1de1169c-e6dd-486f-e063-6394a90a3b59
34390-5
HUMAN OTC DRUG LABEL
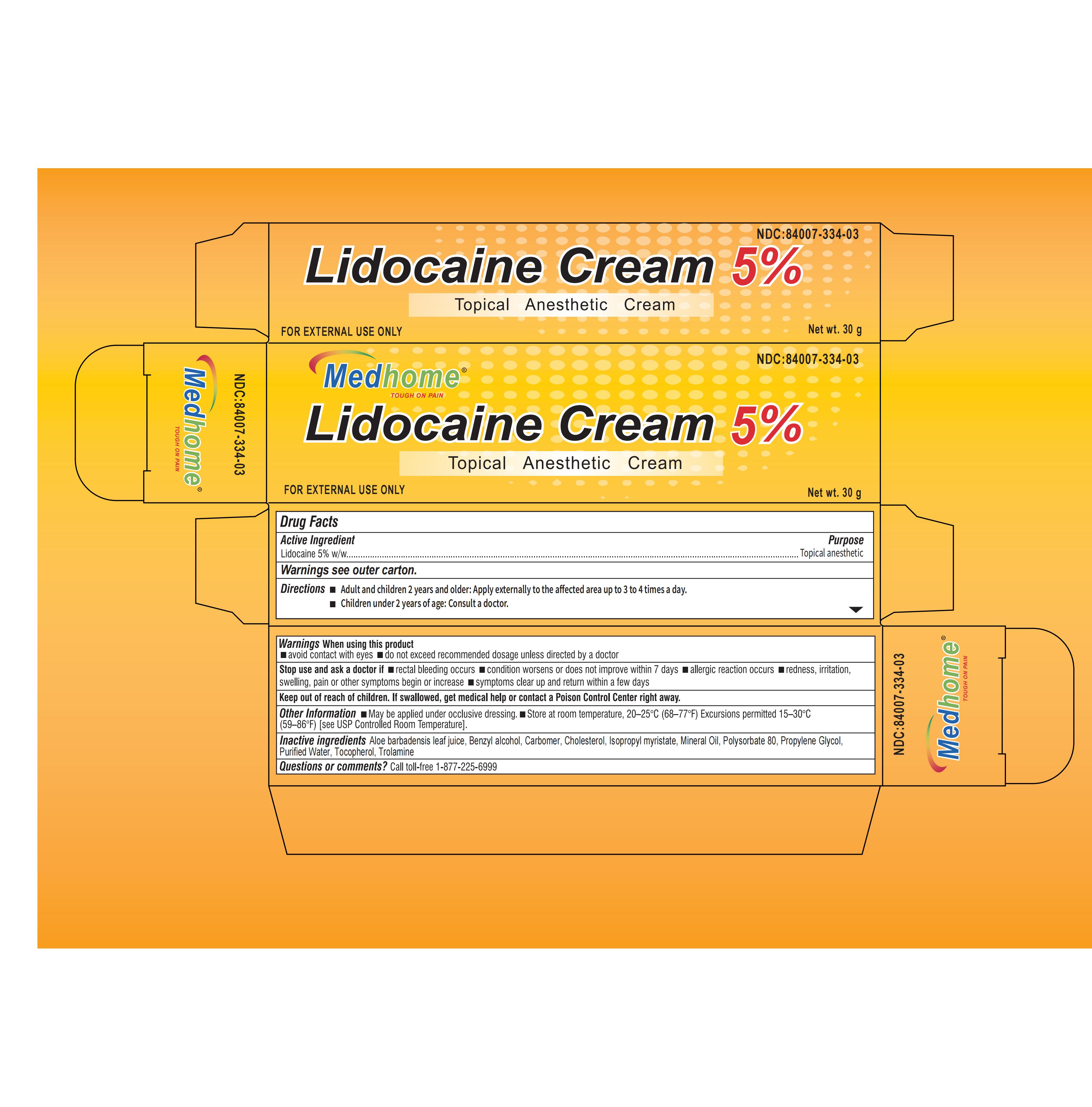
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 5%
Purpose
Local Anesthetic
Medication Information
Purpose
Local Anesthetic
Description
Lidocaine 5%
Use
For temporary relief of local discomfort, itching, pain, soreness, or burning related to the tattoo process
Package
Stop Use
Do not exceed the recommended daily dosage unless directed by a doctor.
Avoid contact with eyes. lf this happens, rinse thoroughly with water.
lf symptoms persist for more than 7 days or subside but occur again within 3 days, stop use and consult a physician.
Stop use and seek medical attention if signs of an allergy occur, such as redness,iritation, sweling, pain, or other severe symptoms. fsymptoms do not subside completely within 7 days, seek medica help.
Warnings
For external use only.
Ask Doctor
Stop use and sek meditcal attention if signs of an alerqy occur,such as rednes,irtation,sweling, pain, or other severe symptoms.If symptoms do not subside completely within 7 days, seek medical help.
Directions
Before applying the numbing cream, thoroughly wash your hands and wear disposable tattoo gloves.
Clean the area of the body receivina the tattoo,Dry completely.
Apply a generous amount of numbing cream to the cleaned area and rub it inthoroughly in a circlar motion for30 to 60 seconds,Then. cover with plastic wrap.
Leave the cream and wrap in place for 30 to 45 minutes,Wipe away the cream and thoroughly clean the area to prepare for a pain-free tattoo.
Do Not Use
Do not exceed the recommended daily dosage unless directed by a doctor
Active Ingredient
Lidocaine 5%
Other Information
Store between 15-30℃(59-86F). Avoid freezing and excessive heat above 40 ℃(104°F).
Inactive Ingredients
Water,Carbomer934, propylene Glycol,soybean Lecithin,.Alpha.-Tocopherol Acetate,Benzyl Alcohol.
Keep Out of Reach of Children
Keep out of reach of children
Structured Label Content
Use
For temporary relief of local discomfort, itching, pain, soreness, or burning related to the tattoo process
Package
Purpose
Local Anesthetic
Stop Use
Do not exceed the recommended daily dosage unless directed by a doctor.
Avoid contact with eyes. lf this happens, rinse thoroughly with water.
lf symptoms persist for more than 7 days or subside but occur again within 3 days, stop use and consult a physician.
Stop use and seek medical attention if signs of an allergy occur, such as redness,iritation, sweling, pain, or other severe symptoms. fsymptoms do not subside completely within 7 days, seek medica help.
Warnings
For external use only.
Ask Doctor
Stop use and sek meditcal attention if signs of an alerqy occur,such as rednes,irtation,sweling, pain, or other severe symptoms.If symptoms do not subside completely within 7 days, seek medical help.
Directions
Before applying the numbing cream, thoroughly wash your hands and wear disposable tattoo gloves.
Clean the area of the body receivina the tattoo,Dry completely.
Apply a generous amount of numbing cream to the cleaned area and rub it inthoroughly in a circlar motion for30 to 60 seconds,Then. cover with plastic wrap.
Leave the cream and wrap in place for 30 to 45 minutes,Wipe away the cream and thoroughly clean the area to prepare for a pain-free tattoo.
Do Not Use (Do not use)
Do not exceed the recommended daily dosage unless directed by a doctor
Active Ingredient
Lidocaine 5%
Other Information (Other information)
Store between 15-30℃(59-86F). Avoid freezing and excessive heat above 40 ℃(104°F).
Inactive Ingredients (Inactive ingredients)
Water,Carbomer934, propylene Glycol,soybean Lecithin,.Alpha.-Tocopherol Acetate,Benzyl Alcohol.
Keep Out of Reach of Children (Keep Out Of Reach Of Children)
Keep out of reach of children
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:40.537999 · Updated: 2026-03-14T23:11:23.873993