Childrens
1daa3c05-eaeb-4219-a959-4959750a30a0
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Expectorant
Medication Information
Purpose
Expectorant
Description
Drug Facts
Uses
helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if cough lasts more than 7 days, comes back, or occurs with fever, rash, or persistent headache. These could be signs of a serious illness.
Directions
- do not take more than 6 doses in any 24-hour period
- measure only with dosing cup provided
- do not use dosing cup with other products
- mL = milliliter
| Age | Dose |
|---|---|
| children 6 years to under 12 years | 5 mL – 10 mL every 4 hours |
| children 4 years to under 6 years | 2.5 mL – 5 mL every 4 hours |
| children under 4 years | do not use |
Questions?
1-866-MUCINEX (1-866-682-4639)
You may also report side effects to this phone number.
Other Information
- tamper evident: do not use if printed neckband on bottle cap is broken or missing
- store between 20-25°C (68-77°F)
- do not refrigerate
- dosing cup provided
Inactive Ingredients
acesulfame potassium, anhydrous citric acid, FD&C blue #1, FD&C red #40, flavor, glycerin, maltitol, propylene glycol, purified water, saccharin sodium, sodium benzoate, sorbitol, sucralose, xanthan gum
Active Ingredient (in Each 5 Ml)
Guaifenesin, USP 100 mg
Ask A Doctor Before Use If the Child Has
- cough that occurs with too much phlegm (mucus)
- persistent or chronic cough such as occurs with asthma
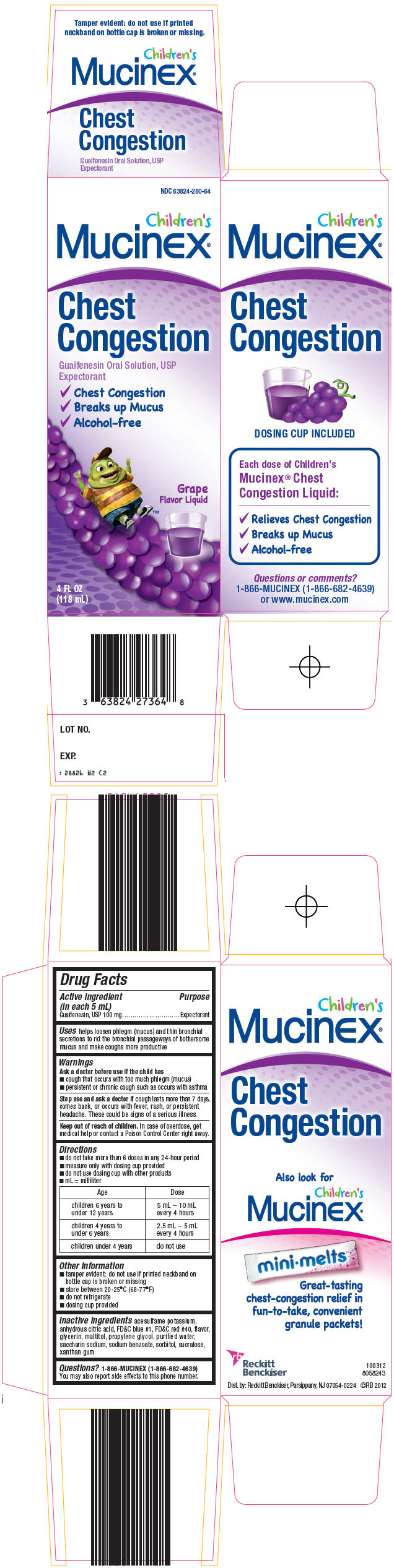
Principal Display Panel 118 Ml Bottle Carton
NDC 63824-280-64
Children's
Mucinex®
Chest
Congestion
Guaifenesin Oral Solution, USP
Expectorant
-
✓Chest Congestion
-
✓Breaks up Mucus
-
✓Alcohol-free
Grape
Flavor Liquid
4 FL OZ
(118 mL)
Structured Label Content
Uses
helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if cough lasts more than 7 days, comes back, or occurs with fever, rash, or persistent headache. These could be signs of a serious illness.
Purpose
Expectorant
Directions
- do not take more than 6 doses in any 24-hour period
- measure only with dosing cup provided
- do not use dosing cup with other products
- mL = milliliter
| Age | Dose |
|---|---|
| children 6 years to under 12 years | 5 mL – 10 mL every 4 hours |
| children 4 years to under 6 years | 2.5 mL – 5 mL every 4 hours |
| children under 4 years | do not use |
Questions?
1-866-MUCINEX (1-866-682-4639)
You may also report side effects to this phone number.
Other Information (Other information)
- tamper evident: do not use if printed neckband on bottle cap is broken or missing
- store between 20-25°C (68-77°F)
- do not refrigerate
- dosing cup provided
Inactive Ingredients (Inactive ingredients)
acesulfame potassium, anhydrous citric acid, FD&C blue #1, FD&C red #40, flavor, glycerin, maltitol, propylene glycol, purified water, saccharin sodium, sodium benzoate, sorbitol, sucralose, xanthan gum
Active Ingredient (in Each 5 Ml) (Active ingredient (in each 5 mL))
Guaifenesin, USP 100 mg
Ask A Doctor Before Use If the Child Has (Ask a doctor before use if the child has)
- cough that occurs with too much phlegm (mucus)
- persistent or chronic cough such as occurs with asthma
Principal Display Panel 118 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 118 mL Bottle Carton)
NDC 63824-280-64
Children's
Mucinex®
Chest
Congestion
Guaifenesin Oral Solution, USP
Expectorant
-
✓Chest Congestion
-
✓Breaks up Mucus
-
✓Alcohol-free
Grape
Flavor Liquid
4 FL OZ
(118 mL)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:33.098720 · Updated: 2026-03-14T22:53:19.872567