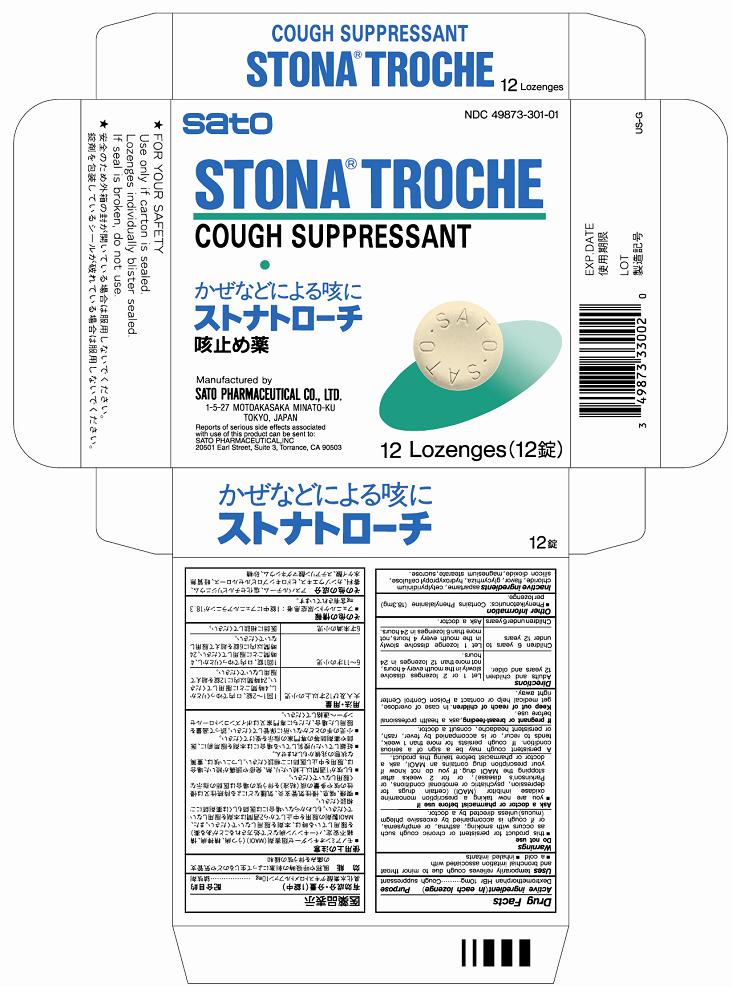
Stona Troche
1ccc9e56-32a4-450f-8412-5814b6aef285
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredients (in each lozenge) Dextromethorphan HBr 10mg
Medication Information
Warnings and Precautions
Enter section text here
Indications and Usage
Uses temporarily relieves cough due to minor throat and bronchial irritation associated with
■a cold ■inhaled irritants
Dosage and Administration
Directions
Adults and children 12 years and older - Let 1 or 2 lozenges dissolve slowly in the mouth every 4 hours, not more than 12 lozenges in 24 hours.
Children 6 years to under 12 years - Let 1 lozenge dissolve slowly in the mouth every 4 hours, not more than 6 lozenges in 24 hours.
Children under 6 years - Ask a doctor
■once or twice daily, preferably morning and evening, when needed or as directed by a doctor
■start initial dosage with minimum dose, then adjust it to suit to bowel condition
Description
Active ingredients (in each lozenge) Dextromethorphan HBr 10mg
Section 50565-1
Keep out of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Section 50568-5
Ask a doctor or pharmacist before use if
■you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product. A persistent cough may be a sign of a serious condition. If cough persists for more than 1 week, tends to recur, or is accompanied by fever, rash, or persistent headache, consult a doctor.
Section 50570-1
Do not use
■this product for persistent or chronic cough such as occurs with smoking, asthema, or emphysem, or if cough is accompanied by excessive phlegm (mucus) unless directed by a doctor.
Section 51727-6
Inactive ingredients aspartame, cetylpyridinium chloride, flavor, glycyrrhiza, hydroxypropyl cellulose, silicon dioxide, magnesium stearate, sucrose.
Section 51945-4
stonatrochecart.jpg
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Purpose
Cough suppressant
Section 55106-9
Active ingredients (in each lozenge)
Dextromethorphan HBr 10mg
Section 60561-8
Other information
■Phenylketonurics: Contains Phenylalanine (18.3mg) per lozenge.
Structured Label Content
Indications and Usage (34067-9)
Uses temporarily relieves cough due to minor throat and bronchial irritation associated with
■a cold ■inhaled irritants
Dosage and Administration (34068-7)
Directions
Adults and children 12 years and older - Let 1 or 2 lozenges dissolve slowly in the mouth every 4 hours, not more than 12 lozenges in 24 hours.
Children 6 years to under 12 years - Let 1 lozenge dissolve slowly in the mouth every 4 hours, not more than 6 lozenges in 24 hours.
Children under 6 years - Ask a doctor
■once or twice daily, preferably morning and evening, when needed or as directed by a doctor
■start initial dosage with minimum dose, then adjust it to suit to bowel condition
Warnings and Precautions (34071-1)
Enter section text here
Section 50565-1 (50565-1)
Keep out of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if
■you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product. A persistent cough may be a sign of a serious condition. If cough persists for more than 1 week, tends to recur, or is accompanied by fever, rash, or persistent headache, consult a doctor.
Section 50570-1 (50570-1)
Do not use
■this product for persistent or chronic cough such as occurs with smoking, asthema, or emphysem, or if cough is accompanied by excessive phlegm (mucus) unless directed by a doctor.
Section 51727-6 (51727-6)
Inactive ingredients aspartame, cetylpyridinium chloride, flavor, glycyrrhiza, hydroxypropyl cellulose, silicon dioxide, magnesium stearate, sucrose.
Section 51945-4 (51945-4)
stonatrochecart.jpg
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Purpose
Cough suppressant
Section 55106-9 (55106-9)
Active ingredients (in each lozenge)
Dextromethorphan HBr 10mg
Section 60561-8 (60561-8)
Other information
■Phenylketonurics: Contains Phenylalanine (18.3mg) per lozenge.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:24.379253 · Updated: 2026-03-14T23:01:55.379030