Primaquine Phosphate Tablets, Usp
1bfbf4ae-81b8-4160-a00d-6322aadd4b59
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
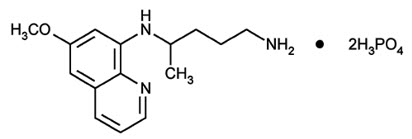
Primaquine phosphate is 8-[(4-amino-1-methylbutyl) amino]-6-methoxyquinoline phosphate, a synthetic compound with potent antimalarial activity. The molecular formula of Primaquine phosphate is C 15 H 21 N 3 O∙2H 3 PO 4 and its molecular weight is 455.34. The structural formula of Primaquine phosphate is: Figure 1: Primaquine phosphate structure. Each Primaquine phosphate tablet contains 26.3 mg of primaquine phosphate (equivalent to 15 mg of primaquine base). The dosage is customarily expressed in terms of the base. Inactive Ingredients: Carnauba Wax, Hydroxypropyl Methylcellulose, Lactose, Magnesium Stearate, Microcrystalline Cellulose, Polyethylene Glycol 400, Polysorbate 80, Pregelatinized Starch, Red Ferric Oxide, Talc, Titanium Dioxide.
Indications and Usage
Primaquine phosphate Tablets are indicated for the radical cure (prevention of relapse) of vivax malaria.
Dosage and Administration
Primaquine phosphate Tablets are recommended only for the radical cure of vivax malaria, the prevention of relapse in vivax malaria, or following the termination of chloroquine phosphate suppressive therapy in an area where vivax malaria is endemic. Patients suffering from an attack of vivax malaria or having parasitized red blood cells should receive a course of chloroquine phosphate, which quickly destroys the erythrocytic parasites and terminates the paroxysm. Primaquine phosphate Tablets should be administered concurrently to eradicate the exoerythrocytic parasites in adults at a dosage of 1 tablet (equivalent to 15 mg base) daily for 14 days. Primaquine phosphate Tablets can be taken with or without food. Administration of Primaquine phosphate Tablets with food may reduce the incidence of gastrointestinal symptoms.
Contraindications
Known hypersensitivity reactions to primaquine phosphate, other 8- aminoquinolones, or to any component in Primaquine phosphate Tablets. Severe glucose-6-phosphate dehydrogenase (G6PD) deficiency (see WARNINGS, Hemolytic Anemia ). Pregnant women (see WARNINGS, Pregnancy ). Breastfeeding by a lactating woman when the infant is found to be G6PD deficient or if G6PD status is unknown (see WARNINGS, Nursing Mothers ). Because quinacrine hydrochloride appears to potentiate the toxicity of antimalarial compounds which are structurally related to primaquine, the use of quinacrine in patients receiving Primaquine phosphate Tablets is contraindicated. Similarly, Primaquine phosphate Tablets should not be administered to patients who have received quinacrine recently, as toxicity is increased.
Adverse Reactions
Gastrointestinal: Nausea, vomiting, epigastric distress, abdominal cramps. Hematologic: Leukopenia, hemolytic anemia, decreased hemoglobin, methemoglobinemia. Hemolytic anemia occurs commonly in patients with G6PD deficiency and may be severe or fatal in patients with severe G6PD deficiency (see WARNINGS ). Methemoglobin levels are usually <10%, but methemoglobinemia may be severe in nicotinamide adenine dinucleotide (NADH) methemoglobin reductase deficient individuals or in patients with other risk factors (see PRECAUTIONS ). Leukopenia was observed in patients with rheumatoid arthritis or lupus erythematosus (see PRECAUTIONS ). Cardiac: Cardiac arrhythmia and QT interval prolongation (see PRECAUTIONS , OVERDOSAGE ). Nervous System: Dizziness. Skin and Soft Tissue: Rash, pruritus.
How Supplied
Primaquine phosphate Tablets are supplied as pink, convex, discoid, film-coated tablets of 26.3 mg (= 15 mg base), printed with a "W" and "P97" on one side. Available in bottles of 100 tablets. (NDC 0024-1596-01)
Medication Information
Indications and Usage
Primaquine phosphate Tablets are indicated for the radical cure (prevention of relapse) of vivax malaria.
Dosage and Administration
Primaquine phosphate Tablets are recommended only for the radical cure of vivax malaria, the prevention of relapse in vivax malaria, or following the termination of chloroquine phosphate suppressive therapy in an area where vivax malaria is endemic. Patients suffering from an attack of vivax malaria or having parasitized red blood cells should receive a course of chloroquine phosphate, which quickly destroys the erythrocytic parasites and terminates the paroxysm. Primaquine phosphate Tablets should be administered concurrently to eradicate the exoerythrocytic parasites in adults at a dosage of 1 tablet (equivalent to 15 mg base) daily for 14 days.
Primaquine phosphate Tablets can be taken with or without food. Administration of Primaquine phosphate Tablets with food may reduce the incidence of gastrointestinal symptoms.
Contraindications
Known hypersensitivity reactions to primaquine phosphate, other 8- aminoquinolones, or to any component in Primaquine phosphate Tablets.
Severe glucose-6-phosphate dehydrogenase (G6PD) deficiency (see WARNINGS, Hemolytic Anemia).
Pregnant women (see WARNINGS, Pregnancy).
Breastfeeding by a lactating woman when the infant is found to be G6PD deficient or if G6PD status is unknown (see WARNINGS, Nursing Mothers).
Because quinacrine hydrochloride appears to potentiate the toxicity of antimalarial compounds which are structurally related to primaquine, the use of quinacrine in patients receiving Primaquine phosphate Tablets is contraindicated. Similarly, Primaquine phosphate Tablets should not be administered to patients who have received quinacrine recently, as toxicity is increased.
Adverse Reactions
Gastrointestinal: Nausea, vomiting, epigastric distress, abdominal cramps.
Hematologic: Leukopenia, hemolytic anemia, decreased hemoglobin, methemoglobinemia.
Hemolytic anemia occurs commonly in patients with G6PD deficiency and may be severe or fatal in patients with severe G6PD deficiency (see WARNINGS).
Methemoglobin levels are usually <10%, but methemoglobinemia may be severe in nicotinamide adenine dinucleotide (NADH) methemoglobin reductase deficient individuals or in patients with other risk factors (see PRECAUTIONS).
Leukopenia was observed in patients with rheumatoid arthritis or lupus erythematosus (see PRECAUTIONS).
Cardiac: Cardiac arrhythmia and QT interval prolongation (see PRECAUTIONS, OVERDOSAGE).
Nervous System: Dizziness.
Skin and Soft Tissue: Rash, pruritus.
How Supplied
Primaquine phosphate Tablets are supplied as pink, convex, discoid, film-coated tablets of 26.3 mg (= 15 mg base), printed with a "W" and "P97" on one side.
Available in bottles of 100 tablets. (NDC 0024-1596-01)
Description
Primaquine phosphate is 8-[(4-amino-1-methylbutyl) amino]-6-methoxyquinoline phosphate, a synthetic compound with potent antimalarial activity. The molecular formula of Primaquine phosphate is C15H21N3O∙2H3PO4 and its molecular weight is 455.34. The structural formula of Primaquine phosphate is:
Figure 1: Primaquine phosphate structure.
Each Primaquine phosphate tablet contains 26.3 mg of primaquine phosphate (equivalent to 15 mg of primaquine base). The dosage is customarily expressed in terms of the base.
Inactive Ingredients: Carnauba Wax, Hydroxypropyl Methylcellulose, Lactose, Magnesium Stearate, Microcrystalline Cellulose, Polyethylene Glycol 400, Polysorbate 80, Pregelatinized Starch, Red Ferric Oxide, Talc, Titanium Dioxide.
Section 42229-5
Rx Only
Revised February 2026
Manufactured for:
sanofi-aventis U.S. LLC
Morristown, NJ 07960
A SANOFI COMPANY
©2026 sanofi-aventis U.S. LLC
Section 44425-7
Store at 25° C (77° F); excursions permitted to 15° C – 30° C (59° F – 86° F) [see USP Controlled Room Temperature]
Dispense in tight, light-resistant container as defined in the USP/NF.
Pregnancy
Safe usage of Primaquine phosphate Tablets in pregnancy has not been established. Primaquine is contraindicated in pregnant women. The use of Primaquine phosphate Tablets during pregnancy may cause hemolytic anemia in a G6PD-deficient fetus. Even if a pregnant woman has normal levels of G6PD, the fetus could be G6PD-deficient (see CONTRAINDICATIONS). Animal data show toxicity to reproduction and embryofetal development. (See PRECAUTIONS, Animal Pharmacology and/or Animal Toxicology).
Nonclinical data from studies conducted in bacteria and in animals treated with primaquine show evidence of gene mutations and chromosomal/DNA damage, teratogenicity, and injury to embryos and developing fetuses when primaquine is administered to pregnant animals. Inform patients of the potential for adverse genetic and reproductive effects associated with primaquine treatment (see PRECAUTIONS, Carcinogenesis, Mutagenesis, and Impairment of Fertility, and Animal Pharmacology and/or Animal Toxicology).
Leukopenia
Primaquine may cause leukopenia in patients with established granulocytopenia, such as rheumatoid arthritis and lupus erythematosus. Avoid concurrent administration of bone-marrow depressants.
Discontinue the use of primaquine promptly if there is a sudden decrease in leukocyte count.
Management
Treatment of overdosage consists of institution of appropriate symptomatic and/or supportive therapy. Consider contacting a poison center or a medical toxicologist for overdosage management recommendations.
Quinacrine
Concurrent use of quinacrine (mepacrine) and Primaquine phosphate Tablets are contraindicated. Increased toxicity was seen when quinacrine was used with pamaquine, another 8-aminoquinoline (see CONTRAINDICATIONS).
References
- Shubber EK, Jacobson-Kram D, Williams JR. Comparison of the Ames assay and the induction of sister chromatid exchanges: results with ten pharmaceuticals and five selected agents. Cell Biol Toxicol. 1986;2:379–99.
- Chatterjee T, Muhkopadhyay A, Khan KA, Giri AK. Comparative mutagenic and genotoxic effects of three antimalarial drugs, chloroquine, primaquine and amodiaquine. Mutagenesis. 1998;13:619–24.
- Marss TC. Bright JE, Morris BC. Methemoglobinogenic potential of primaquine and its mutagenicity in the Ames test. Toxicol Lett. 1987;36:281–7.
- Ono T, Norimatsu M, Yoshimura H. Mutagenic evaluation of primaquine, pentaquine and pamaquine in the Salmonella/mammalian microsome assay. Mutat Res. 1994;325:7–10.
- Giovanella F, Ferreira GK, de Prá1 SDT, et al. Effects of primaquine and chloroquine on oxidative stress parameters in rats. An Acad Bras Cienc (Annals of the Brazilian Academy of Sciences). 2015;87:1487–1496.
- Trutter JA, Reno FE, Durloo RS. Teratogenicity studies with a candidate antileishmanial drug. The Toxicologist. 1983;3:65.
- Beveridge E, Caldwell IC, Latter VS, Neal RA, Udall V, Waldron MM. The activity against Trypanosoma cruzi and cutaneous leishmaniasis, and toxicity, of moxipraquine (349C59). Trans R Soc Trop Med Hyg. 1980;74:43–51.
- Bennett JW, Pybus BS, Yadava A, Tosh D, Sousa JC, McCarthy WF, et al. Primaquine failure and cytochrome P-450 2D6 in Plasmodium vivax malaria. N Engl J Med.2013;369(14):1381–82
Resistance
Development of resistance to primaquine phosphate in Plasmodium species has not been well studied.
Contraception
Patients should avoid pregnancy during treatment. Advise sexually active females of childbearing potential to use effective contraception (methods that result in less than 1% pregnancy rates) when using primaquine and for 6 months after stopping treatment). Advise treated males whose partners may become pregnant, to use a condom while on treatment and for 3 months after stopping treatment with primaquine.
Geriatric Use
Clinical studies of Primaquine phosphate Tablets did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Primaquine phosphate Tablets are contraindicated in breastfeeding women when the infant is found to be G6PD deficient or the G6PD status of the infant is unknown (see CONTRAINDICATIONS and WARNINGS, Nursing Mothers).
Nursing Mothers
A breastfed infant with G6PD deficiency is at risk for hemolytic anemia from exposure to primaquine. Infant G6PD status should be checked before breastfeeding begins. Primaquine phosphate Tablets are contraindicated in breastfeeding women when the infant is found to be G6PD deficient or the G6PD status of the infant is unknown (see CONTRAINDICATIONS). Advise the woman with a G6PD-deficient infant or if the G6PD status of the infant is unknown not to breastfeed.
The presence of primaquine and its major metabolite in breast milk and infant plasma were evaluated in a published study of 21 G6PD-normal lactating women and their G6PD-normal infants aged 28 days or older. After repeat administration of a 0.5 mg/kg/day primaquine base dose for 14 days in the lactating women, low concentrations of primaquine and carboxyprimaquine were measured both in breast milk and in infant plasma. The estimated infant ingested dose was found to be less than 1% of a 0.5 mg/kg/day primaquine base dose determined from an observed milk to maternal plasma AUC ratio of 0.34 (range: 0.12 to 0.64) and assuming an infant milk consumption of 150 mL/kg/day. Infant primaquine concentrations in plasma were below measurement thresholds (2.28 ng/mL) in all but 1 infant capillary plasma sample (2.6 ng/mL), and carboxyprimaquine concentrations in plasma were likewise unmeasurable in the majority of infant samples (range, 4.88 ng/mL [measurement threshold] to maximum value 25.8 ng/mL). There is no information on the effects of Primaquine phosphate Tablets on the breastfed infant, or the effects on milk production.
P Gp Substrates
Refer to the Prescribing Information for a P-gp substrate where minimal concentration changes may lead to serious adverse reactions (e.g., digoxin and dabigatran) for the recommended dosage modification and/or monitoring.
In vitro observations suggest that primaquine inhibits the P-gp membrane transporter. Therefore, there is a potential for increased concentrations of drugs that are P-gp substrates when co-administered with Primaquine phosphate Tablets. Increase monitoring for adverse reactions associated with P-gp substrate drugs where minimal concentration changes may lead to serious adverse reactions when concomitantly administered with Primaquine phosphate Tablets.
Clinical Studies
Persons with acute attacks of vivax malaria, provoked by the release of erythrocytic forms of the parasite, respond readily to therapy, particularly to chloroquine phosphate. Primaquine eliminates tissue (exoerythrocytic) infection and prevents relapses in experimentally induced vivax malaria in human volunteers and in persons with naturally occurring infections and is a valuable adjunct to conventional therapy in vivax malaria.
Elderly Patients
There are no pharmacokinetics studies in patients older than 52 years of age.
Hemolytic Anemia
Hemolytic reactions (moderate to severe) may occur in individuals with G6PD deficiency and in individuals with a family or personal history of favism. Areas of high prevalence of G6PD deficiency are Africa, Southern Europe, Mediterranean region, Middle East, South-East Asia, and Oceania. People from these regions have a greater tendency to develop hemolytic anemia due to a congenital deficiency of erythrocytic G6PD while receiving primaquine and related drugs.
Due to the risk of hemolytic anemia in patients with G6PD deficiency, G6PD testing must be performed before using primaquine. Before initiating treatment, obtain baseline hemoglobin and hematocrit. In case of severe anemia, postpone the G6PD test and decision on treatment with primaquine until recovery.
Due to the limitations of G6PD tests, physicians need to be aware of residual risk of hemolysis and adequate medical support and follow-up to manage hemolytic risk should be available. This is of particular importance in individuals with a personal or family history of hemolytic anemia.
Pharmacogenomics
Published clinical reports indicate that primaquine is a CYP2D6 substrate. Experiments in mice indicate primaquine activity likely depends on the formation of CYP2D6 metabolite(s). CYP2D6 has variants that affect CYP2D6 metabolic function. CYP2D6 poor metabolizers are individuals with two nonfunctional alleles (e.g., CYP2D6*5/*5), and as a result have no CYP2D6 activity. CYP2D6 intermediate metabolizers are individuals with a combination of nonfunctional, reduced, or normal function alleles, and as a result have reduced CYP2D6 activity (e.g., CYP2D6*1/*5, CYP2D6*4/*10).
Individuals who are CYP2D6 intermediate or poor metabolizers exhibit a prolonged primaquine half-life and increased primaquine plasma concentrations when compared to individuals who are CYP2D6 normal metabolizers. CYP2D6 metabolizer status may be associated with variability in clinical response to Primaquine phosphate Tablets (see PRECAUTIONS).
Pharmacokinetics
Following single oral dosing, the Cmax and AUC of primaquine increase approximately dose-proportionally over a primaquine base dose range of 15 mg to 45 mg (3 times the approved dose).
The pharmacokinetic parameters and properties of primaquine and carboxyprimaquine (main circulating metabolite not expected to be active) in patients with P. vivax malaria following Oral Administration of Primaquine phosphate Tablets are provided in TABLE 1.
| PK Parameter 15 mg once daily in adult patients (18 years of age and older) with P. vivax malaria, unless otherwise specified
|
Day | Primaquine | Carboxyprimaquine |
|---|---|---|---|
| Cmax=maximum plasma concentration; AUC=area under the plasma concentration-time curve from time zero up to infinity; MAO-A = monoamine oxidase A | |||
| Cmax (ng/mL) | 1 | 50.7 ± 21.2 | 291 ± 52 |
| Cmax (ng/mL) | 14 | 49.7 ± 14.4 | 432 ±112 |
| AUC or AUC0–24 (µg/mL*h) AUC for primaquine, AUC0–24 for carboxyprimaquine
|
1 | 0.48 ± 0.26 | 5.15 ± 1.01 |
| AUC or AUC0–24 (µg/mL*h) | 14 | 0.49 ± 0.19 | 7.24 ± 1.82 |
| Primaquine | |||
| Absorption | |||
| Bioavailability Healthy participants
|
>70 % | ||
| Tmax | 2.3 ± 1.1 hours | ||
| Effect of food on Primaquine Phosphate Tablet (relative to fasting) Values refer to increase in mean systemic exposure with bread and butter: 82% fat, ~28g fat after single dose of 30 mg Primaquine in healthy participants Geometric mean [95% confidence interval] |
↑ 14% [3, 27] (AUC); ↑ 26% [12, 40] (Cmax) | ||
| Distribution | |||
| % Bound to human plasma proteins | 74% (mainly to alpha 1 acid glycoprotein) | ||
| Volume of distribution (V) IV dose administration of [14C]-primaquine in healthy participants
|
243 ± 69 L | ||
| Metabolism | |||
| Metabolic pathways | -Oxidative deamination, MAO-A -Hydroxylation of the quinoline ring, CYP2D6 -Direct conjugations |
||
| Elimination | |||
| Major route of elimination | Metabolism | ||
| Apparent Clearance (CL/F) | 37.6 ± 14.7 L/hr | ||
| Mean terminal half-life (t1/2) The mean terminal half-life of carboxyprimaquine is approximately 22 hours
|
5.6 ± 1.0 hours | ||
| % of dose excreted in urine Oral administration of [14C]-primaquine in healthy participants; no data in feces
, The main circulating metabolite, carboxyprimaquine is subjected to further metabolism and not eliminated through urine
|
64%, (including 3.6% of primaquine, the remnant being metabolites other than carboxyprimaquine) |
Renal Impairment
The efficacy and safety of Primaquine phosphate Tablets after repeated dosing have not been assessed in patients with renal impairment. Because of limited data, there is no specific dosing adjustment. If Primaquine phosphate Tablets are administered to such patients, monitoring of efficacy and for primaquine-related adverse reactions is needed, in particular in patients with severe renal impairment (see CLINICAL PHARMACOLOGY).
Renal Impairment
Single dose pharmacokinetics studies performed in patients with chronic severe (eGFR 15 to 29 mL/min) or end-stage (< 15 mL/min) renal impairment indicate higher primaquine Cmax (up to 1.7-fold higher as compared to healthy subjects) but no evidence of major difference in AUC or t1/2. It is not known whether after repeated dosing there could be an accumulation of metabolites that are mainly excreted by renal route.
Cyp1a2 Substrates
Published clinical and non-clinical reports indicate primaquine inhibits CYP1A2 enzyme activity and thus may lead to increased exposure of CYP1A2 substrate drugs (e.g., duloxetine, alosetron, theophylline and tizanidine) when co-administered with Primaquine phosphate Tablets. Since data are limited, no predictions can be made regarding the extent of the impact on CYP1A2 substrate drug exposures. Increase monitoring for adverse reactions associated with the CYP1A2 substrate drug when concurrently administered with Primaquine phosphate tablets.
Cyp3a4 Substrates
Refer to the Prescribing Information for a CYP3A substrate where minimal concentration changes may lead to serious adverse reactions (e.g. rivaroxaban, calcineurin inhibitors, ergot derivatives, tyrosine kinase inhibitors) for the recommended dosage modification and/or monitoring.
Published clinical reports indicate primaquine may inhibit CYP3A4 enzyme activity and thus may lead to increased exposure of oral CYP3A4 substrate drugs when co-administered with Primaquine phosphate Tablets. Since data are limited, no predictions can be made regarding the extent of the impact on oral CYP3A4 substrate drug exposures. Increase monitoring for adverse reactions associated with CYP3A4 substrate drugs that prolong the QT interval (e.g., pimozide) or where minimal concentration changes may lead to serious adverse reactions when concurrently administered with Primaquine phosphate Tablets.
Methemoglobinemia
Primaquine may cause a transient increase in methemoglobin levels up to 10% in patients without risk factors (see ADVERSE REACTIONS). Methemoglobinemia may be severe in patients who are deficient in nicotinamide adenine dinucleotide (NADH), methemoglobin reductase or treated with methemoglobinemia-inducing drugs such as dapsone or sulfonamide, (see PRECAUTIONS, Drug Interactions). Monitor methemoglobin levels closely in these cases.
Advise all patients to seek immediate medical attention if signs of methemoglobinemia occur such as bluish lips or nails.
Pregnancy Testing
Sexually active females of reproductive potential should have a pregnancy test prior to starting treatment with primaquine.
Hepatic Impairment
Efficacy and safety of Primaquine phosphate Tablets after repeated dosing have not been assessed in patients with hepatic impairment. Primaquine is metabolized in the liver to generate active metabolites, and it is not known if efficacy could be affected in patients with hepatic impairment. Because of limited data, there is no specific dosing adjustment. If Primaquine phosphate Tablets are administered to such patients, monitoring of efficacy and for primaquine-related adverse reactions is needed, in particular in patients with severe hepatic impairment (see CLINICAL PHARMACOLOGY).
Hepatic Impairment
Single dose pharmacokinetics study performed in patients with mild or moderate hepatic impairment indicate that only moderate hepatic dysfunction impacted significantly the PK of primaquine with a 3-fold lower primaquine Cmax in patients with moderate hepatic dysfunction as compared to healthy subjects. The primaquine AUC was not significantly modified.
No data are available after repeated dosing in patients with hepatic impairment. It is not known whether in patients with hepatic impairment, accumulation of primaquine and its metabolites could occur or if there could be an impact on generation of metabolites contributing to pharmacological activity.
Signs and Symptoms
Symptoms of overdosage of primaquine phosphate include abdominal cramps, vomiting, burning epigastric distress, central nervous system disturbances including headache, insomnia, and cardiovascular disturbances, including cardiac arrhythmia and QT interval prolongation, methemoglobinemia (indicated by cyanosis), moderate leukocytosis or leukopenia, granulocytopenia, and anemia. Acute hemolysis may occur with particular severity in G6PD deficient patients.
Mechanism of Action
Primaquine phosphate is an 8-aminoquinoline antimalarial drug. The mechanism of action has not been fully established. The major assumptions are an inhibition of the mitochondrial system of dormant parasites, and an oxidative stress generated through reactive metabolites in infected cells. In humans, primaquine phosphate activity is probably related to hydroxylated metabolites generated intrahepatically by CYP2D6.
Gender and Ethnicity
No gender nor ethnicity effect has been evidenced in studies conducted to date.
Antimicrobial Activity
Primaquine phosphate is active against the dormant liver forms of P.vivax, namely hypnozoites, as well as exoerythrocytic stages of the parasite. Thereby, it prevents the development of the blood (erythrocytic) forms of the parasite which are responsible for relapses in vivax malaria. Primaquine phosphate is also active against gametocytes of Plasmodium falciparum.
Strong Cyp2d6 Inhibitors
Published clinical and non-clinical reports indicate reduced CYP2D6 activity may decrease the formation of active metabolites of primaquine, which may reduce antimalarial efficacy of Primaquine phosphate Tablets (see CLINICAL PHARMACOLOGY, Pharmacogenomics). Where possible, consider alternative medications that are not strong CYP2D6 inhibitors. If concurrent use with Primaquine phosphate Tablets is necessary, increase monitoring for possible relapse.
Concomitant use of an MAO-A inhibitor in patients with reduced or absent CYP2D6 activity (e.g., strong CYP2D6 inhibitor, CYP2D6 intermediate or poor metabolizer) is expected to increase primaquine exposure which may increase the risk of adverse reactions (see CLINICAL PHARMACOLOGY, Pharmacogenomics). Defer initiation of MAO-A inhibitor therapy or consider alternative drug therapy in patients with reduced or absent CYP2D6 activity until primaquine treatment is completed. If concurrent use with Primaquine phosphate Tablets is necessary, increase patient monitoring for potential adverse reactions (see PRECAUTIONS).
Qt Interval Prolonging Drugs
The pharmacodynamic interaction potential to prolong the QT interval of the electrocardiogram between Primaquine phosphate Tablets and other drugs that effect cardiac conduction is unknown. If Primaquine phosphate Tablets are used concomitantly with other drugs that prolong the QT interval, close and frequent electrocardiogram monitoring is advised (see PRECAUTIONS, ADVERSE REACTIONS, and OVERDOSAGE).
Patients With G6pd Deficiency
Primaquine phosphate Tablets are contraindicated in patients with severe G6PD deficiency (see CONTRAINDICATIONS).
In case of mild to moderate G6PD deficiency, a decision to prescribe primaquine must be based on an assessment of the risks and benefits of using primaquine. If primaquine administration is considered, baseline hematocrit and hemoglobin must be checked before treatment and close hematological monitoring (e.g., at day 3 and 8) is required. Adequate medical support to manage hemolytic risk should be available.
Patients Without G6pd Deficiency
In G6PD normal patients it is also advisable to perform routine blood examinations (particularly blood cell counts and hemoglobin determinations) during therapy.
Patients With Unknown G6pd Status
When the G6PD status is unknown and G6PD testing is not available, a decision to prescribe primaquine must be based on an assessment of the risks and benefits of using primaquine. Risk factors for G6PD deficiency or favism must be assessed. Baseline hematocrit and hemoglobin must be checked before treatment and close hematological monitoring (e.g., at day 3 and 8) is required. Adequate medical support to manage hemolytic risk should be available.
Risk of Hemolysis With Other Drugs
Avoid the concurrent administration of hemolytic agents in all patients (see CLINICAL PHARMACOLOGY, Drug Interactions). Warn patients to discontinue the use of Primaquine phosphate Tablets promptly if signs suggestive of hemolytic anemia occur (such as darkening of the urine, pale skin, shortness of breath, dizziness, and fatigue) and to contact their healthcare professional immediately.
Potential Prolongation of Qt Interval
Due to potential for QT interval prolongation, monitor ECG when using primaquine in patients with cardiac disease, long QT syndrome, a history of ventricular arrhythmias, uncorrected hypokalemia and/or hypomagnesemia, or bradycardia (<50 bpm), and during concomitant administration with QT interval prolonging agents (see PRECAUTIONS, Drug Interactions, ADVERSE REACTIONS, and OVERDOSAGE).
Animal Pharmacology And/or Animal Toxicology
Literature data on reproductive toxicology identified embryo-fetal development toxicity. In studies in rats, teratogenic effects on fetus were observed (see WARNINGS, Usage in Pregnancy).
In the first reproductive toxicity study6, primaquine was administered orally to rats between gestation day (GD) 6 and GD15 at dose levels of 10.3, 30.8 and 61.5 mg/kg/day (as base) (representing approximatively 7, 20 and 40 times the human dose [HD] on a body surface area comparison) when considering a human body weight of 60 kg). High dose levels induced death of pregnant females in almost all cases, while lower dose levels caused maternal toxicity. At cesarean section, embryo resorption, a decrease in fetal survival rate and body size, internal abnormalities (including hydrocephalia, heterotaxia), and an increase in skeletal variations were observed at the mid dose-level. There were no fetal abnormalities at the low dose level providing a potential safety margin of at least 7 times the recommended clinical dose.
For the second reproductive toxicity study7, 6 to10 animals per group were used. Dose levels of 0.57, 5.7, 11.4 and 34 mg/kg/day of primaquine (as base) (representing approximatively 0.4, 4, 7 and 22 times the HD on a body surface area comparison) were administered orally to Sprague Dawley rats between GD8 and GD16, or of 57 mg/kg only once on GD13 (representing more than 37 times the HD on a body surface area comparison). A total of 1/7 and 4/6 pregnant females at 34 mg/kg/day and at 57 mg/kg, respectively, died. Primaquine-associated teratogenic malformations (including cleft palate and small chin) were observed in 4/54 fetuses in the 57 mg/kg single-dose group.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity studies have been conducted with primaquine. No fertility studies have been conducted with primaquine. Primaquine is reported in the literature to be a weak genotoxic agent which elicits both gene mutations1, chromosomal damage and DNA strand breaks2. The publications reported positive results in the in vitro reverse gene mutation assays using bacteria (Ames test) 3,4 and in the in vivo studies using rodents (mouse bone marrow cell sister chromatid exchange, mouse bone marrow cell chromosome abnormality, and rat DNA strand breaks in multiple organs) 2, 5. The genotoxicity data obtained in vitro and in rodent models are suggestive of a human risk for genotoxicity with primaquine administration (see WARNINGS, Usage in Pregnancy).
Hemolytic Agents and Methemoglobinemia Inducing Drugs
The concurrent administration of hemolytic agents or methemoglobinemia-inducing drugs and primaquine should be avoided (see PRECAUTIONS). If the concurrent administration cannot be avoided, close blood monitoring is required.
Principal Display Panel 26.3 Mg Tablet Bottle Label
P-425
NDC 0024-1596-01
Rx only
Primaquine phosphate
Tablets, USP
26.3 mg (=15 mg base)
Store at 25° C (77° F); excursions
permitted to 15° C - 30° C
(59° F - 86° F) [see USP Controlled
Room Temperature].
NSN 6505-01-348-2465
100 tablets
sanofi
Effect of Other Drugs On the Pharmacokinetics of Primaquine
In vitro data suggest primaquine is not a substrate of either P-gp or BCRP membrane transporters.
Effect of Primaquine On the Pharmacokinetics of Other Drugs
In vitro data suggest primaquine has the potential to inhibit CYP1A2 enzyme activity, but no or low potential to inhibit MAO-A, MAO-B, or CYP450 isoforms 2A6, 2C8, 2C9, 2C19, 2D6, 3A4 enzymes involved in drug biotransformation.
In vitro data suggest primaquine has the potential to inhibit the P-gp membrane transporter.
Strong Cyp2d6 Inhibitors, Cyp2d6 Poor Or Intermediate Metabolizers, and Treatment Failure
Based on published non-clinical reports, primaquine activity likely depends on the formation of CYP2D6 metabolite(s). Therefore, CYP2D6 genetic variation or drugs that impact CYP2D6 activity may be associated with variability in clinical response to Primaquine phosphate Tablets.
Limited published clinical data reported higher treatment failure rates in patients who were CYP2D6 poor or intermediate metabolizers than in patients who were CYP2D6 normal metabolizers (see CLINICAL PHARMACOLOGY).
When possible, consider alternative medications that are not strong CYP2D6 inhibitors. Increase monitoring for possible relapse in patients with concurrent strong CYP2D6 inhibitor usage or in patients who are CYP2D6 poor or intermediate metabolizers (see PRECAUTIONS, Drug Interactions).
In case of treatment failure, after checking patient's compliance to treatment, reassess use of CYP2D6 inhibitors and assess the patient's CYP2D6 metabolizer status, if feasible. For patients who are CYP2D6 poor or intermediate metabolizers, alternative treatment should be considered.
Structured Label Content
Section 42229-5 (42229-5)
Rx Only
Revised February 2026
Manufactured for:
sanofi-aventis U.S. LLC
Morristown, NJ 07960
A SANOFI COMPANY
©2026 sanofi-aventis U.S. LLC
Section 44425-7 (44425-7)
Store at 25° C (77° F); excursions permitted to 15° C – 30° C (59° F – 86° F) [see USP Controlled Room Temperature]
Dispense in tight, light-resistant container as defined in the USP/NF.
Pregnancy
Safe usage of Primaquine phosphate Tablets in pregnancy has not been established. Primaquine is contraindicated in pregnant women. The use of Primaquine phosphate Tablets during pregnancy may cause hemolytic anemia in a G6PD-deficient fetus. Even if a pregnant woman has normal levels of G6PD, the fetus could be G6PD-deficient (see CONTRAINDICATIONS). Animal data show toxicity to reproduction and embryofetal development. (See PRECAUTIONS, Animal Pharmacology and/or Animal Toxicology).
Nonclinical data from studies conducted in bacteria and in animals treated with primaquine show evidence of gene mutations and chromosomal/DNA damage, teratogenicity, and injury to embryos and developing fetuses when primaquine is administered to pregnant animals. Inform patients of the potential for adverse genetic and reproductive effects associated with primaquine treatment (see PRECAUTIONS, Carcinogenesis, Mutagenesis, and Impairment of Fertility, and Animal Pharmacology and/or Animal Toxicology).
Leukopenia
Primaquine may cause leukopenia in patients with established granulocytopenia, such as rheumatoid arthritis and lupus erythematosus. Avoid concurrent administration of bone-marrow depressants.
Discontinue the use of primaquine promptly if there is a sudden decrease in leukocyte count.
Management
Treatment of overdosage consists of institution of appropriate symptomatic and/or supportive therapy. Consider contacting a poison center or a medical toxicologist for overdosage management recommendations.
Quinacrine
Concurrent use of quinacrine (mepacrine) and Primaquine phosphate Tablets are contraindicated. Increased toxicity was seen when quinacrine was used with pamaquine, another 8-aminoquinoline (see CONTRAINDICATIONS).
References (REFERENCES)
- Shubber EK, Jacobson-Kram D, Williams JR. Comparison of the Ames assay and the induction of sister chromatid exchanges: results with ten pharmaceuticals and five selected agents. Cell Biol Toxicol. 1986;2:379–99.
- Chatterjee T, Muhkopadhyay A, Khan KA, Giri AK. Comparative mutagenic and genotoxic effects of three antimalarial drugs, chloroquine, primaquine and amodiaquine. Mutagenesis. 1998;13:619–24.
- Marss TC. Bright JE, Morris BC. Methemoglobinogenic potential of primaquine and its mutagenicity in the Ames test. Toxicol Lett. 1987;36:281–7.
- Ono T, Norimatsu M, Yoshimura H. Mutagenic evaluation of primaquine, pentaquine and pamaquine in the Salmonella/mammalian microsome assay. Mutat Res. 1994;325:7–10.
- Giovanella F, Ferreira GK, de Prá1 SDT, et al. Effects of primaquine and chloroquine on oxidative stress parameters in rats. An Acad Bras Cienc (Annals of the Brazilian Academy of Sciences). 2015;87:1487–1496.
- Trutter JA, Reno FE, Durloo RS. Teratogenicity studies with a candidate antileishmanial drug. The Toxicologist. 1983;3:65.
- Beveridge E, Caldwell IC, Latter VS, Neal RA, Udall V, Waldron MM. The activity against Trypanosoma cruzi and cutaneous leishmaniasis, and toxicity, of moxipraquine (349C59). Trans R Soc Trop Med Hyg. 1980;74:43–51.
- Bennett JW, Pybus BS, Yadava A, Tosh D, Sousa JC, McCarthy WF, et al. Primaquine failure and cytochrome P-450 2D6 in Plasmodium vivax malaria. N Engl J Med.2013;369(14):1381–82
Resistance
Development of resistance to primaquine phosphate in Plasmodium species has not been well studied.
Description (DESCRIPTION)
Primaquine phosphate is 8-[(4-amino-1-methylbutyl) amino]-6-methoxyquinoline phosphate, a synthetic compound with potent antimalarial activity. The molecular formula of Primaquine phosphate is C15H21N3O∙2H3PO4 and its molecular weight is 455.34. The structural formula of Primaquine phosphate is:
Figure 1: Primaquine phosphate structure.
Each Primaquine phosphate tablet contains 26.3 mg of primaquine phosphate (equivalent to 15 mg of primaquine base). The dosage is customarily expressed in terms of the base.
Inactive Ingredients: Carnauba Wax, Hydroxypropyl Methylcellulose, Lactose, Magnesium Stearate, Microcrystalline Cellulose, Polyethylene Glycol 400, Polysorbate 80, Pregelatinized Starch, Red Ferric Oxide, Talc, Titanium Dioxide.
How Supplied (HOW SUPPLIED)
Primaquine phosphate Tablets are supplied as pink, convex, discoid, film-coated tablets of 26.3 mg (= 15 mg base), printed with a "W" and "P97" on one side.
Available in bottles of 100 tablets. (NDC 0024-1596-01)
Contraception
Patients should avoid pregnancy during treatment. Advise sexually active females of childbearing potential to use effective contraception (methods that result in less than 1% pregnancy rates) when using primaquine and for 6 months after stopping treatment). Advise treated males whose partners may become pregnant, to use a condom while on treatment and for 3 months after stopping treatment with primaquine.
Geriatric Use
Clinical studies of Primaquine phosphate Tablets did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Primaquine phosphate Tablets are contraindicated in breastfeeding women when the infant is found to be G6PD deficient or the G6PD status of the infant is unknown (see CONTRAINDICATIONS and WARNINGS, Nursing Mothers).
Nursing Mothers
A breastfed infant with G6PD deficiency is at risk for hemolytic anemia from exposure to primaquine. Infant G6PD status should be checked before breastfeeding begins. Primaquine phosphate Tablets are contraindicated in breastfeeding women when the infant is found to be G6PD deficient or the G6PD status of the infant is unknown (see CONTRAINDICATIONS). Advise the woman with a G6PD-deficient infant or if the G6PD status of the infant is unknown not to breastfeed.
The presence of primaquine and its major metabolite in breast milk and infant plasma were evaluated in a published study of 21 G6PD-normal lactating women and their G6PD-normal infants aged 28 days or older. After repeat administration of a 0.5 mg/kg/day primaquine base dose for 14 days in the lactating women, low concentrations of primaquine and carboxyprimaquine were measured both in breast milk and in infant plasma. The estimated infant ingested dose was found to be less than 1% of a 0.5 mg/kg/day primaquine base dose determined from an observed milk to maternal plasma AUC ratio of 0.34 (range: 0.12 to 0.64) and assuming an infant milk consumption of 150 mL/kg/day. Infant primaquine concentrations in plasma were below measurement thresholds (2.28 ng/mL) in all but 1 infant capillary plasma sample (2.6 ng/mL), and carboxyprimaquine concentrations in plasma were likewise unmeasurable in the majority of infant samples (range, 4.88 ng/mL [measurement threshold] to maximum value 25.8 ng/mL). There is no information on the effects of Primaquine phosphate Tablets on the breastfed infant, or the effects on milk production.
P Gp Substrates (P-gp Substrates)
Refer to the Prescribing Information for a P-gp substrate where minimal concentration changes may lead to serious adverse reactions (e.g., digoxin and dabigatran) for the recommended dosage modification and/or monitoring.
In vitro observations suggest that primaquine inhibits the P-gp membrane transporter. Therefore, there is a potential for increased concentrations of drugs that are P-gp substrates when co-administered with Primaquine phosphate Tablets. Increase monitoring for adverse reactions associated with P-gp substrate drugs where minimal concentration changes may lead to serious adverse reactions when concomitantly administered with Primaquine phosphate Tablets.
Clinical Studies (CLINICAL STUDIES)
Persons with acute attacks of vivax malaria, provoked by the release of erythrocytic forms of the parasite, respond readily to therapy, particularly to chloroquine phosphate. Primaquine eliminates tissue (exoerythrocytic) infection and prevents relapses in experimentally induced vivax malaria in human volunteers and in persons with naturally occurring infections and is a valuable adjunct to conventional therapy in vivax malaria.
Elderly Patients (Elderly patients)
There are no pharmacokinetics studies in patients older than 52 years of age.
Hemolytic Anemia
Hemolytic reactions (moderate to severe) may occur in individuals with G6PD deficiency and in individuals with a family or personal history of favism. Areas of high prevalence of G6PD deficiency are Africa, Southern Europe, Mediterranean region, Middle East, South-East Asia, and Oceania. People from these regions have a greater tendency to develop hemolytic anemia due to a congenital deficiency of erythrocytic G6PD while receiving primaquine and related drugs.
Due to the risk of hemolytic anemia in patients with G6PD deficiency, G6PD testing must be performed before using primaquine. Before initiating treatment, obtain baseline hemoglobin and hematocrit. In case of severe anemia, postpone the G6PD test and decision on treatment with primaquine until recovery.
Due to the limitations of G6PD tests, physicians need to be aware of residual risk of hemolysis and adequate medical support and follow-up to manage hemolytic risk should be available. This is of particular importance in individuals with a personal or family history of hemolytic anemia.
Pharmacogenomics
Published clinical reports indicate that primaquine is a CYP2D6 substrate. Experiments in mice indicate primaquine activity likely depends on the formation of CYP2D6 metabolite(s). CYP2D6 has variants that affect CYP2D6 metabolic function. CYP2D6 poor metabolizers are individuals with two nonfunctional alleles (e.g., CYP2D6*5/*5), and as a result have no CYP2D6 activity. CYP2D6 intermediate metabolizers are individuals with a combination of nonfunctional, reduced, or normal function alleles, and as a result have reduced CYP2D6 activity (e.g., CYP2D6*1/*5, CYP2D6*4/*10).
Individuals who are CYP2D6 intermediate or poor metabolizers exhibit a prolonged primaquine half-life and increased primaquine plasma concentrations when compared to individuals who are CYP2D6 normal metabolizers. CYP2D6 metabolizer status may be associated with variability in clinical response to Primaquine phosphate Tablets (see PRECAUTIONS).
Pharmacokinetics
Following single oral dosing, the Cmax and AUC of primaquine increase approximately dose-proportionally over a primaquine base dose range of 15 mg to 45 mg (3 times the approved dose).
The pharmacokinetic parameters and properties of primaquine and carboxyprimaquine (main circulating metabolite not expected to be active) in patients with P. vivax malaria following Oral Administration of Primaquine phosphate Tablets are provided in TABLE 1.
| PK Parameter 15 mg once daily in adult patients (18 years of age and older) with P. vivax malaria, unless otherwise specified
|
Day | Primaquine | Carboxyprimaquine |
|---|---|---|---|
| Cmax=maximum plasma concentration; AUC=area under the plasma concentration-time curve from time zero up to infinity; MAO-A = monoamine oxidase A | |||
| Cmax (ng/mL) | 1 | 50.7 ± 21.2 | 291 ± 52 |
| Cmax (ng/mL) | 14 | 49.7 ± 14.4 | 432 ±112 |
| AUC or AUC0–24 (µg/mL*h) AUC for primaquine, AUC0–24 for carboxyprimaquine
|
1 | 0.48 ± 0.26 | 5.15 ± 1.01 |
| AUC or AUC0–24 (µg/mL*h) | 14 | 0.49 ± 0.19 | 7.24 ± 1.82 |
| Primaquine | |||
| Absorption | |||
| Bioavailability Healthy participants
|
>70 % | ||
| Tmax | 2.3 ± 1.1 hours | ||
| Effect of food on Primaquine Phosphate Tablet (relative to fasting) Values refer to increase in mean systemic exposure with bread and butter: 82% fat, ~28g fat after single dose of 30 mg Primaquine in healthy participants Geometric mean [95% confidence interval] |
↑ 14% [3, 27] (AUC); ↑ 26% [12, 40] (Cmax) | ||
| Distribution | |||
| % Bound to human plasma proteins | 74% (mainly to alpha 1 acid glycoprotein) | ||
| Volume of distribution (V) IV dose administration of [14C]-primaquine in healthy participants
|
243 ± 69 L | ||
| Metabolism | |||
| Metabolic pathways | -Oxidative deamination, MAO-A -Hydroxylation of the quinoline ring, CYP2D6 -Direct conjugations |
||
| Elimination | |||
| Major route of elimination | Metabolism | ||
| Apparent Clearance (CL/F) | 37.6 ± 14.7 L/hr | ||
| Mean terminal half-life (t1/2) The mean terminal half-life of carboxyprimaquine is approximately 22 hours
|
5.6 ± 1.0 hours | ||
| % of dose excreted in urine Oral administration of [14C]-primaquine in healthy participants; no data in feces
, The main circulating metabolite, carboxyprimaquine is subjected to further metabolism and not eliminated through urine
|
64%, (including 3.6% of primaquine, the remnant being metabolites other than carboxyprimaquine) |
Renal Impairment
The efficacy and safety of Primaquine phosphate Tablets after repeated dosing have not been assessed in patients with renal impairment. Because of limited data, there is no specific dosing adjustment. If Primaquine phosphate Tablets are administered to such patients, monitoring of efficacy and for primaquine-related adverse reactions is needed, in particular in patients with severe renal impairment (see CLINICAL PHARMACOLOGY).
Renal Impairment (Renal impairment)
Single dose pharmacokinetics studies performed in patients with chronic severe (eGFR 15 to 29 mL/min) or end-stage (< 15 mL/min) renal impairment indicate higher primaquine Cmax (up to 1.7-fold higher as compared to healthy subjects) but no evidence of major difference in AUC or t1/2. It is not known whether after repeated dosing there could be an accumulation of metabolites that are mainly excreted by renal route.
Adverse Reactions (ADVERSE REACTIONS)
Gastrointestinal: Nausea, vomiting, epigastric distress, abdominal cramps.
Hematologic: Leukopenia, hemolytic anemia, decreased hemoglobin, methemoglobinemia.
Hemolytic anemia occurs commonly in patients with G6PD deficiency and may be severe or fatal in patients with severe G6PD deficiency (see WARNINGS).
Methemoglobin levels are usually <10%, but methemoglobinemia may be severe in nicotinamide adenine dinucleotide (NADH) methemoglobin reductase deficient individuals or in patients with other risk factors (see PRECAUTIONS).
Leukopenia was observed in patients with rheumatoid arthritis or lupus erythematosus (see PRECAUTIONS).
Cardiac: Cardiac arrhythmia and QT interval prolongation (see PRECAUTIONS, OVERDOSAGE).
Nervous System: Dizziness.
Skin and Soft Tissue: Rash, pruritus.
Contraindications (CONTRAINDICATIONS)
Known hypersensitivity reactions to primaquine phosphate, other 8- aminoquinolones, or to any component in Primaquine phosphate Tablets.
Severe glucose-6-phosphate dehydrogenase (G6PD) deficiency (see WARNINGS, Hemolytic Anemia).
Pregnant women (see WARNINGS, Pregnancy).
Breastfeeding by a lactating woman when the infant is found to be G6PD deficient or if G6PD status is unknown (see WARNINGS, Nursing Mothers).
Because quinacrine hydrochloride appears to potentiate the toxicity of antimalarial compounds which are structurally related to primaquine, the use of quinacrine in patients receiving Primaquine phosphate Tablets is contraindicated. Similarly, Primaquine phosphate Tablets should not be administered to patients who have received quinacrine recently, as toxicity is increased.
Cyp1a2 Substrates (CYP1A2 Substrates)
Published clinical and non-clinical reports indicate primaquine inhibits CYP1A2 enzyme activity and thus may lead to increased exposure of CYP1A2 substrate drugs (e.g., duloxetine, alosetron, theophylline and tizanidine) when co-administered with Primaquine phosphate Tablets. Since data are limited, no predictions can be made regarding the extent of the impact on CYP1A2 substrate drug exposures. Increase monitoring for adverse reactions associated with the CYP1A2 substrate drug when concurrently administered with Primaquine phosphate tablets.
Cyp3a4 Substrates (CYP3A4 Substrates)
Refer to the Prescribing Information for a CYP3A substrate where minimal concentration changes may lead to serious adverse reactions (e.g. rivaroxaban, calcineurin inhibitors, ergot derivatives, tyrosine kinase inhibitors) for the recommended dosage modification and/or monitoring.
Published clinical reports indicate primaquine may inhibit CYP3A4 enzyme activity and thus may lead to increased exposure of oral CYP3A4 substrate drugs when co-administered with Primaquine phosphate Tablets. Since data are limited, no predictions can be made regarding the extent of the impact on oral CYP3A4 substrate drug exposures. Increase monitoring for adverse reactions associated with CYP3A4 substrate drugs that prolong the QT interval (e.g., pimozide) or where minimal concentration changes may lead to serious adverse reactions when concurrently administered with Primaquine phosphate Tablets.
Methemoglobinemia
Primaquine may cause a transient increase in methemoglobin levels up to 10% in patients without risk factors (see ADVERSE REACTIONS). Methemoglobinemia may be severe in patients who are deficient in nicotinamide adenine dinucleotide (NADH), methemoglobin reductase or treated with methemoglobinemia-inducing drugs such as dapsone or sulfonamide, (see PRECAUTIONS, Drug Interactions). Monitor methemoglobin levels closely in these cases.
Advise all patients to seek immediate medical attention if signs of methemoglobinemia occur such as bluish lips or nails.
Pregnancy Testing
Sexually active females of reproductive potential should have a pregnancy test prior to starting treatment with primaquine.
Hepatic Impairment
Efficacy and safety of Primaquine phosphate Tablets after repeated dosing have not been assessed in patients with hepatic impairment. Primaquine is metabolized in the liver to generate active metabolites, and it is not known if efficacy could be affected in patients with hepatic impairment. Because of limited data, there is no specific dosing adjustment. If Primaquine phosphate Tablets are administered to such patients, monitoring of efficacy and for primaquine-related adverse reactions is needed, in particular in patients with severe hepatic impairment (see CLINICAL PHARMACOLOGY).
Hepatic Impairment (Hepatic impairment)
Single dose pharmacokinetics study performed in patients with mild or moderate hepatic impairment indicate that only moderate hepatic dysfunction impacted significantly the PK of primaquine with a 3-fold lower primaquine Cmax in patients with moderate hepatic dysfunction as compared to healthy subjects. The primaquine AUC was not significantly modified.
No data are available after repeated dosing in patients with hepatic impairment. It is not known whether in patients with hepatic impairment, accumulation of primaquine and its metabolites could occur or if there could be an impact on generation of metabolites contributing to pharmacological activity.
Signs and Symptoms (Signs and symptoms)
Symptoms of overdosage of primaquine phosphate include abdominal cramps, vomiting, burning epigastric distress, central nervous system disturbances including headache, insomnia, and cardiovascular disturbances, including cardiac arrhythmia and QT interval prolongation, methemoglobinemia (indicated by cyanosis), moderate leukocytosis or leukopenia, granulocytopenia, and anemia. Acute hemolysis may occur with particular severity in G6PD deficient patients.
Mechanism of Action
Primaquine phosphate is an 8-aminoquinoline antimalarial drug. The mechanism of action has not been fully established. The major assumptions are an inhibition of the mitochondrial system of dormant parasites, and an oxidative stress generated through reactive metabolites in infected cells. In humans, primaquine phosphate activity is probably related to hydroxylated metabolites generated intrahepatically by CYP2D6.
Gender and Ethnicity (Gender and ethnicity)
No gender nor ethnicity effect has been evidenced in studies conducted to date.
Indications and Usage (INDICATIONS AND USAGE)
Primaquine phosphate Tablets are indicated for the radical cure (prevention of relapse) of vivax malaria.
Antimicrobial Activity
Primaquine phosphate is active against the dormant liver forms of P.vivax, namely hypnozoites, as well as exoerythrocytic stages of the parasite. Thereby, it prevents the development of the blood (erythrocytic) forms of the parasite which are responsible for relapses in vivax malaria. Primaquine phosphate is also active against gametocytes of Plasmodium falciparum.
Strong Cyp2d6 Inhibitors (Strong CYP2D6 Inhibitors)
Published clinical and non-clinical reports indicate reduced CYP2D6 activity may decrease the formation of active metabolites of primaquine, which may reduce antimalarial efficacy of Primaquine phosphate Tablets (see CLINICAL PHARMACOLOGY, Pharmacogenomics). Where possible, consider alternative medications that are not strong CYP2D6 inhibitors. If concurrent use with Primaquine phosphate Tablets is necessary, increase monitoring for possible relapse.
Concomitant use of an MAO-A inhibitor in patients with reduced or absent CYP2D6 activity (e.g., strong CYP2D6 inhibitor, CYP2D6 intermediate or poor metabolizer) is expected to increase primaquine exposure which may increase the risk of adverse reactions (see CLINICAL PHARMACOLOGY, Pharmacogenomics). Defer initiation of MAO-A inhibitor therapy or consider alternative drug therapy in patients with reduced or absent CYP2D6 activity until primaquine treatment is completed. If concurrent use with Primaquine phosphate Tablets is necessary, increase patient monitoring for potential adverse reactions (see PRECAUTIONS).
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Primaquine phosphate Tablets are recommended only for the radical cure of vivax malaria, the prevention of relapse in vivax malaria, or following the termination of chloroquine phosphate suppressive therapy in an area where vivax malaria is endemic. Patients suffering from an attack of vivax malaria or having parasitized red blood cells should receive a course of chloroquine phosphate, which quickly destroys the erythrocytic parasites and terminates the paroxysm. Primaquine phosphate Tablets should be administered concurrently to eradicate the exoerythrocytic parasites in adults at a dosage of 1 tablet (equivalent to 15 mg base) daily for 14 days.
Primaquine phosphate Tablets can be taken with or without food. Administration of Primaquine phosphate Tablets with food may reduce the incidence of gastrointestinal symptoms.
Qt Interval Prolonging Drugs (QT Interval Prolonging Drugs)
The pharmacodynamic interaction potential to prolong the QT interval of the electrocardiogram between Primaquine phosphate Tablets and other drugs that effect cardiac conduction is unknown. If Primaquine phosphate Tablets are used concomitantly with other drugs that prolong the QT interval, close and frequent electrocardiogram monitoring is advised (see PRECAUTIONS, ADVERSE REACTIONS, and OVERDOSAGE).
Patients With G6pd Deficiency (Patients with G6PD Deficiency)
Primaquine phosphate Tablets are contraindicated in patients with severe G6PD deficiency (see CONTRAINDICATIONS).
In case of mild to moderate G6PD deficiency, a decision to prescribe primaquine must be based on an assessment of the risks and benefits of using primaquine. If primaquine administration is considered, baseline hematocrit and hemoglobin must be checked before treatment and close hematological monitoring (e.g., at day 3 and 8) is required. Adequate medical support to manage hemolytic risk should be available.
Patients Without G6pd Deficiency (Patients without G6PD Deficiency)
In G6PD normal patients it is also advisable to perform routine blood examinations (particularly blood cell counts and hemoglobin determinations) during therapy.
Patients With Unknown G6pd Status (Patients with Unknown G6PD Status)
When the G6PD status is unknown and G6PD testing is not available, a decision to prescribe primaquine must be based on an assessment of the risks and benefits of using primaquine. Risk factors for G6PD deficiency or favism must be assessed. Baseline hematocrit and hemoglobin must be checked before treatment and close hematological monitoring (e.g., at day 3 and 8) is required. Adequate medical support to manage hemolytic risk should be available.
Risk of Hemolysis With Other Drugs (Risk of Hemolysis with Other Drugs)
Avoid the concurrent administration of hemolytic agents in all patients (see CLINICAL PHARMACOLOGY, Drug Interactions). Warn patients to discontinue the use of Primaquine phosphate Tablets promptly if signs suggestive of hemolytic anemia occur (such as darkening of the urine, pale skin, shortness of breath, dizziness, and fatigue) and to contact their healthcare professional immediately.
Potential Prolongation of Qt Interval (Potential Prolongation of QT Interval)
Due to potential for QT interval prolongation, monitor ECG when using primaquine in patients with cardiac disease, long QT syndrome, a history of ventricular arrhythmias, uncorrected hypokalemia and/or hypomagnesemia, or bradycardia (<50 bpm), and during concomitant administration with QT interval prolonging agents (see PRECAUTIONS, Drug Interactions, ADVERSE REACTIONS, and OVERDOSAGE).
Animal Pharmacology And/or Animal Toxicology (Animal Pharmacology and/or Animal Toxicology)
Literature data on reproductive toxicology identified embryo-fetal development toxicity. In studies in rats, teratogenic effects on fetus were observed (see WARNINGS, Usage in Pregnancy).
In the first reproductive toxicity study6, primaquine was administered orally to rats between gestation day (GD) 6 and GD15 at dose levels of 10.3, 30.8 and 61.5 mg/kg/day (as base) (representing approximatively 7, 20 and 40 times the human dose [HD] on a body surface area comparison) when considering a human body weight of 60 kg). High dose levels induced death of pregnant females in almost all cases, while lower dose levels caused maternal toxicity. At cesarean section, embryo resorption, a decrease in fetal survival rate and body size, internal abnormalities (including hydrocephalia, heterotaxia), and an increase in skeletal variations were observed at the mid dose-level. There were no fetal abnormalities at the low dose level providing a potential safety margin of at least 7 times the recommended clinical dose.
For the second reproductive toxicity study7, 6 to10 animals per group were used. Dose levels of 0.57, 5.7, 11.4 and 34 mg/kg/day of primaquine (as base) (representing approximatively 0.4, 4, 7 and 22 times the HD on a body surface area comparison) were administered orally to Sprague Dawley rats between GD8 and GD16, or of 57 mg/kg only once on GD13 (representing more than 37 times the HD on a body surface area comparison). A total of 1/7 and 4/6 pregnant females at 34 mg/kg/day and at 57 mg/kg, respectively, died. Primaquine-associated teratogenic malformations (including cleft palate and small chin) were observed in 4/54 fetuses in the 57 mg/kg single-dose group.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity studies have been conducted with primaquine. No fertility studies have been conducted with primaquine. Primaquine is reported in the literature to be a weak genotoxic agent which elicits both gene mutations1, chromosomal damage and DNA strand breaks2. The publications reported positive results in the in vitro reverse gene mutation assays using bacteria (Ames test) 3,4 and in the in vivo studies using rodents (mouse bone marrow cell sister chromatid exchange, mouse bone marrow cell chromosome abnormality, and rat DNA strand breaks in multiple organs) 2, 5. The genotoxicity data obtained in vitro and in rodent models are suggestive of a human risk for genotoxicity with primaquine administration (see WARNINGS, Usage in Pregnancy).
Hemolytic Agents and Methemoglobinemia Inducing Drugs (Hemolytic Agents and Methemoglobinemia-Inducing Drugs)
The concurrent administration of hemolytic agents or methemoglobinemia-inducing drugs and primaquine should be avoided (see PRECAUTIONS). If the concurrent administration cannot be avoided, close blood monitoring is required.
Principal Display Panel 26.3 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 26.3 mg Tablet Bottle Label)
P-425
NDC 0024-1596-01
Rx only
Primaquine phosphate
Tablets, USP
26.3 mg (=15 mg base)
Store at 25° C (77° F); excursions
permitted to 15° C - 30° C
(59° F - 86° F) [see USP Controlled
Room Temperature].
NSN 6505-01-348-2465
100 tablets
sanofi
Effect of Other Drugs On the Pharmacokinetics of Primaquine (Effect of other Drugs on the Pharmacokinetics of primaquine)
In vitro data suggest primaquine is not a substrate of either P-gp or BCRP membrane transporters.
Effect of Primaquine On the Pharmacokinetics of Other Drugs (Effect of primaquine on the Pharmacokinetics of other drugs)
In vitro data suggest primaquine has the potential to inhibit CYP1A2 enzyme activity, but no or low potential to inhibit MAO-A, MAO-B, or CYP450 isoforms 2A6, 2C8, 2C9, 2C19, 2D6, 3A4 enzymes involved in drug biotransformation.
In vitro data suggest primaquine has the potential to inhibit the P-gp membrane transporter.
Strong Cyp2d6 Inhibitors, Cyp2d6 Poor Or Intermediate Metabolizers, and Treatment Failure (Strong CYP2D6 Inhibitors, CYP2D6 Poor or Intermediate Metabolizers, and Treatment Failure)
Based on published non-clinical reports, primaquine activity likely depends on the formation of CYP2D6 metabolite(s). Therefore, CYP2D6 genetic variation or drugs that impact CYP2D6 activity may be associated with variability in clinical response to Primaquine phosphate Tablets.
Limited published clinical data reported higher treatment failure rates in patients who were CYP2D6 poor or intermediate metabolizers than in patients who were CYP2D6 normal metabolizers (see CLINICAL PHARMACOLOGY).
When possible, consider alternative medications that are not strong CYP2D6 inhibitors. Increase monitoring for possible relapse in patients with concurrent strong CYP2D6 inhibitor usage or in patients who are CYP2D6 poor or intermediate metabolizers (see PRECAUTIONS, Drug Interactions).
In case of treatment failure, after checking patient's compliance to treatment, reassess use of CYP2D6 inhibitors and assess the patient's CYP2D6 metabolizer status, if feasible. For patients who are CYP2D6 poor or intermediate metabolizers, alternative treatment should be considered.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:52.935557 · Updated: 2026-03-14T22:50:42.037952