time of use.
- Protect from light until use.
- Do not freeze.
- Do not shake.
- Not made with natural rubber latex.
11 Description (11 DESCRIPTION)
Guselkumab, an interleukin-23 antagonist, is a human immunoglobulin G1 lambda (IgG1λ) monoclonal antibody. Guselkumab is produced in a mammalian cell line using recombinant DNA technology and has an approximate molecular weight of approximately 147 kDa.
TREMFYA® (guselkumab) injection is a sterile, preservative free, clear, and colorless to light yellow solution.
5.2 Infections
TREMFYA may increase the risk of infection [see Adverse Reactions (6.1)].
In placebo-controlled clinical trials of up to 48 weeks in subjects with ulcerative colitis and Crohn’s disease, serious infections occurred in ≤ 2% of subjects who received TREMFYA. In the 16-week placebo-controlled trials in subjects with plaque psoriasis, the rate of serious infections for the TREMFYA group and the placebo group was ≤ 0.2%. A similar rate of serious infections was seen in placebo-controlled trials in subjects with psoriatic arthritis. The overall rates of infections were similar between subjects in the TREMFYA groups and subjects in the placebo groups in clinical trials for all indicated populations [see Adverse Reactions (6.1)].
Treatment with TREMFYA should not be initiated in patients with any clinically important active infection until the infection resolves or is adequately treated.
In patients with a chronic infection or a history of recurrent infection, consider the risks and benefits prior to prescribing TREMFYA. Instruct patients to seek medical help if signs or symptoms of clinically important chronic or acute infection occur. If a patient develops a clinically important or serious infection or is not responding to standard therapy, monitor the patient closely and discontinue TREMFYA until the infection resolves.
5.3 Tuberculosis
Evaluate patients for tuberculosis (TB) infection prior to initiating TREMFYA treatment. Do not administer TREMFYA to patients with active TB infection. Initiate treatment of latent TB prior to administering TREMFYA. Consider anti-TB therapy prior to initiating TREMFYA in patients with a past history of latent or active TB in whom an adequate course of treatment cannot be confirmed. Monitor all patients for signs and symptoms of active TB during and after TREMFYA treatment.
In clinical trials, 105 subjects with plaque psoriasis, 71 subjects with psoriatic arthritis, 43 subjects with ulcerative colitis, and 36 subjects with Crohn's disease with latent TB who were concurrently treated with TREMFYA, and appropriate TB prophylaxis did not develop active TB. In clinical trials of TREMFYA in subjects with Crohn's disease, active TB was reported in 2 subjects during treatment with TREMFYA [see Adverse Reactions (6.1)].
5.5 Immunizations
Avoid use of live vaccines in patients treated with TREMFYA. Medications that interact with the immune system may increase the risk of infection following administration of live vaccines. Prior to initiating therapy with TREMFYA, complete all age-appropriate vaccinations according to current immunization guidelines. No data are available on the response to live or inactive vaccines.
8.5 Geriatric Use
Of the 5723 subjects with plaque psoriasis, psoriatic arthritis, ulcerative colitis, or Crohn's disease exposed to TREMFYA, a total of 313 subjects were 65 years or older, and 32 subjects were 75 years or older. Clinical studies of TREMFYA, within each indication, did not include sufficient numbers of subjects 65 years of age and older to determine whether they respond differently from younger adult subjects.
No clinically meaningful differences in the pharmacokinetics of guselkumab were observed based on age [see Clinical Pharmacology (12.3)].
5.4 Hepatotoxicity
A serious adverse reaction of drug-induced liver injury was reported in a clinical trial subject with Crohn's disease following three doses of a higher than the recommended induction regimen. This subject had peak alanine aminotransferase (ALT) of 18x the upper limit of normal (ULN), aspartate aminotransferase (AST) of 11x ULN, and total bilirubin of 2.4x ULN. TREMFYA was subsequently discontinued, and the liver test abnormalities resolved following administration of corticosteroids [see Adverse Reactions (6.1)].
In patients with ulcerative colitis or Crohn's disease, evaluate liver enzymes and bilirubin at baseline, for at least 16 weeks of treatment, and periodically thereafter according to routine patient management.
In patients with plaque psoriasis or psoriatic arthritis, if clinically indicated, evaluate liver enzymes and bilirubin at baseline, and periodically thereafter according to routine patient management.
Consider other treatment options in patients with evidence of acute liver disease or cirrhosis. Prompt investigation of the cause of liver enzyme elevation is recommended to identify potential cases of drug-induced liver injury. Interrupt treatment if drug-induced liver injury is suspected, until this diagnosis is excluded. Instruct patients to seek immediate medical attention if they experience symptoms suggestive of hepatic dysfunction.
1.4 Crohn's Disease
TREMFYA is indicated for the treatment of adult patients with moderately to severely active Crohn's disease.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of guselkumab or of other guselkumab products.
4 Contraindications (4 CONTRAINDICATIONS)
TREMFYA is contraindicated in patients with a history of serious hypersensitivity reaction to guselkumab or to any of the excipients [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of labeling:
- Hypersensitivity Reactions [see Contraindications (4) and Warnings and Precautions (5.1)]
- Infections [see Warnings and Precautions (5.2)]
- Tuberculosis [see Warnings and Precautions (5.3)]
- Hepatotoxicity [see Warnings and Precautions (5.4)]
1.1 Plaque Psoriasis
TREMFYA is indicated for the treatment of adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with moderate-to-severe plaque psoriasis and who are candidates for systemic therapy or phototherapy.
Instructions for Use (INSTRUCTIONS FOR USE)
TREMFYA® [trem fye' ah]
(guselkumab)
injection, for subcutaneous use
200 mg/2mL (100 mg/mL) Prefilled Syringe
This Instructions for Use contains information on how to inject TREMFYA.
| SINGLE-DOSE |
Important Information You Need to Know Before Injecting TREMFYA
TREMFYA comes in a single-dose prefilled syringe containing one 200 mg dose.
| Your healthcare provider will tell you if you will need to use 1 or 2 prefilled syringes. |
If your healthcare provider decides that you or a caregiver may be able to give your injections of TREMFYA at home, you should receive training on the correct way to prepare and inject TREMFYA before using the prefilled syringe.
Read this Instructions for Use before using your TREMFYA prefilled syringe and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your healthcare provider about your medical condition or your treatment.
Each TREMFYA prefilled syringe can only be used one time. Throw the used prefilled syringe away (see Step 4) after one dose, even if there is still medicine left in it. Do not reuse your TREMFYA prefilled syringe.
The TREMFYA prefilled syringe is intended for injection under the skin, not into the muscle or vein. After injection, the needle will retract into the device and lock into place.
Storage information
Store in refrigerator between 36°F to 46°F (2°C to 8°C).
Do not freeze TREMFYA prefilled syringe.
Do not shake your TREMFYA prefilled syringe.
Keep TREMFYA prefilled syringe and all medicines out of reach of children.
Keep TREMFYA prefilled syringe in the original carton to protect from light and physical damage.
Prefilled syringe parts
1. Get ready
Check your dose to see if you will need to use 1 or 2 prefilled syringes and inspect carton(s)
Remove the carton(s) from the refrigerator.
Check the expiration ('EXP') date. Do not use your prefilled syringe if the expiration date has passed or if the seal on the carton is broken. Contact your healthcare provider or pharmacist for a new prefilled syringe.
Allow TREMFYA to come to room temperature
Let the cartons sit on a flat surface at room temperature for approximately 30 minutes before use.
Do not warm the prefilled syringe(s) any other way.
2. Prepare to inject TREMFYA
Take the prefilled syringe out of the carton.
Inspect liquid to see that it is clear and colorless to slightly yellow
Check the liquid in the viewing window. It should be clear and colorless to slightly yellow and may contain tiny white or clear particles. You may also see air bubbles. This is normal.
Do not inject if the liquid is:
- cloudy or
- discolored or
- has large particles
Do not use the prefilled syringe if it is dropped. Call your healthcare provider or pharmacist for a new prefilled syringe.
Choose injection site
Select a site from the following areas for your injection:
- Front of thighs
- Lower stomach area (lower abdomen), except for a 2-inch area right around your navel (belly-button)
- Back of upper arms (only if someone else is giving you the injection)
If you need to give 2 injections to complete your dose, choose different areas or leave at least 2 inches between injection sites.
Do not inject into skin that is tender, bruised, red, scaly, thick or hard. Avoid areas with scars or stretch marks.
Wash hands and clean injection site
Wash your hands well with soap and warm water.
Wipe your chosen injection site with an alcohol swab and allow it to dry.
Do not touch, fan, or blow on the injection site after you have cleaned it.
3. Inject TREMFYA
Remove needle cover when you are ready to inject
Hold the prefilled syringe by the body and pull needle cover straight off. It is normal to see a few drops of liquid.
Inject TREMFYA within 5 minutes of removing the needle cover.
Do not put needle cover back on, as this may damage the needle or cause a needle stick injury.
Do not touch needle or let it touch any surface.
Do not use the prefilled syringe if it is dropped. Call your healthcare provider or pharmacist for a new prefilled syringe.
Do not hold or pull the plunger at any time.
Pinch injection site and insert needle at about a 45-degree angle
It is important to pinch enough skin to inject under the skin and not into muscle.
Insert needle with a quick dart-like motion.
Slowly press plunger all the way down until it stops to inject all of the liquid
You will feel some resistance as you press the plunger, this is normal.
Release pressure from plunger to remove the needle from the skin
The needle will retract into the device and lock into place.
If your prescribed dose requires two injections, repeat Steps 2 to 4 with the second prefilled syringe.
4. After your injection
Check injection site
There may be a small amount of blood or liquid at the injection site. Gently hold pressure over the injection site with a cotton ball or gauze pad until any bleeding stops.
Do not rub the injection site.
If needed, cover the injection site with a bandage.
Dispose of your prefilled syringe
Put the used prefilled syringe in an FDA-cleared sharps disposal container right away after use.
Do not throw away (dispose of) your prefilled syringe in your household trash.
Do not recycle your used sharps disposal container.
For more information, see Disposing of TREMFYA prefilled syringe.
Disposing of TREMFYA prefilled syringe
If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- made of heavy-duty plastic
- can be closed with a tight-fitting, puncture resistant lid, without sharps being able to come out
- upright and stable during use
- leak-resistant
- properly labeled to warn of hazardous waste inside the container
When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes.
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: www.fda.gov/safesharpsdisposal. You may also consult your pharmacist.
Need help?
Call your healthcare provider to talk about any questions you may have. For additional assistance or to share your feedback, call 800-526-7736.
Manufactured by:
Janssen Biotech, Inc.
Horsham, PA 19044, USA
US License No. 1864
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Approved: August / 2025
Instructions for Use
TREMFYA®
(trem fye' ah)
(guselkumab)
Prefilled Syringe
SINGLE-DOSE
Important
TREMFYA comes as a single-dose prefilled syringe containing one 100 mg dose. Each TREMFYA prefilled syringe can only be used one time. Throw the used prefilled syringe away (See Step 3) after one dose, even if there is medicine left in it. Do not reuse your TREMFYA prefilled syringe.
If your healthcare provider decides that you or a caregiver may be able to give your injections of TREMFYA at home, you should receive training on the right way to prepare and inject TREMFYA using the prefilled syringe before attempting to inject. Do not try to inject yourself until you have been shown the right way to give the injections by your healthcare provider.
For children, TREMFYA should be administered by a healthcare provider or an adult caregiver who has been shown the right way to give the injections by your healthcare provider.
Read this Instructions for Use before using your TREMFYA prefilled syringe and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your healthcare provider about your medical condition or your treatment.
The TREMFYA prefilled syringe is intended for injection under the skin, not into the muscle or vein. After injection, the needle will retract into the body of the device and lock into place.
Storage information
Store in refrigerator at 36 °F to 46 °F (2 °C to 8 °C). Do not freeze TREMFYA prefilled syringe.
Keep TREMFYA prefilled syringe and all medicines out of reach of children.
Do not shake your TREMFYA prefilled syringe.
Keep TREMFYA prefilled syringe in the original carton to protect from light and physical damage.
Prefilled syringe parts
Before use
After use
|
You will need these supplies:
Not provided in the TREMFYA prefilled syringe carton:
|
1. Prepare for your injection
Inspect carton
Remove your TREMFYA prefilled syringe carton from the refrigerator.
Keep the prefilled syringe in the carton and let it sit on a flat surface at room temperature for at least 30 minutes before use.
Do not warm the prefilled syringe any other way.
Check the expiration date ('EXP') on the back panel of the carton.
Do not use your prefilled syringe if the expiration date has passed.
Do not inject TREMFYA if the perforations on the carton are broken. Call your healthcare provider or pharmacist for a refill.
Choose injection site
Select from the following areas for your injection:
- Front of thighs (recommended)
- Lower stomach area (lower abdomen), except for a 2-inch area right around your navel (belly-button)
- Back of upper arms (only if someone else is giving you the injection)
Do not inject into skin that is tender, bruised, red, hard, thick, scaly or affected by psoriasis.
Clean injection site
Wash your hands well with soap and warm water.
Wipe your chosen injection site with an alcohol swab and allow it to dry.
Do not touch, fan, or blow on the injection site after you have cleaned it.
Inspect liquid
Take your TREMFYA prefilled syringe out of the carton.
Check the TREMFYA prefilled syringe liquid in the viewing window. It should be clear to slightly yellow and may contain tiny white or clear particles. You may also see one or more air bubbles. This is normal.
Do not inject if the liquid is cloudy or discolored, or has large particles. Call your healthcare provider or pharmacist for a refill.
2. Inject TREMFYA using prefilled syringe
Remove needle cover
Hold your prefilled syringe by the body and pull needle cover straight off. It is normal to see a drop of liquid.
Inject TREMFYA within 5 minutes of removing the needle cover.
Do not put needle cover back on, as this may damage the needle or cause a needle stick injury.
Do not touch needle or let it touch any surface.
Do not use a TREMFYA prefilled syringe if it is dropped. Call your healthcare provider or pharmacist for a refill.
Position fingers and insert needle
Place your thumb, index and middle fingers directly under the finger flange, as shown.
Do not touch plunger or area above finger flange as this may cause the needle safety device to activate.
Use your other hand to pinch skin at the injection site. Position syringe at about a 45 degree angle to the skin.
It is important to pinch enough skin to inject under the skin and not into the muscle.
Insert needle with a quick, dart-like motion.
Release pinch and reposition hand
Use your free hand to grasp the body of the prefilled syringe.
Press plunger
Place thumb from the opposite hand on the plunger and press the plunger all the way down until it stops.
Release pressure from plunger
The safety guard will cover the needle and lock into place, removing the needle from your skin.
3. After your injection
Dispose of your prefilled syringe
Put your used TREMFYA prefilled syringe in an FDA-cleared sharps disposal container right away after use.
Do not throw away (dispose of) your TREMFYA prefilled syringe in your household trash.
Do not recycle your used sharps disposal container.
For more information, see "How should I dispose of the used prefilled syringe?"
Check injection site
There may be a small amount of blood or liquid at the injection site. Hold pressure over your skin with a cotton ball or gauze pad until any bleeding stops.
Do not rub the injection site.
If needed, cover injection site with a bandage.
Need help?
Call your healthcare provider to talk about any questions you may have. For additional assistance or to share your feedback call 1-800-526-7736.
How should I dispose of the used prefilled syringe?
If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out
- upright and stable during use
- leak-resistant
- properly labeled to warn of hazardous waste inside the container
When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes.
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: www.fda.gov/safesharpsdisposal
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by:
Janssen Biotech, Inc.
Horsham, PA 19044, USA
U.S. License No. 1864
Approved: September 2025
12.2 Pharmacodynamics
In evaluated adult subjects with moderate-to-severe plaque psoriasis, guselkumab reduced serum levels of IL-17A, IL-17F and IL-22 relative to pre-treatment levels based on exploratory analyses of the pharmacodynamic markers.
In evaluated adult subjects with active psoriatic arthritis, serum levels of acute phase proteins C-reactive protein, serum amyloid A and IL-6, and Th17 effector cytokines IL-17A, IL-17F and IL-22 were elevated at baseline. Serum levels of these proteins measured at Week 4 and Week 24 were decreased compared to baseline following guselkumab treatment at Week 0, Week 4, and every 8 weeks thereafter.
The relationship between these pharmacodynamic markers and the mechanism(s) by which guselkumab exerts its clinical effects is unknown.
12.3 Pharmacokinetics
Guselkumab exhibited linear pharmacokinetics in healthy adult subjects and adult subjects with plaque psoriasis following subcutaneous injections. In subjects with plaque psoriasis, following subcutaneous administration of 100 mg of TREMFYA at Weeks 0 and 4, and every 8 weeks thereafter, mean steady-state trough serum guselkumab concentration was approximately 1.2 mcg/mL.
The pharmacokinetics of guselkumab in adult subjects with psoriatic arthritis was similar to that in adult subjects with plaque psoriasis. Following subcutaneous administration of 100 mg of TREMFYA at Weeks 0, 4, and every 8 weeks thereafter, mean steady-state trough serum guselkumab concentration was approximately 1.2 mcg/mL.
Following subcutaneous maintenance dosing of 100 mg TREMFYA every 8 weeks or 200 mg TREMFYA every 4 weeks in adult subjects with ulcerative colitis, mean steady-state trough serum guselkumab concentrations were approximately 1.4 mcg/mL and 10.7 mcg/mL, respectively.
Following subcutaneous maintenance dosing of 100 mg TREMFYA every 8 weeks, or 200 mg TREMFYA every 4 weeks, in adult subjects with Crohn's disease mean steady-state trough serum guselkumab concentrations were approximately 1.2 mcg/mL and 10.1 mcg/mL, respectively.
7.1 Cyp450 Substrates (7.1 CYP450 Substrates)
The formation of CYP450 enzymes can be altered by increased levels of certain cytokines (e.g., IL-1, IL-6, IL-10, TNFα, interferon) during chronic inflammation.
Results from an exploratory drug-drug interaction trial in subjects with moderate-to-severe plaque psoriasis suggested a low potential for clinically relevant drug interactions for drugs metabolized by CYP3A4, CYP2C9, CYP2C19 and CYP1A2 but the interaction potential cannot be ruled out for drugs metabolized by CYP2D6. However, the results were highly variable because of the limited number of subjects in the trial.
Upon initiation of TREMFYA in patients who are receiving concomitant CYP450 substrates, particularly those with a narrow therapeutic index, consider monitoring for therapeutic effect or drug concentration and consider dosage adjustment as needed [see Clinical Pharmacology (12.3)].
1.3 Ulcerative Colitis
TREMFYA is indicated for the treatment of adult patients with moderately to severely active ulcerative colitis.
1 Indications and Usage (1 INDICATIONS AND USAGE)
TREMFYA is an interleukin-23 antagonist indicated for the treatment of:
- adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with moderate-to-severe plaque psoriasis and who are candidates for systemic therapy or phototherapy. (1.1)
- adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with active psoriatic arthritis. (1.2)
- adults with moderately to severely active ulcerative colitis. (1.3)
- adults with moderately to severely active Crohn's disease. (1.4)
1.2 Psoriatic Arthritis
TREMFYA is indicated for the treatment of adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with active psoriatic arthritis.
12.1 Mechanism of Action
Guselkumab is a human monoclonal IgG1λ antibody that selectively binds to the p19 subunit of interleukin 23 (IL-23) and inhibits its interaction with the IL-23 receptor. IL-23 is a naturally occurring cytokine that is involved in normal inflammatory and immune responses. Guselkumab inhibits the release of proinflammatory cytokines and chemokines.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity Reactions: Serious hypersensitivity reactions, including anaphylaxis, may occur. (5.1)
- Infections: TREMFYA may increase the risk of infections. Do not initiate treatment with TREMFYA in patients with clinically important active infection until the infection resolves or is adequately treated. If such an infection develops, discontinue TREMFYA until the infection resolves. (5.2)
- Tuberculosis (TB): Evaluate for TB prior to initiating treatment with TREMFYA. Monitor patients for signs and symptoms of active TB during and after treatment with TREMFYA. (5.3)
- Hepatotoxicity: Drug-induced liver injury has been reported. For the treatment of ulcerative colitis or Crohn’s disease, evaluate liver enzymes and bilirubin levels at baseline, for at least 16 weeks of treatment, and periodically thereafter according to routine patient management. For the treatment of plaque psoriasis or psoriatic arthritis, if clinically indicated, evaluate liver enzymes and bilirubin at baseline, and periodically thereafter according to routine patient management. Interrupt treatment if drug-induced liver injury is suspected, until this diagnosis is excluded. (5.4)
- Immunizations: Avoid use of live vaccines. (5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For the treatment of ulcerative colitis or Crohn’s disease: Obtain liver enzymes and bilirubin levels prior to initiating treatment with TREMFYA. (2.1, 5.4).
- For the treatment of plaque psoriasis or psoriatic arthritis, if clinically indicated, evaluate liver enzymes and bilirubin at baseline prior to initiating treatment with TREMFYA (2.1, 5.4).
- Complete all age-appropriate vaccinations as recommended by current immunization guidelines prior to treatment initiation. (2.1)
Recommended Dosage
Plaque Psoriasis
Adults
- 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. (2.2)
Pediatric Patients 6 Years of Age and Older Who Also Weigh at Least 40 kg
- 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. (2.2)
Psoriatic Arthritis
Adults
- 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. TREMFYA can be used alone or in combination with a conventional DMARD (e.g., methotrexate). (2.3)
Pediatric Patients 6 Years of Age and Older Who Also Weigh at Least 40 kg
- 100 mg administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter. TREMFYA may be administered alone or in combination with a conventional disease-modifying antirheumatic drug (e.g., methotrexate). (2.3)
Ulcerative Colitis and Crohn’s Disease
- Induction: 200 mg administered by intravenous infusion over at least one hour at Week 0, Week 4, and Week 8 or 400 mg administered by subcutaneous injection at Week 0, Week 4, and Week 8. (2.4)
- Maintenance: 100 mg administered by subcutaneous injection at Week 16, and every 8 weeks thereafter, or 200 mg administered by subcutaneous injection at Week 12, and every 4 weeks thereafter. Use the lowest effective recommended dosage to maintain therapeutic response. (2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
TREMFYA is a clear and colorless to light yellow solution.
6.2 Postmarketing Experience
The following adverse reactions have been reported during post-approval of TREMFYA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to TREMFYA exposure.
Immune system disorders: Hypersensitivity, including anaphylaxis [see Warnings and Precautions (5.1)]
Skin and subcutaneous tissue disorders: Rash [see Warnings and Precautions (5.1)]
5.1 Hypersensitivity Reactions
Serious hypersensitivity reactions, including anaphylaxis, have been reported with post market use of TREMFYA. Some cases required hospitalization. If a serious hypersensitivity reaction occurs, discontinue TREMFYA and initiate appropriate therapy.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patients and/or caregivers to read the FDA-approved patient labeling (Medication Guide and Instructions for Use) before starting TREMFYA therapy, and each time the prescription is renewed, as there may be new information they need to know.
Principal Display Panel 100 Mg/ml Pen Carton (PRINCIPAL DISPLAY PANEL - 100 mg/mL Pen Carton)
NDC 57894-640-06
Tremfya® PEN
(guselkumab)
Injection
100 mg/mL
FOR SUBCUTANEOUS USE ONLY
Rx only
One single-dose prefilled pen
Discard unused portion
Attention: Dispense the enclosed
Medication Guide to each patient.
Principal Display Panel 200 Mg/2 Ml Pen Carton (PRINCIPAL DISPLAY PANEL - 200 mg/2 mL Pen Carton)
NDC 57894-651-02
Tremfya® PEN
(guselkumab)
Injection
200 mg/2 mL
FOR SUBCUTANEOUS USE ONLY
Rx only
One single-dose prefilled pen
Discard unused portion
ATTENTION: Dispense the enclosed
Medication Guide to each patient.
Principal Display Panel 200 Mg/20 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 200 mg/20 mL Vial Carton)
NDC 57894-650-02
Tremfya®
(guselkumab)
Injection
200 mg/20 mL
(10 mg/mL)
FOR INTRAVENOUS
INFUSION ONLY
Attention: Dispense the
enclosed Medication Guide
to each patient.
Rx only
Single-dose vial
Discard unused portion
14.4 Clinical Studies in Adults With Crohn's Disease (14.4 Clinical Studies in Adults with Crohn's Disease)
The efficacy and safety of TREMFYA were assessed in three randomized, double-blind, placebo-controlled trials that enrolled adult subjects with moderately to severely active Crohn's disease who had a history of inadequate response, loss of response, or intolerance to oral corticosteroids, immunomodulators (azathioprine, 6-mercaptopurine, methotrexate), and/or biologic therapy (TNF blockers or vedolizumab). Moderately to severely active Crohn's disease was defined as a Crohn's Disease Activity Index (CDAI) score of ≥220 and a Simple Endoscopic Score for Crohn's Disease (SES-CD) of ≥6 (or ≥4 for subjects with isolated ileal disease). Subjects were permitted to use stable doses of oral corticosteroids (prednisone ≤40 mg/day or equivalent), immunomodulators (azathioprine, 6-mercaptopurine, methotrexate), and/or aminosalicylates.
Principal Display Panel 200 Mg/2 Ml Syringe Carton (PRINCIPAL DISPLAY PANEL - 200 mg/2 mL Syringe Carton)
NDC 57894-651-22
Tremfya®
(guselkumab)
Injection
200 mg/2 mL
FOR SUBCUTANEOUS USE ONLY
Rx only
One single-dose prefilled syringe
Discard unused portion
ATTENTION: Dispense the enclosed
Medication Guide to each patient.
2.3 Recommended Dosage for Active Psoriatic Arthritis
Administer TREMFYA by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter.
TREMFYA may be administered alone or in combination with a conventional disease-modifying antirheumatic drug (e.g., methotrexate).
Adults
The recommended dose is 100 mg.
Pediatric Patients 6 Years of Age and Older Who Also Weigh at Least 40 kg
The recommended dose is 100 mg.
14.3 Clinical Studies in Adults With Ulcerative Colitis (14.3 Clinical Studies in Adults with Ulcerative Colitis)
The efficacy and safety of TREMFYA was assessed in three randomized, double-blind, placebo-controlled trials (UC1, UC2, and UC 4) that enrolled adult subjects with moderately to severely active ulcerative colitis. Disease activity was assessed by the modified Mayo score (mMS), a 3-component Mayo score (0 –9) which consists of the following subscores (0 to 3 for each subscore): stool frequency (SFS), rectal bleeding (RBS), and findings on centrally reviewed endoscopy (ES). An ES of 2 was defined by marked erythema, lack of vascular pattern, friability, and/or erosions; an ES of 3 was defined by spontaneous bleeding and ulceration. Enrolled subjects with a mMS between 5 and 9 and an ES of 2 or 3 were classified as having moderately to severely active ulcerative colitis. Subjects with inadequate response, loss of response, or intolerance to corticosteroids, immunomodulators (azathioprine, 6-mercaptopurine), biologic therapy (TNF blockers, vedolizumab), and/or Janus kinase (JAK) inhibitors were enrolled. In UC4, subjects with inadequate response, loss of response, or intolerance to sphingosine-1-phosphate receptor modulators (S1PRM) were also enrolled.
14.2 Clinical Studies in Adults With Psoriatic Arthritis (14.2 Clinical Studies in Adults with Psoriatic Arthritis)
The safety and efficacy of TREMFYA were assessed in 2 randomized, double-blind, placebo-controlled trials (PsA1 [NCT03162796] and PsA2 [NCT03158285]) in 1120 adult subjects with active psoriatic arthritis (PsA) (≥3 swollen joints, ≥3 tender joints, and a C-reactive protein (CRP) level of ≥0.3 mg/dL in PsA1 and ≥5 swollen joints, ≥5 tender joints, and a CRP level of ≥0.6 mg/dL in PsA2) who had inadequate response to standard therapies (e.g., conventional DMARDs [cDMARDs]), apremilast, or nonsteroidal anti-inflammatory drugs [NSAIDs]). Subjects in these trials had a diagnosis of PsA for at least 6 months based on the Classification criteria for Psoriatic Arthritis (CASPAR) and a median duration of PsA of 4 years at baseline.
In trial PsA1, approximately 31% of subjects had been previously treated with up to 2 anti-tumor necrosis factor alpha (anti-TNFα) agents whereas in trial PsA2, all subjects were biologic naïve. Approximately 58% of subjects from both trials had concomitant methotrexate (MTX) use. Subjects with different subtypes of PsA were enrolled in both trials, including polyarticular arthritis with the absence of rheumatoid nodules (40%), spondylitis with peripheral arthritis (30%), asymmetric peripheral arthritis (23%), distal interphalangeal involvement (7%) and arthritis mutilans (1%). At baseline, over 65% and 42% of the subjects had enthesitis and dactylitis, respectively and 79% had ≥3% body surface area (BSA) psoriasis skin involvement.
Trial PsA1 evaluated 381 subjects who were treated with placebo SC, TREMFYA 100 mg SC at Weeks 0, 4 and every 8 weeks (q8w) thereafter, or TREMFYA 100 mg SC every 4 weeks (q4w). Trial PsA2 evaluated 739 subjects who were treated with placebo SC, TREMFYA 100 mg SC at Weeks 0, 4 and q8w thereafter, or TREMFYA 100 mg SC q4w. The primary endpoint in both trials was the percentage of subjects achieving an ACR20 response at Week 24.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Animal studies have not been conducted to evaluate the carcinogenic or mutagenic potential of TREMFYA.
No effects on fertility parameters were observed after male guinea pigs were subcutaneously administered guselkumab at a dose of 25 mg/kg twice weekly (6 times the exposure (AUC) in humans administered 200 mg intravenously and 5 times the exposure (AUC) at the 400 mg subcutaneous dose).
No effects on fertility parameters were observed after female guinea pigs were subcutaneously administered guselkumab at doses up to 100 mg/kg twice weekly (12 times the exposure (AUC) in humans administered 200 mg intravenously and 10 times the exposure (AUC) at the 400 mg subcutaneous dose).
2.2 Recommended Dosage for Moderate to Severe Plaque Psoriasis (2.2 Recommended Dosage for Moderate-to-Severe Plaque Psoriasis)
Administer TREMFYA by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter.
Adults
The recommended dose is 100 mg.
Pediatric Patients 6 Years of Age and Older Who Also Weigh at Least 40 kg
The recommended dose is 100 mg.
Principal Display Panel 200 Mg/2 Ml Pen Carton Ndc 57894 651 04 (PRINCIPAL DISPLAY PANEL - 200 mg/2 mL Pen Carton - NDC 57894-651-04)
NDC 57894-651-04
Rx only
Tremfya® PEN
(guselkumab)
Injection
200 mg/2 mL
FOR SUBCUTANEOUS USE ONLY
Induction Pack for Crohn's Disease
and Ulcerative Colitis
(includes 1 induction dose)
The entire carton is to
be dispensed as a unit.
Contains: Two cartons each containing
one single-dose prefilled pen.
Discard unused portion.
ATTENTION: Dispense the enclosed
Medication Guide to each patient.
Principal Display Panel 100 Mg/ml Syringe Carton Ndc 57894 640 01 (PRINCIPAL DISPLAY PANEL - 100 mg/mL Syringe Carton - NDC 57894-640-01)
Tremfya®
(guselkumab)
Injection
100 mg/mL
FOR SUBCUTANEOUS USE ONLY
Rx only
NDC 57894-640-01
One single-dose prefilled syringe
Discard unused portion
Contains no preservative
ATTENTION: Dispense the enclosed
Medication Guide to each patient
Principal Display Panel 100 Mg/ml Syringe Carton Ndc 57894 640 11 (PRINCIPAL DISPLAY PANEL - 100 mg/mL Syringe Carton - NDC 57894-640-11)
Tremfya®
(guselkumab)
Injection
100 mg/mL
FOR SUBCUTANEOUS USE ONLY
Rx only
NDC 57894-640-11
Single-dose One-Press
patient-controlled injector
Discard unused portion
Contains no preservative
ATTENTION: Dispense the enclosed
Medication Guide to each patient
2.5 Preparation and Administration Instructions for Subcutaneous Injection
TREMFYA is available for subcutaneous use in the following presentations: prefilled pen (TREMFYA PEN), One-Press injector, and prefilled syringes [see Dosage Forms and Strengths (3) and How Supplied/Storage and Handling (16)].
- Each prefilled pen, One-Press injector, or prefilled syringe is for one time use in one patient only. Instruct patients to inject the full amount: 100 mg or 200 mg of TREMFYA (1 mL or 2 mL, respectively).
-
TREMFYA is intended for use under the guidance and supervision of a healthcare professional. After proper training in subcutaneous injection technique:
Adults- Adults may self-inject with the TREMFYA prefilled syringe, One-Press injector, and prefilled pen.
- Inject into the front of the thighs, the lower abdomen except for the 2 inches around the navel, or the back of the upper arms (healthcare professional or caregiver only).
- Pediatric self-administration is not recommended. Administration of TREMFYA to pediatric patients with the prefilled syringe, One-Press injector and prefilled pen should be performed by a healthcare provider or by a caregiver who has received training and demonstrated proper subcutaneous injection technique.
- Inject into the front of the thighs or the lower abdomen except for the 2 inches around the navel. For the prefilled syringe only, injection can also be given in the back of the upper arms.
- Before injection, remove TREMFYA from the refrigerator and allow to reach room temperature up to 25 °C (77 °F) (30 minutes) without removing the needle cap.
- Do not inject TREMFYA into areas where the skin is tender, bruised, red, hard, thick, scaly, or affected by psoriasis [see Instructions for Use].
- The TREMFYA Instructions for Use contains more detailed patient instructions on the preparation and administration of TREMFYA [see Instructions for Use].
- If a dose is missed, administer the dose as soon as possible. Thereafter, resume dosing at the regular scheduled time.
- Inspect TREMFYA visually for particulate matter and discoloration prior to administration. TREMFYA is a clear and colorless to light yellow solution that may contain small translucent particles. Do not use if the liquid contains large particles, is discolored or cloudy. TREMFYA does not contain preservatives; therefore, discard any unused product remaining in the prefilled pen, One-Press injector, or prefilled syringe.
2.1 Recommended Evaluations and Immunizations Prior to Treatment Initiation
- Evaluate patients for tuberculosis (TB) infection prior to initiating treatment with TREMFYA [see Warnings and Precautions (5.3)].
- For the treatment of ulcerative colitis or Crohn’s disease, obtain liver enzymes and bilirubin levels prior to initiating treatment with TREMFYA [see Warnings and Precautions (5.4)].
- For the treatment of plaque psoriasis or psoriatic arthritis, if clinically indicated, evaluate liver enzymes and bilirubin prior to initiating treatment with TREMFYA [see Warnings and Precautions (5.4)].
- Complete all age-appropriate vaccinations according to current immunization guidelines [see Warnings and Precautions (5.5)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T02:18:22.154540 · Updated: 2026-03-14T22:49:49.558846