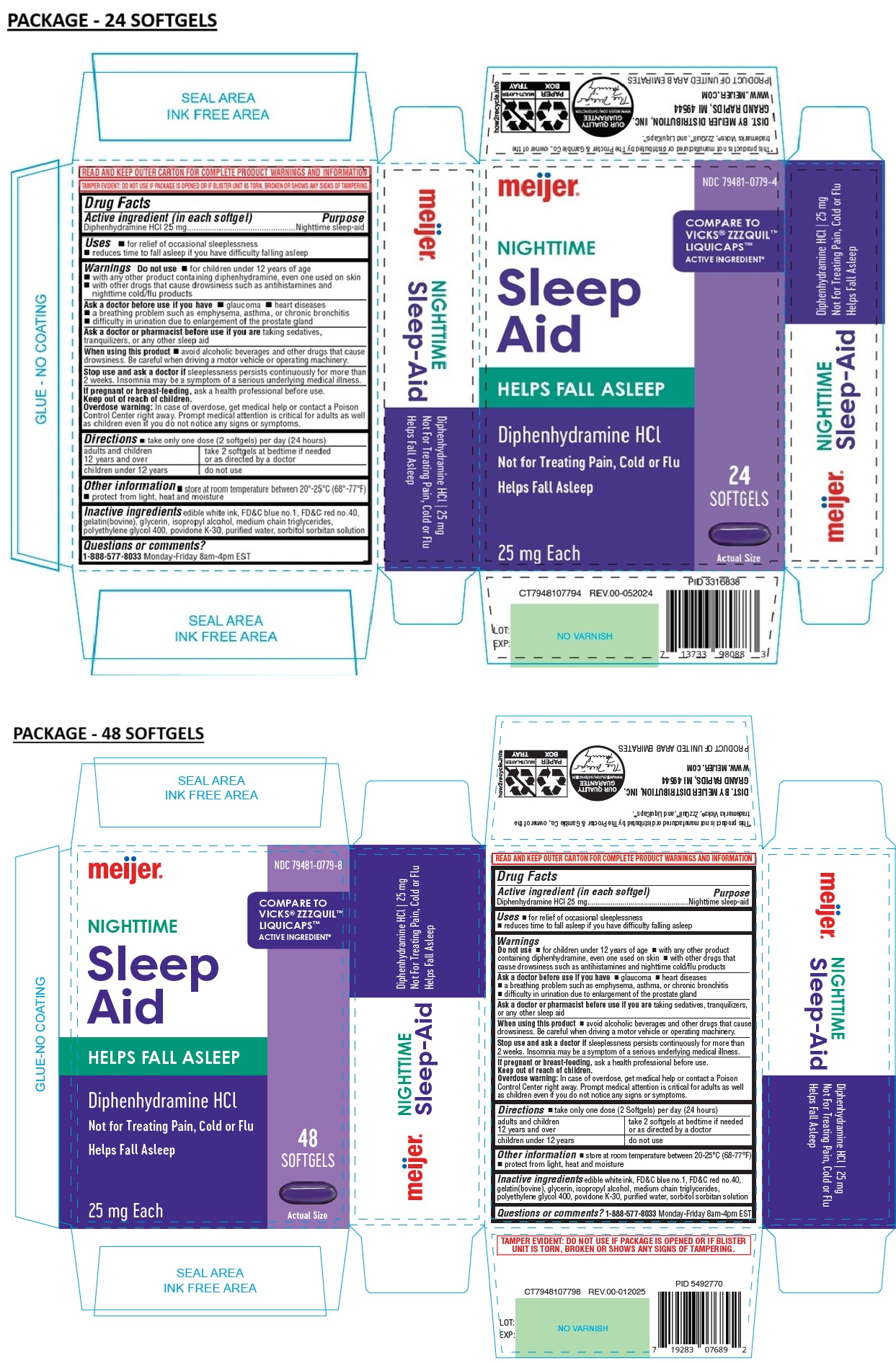
Meijer Nighttime Sleep Aid
1bede4cd-bf55-8b84-e063-6394a90aa5c9
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Diphenhydramine HCl 25 mg
Purpose
Nighttime sleep-aid
Medication Information
Purpose
Nighttime sleep-aid
Description
Diphenhydramine HCl 25 mg
Uses
• for relief of occasional sleeplessness
• reduces time to fall asleep if you have difficulty falling asleep
Section 42229-5
COMPARE TO VICKS® ZZZQUIL™ LIQUICAPS™ ACTIVE INGREDIENT*
HELPS FALL ASLEEP
Not for Treating Pain, Cold or Flu
| READ AND KEEP OUTER CARTON FOR COMPLETE PRODUCT WARNINGS AND INFORMATION |
| TAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED OR IF BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING. |
*This product is not manufactured or distributed by The Procter & Gamble Co., owner of the trademarks Vicks®, ZzzQuil™, and LiquiCaps™.
DIST. BY MEIJER DISTRIBUTION, INC.
GRAND RAPIDS, MI 49544
WWW.MEIJER.COM
PRODUCT OF UNITED ARAB EMIRATES
Section 50565-1
Keep out of reach of children.
Overdose warning: In case of overdose, get medical help or contact a Poison Control Center right away. Prompt medical attention is critical for adults as well as children even if you do not notice any signs or symptoms.
Warnings
Do not use • for children under 12 years of age
• with any other product containing diphenhydramine, even one used on skin
• with other drugs that cause drowsiness such as antihistamines and nighttime cold/flu products
Ask a doctor before use if you have • glaucoma • heart diseases
• a breathing problem such as emphysema, asthma, or chronic bronchitis
• difficulty in urination due to enlargement of the prostate gland
Ask a doctor or pharmacist before use if you are taking sedatives, tranquilizers, or any other sleep aid
When using this product • avoid alcoholic beverages and other drugs that cause drowsiness. Be careful when driving a motor vehicle or operating machinery.
Stop use and ask a doctor if sleeplessness persists continuously for more than 2 weeks. Insomnia may be a symptom of a serious underlying medical illness.
If pregnant or breast-feeding, ask a health professional before use.
Packaging
Directions
• take only one dose (2 softgels) per day (24 hours)
| adults and children 12 years and over | take 2 softgels at bedtime if needed or as directed by a doctor |
| children under 12 years | do not use |
Drug Facts
Other Information
• store at room temperature between 20°-25°C (68°-77°F)
• protect from light, heat and moisture
Inactive Ingredients
edible white ink, FD&C blue no.1, FD&C red no.40, gelatin(bovine), glycerin, isopropyl alcohol, medium chain triglycerides, polyethylene glycol 400, povidone K-30, purified water, sorbitol sorbitan solution
Questions Or Comments?
1-888-577-8033 Monday-Friday 8am-4pm EST
Active Ingredient (in Each Softgel)
Diphenhydramine HCl 25 mg
Structured Label Content
Uses
• for relief of occasional sleeplessness
• reduces time to fall asleep if you have difficulty falling asleep
Section 42229-5 (42229-5)
COMPARE TO VICKS® ZZZQUIL™ LIQUICAPS™ ACTIVE INGREDIENT*
HELPS FALL ASLEEP
Not for Treating Pain, Cold or Flu
| READ AND KEEP OUTER CARTON FOR COMPLETE PRODUCT WARNINGS AND INFORMATION |
| TAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED OR IF BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING. |
*This product is not manufactured or distributed by The Procter & Gamble Co., owner of the trademarks Vicks®, ZzzQuil™, and LiquiCaps™.
DIST. BY MEIJER DISTRIBUTION, INC.
GRAND RAPIDS, MI 49544
WWW.MEIJER.COM
PRODUCT OF UNITED ARAB EMIRATES
Section 50565-1 (50565-1)
Keep out of reach of children.
Overdose warning: In case of overdose, get medical help or contact a Poison Control Center right away. Prompt medical attention is critical for adults as well as children even if you do not notice any signs or symptoms.
Purpose
Nighttime sleep-aid
Warnings
Do not use • for children under 12 years of age
• with any other product containing diphenhydramine, even one used on skin
• with other drugs that cause drowsiness such as antihistamines and nighttime cold/flu products
Ask a doctor before use if you have • glaucoma • heart diseases
• a breathing problem such as emphysema, asthma, or chronic bronchitis
• difficulty in urination due to enlargement of the prostate gland
Ask a doctor or pharmacist before use if you are taking sedatives, tranquilizers, or any other sleep aid
When using this product • avoid alcoholic beverages and other drugs that cause drowsiness. Be careful when driving a motor vehicle or operating machinery.
Stop use and ask a doctor if sleeplessness persists continuously for more than 2 weeks. Insomnia may be a symptom of a serious underlying medical illness.
If pregnant or breast-feeding, ask a health professional before use.
Packaging
Directions
• take only one dose (2 softgels) per day (24 hours)
| adults and children 12 years and over | take 2 softgels at bedtime if needed or as directed by a doctor |
| children under 12 years | do not use |
Drug Facts
Other Information (Other information)
• store at room temperature between 20°-25°C (68°-77°F)
• protect from light, heat and moisture
Inactive Ingredients (Inactive ingredients)
edible white ink, FD&C blue no.1, FD&C red no.40, gelatin(bovine), glycerin, isopropyl alcohol, medium chain triglycerides, polyethylene glycol 400, povidone K-30, purified water, sorbitol sorbitan solution
Questions Or Comments? (Questions or comments?)
1-888-577-8033 Monday-Friday 8am-4pm EST
Active Ingredient (in Each Softgel) (Active ingredient (in each softgel))
Diphenhydramine HCl 25 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:42.421626 · Updated: 2026-03-14T23:07:53.934759