Sohmed™ Allergy
1e650657-83e5-4704-863b-c420390a2247
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Antihistamine
Medication Information
Purpose
Antihistamine
Description
Drug Facts
Uses
- temporarily relieves symptoms due to hay fever or other upper respiratory allergies :
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
- temporarily relieves symptoms due to the common cold:
- runny nose
- sneezing
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children.
Section 50568-5
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers
Section 50570-1
Do not use with any other product containing diphenhydramine, even one used on skin.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Directions
- take every 4 to 6 hours
- do not take more than 6 times in 24 hours
| adults and children 12 years and over | 1 to 2 caplets |
| children 6 to under 12 years | 1 caplet |
| children under 6 years | consult a doctor |
Overdose Warning
In case of overdose, get medical help or contact a Poison Control Center right away.
Other Information
- Store between 20°-25°C (68°-77°F).
Avoid high humidity. Protect from light - See end flap for expiration date and lot number
Inactive Ingredients
croscarmellose sodium, D&C Red #27, hydroxypropylmethyl cellulose, lactose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, silicon dioxide, titanium dioxide, May contain polysorbate.
Question Or Comments?
1 (856) 2863646
When Using This Product
- marked drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
Active Ingredients (in Each Caplet)
Diphenhydramine HCl 25 mg
Ask A Doctor Before Use If You Have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
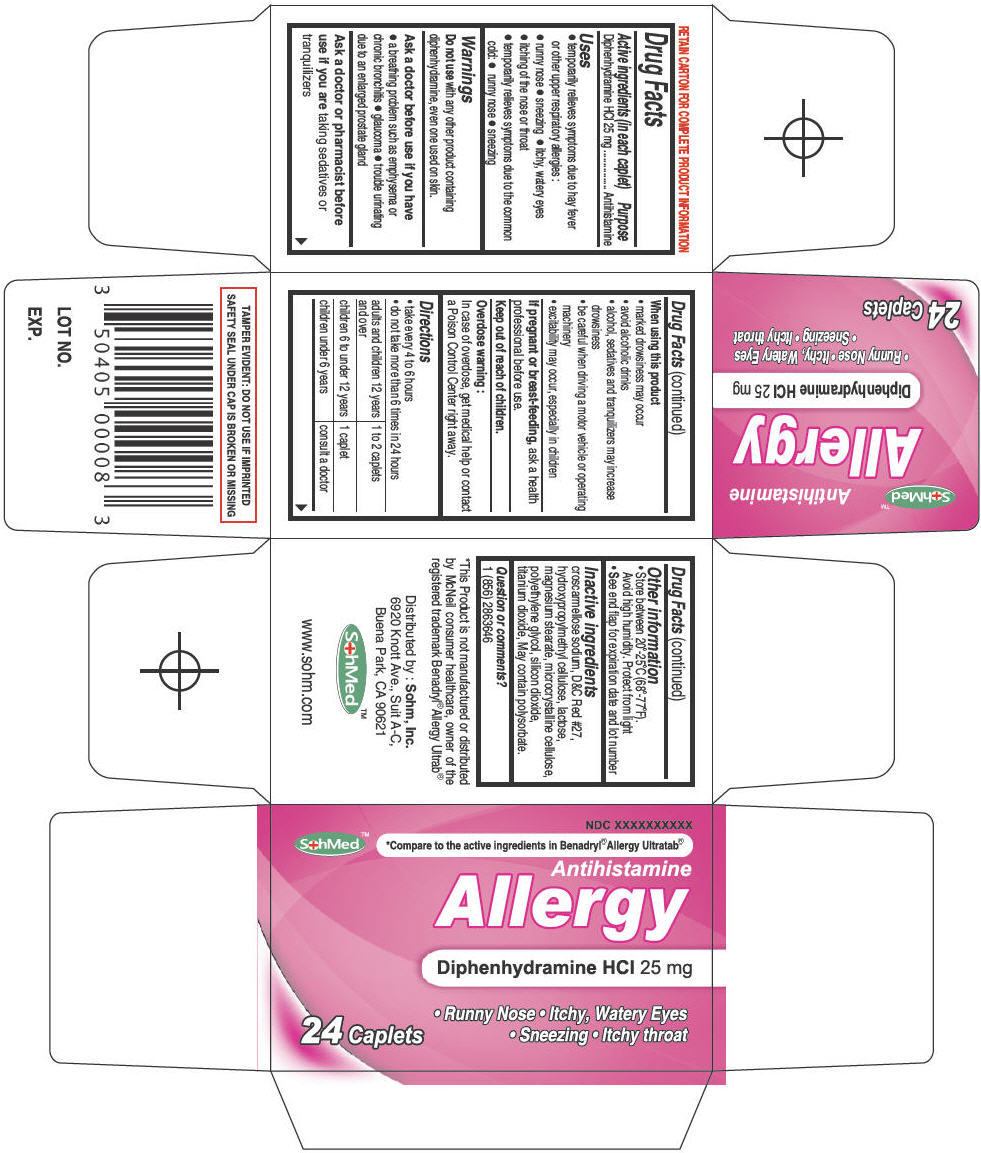
Principal Display Panel 24 Caplet Bottle Carton
NDC XXXXXXXXXX
SohMed™
*Compare to the active ingredients in Benadryl® Allergy Ultratab®
Antihistamine
Allergy
Diphenhydramine HCl 25 mg
24 Caplets
• Runny Nose • Itchy, Watery Eyes
• Sneezing • Itchy throat
Structured Label Content
Uses
- temporarily relieves symptoms due to hay fever or other upper respiratory allergies :
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
- temporarily relieves symptoms due to the common cold:
- runny nose
- sneezing
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers
Section 50570-1 (50570-1)
Do not use with any other product containing diphenhydramine, even one used on skin.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Antihistamine
Directions
- take every 4 to 6 hours
- do not take more than 6 times in 24 hours
| adults and children 12 years and over | 1 to 2 caplets |
| children 6 to under 12 years | 1 caplet |
| children under 6 years | consult a doctor |
Overdose Warning (Overdose warning)
In case of overdose, get medical help or contact a Poison Control Center right away.
Other Information (Other information)
- Store between 20°-25°C (68°-77°F).
Avoid high humidity. Protect from light - See end flap for expiration date and lot number
Inactive Ingredients (Inactive ingredients)
croscarmellose sodium, D&C Red #27, hydroxypropylmethyl cellulose, lactose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, silicon dioxide, titanium dioxide, May contain polysorbate.
Question Or Comments? (Question or comments?)
1 (856) 2863646
When Using This Product (When using this product)
- marked drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
Active Ingredients (in Each Caplet) (Active ingredients (in each caplet))
Diphenhydramine HCl 25 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
Principal Display Panel 24 Caplet Bottle Carton (PRINCIPAL DISPLAY PANEL - 24 Caplet Bottle Carton)
NDC XXXXXXXXXX
SohMed™
*Compare to the active ingredients in Benadryl® Allergy Ultratab®
Antihistamine
Allergy
Diphenhydramine HCl 25 mg
24 Caplets
• Runny Nose • Itchy, Watery Eyes
• Sneezing • Itchy throat
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:41.569809 · Updated: 2026-03-14T22:53:35.209708