Glatiramer Acetate Injection, For Subcutaneous Use
1b3a60de-2191-4b15-be69-01329da87711
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
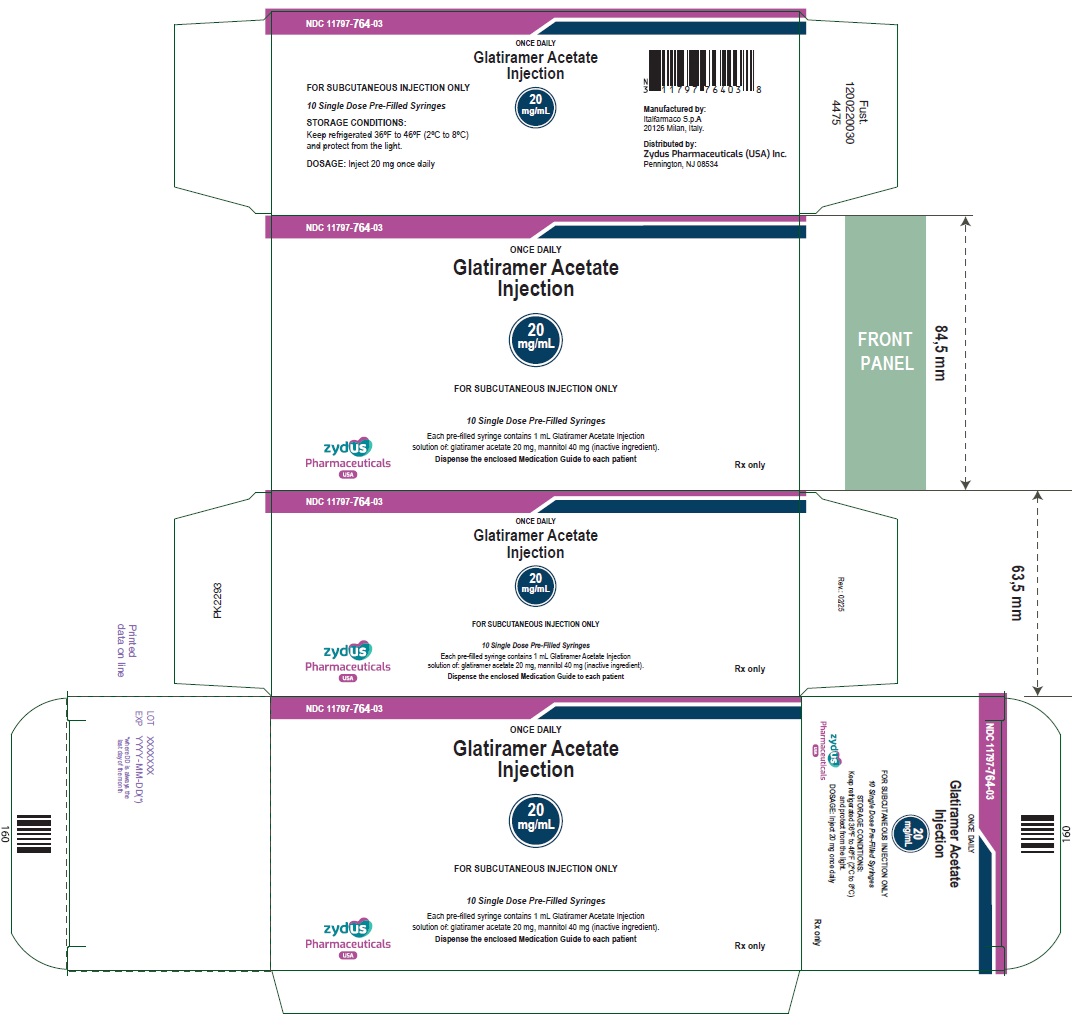
NDC 11797-764-03 ONCE DAILY Glatiramer Acetate Injection 20 mg/mL FOR SUBCUTANEOUS INJECTION ONLY 30 pre-filled syringe (3 carton box of 10 syringes) Each pre-filled syringe contains 1 mL Glatiramer Acetate Injection solution of: glatiramer acetate 20 mg, mannitol 40 mg (inactive ingredient). Dispense the enclosed Medication Guide to each patient zydus Pharmaceuticals USA Rx only NDC 11797-765-03 THREE TIMES A WEEK Glatiramer Acetate Injection 40 mg/mL FOR SUBCUTANEOUS INJECTION ONLY 12 Single Dose Pre-Filled Syringes Each pre-filled syringe contains 1 mL Glatiramer Acetate Injection solution of: glatiramer acetate 40 mg, mannitol 40 mg (inactive ingredient). Dispense the enclosed Medication Guide to each patient zydus Pharmaceuticals USA Rx only
Medication Information
Description
NDC 11797-764-03 ONCE DAILY Glatiramer Acetate Injection 20 mg/mL FOR SUBCUTANEOUS INJECTION ONLY 30 pre-filled syringe (3 carton box of 10 syringes) Each pre-filled syringe contains 1 mL Glatiramer Acetate Injection solution of: glatiramer acetate 20 mg, mannitol 40 mg (inactive ingredient). Dispense the enclosed Medication Guide to each patient zydus Pharmaceuticals USA Rx only NDC 11797-765-03 THREE TIMES A WEEK Glatiramer Acetate Injection 40 mg/mL FOR SUBCUTANEOUS INJECTION ONLY 12 Single Dose Pre-Filled Syringes Each pre-filled syringe contains 1 mL Glatiramer Acetate Injection solution of: glatiramer acetate 40 mg, mannitol 40 mg (inactive ingredient). Dispense the enclosed Medication Guide to each patient zydus Pharmaceuticals USA Rx only
Package Label.principal Display Panel
NDC 11797-764-03
ONCE DAILY
Glatiramer Acetate
Injection
20
mg/mL
FOR SUBCUTANEOUS INJECTION ONLY
30 pre-filled syringe (3 carton box of 10 syringes)
Each pre-filled syringe contains 1 mL Glatiramer Acetate Injection
solution of: glatiramer acetate 20 mg, mannitol 40 mg (inactive ingredient).
Dispense the enclosed Medication Guide to each patient
zydus
Pharmaceuticals
USA
Rx only
NDC 11797-765-03
THREE TIMES A WEEK
Glatiramer Acetate
Injection
40
mg/mL
FOR SUBCUTANEOUS INJECTION ONLY
12 Single Dose Pre-Filled Syringes
Each pre-filled syringe contains 1 mL Glatiramer Acetate Injection
solution of: glatiramer acetate 40 mg, mannitol 40 mg (inactive ingredient).
Dispense the enclosed Medication Guide to each patient
zydus
Pharmaceuticals
USA
Rx only
Structured Label Content
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
NDC 11797-764-03
ONCE DAILY
Glatiramer Acetate
Injection
20
mg/mL
FOR SUBCUTANEOUS INJECTION ONLY
30 pre-filled syringe (3 carton box of 10 syringes)
Each pre-filled syringe contains 1 mL Glatiramer Acetate Injection
solution of: glatiramer acetate 20 mg, mannitol 40 mg (inactive ingredient).
Dispense the enclosed Medication Guide to each patient
zydus
Pharmaceuticals
USA
Rx only
NDC 11797-765-03
THREE TIMES A WEEK
Glatiramer Acetate
Injection
40
mg/mL
FOR SUBCUTANEOUS INJECTION ONLY
12 Single Dose Pre-Filled Syringes
Each pre-filled syringe contains 1 mL Glatiramer Acetate Injection
solution of: glatiramer acetate 40 mg, mannitol 40 mg (inactive ingredient).
Dispense the enclosed Medication Guide to each patient
zydus
Pharmaceuticals
USA
Rx only
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:54:40.413087 · Updated: 2026-03-14T22:49:26.517746