Quinapril And Hydrochlorothiazide Tablets, Usp
1de5420c-23d6-40c5-ba24-39143c0f90fb
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: FETAL TOXICITY When pregnancy is detected, discontinue quinapril and hydrochlorothiazide tablets as soon as possible. Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. See Warnings: Fetal Toxicity
Contraindications
Quinapril and hydrochlorothiazide tablets are contraindicated in patients who are hypersensitive to quinapril or hydrochlorothiazide and in patients with a history of angioedema related to previous treatment with an ACE inhibitor. Quinapril and hydrochlorothiazide tablets is contraindicated in combination with a neprilysin inhibitor (e.g., sacubitril). Do not administer Quinapril and hydrochlorothiazide tablets within 36 hours of switching to or from sacubitril/valsartan, a neprilysin inhibitor (see WARNINGS and PRECAUTIONS). Because of the hydrochlorothiazide components, this product is contraindicated in patients with anuria or hypersensitivity to other sulfonamide-derived drugs. Do not co-administer quinapril and hydrochlorothiazide with aliskiren: in patients with diabetes.
Medication Information
Recent Major Changes
WARNING: FETAL TOXICITY
- When pregnancy is detected, discontinue quinapril and hydrochlorothiazide tablets as soon as possible.
- Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. See Warnings: Fetal Toxicity
Contraindications
Quinapril and hydrochlorothiazide tablets are contraindicated in patients who are hypersensitive to quinapril or hydrochlorothiazide and in patients with a history of angioedema related to previous treatment with an ACE inhibitor.
Quinapril and hydrochlorothiazide tablets is contraindicated in combination with a neprilysin inhibitor (e.g., sacubitril). Do not administer Quinapril and hydrochlorothiazide tablets within 36 hours of switching to or from sacubitril/valsartan, a neprilysin inhibitor (see WARNINGS and PRECAUTIONS).
Because of the hydrochlorothiazide components, this product is contraindicated in patients with anuria or hypersensitivity to other sulfonamide-derived drugs.
Do not co-administer quinapril and hydrochlorothiazide with aliskiren:
- in patients with diabetes.
Description
WARNING: FETAL TOXICITY When pregnancy is detected, discontinue quinapril and hydrochlorothiazide tablets as soon as possible. Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. See Warnings: Fetal Toxicity
Section 42229-5
Quinapril and hydrochlorothiazide tablets, USP are fixed-combination tablet that combines an angiotensin-converting enzyme (ACE) inhibitor, quinapril hydrochloride, and a thiazide diuretic, hydrochlorothiazide.
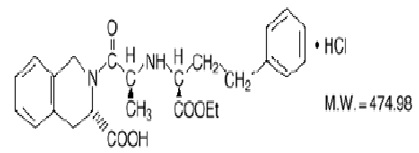
Quinapril hydrochloride, USP is chemically described as [3S-[2[R*(R*)], 3R*]]-2-[2-[[1-(ethoxycarbonyl)-3-phenylpropyl]amino]-1-oxopropyl]-1,2,3,4-tetrahydro-3-isoquinolinecarboxylic acid, monohydrochloride. Its empirical formula is C25H30N2O5. HCl and its structural formula is:
Quinapril hydrochloride, USP is a white to off-white amorphous powder that is freely soluble in aqueous solvents.
Hydrochlorothiazide, USP is chemically described as: 6-Chloro-3,4-dihydro-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide. Its empirical formula is C7H8CIN3O4S2 and its structural formula is:
Hydrochlorothiazide, USP is a white to off-white, crystalline powder which is slightly soluble in water but freely soluble in sodium hydroxide solution.
Quinapril and hydrochlorothiazide tablets, USP are available for oral use as fixed combination tablets in three strengths of quinapril with hydrochlorothiazide: 10 mg with 12.5 mg (quinapril and hydrochlorothiazide 10/12.5), 20 mg with 12.5 mg (quinapril and hydrochlorothiazide 20/12.5), and 20 mg with 25 mg (quinapril and hydrochlorothiazide 20/25). Inactive ingredients: lactose, magnesium carbonate, crospovidone, povidone, magnesium stearate and opadry pink 03B14436 (hypromellose, titanium dioxide, polyethylene glycol and iron oxide red).
Package Label.principal Display Panel
NDC 69097-828-05 Rx Only
Quinapril and
Hydrochlorothiazide
Tablets, USP
10 mg/12.5 mg*
90 Tablets
Cipla
NDC 69097-829-05 Rx Only
Quinapril and
Hydrochlorothiazide
Tablets, USP
20 mg/12.5 mg*
90 Tablets
Cipla
NDC 69097-831-05 Rx Only
Quinapril and
Hydrochlorothiazide
Tablets, USP
20 mg/25 mg*
90 Tablets
Cipla
Structured Label Content
Recent Major Changes (34066-1)
WARNING: FETAL TOXICITY
- When pregnancy is detected, discontinue quinapril and hydrochlorothiazide tablets as soon as possible.
- Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. See Warnings: Fetal Toxicity
Section 42229-5 (42229-5)
Quinapril and hydrochlorothiazide tablets, USP are fixed-combination tablet that combines an angiotensin-converting enzyme (ACE) inhibitor, quinapril hydrochloride, and a thiazide diuretic, hydrochlorothiazide.
Quinapril hydrochloride, USP is chemically described as [3S-[2[R*(R*)], 3R*]]-2-[2-[[1-(ethoxycarbonyl)-3-phenylpropyl]amino]-1-oxopropyl]-1,2,3,4-tetrahydro-3-isoquinolinecarboxylic acid, monohydrochloride. Its empirical formula is C25H30N2O5. HCl and its structural formula is:
Quinapril hydrochloride, USP is a white to off-white amorphous powder that is freely soluble in aqueous solvents.
Hydrochlorothiazide, USP is chemically described as: 6-Chloro-3,4-dihydro-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide. Its empirical formula is C7H8CIN3O4S2 and its structural formula is:
Hydrochlorothiazide, USP is a white to off-white, crystalline powder which is slightly soluble in water but freely soluble in sodium hydroxide solution.
Quinapril and hydrochlorothiazide tablets, USP are available for oral use as fixed combination tablets in three strengths of quinapril with hydrochlorothiazide: 10 mg with 12.5 mg (quinapril and hydrochlorothiazide 10/12.5), 20 mg with 12.5 mg (quinapril and hydrochlorothiazide 20/12.5), and 20 mg with 25 mg (quinapril and hydrochlorothiazide 20/25). Inactive ingredients: lactose, magnesium carbonate, crospovidone, povidone, magnesium stearate and opadry pink 03B14436 (hypromellose, titanium dioxide, polyethylene glycol and iron oxide red).
Contraindications (CONTRAINDICATIONS)
Quinapril and hydrochlorothiazide tablets are contraindicated in patients who are hypersensitive to quinapril or hydrochlorothiazide and in patients with a history of angioedema related to previous treatment with an ACE inhibitor.
Quinapril and hydrochlorothiazide tablets is contraindicated in combination with a neprilysin inhibitor (e.g., sacubitril). Do not administer Quinapril and hydrochlorothiazide tablets within 36 hours of switching to or from sacubitril/valsartan, a neprilysin inhibitor (see WARNINGS and PRECAUTIONS).
Because of the hydrochlorothiazide components, this product is contraindicated in patients with anuria or hypersensitivity to other sulfonamide-derived drugs.
Do not co-administer quinapril and hydrochlorothiazide with aliskiren:
- in patients with diabetes.
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
NDC 69097-828-05 Rx Only
Quinapril and
Hydrochlorothiazide
Tablets, USP
10 mg/12.5 mg*
90 Tablets
Cipla
NDC 69097-829-05 Rx Only
Quinapril and
Hydrochlorothiazide
Tablets, USP
20 mg/12.5 mg*
90 Tablets
Cipla
NDC 69097-831-05 Rx Only
Quinapril and
Hydrochlorothiazide
Tablets, USP
20 mg/25 mg*
90 Tablets
Cipla
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:36:29.027193 · Updated: 2026-03-14T21:46:26.671613