Loma Lux Psoriasis
1b88d668-388b-43b0-b968-eda35d536191
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
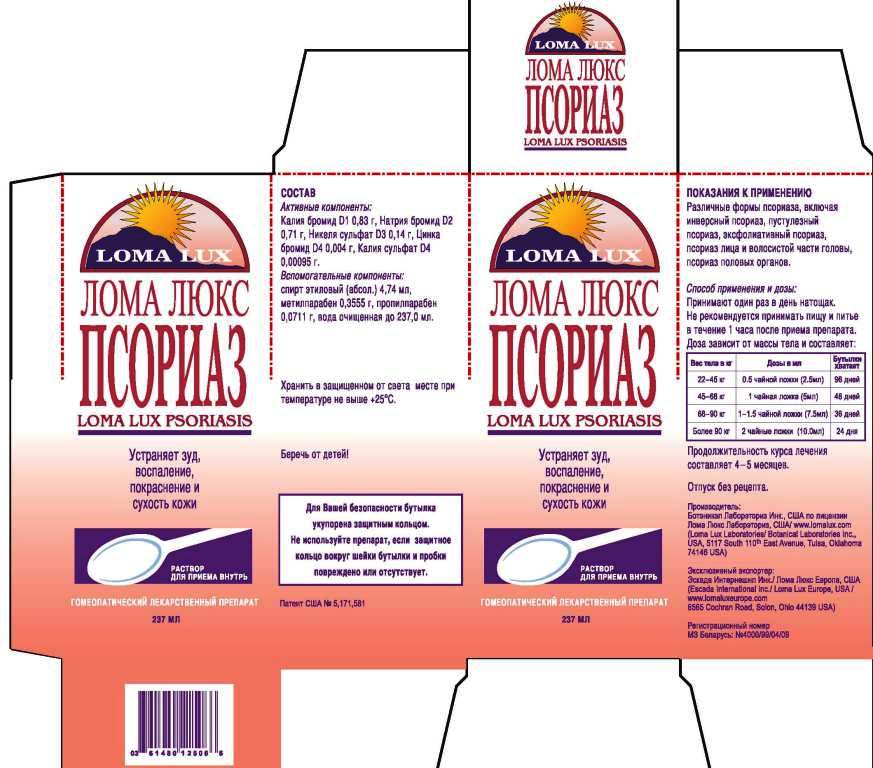
ACTIVE INGREDIENTS: Kali Bromatum 1X, Natrum bromatum 2X, Niccolum sulphuricum 3X, Kali sulphuricum 4X, Zincum bromatum 4X.
Medication Information
Warnings and Precautions
WARNINGS: Do not use if pregnant or nursing.
If allergic to nickel or costume jewelry, use only under the advice and supervision of a physician.
If symptoms worsen, contact a physician.
Keep out or the reach of children.
CAUTION: Use only as directed.
Do not give to children under ten years old or use in the presence of kidney disease.
If skin rash appears or if nervous symptoms persist, recur frequently or are unusual, discontinue use and consult a physician.
Indications and Usage
For relief from the scaling, itching and redness of Psoriasis and Seborrhea.
Dosage and Administration
DIRECTIONS: Take medication orally at the beginning of the day before eating or drinking anything other than water. Take nothing but water for one hour after taking medication in order to improve absorption. Continue LOMA LUX PSORIASIS as long as it is beneficial to your overall condition. Recommended minimum duration of treatment is 4-5 months.
Weight Daily Dosage Bottle Lasts
50-100 lbs 1/2 tsp 96 days
100-150 lbs 1 tsp 48 days
150-200 lbs 1 1/2 tsp 36 days
Over 200 lbs 2 tsp 24 days
Description
ACTIVE INGREDIENTS: Kali Bromatum 1X, Natrum bromatum 2X, Niccolum sulphuricum 3X, Kali sulphuricum 4X, Zincum bromatum 4X.
Section 50565-1
KEEP OUT OF THE REACH OF CHILDREN.
Section 51727-6
INACTIVE INGREDIENTS: Purified water, USP alcohol 2%, methylparaben 0.15%, propylparaben 0.03%.
Section 51945-4
LOMA LUX
LOMA LUX PSORIASIS
For relief from the scaling, itching and redness of Psoriasis and Seborrhea
TAKE ORALLY
HOMEOPATHIC MEDICINE
8 FL. OZ. (237 ml)
Section 53413-1
Manufactured for and dist. by:
LOMA LUX LABORATORIES
P.O. BOX 702418
TULSA, OK 74170-2418
Questions? Call us!
800-316-9636
More information? Visit our web site:
www.lomalux.com
Section 55105-1
For relief from the scaling, itching and redness of Psoriasis and Seborrhea.
Section 55106-9
ACTIVE INGREDIENTS: Kali Bromatum 1X, Natrum bromatum 2X, Niccolum sulphuricum 3X, Kali sulphuricum 4X, Zincum bromatum 4X.
Structured Label Content
Indications and Usage (34067-9)
For relief from the scaling, itching and redness of Psoriasis and Seborrhea.
Dosage and Administration (34068-7)
DIRECTIONS: Take medication orally at the beginning of the day before eating or drinking anything other than water. Take nothing but water for one hour after taking medication in order to improve absorption. Continue LOMA LUX PSORIASIS as long as it is beneficial to your overall condition. Recommended minimum duration of treatment is 4-5 months.
Weight Daily Dosage Bottle Lasts
50-100 lbs 1/2 tsp 96 days
100-150 lbs 1 tsp 48 days
150-200 lbs 1 1/2 tsp 36 days
Over 200 lbs 2 tsp 24 days
Warnings and Precautions (34071-1)
WARNINGS: Do not use if pregnant or nursing.
If allergic to nickel or costume jewelry, use only under the advice and supervision of a physician.
If symptoms worsen, contact a physician.
Keep out or the reach of children.
CAUTION: Use only as directed.
Do not give to children under ten years old or use in the presence of kidney disease.
If skin rash appears or if nervous symptoms persist, recur frequently or are unusual, discontinue use and consult a physician.
Section 50565-1 (50565-1)
KEEP OUT OF THE REACH OF CHILDREN.
Section 51727-6 (51727-6)
INACTIVE INGREDIENTS: Purified water, USP alcohol 2%, methylparaben 0.15%, propylparaben 0.03%.
Section 51945-4 (51945-4)
LOMA LUX
LOMA LUX PSORIASIS
For relief from the scaling, itching and redness of Psoriasis and Seborrhea
TAKE ORALLY
HOMEOPATHIC MEDICINE
8 FL. OZ. (237 ml)
Section 53413-1 (53413-1)
Manufactured for and dist. by:
LOMA LUX LABORATORIES
P.O. BOX 702418
TULSA, OK 74170-2418
Questions? Call us!
800-316-9636
More information? Visit our web site:
www.lomalux.com
Section 55105-1 (55105-1)
For relief from the scaling, itching and redness of Psoriasis and Seborrhea.
Section 55106-9 (55106-9)
ACTIVE INGREDIENTS: Kali Bromatum 1X, Natrum bromatum 2X, Niccolum sulphuricum 3X, Kali sulphuricum 4X, Zincum bromatum 4X.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:09:09.037821 · Updated: 2026-03-14T23:14:18.528193