Fomepizole Injection, 1.5 Grams Per 1.5 Ml (1 Gram Per Ml)
1dea37fd-ec17-416e-b28e-980a87d49e1a
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
SAGENT ® Rx only Caution: Must be diluted prior to use. Do not use polycarbonate syringes or polycarbonate-containing needles (including polycarbonate filter needles) with fomepizole injection.
Indications and Usage
Fomepizole injection is indicated as an antidote for ethylene glycol (such as antifreeze) or methanol poisoning, or for use in suspected ethylene glycol or methanol ingestion, either alone or in combination with hemodialysis (see DOSAGE AND ADMINISTRATION ).
Dosage and Administration
Do not use polycarbonate syringes or polycarbonate-containing needles (including polycarbonate filter needles) when diluting or administering fomepizole injection. Fomepizole can interact with polycarbonate, compromising the integrity of the syringe and/or needle component containing polycarbonate.
Contraindications
Fomepizole should not be administered to patients with a documented serious hypersensitivity reaction to fomepizole or other pyrazoles.
Adverse Reactions
The most frequent adverse events reported as drug-related or unknown relationship to study drug in the 78 patients and 63 normal volunteers who received fomepizole injection were headache (14%), nausea (11%), and dizziness, increased drowsiness, and bad taste/metallic taste (6% each). All other adverse events in this population were reported in approximately 3% or fewer of those receiving fomepizole injection and were as follows: Body as a Whole: Abdominal pain, fever, multiorgan system failure, pain during fomepizole injection, inflammation at injection site, lumbalgia/backache, hangover Cardiovascular: Sinus bradycardia/bradycardia, phlebosclerosis, tachycardia, phlebitis, shock, hypotension Gastrointestinal: Vomiting, diarrhea, dyspepsia, heartburn, decreased appetite, transient transaminitis Hemic/Lymphatic: Eosinophilia/hypereosinophilia, lymphangitis, disseminated intravascular coagulation, anemia Nervous: Lightheadedness, seizure, agitation, feeling drunk, facial flush, vertigo, nystagmus, anxiety, "felt strange", decreased environmental awareness Respiratory: Hiccups, pharyngitis Skin/Appendages: Application site reaction, rash Special Senses: Abnormal smell, speech/visual disturbances, transient blurred vision, roar in ear Urogenital: Anuria To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
How Supplied
Fomepizole Injection is a clear, colorless to yellow solution for intravenous use supplied as follows: NDC Fomepizole Injection (1 gram per mL) Package Factor 25021-829-02 1.5 grams per 1.5 mL Single-Dose Vial 1 vial per carton
Medication Information
Indications and Usage
Fomepizole injection is indicated as an antidote for ethylene glycol (such as antifreeze) or methanol poisoning, or for use in suspected ethylene glycol or methanol ingestion, either alone or in combination with hemodialysis (see DOSAGE AND ADMINISTRATION ).
Dosage and Administration
Do not use polycarbonate syringes or polycarbonate-containing needles (including polycarbonate filter needles) when diluting or administering fomepizole injection. Fomepizole can interact with polycarbonate, compromising the integrity of the syringe and/or needle component containing polycarbonate.
Contraindications
Fomepizole should not be administered to patients with a documented serious hypersensitivity reaction to fomepizole or other pyrazoles.
Adverse Reactions
The most frequent adverse events reported as drug-related or unknown relationship to study drug in the 78 patients and 63 normal volunteers who received fomepizole injection were headache (14%), nausea (11%), and dizziness, increased drowsiness, and bad taste/metallic taste (6% each). All other adverse events in this population were reported in approximately 3% or fewer of those receiving fomepizole injection and were as follows:
Body as a Whole: Abdominal pain, fever, multiorgan system failure, pain during fomepizole injection, inflammation at injection site, lumbalgia/backache, hangover
Cardiovascular: Sinus bradycardia/bradycardia, phlebosclerosis, tachycardia, phlebitis, shock, hypotension
Gastrointestinal: Vomiting, diarrhea, dyspepsia, heartburn, decreased appetite, transient transaminitis
Hemic/Lymphatic: Eosinophilia/hypereosinophilia, lymphangitis, disseminated intravascular coagulation, anemia
Nervous: Lightheadedness, seizure, agitation, feeling drunk, facial flush, vertigo, nystagmus, anxiety, "felt strange", decreased environmental awareness
Respiratory: Hiccups, pharyngitis
Skin/Appendages: Application site reaction, rash
Special Senses: Abnormal smell, speech/visual disturbances, transient blurred vision, roar in ear
Urogenital: Anuria
To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
How Supplied
Fomepizole Injection is a clear, colorless to yellow solution for intravenous use supplied as follows:
| NDC | Fomepizole Injection (1 gram per mL) | Package Factor |
| 25021-829-02 | 1.5 grams per 1.5 mL Single-Dose Vial | 1 vial per carton |
Description
Fomepizole Injection is intended for intravenous use.
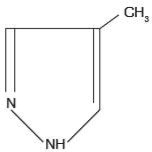
Fomepizole Injection is a competitive inhibitor of alcohol dehydrogenase. The chemical name of fomepizole is 4-methylpyrazole. It has the molecular formula C4H6N2 and a molecular weight of 82.1.
The structural formula is:
It is a clear to yellow liquid at room temperature. Its melting point is 25°C (77°F) and it may present as solid at room temperature. Fomepizole is soluble in water and very soluble in ethanol, diethyl ether, and chloroform. Each vial contains 1.5 mL (1 gram per mL) of fomepizole.
Section 34072-9
General: Fomepizole injection should not be given undiluted or by bolus injection. Venous irritation and phlebosclerosis were noted in two of six normal volunteers given bolus injections (over 5 minutes) of fomepizole injection at a concentration of 25 mg/mL.
Do not use polycarbonate syringes or polycarbonate-containing needles (including polycarbonate filter needles) when diluting or administering fomepizole injection. Fomepizole can interact with polycarbonate, compromising the integrity of the syringe and/or needle component containing polycarbonate.
Minor allergic reactions (mild rash, eosinophilia) have been reported in a few patients receiving fomepizole (see ADVERSE REACTIONS ). Therefore, patients should be monitored for signs of allergic reactions.
Section 34073-7
Drug Interactions: Oral doses of fomepizole (10 to 20 mg/kg), via alcohol dehydrogenase inhibition, significantly reduced the rate of elimination of ethanol (by approximately 40%) given to healthy volunteers in moderate doses. Similarly, ethanol decreased the rate of elimination of fomepizole (by approximately 50%) by the same mechanism.
Reciprocal interactions may occur with concomitant use of fomepizole and drugs that increase or inhibit the cytochrome P450 system (e.g., phenytoin, carbamazepine, cimetidine, ketoconazole), though this has not been studied.
Section 34075-2
Laboratory Tests: In addition to specific antidote treatment with fomepizole, patients intoxicated with ethylene glycol or methanol must be managed for metabolic acidosis, acute renal failure (ethylene glycol), adult respiratory distress syndrome, visual disturbances (methanol), and hypocalcemia. Fluid therapy and sodium bicarbonate administration are potential supportive therapies. In addition, potassium and calcium supplementation and oxygen administration are usually necessary. Hemodialysis is necessary in the anuric patient, or in patients with severe metabolic acidosis or azotemia (see DOSAGE AND ADMINISTRATION ). Treatment success should be assessed by frequent measurements of blood gases, pH, electrolytes, BUN, creatinine, and urinalysis, in addition to other laboratory tests as indicated by individual patient conditions. At frequent intervals throughout the treatment, patients poisoned with ethylene glycol should be monitored for ethylene glycol concentrations in serum and urine, and the presence of urinary oxalate crystals. Similarly, serum methanol concentrations should be monitored in patients poisoned with methanol. Electrocardiography should be performed because acidosis and electrolyte imbalances can affect the cardiovascular system. In the comatose patient, electroencephalography may also be required. In addition, hepatic enzymes and white blood cell counts should be monitored during treatment, as transient increases in serum transaminase concentrations and eosinophilia have been noted with repeated fomepizole dosing.
Section 34080-2
Nursing Mothers: It is not known whether fomepizole is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when fomepizole is administered to a nursing woman.
Section 34081-0
Pediatric Use: Safety and effectiveness in pediatric patients have not been established.
Section 34082-8
Geriatric Use: Safety and effectiveness in geriatric patients have not been established.
Section 34083-6
Carcinogenesis, Mutagenesis and Impairment of Fertility: There have been no long term studies performed in animals to evaluate carcinogenic potential.
There was a positive Ames test result in the Escherichia coli tester strain WP2uvrA and the Salmonella typhimurium tester strain TA102 in the absence of metabolic activation.
There was no evidence of a clastogenic effect in the in vivo mouse micronucleus assay.
In rats, fomepizole (110 mg/kg) administered orally for 40 to 42 days resulted in decreased testicular mass (approximately 8% reduction). This dose is approximately 0.6 times the human maximum daily exposure based on surface area (mg/m2). This reduction was similar for rats treated with either ethanol or fomepizole alone. When fomepizole was given in combination with ethanol, the decrease in testicular mass was significantly greater (approximately 30% reduction) compared to those rats treated exclusively with fomepizole or ethanol.
Section 42228-7
Pregnancy: Animal reproduction studies have not been conducted with fomepizole. It is also not known whether fomepizole can cause fetal harm when administered to pregnant women or can affect reproduction capacity. Fomepizole should be given to pregnant women only if clearly needed.
Section 42229-5
SAGENT®
Rx only
Caution: Must be diluted prior to use. Do not use polycarbonate syringes or polycarbonate-containing needles (including polycarbonate filter needles) with fomepizole injection.
Section 43679-0
Mechanism of Action: Fomepizole is a competitive inhibitor of alcohol dehydrogenase. Alcohol dehydrogenase catalyzes the oxidation of ethanol to acetaldehyde. Alcohol dehydrogenase also catalyzes the initial steps in the metabolism of ethylene glycol and methanol to their toxic metabolites.
Ethylene glycol, the main component of most antifreezes and coolants, is metabolized to glycolaldehyde, which undergoes subsequent sequential oxidations to yield glycolate, glyoxylate, and oxalate. Glycolate and oxalate are the metabolic by products primarily responsible for the metabolic acidosis and renal damage seen in ethylene glycol toxicosis. The lethal dose of ethylene glycol in humans is approximately 1.4 mL/kg.
Methanol, the main component of windshield wiper fluid, is slowly metabolized via alcohol dehydrogenase to formaldehyde with subsequent oxidation via formaldehyde dehydrogenase to yield formic acid. Formic acid is primarily responsible for the metabolic acidosis and visual disturbances (e.g., decreased visual acuity and potential blindness) associated with methanol poisoning. A lethal dose of methanol in humans is approximately 1 to 2 mL/kg.
Fomepizole has been shown in vitro to block alcohol dehydrogenase enzyme activity in dog, monkey, and human liver. The concentration of fomepizole at which alcohol dehydrogenase is inhibited by 50% in vitro is approximately 0.1 mcmol/L.
In a study of dogs given a lethal dose of ethylene glycol, three animals each were administered fomepizole, ethanol, or left untreated (control group). The three animals in the untreated group became progressively obtunded, moribund, and died. At necropsy, all three dogs had severe renal tubular damage. Fomepizole or ethanol, given 3 hours after ethylene glycol ingestion, attenuated the metabolic acidosis and prevented the renal tubular damage associated with ethylene glycol intoxication.
Several studies have demonstrated that fomepizole plasma concentrations of approximately 10 mcmol/L (0.82 mg/L) in monkeys are sufficient to inhibit methanol metabolism to formate, which is also mediated by alcohol dehydrogenase. Based on these results, concentrations of fomepizole in humans in the range of 100 to 300 mcmol/L (8.6 to 24.6 mg/L) have been targeted to assure adequate plasma concentrations for the effective inhibition of alcohol dehydrogenase.
In healthy volunteers, oral doses of fomepizole (10 to 20 mg/kg) significantly reduced the rate of elimination of moderate doses of ethanol, which is also metabolized through the action of alcohol dehydrogenase (see PRECAUTIONS, Drug Interactions ).
Section 43682-4
Pharmacokinetics: The plasma half-life of fomepizole varies with dose, even in patients with normal renal function, and has not been calculated.
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-829-02
Rx only
Fomepizole Injection
1.5 grams per 1.5 mL
(1 gram per mL)
For Intravenous Infusion Only
CAUTION: Must be diluted before use.
1.5 Single-Dose Vial
Overdosage
Nausea, dizziness, and vertigo were noted in healthy volunteers receiving 50 and 100 mg/kg doses of fomepizole (at plasma concentrations of 290 to 520 mcmol/L, 23.8 to 42.6 mg/L). These doses are 3 to 6 times the recommended dose. This dose-dependent CNS effect was short-lived in most subjects and lasted up to 30 hours in one subject.
Fomepizole is dialyzable, and hemodialysis may be useful in treating cases of overdosage.
Storage Conditions
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
SAGENT®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60195 (USA)
Made in India
©2023 Sagent Pharmaceuticals
August 2023
SAGENT Pharmaceuticals ®
Structured Label Content
Section 34072-9 (34072-9)
General: Fomepizole injection should not be given undiluted or by bolus injection. Venous irritation and phlebosclerosis were noted in two of six normal volunteers given bolus injections (over 5 minutes) of fomepizole injection at a concentration of 25 mg/mL.
Do not use polycarbonate syringes or polycarbonate-containing needles (including polycarbonate filter needles) when diluting or administering fomepizole injection. Fomepizole can interact with polycarbonate, compromising the integrity of the syringe and/or needle component containing polycarbonate.
Minor allergic reactions (mild rash, eosinophilia) have been reported in a few patients receiving fomepizole (see ADVERSE REACTIONS ). Therefore, patients should be monitored for signs of allergic reactions.
Section 34073-7 (34073-7)
Drug Interactions: Oral doses of fomepizole (10 to 20 mg/kg), via alcohol dehydrogenase inhibition, significantly reduced the rate of elimination of ethanol (by approximately 40%) given to healthy volunteers in moderate doses. Similarly, ethanol decreased the rate of elimination of fomepizole (by approximately 50%) by the same mechanism.
Reciprocal interactions may occur with concomitant use of fomepizole and drugs that increase or inhibit the cytochrome P450 system (e.g., phenytoin, carbamazepine, cimetidine, ketoconazole), though this has not been studied.
Section 34075-2 (34075-2)
Laboratory Tests: In addition to specific antidote treatment with fomepizole, patients intoxicated with ethylene glycol or methanol must be managed for metabolic acidosis, acute renal failure (ethylene glycol), adult respiratory distress syndrome, visual disturbances (methanol), and hypocalcemia. Fluid therapy and sodium bicarbonate administration are potential supportive therapies. In addition, potassium and calcium supplementation and oxygen administration are usually necessary. Hemodialysis is necessary in the anuric patient, or in patients with severe metabolic acidosis or azotemia (see DOSAGE AND ADMINISTRATION ). Treatment success should be assessed by frequent measurements of blood gases, pH, electrolytes, BUN, creatinine, and urinalysis, in addition to other laboratory tests as indicated by individual patient conditions. At frequent intervals throughout the treatment, patients poisoned with ethylene glycol should be monitored for ethylene glycol concentrations in serum and urine, and the presence of urinary oxalate crystals. Similarly, serum methanol concentrations should be monitored in patients poisoned with methanol. Electrocardiography should be performed because acidosis and electrolyte imbalances can affect the cardiovascular system. In the comatose patient, electroencephalography may also be required. In addition, hepatic enzymes and white blood cell counts should be monitored during treatment, as transient increases in serum transaminase concentrations and eosinophilia have been noted with repeated fomepizole dosing.
Section 34080-2 (34080-2)
Nursing Mothers: It is not known whether fomepizole is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when fomepizole is administered to a nursing woman.
Section 34081-0 (34081-0)
Pediatric Use: Safety and effectiveness in pediatric patients have not been established.
Section 34082-8 (34082-8)
Geriatric Use: Safety and effectiveness in geriatric patients have not been established.
Section 34083-6 (34083-6)
Carcinogenesis, Mutagenesis and Impairment of Fertility: There have been no long term studies performed in animals to evaluate carcinogenic potential.
There was a positive Ames test result in the Escherichia coli tester strain WP2uvrA and the Salmonella typhimurium tester strain TA102 in the absence of metabolic activation.
There was no evidence of a clastogenic effect in the in vivo mouse micronucleus assay.
In rats, fomepizole (110 mg/kg) administered orally for 40 to 42 days resulted in decreased testicular mass (approximately 8% reduction). This dose is approximately 0.6 times the human maximum daily exposure based on surface area (mg/m2). This reduction was similar for rats treated with either ethanol or fomepizole alone. When fomepizole was given in combination with ethanol, the decrease in testicular mass was significantly greater (approximately 30% reduction) compared to those rats treated exclusively with fomepizole or ethanol.
Section 42228-7 (42228-7)
Pregnancy: Animal reproduction studies have not been conducted with fomepizole. It is also not known whether fomepizole can cause fetal harm when administered to pregnant women or can affect reproduction capacity. Fomepizole should be given to pregnant women only if clearly needed.
Section 42229-5 (42229-5)
SAGENT®
Rx only
Caution: Must be diluted prior to use. Do not use polycarbonate syringes or polycarbonate-containing needles (including polycarbonate filter needles) with fomepizole injection.
Section 43679-0 (43679-0)
Mechanism of Action: Fomepizole is a competitive inhibitor of alcohol dehydrogenase. Alcohol dehydrogenase catalyzes the oxidation of ethanol to acetaldehyde. Alcohol dehydrogenase also catalyzes the initial steps in the metabolism of ethylene glycol and methanol to their toxic metabolites.
Ethylene glycol, the main component of most antifreezes and coolants, is metabolized to glycolaldehyde, which undergoes subsequent sequential oxidations to yield glycolate, glyoxylate, and oxalate. Glycolate and oxalate are the metabolic by products primarily responsible for the metabolic acidosis and renal damage seen in ethylene glycol toxicosis. The lethal dose of ethylene glycol in humans is approximately 1.4 mL/kg.
Methanol, the main component of windshield wiper fluid, is slowly metabolized via alcohol dehydrogenase to formaldehyde with subsequent oxidation via formaldehyde dehydrogenase to yield formic acid. Formic acid is primarily responsible for the metabolic acidosis and visual disturbances (e.g., decreased visual acuity and potential blindness) associated with methanol poisoning. A lethal dose of methanol in humans is approximately 1 to 2 mL/kg.
Fomepizole has been shown in vitro to block alcohol dehydrogenase enzyme activity in dog, monkey, and human liver. The concentration of fomepizole at which alcohol dehydrogenase is inhibited by 50% in vitro is approximately 0.1 mcmol/L.
In a study of dogs given a lethal dose of ethylene glycol, three animals each were administered fomepizole, ethanol, or left untreated (control group). The three animals in the untreated group became progressively obtunded, moribund, and died. At necropsy, all three dogs had severe renal tubular damage. Fomepizole or ethanol, given 3 hours after ethylene glycol ingestion, attenuated the metabolic acidosis and prevented the renal tubular damage associated with ethylene glycol intoxication.
Several studies have demonstrated that fomepizole plasma concentrations of approximately 10 mcmol/L (0.82 mg/L) in monkeys are sufficient to inhibit methanol metabolism to formate, which is also mediated by alcohol dehydrogenase. Based on these results, concentrations of fomepizole in humans in the range of 100 to 300 mcmol/L (8.6 to 24.6 mg/L) have been targeted to assure adequate plasma concentrations for the effective inhibition of alcohol dehydrogenase.
In healthy volunteers, oral doses of fomepizole (10 to 20 mg/kg) significantly reduced the rate of elimination of moderate doses of ethanol, which is also metabolized through the action of alcohol dehydrogenase (see PRECAUTIONS, Drug Interactions ).
Section 43682-4 (43682-4)
Pharmacokinetics: The plasma half-life of fomepizole varies with dose, even in patients with normal renal function, and has not been calculated.
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-829-02
Rx only
Fomepizole Injection
1.5 grams per 1.5 mL
(1 gram per mL)
For Intravenous Infusion Only
CAUTION: Must be diluted before use.
1.5 Single-Dose Vial
Overdosage (OVERDOSAGE)
Nausea, dizziness, and vertigo were noted in healthy volunteers receiving 50 and 100 mg/kg doses of fomepizole (at plasma concentrations of 290 to 520 mcmol/L, 23.8 to 42.6 mg/L). These doses are 3 to 6 times the recommended dose. This dose-dependent CNS effect was short-lived in most subjects and lasted up to 30 hours in one subject.
Fomepizole is dialyzable, and hemodialysis may be useful in treating cases of overdosage.
Description (DESCRIPTION)
Fomepizole Injection is intended for intravenous use.
Fomepizole Injection is a competitive inhibitor of alcohol dehydrogenase. The chemical name of fomepizole is 4-methylpyrazole. It has the molecular formula C4H6N2 and a molecular weight of 82.1.
The structural formula is:
It is a clear to yellow liquid at room temperature. Its melting point is 25°C (77°F) and it may present as solid at room temperature. Fomepizole is soluble in water and very soluble in ethanol, diethyl ether, and chloroform. Each vial contains 1.5 mL (1 gram per mL) of fomepizole.
How Supplied (HOW SUPPLIED)
Fomepizole Injection is a clear, colorless to yellow solution for intravenous use supplied as follows:
| NDC | Fomepizole Injection (1 gram per mL) | Package Factor |
| 25021-829-02 | 1.5 grams per 1.5 mL Single-Dose Vial | 1 vial per carton |
Adverse Reactions (ADVERSE REACTIONS)
The most frequent adverse events reported as drug-related or unknown relationship to study drug in the 78 patients and 63 normal volunteers who received fomepizole injection were headache (14%), nausea (11%), and dizziness, increased drowsiness, and bad taste/metallic taste (6% each). All other adverse events in this population were reported in approximately 3% or fewer of those receiving fomepizole injection and were as follows:
Body as a Whole: Abdominal pain, fever, multiorgan system failure, pain during fomepizole injection, inflammation at injection site, lumbalgia/backache, hangover
Cardiovascular: Sinus bradycardia/bradycardia, phlebosclerosis, tachycardia, phlebitis, shock, hypotension
Gastrointestinal: Vomiting, diarrhea, dyspepsia, heartburn, decreased appetite, transient transaminitis
Hemic/Lymphatic: Eosinophilia/hypereosinophilia, lymphangitis, disseminated intravascular coagulation, anemia
Nervous: Lightheadedness, seizure, agitation, feeling drunk, facial flush, vertigo, nystagmus, anxiety, "felt strange", decreased environmental awareness
Respiratory: Hiccups, pharyngitis
Skin/Appendages: Application site reaction, rash
Special Senses: Abnormal smell, speech/visual disturbances, transient blurred vision, roar in ear
Urogenital: Anuria
To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Contraindications (CONTRAINDICATIONS)
Fomepizole should not be administered to patients with a documented serious hypersensitivity reaction to fomepizole or other pyrazoles.
Storage Conditions
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
SAGENT®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60195 (USA)
Made in India
©2023 Sagent Pharmaceuticals
August 2023
SAGENT Pharmaceuticals ®
Indications and Usage (INDICATIONS AND USAGE)
Fomepizole injection is indicated as an antidote for ethylene glycol (such as antifreeze) or methanol poisoning, or for use in suspected ethylene glycol or methanol ingestion, either alone or in combination with hemodialysis (see DOSAGE AND ADMINISTRATION ).
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Do not use polycarbonate syringes or polycarbonate-containing needles (including polycarbonate filter needles) when diluting or administering fomepizole injection. Fomepizole can interact with polycarbonate, compromising the integrity of the syringe and/or needle component containing polycarbonate.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:41.968399 · Updated: 2026-03-14T21:58:55.917732