Freshmaker Antiseptic Hand Wipes
1cbcd01d-604e-4916-9660-82a6b5fe05e5
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
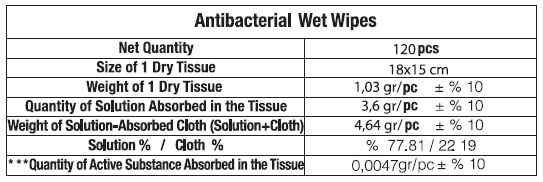
Composition & Product
Identifiers & Packaging
Description
Benzalkonium chloride 0.13%
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Benzalkonium chloride 0.13%
Uses
- hand wipes to decrease bacteria on the skin
- recommended for repeated use
- for use when soap and water are not available
Section 42229-5
ANTISEPTIC AGENT
BENZALKONIUM CHLORIDE
Manufactured by : Fulya Kozmetik Islak Mendil Iml. ve Paz.Ltd.Sti.
Kirac Osmangazi Kucuk Ayazma Street No:17
34522, Esenyurt/ISTANBUL/TURKEY
P: +90 212 689 28 28 Fax: +90 212 689 82 28
Made in TURKEY
Web: http://www.fulyakozmetik.com
e-mail: [email protected]
Lot No:
Expiry:
You may also report any serious side effects to
PO BOX 328, Wood Ridge, NJ 07075-328
Warnings
For external use only
Do not use
- in children less than 2 months of age
- on open skin wounds
When using this product • do not get into eyes. In case of contact, rinse eyes thoroughly with water.
Stop use and ask a doctor if
- irritation and redness develop
- condition persists for more than 72 hours
Packaging
Directions
- open resealable label. Pull out wipes. Close resealable label to prevent drying out
- wipe hands thoroughly with product and allow to dry
- dispose the wipe after use
- supervise children under 6 years of age when using this product to avoid swallowing
- do not flush
drug Facts
Active Ingredient
Benzalkonium chloride 0.13%
Other Information
- store between 15-30˚C(59-86˚F)
- avoid freezing and excessive heat above 40˚C(104˚F)
Inactive Ingredients
benzoic acid, citric acid, cocamidopropyl betaine, dehydroacetic acid, fragrance, glycerin, PEG-7 glyceryl cocoate, phenoxyethanol, polysorbate 20, propylene glycol, tetrasodium EDTA, water
keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away
Structured Label Content
Uses
- hand wipes to decrease bacteria on the skin
- recommended for repeated use
- for use when soap and water are not available
Section 42229-5 (42229-5)
ANTISEPTIC AGENT
BENZALKONIUM CHLORIDE
Manufactured by : Fulya Kozmetik Islak Mendil Iml. ve Paz.Ltd.Sti.
Kirac Osmangazi Kucuk Ayazma Street No:17
34522, Esenyurt/ISTANBUL/TURKEY
P: +90 212 689 28 28 Fax: +90 212 689 82 28
Made in TURKEY
Web: http://www.fulyakozmetik.com
e-mail: [email protected]
Lot No:
Expiry:
You may also report any serious side effects to
PO BOX 328, Wood Ridge, NJ 07075-328
Purpose
Antiseptic
Warnings
For external use only
Do not use
- in children less than 2 months of age
- on open skin wounds
When using this product • do not get into eyes. In case of contact, rinse eyes thoroughly with water.
Stop use and ask a doctor if
- irritation and redness develop
- condition persists for more than 72 hours
Packaging
Directions
- open resealable label. Pull out wipes. Close resealable label to prevent drying out
- wipe hands thoroughly with product and allow to dry
- dispose the wipe after use
- supervise children under 6 years of age when using this product to avoid swallowing
- do not flush
drug Facts (Drug Facts)
Active Ingredient (Active ingredient)
Benzalkonium chloride 0.13%
Other Information (Other information)
- store between 15-30˚C(59-86˚F)
- avoid freezing and excessive heat above 40˚C(104˚F)
Inactive Ingredients
benzoic acid, citric acid, cocamidopropyl betaine, dehydroacetic acid, fragrance, glycerin, PEG-7 glyceryl cocoate, phenoxyethanol, polysorbate 20, propylene glycol, tetrasodium EDTA, water
keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:58:15.541400 · Updated: 2026-03-14T22:57:42.726897