bacitracin zinc and polymyxin b sulfate
1b317249-30eb-4415-8a1c-cfedb4ba3549
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
For the treatment of superficial ocular infections involving the conjunctiva and/or cornea caused by organisms susceptible to bacitracin zinc and polymyxin B sulfate.
Dosage and Administration
Apply the ointment every 3 or 4 hours for 7 to 10 days, depending on the severity of the infection. FOR OPHTHALMIC USE ONLY
Contraindications
This product is contraindicated in those individuals who have shown hypersensitivity to any of its components.
Adverse Reactions
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
Bacitracin zinc and polymyxin B sulfate ophthalmic ointment, USP is available in tubes with an ophthalmic tip applicator in the following size: NDC 24208-555-55 - 3.5 g tube
Description
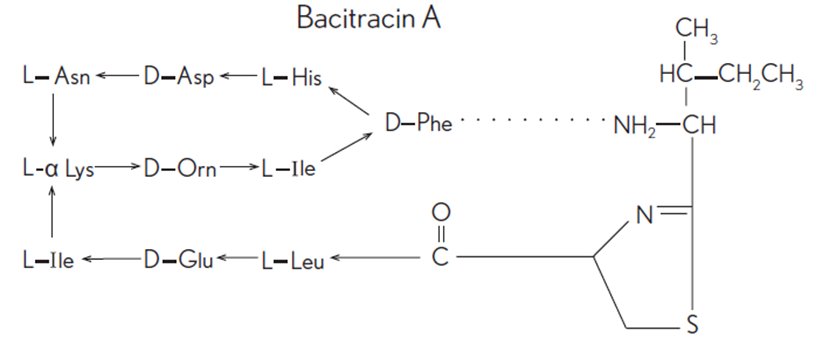
Bacitracin zinc and polymyxin B sulfate ophthalmic ointment, USP is a sterile antimicrobial ointment formulated for ophthalmic use. Bacitracin zinc is the zinc salt of bacitracin, a mixture of related cyclic polypeptides (mainly bacitracin A) produced by the growth of an organism of the licheniformis group of Bacillus subtilis var Tracy. It has a potency of not less than 40 bacitracin units/mg. The structural formula for bacitracin A is: Polymyxin B sulfate is the sulfate salt of polymyxin B 1 and B 2 , which are produced by the growth of Bacillus polymyxa (Prazmowski) Migula (Fam. Bacillaceae). It has a potency of not less than 6,000 polymyxin B units/mg, calculated on an anhydrous basis. The structural formulae are: Each gram contains: Actives: bacitracin zinc equal to 500 bacitracin units and polymyxin B sulfate equal to 10,000 polymyxin B units; Inactives: mineral oil and white petrolatum.
Medication Information
Warnings
Ophthalmic ointments may retard corneal healing.
Indications and Usage
For the treatment of superficial ocular infections involving the conjunctiva and/or cornea caused by organisms susceptible to bacitracin zinc and polymyxin B sulfate.
Dosage and Administration
Apply the ointment every 3 or 4 hours for 7 to 10 days, depending on the severity of the infection.
FOR OPHTHALMIC USE ONLY
Contraindications
This product is contraindicated in those individuals who have shown hypersensitivity to any of its components.
Adverse Reactions
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
Bacitracin zinc and polymyxin B sulfate ophthalmic ointment, USP is available in tubes with an ophthalmic tip applicator in the following size:
NDC 24208-555-55 - 3.5 g tube
Description
Bacitracin zinc and polymyxin B sulfate ophthalmic ointment, USP is a sterile antimicrobial ointment formulated for ophthalmic use.
Bacitracin zinc is the zinc salt of bacitracin, a mixture of related cyclic polypeptides (mainly bacitracin A) produced by the growth of an organism of the licheniformis group of Bacillus subtilisvar Tracy. It has a potency of not less than 40 bacitracin units/mg. The structural formula for bacitracin A is:
Polymyxin B sulfate is the sulfate salt of polymyxin B 1and B 2, which are produced by the growth of Bacillus polymyxa(Prazmowski) Migula (Fam. Bacillaceae). It has a potency of not less than 6,000 polymyxin B units/mg, calculated on an anhydrous basis. The structural formulae are:
Each gram contains: Actives:bacitracin zinc equal to 500 bacitracin units and polymyxin B sulfate equal to 10,000 polymyxin B units; Inactives:mineral oil and white petrolatum.
Principal Display Panel
NDC24208-555-55
Bacitracin Zinc
and Polymyxin B
Sulfate
Ophthalmic
Ointment, USP
(Sterile)
FOR OPHTHALMIC
USE ONLY
Rx only
Net Wt. 3.5 g
(1/8 oz)
BAUSCH + LOMB
9733303
AB07534
Storage:
Store between 15°C to 25°C (59°F to 77°F). KEEP TIGHTLY CLOSED.
KEEP OUT OF REACH OF CHILDREN.
Distributed by:
Bausch & Lomb Americas Inc.
Bridgewater, NJ 08807 USA
Manufactured by:
Bausch & Lomb Incorporated
Tampa, FL 33637 USA
© 2023 Bausch & Lomb Incorporated or its affiliates
Revised: January 2023
9130705
(Folded)
9130605
(Flat)
Precautions
As with other antibiotic preparations, prolonged use may result in overgrowth of nonsusceptible organisms, including fungi. Appropriate measures should be taken if this occurs.
Clinical Pharmacology
Polymyxin B sulfate attacks gram-negative bacilli, including virtually all strains of Pseudomonas aeruginosaand Haemophilus influenzaespecies.
Bacitracin is active against most gram-positive bacilli and cocci including hemolytic streptococci.
Structured Label Content
Warnings (WARNINGS)
Ophthalmic ointments may retard corneal healing.
Indications and Usage (INDICATIONS AND USAGE)
For the treatment of superficial ocular infections involving the conjunctiva and/or cornea caused by organisms susceptible to bacitracin zinc and polymyxin B sulfate.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Apply the ointment every 3 or 4 hours for 7 to 10 days, depending on the severity of the infection.
FOR OPHTHALMIC USE ONLY
Contraindications (CONTRAINDICATIONS)
This product is contraindicated in those individuals who have shown hypersensitivity to any of its components.
Adverse Reactions (ADVERSE REACTIONS)
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied (HOW SUPPLIED)
Bacitracin zinc and polymyxin B sulfate ophthalmic ointment, USP is available in tubes with an ophthalmic tip applicator in the following size:
NDC 24208-555-55 - 3.5 g tube
Description (DESCRIPTION)
Bacitracin zinc and polymyxin B sulfate ophthalmic ointment, USP is a sterile antimicrobial ointment formulated for ophthalmic use.
Bacitracin zinc is the zinc salt of bacitracin, a mixture of related cyclic polypeptides (mainly bacitracin A) produced by the growth of an organism of the licheniformis group of Bacillus subtilisvar Tracy. It has a potency of not less than 40 bacitracin units/mg. The structural formula for bacitracin A is:
Polymyxin B sulfate is the sulfate salt of polymyxin B 1and B 2, which are produced by the growth of Bacillus polymyxa(Prazmowski) Migula (Fam. Bacillaceae). It has a potency of not less than 6,000 polymyxin B units/mg, calculated on an anhydrous basis. The structural formulae are:
Each gram contains: Actives:bacitracin zinc equal to 500 bacitracin units and polymyxin B sulfate equal to 10,000 polymyxin B units; Inactives:mineral oil and white petrolatum.
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
NDC24208-555-55
Bacitracin Zinc
and Polymyxin B
Sulfate
Ophthalmic
Ointment, USP
(Sterile)
FOR OPHTHALMIC
USE ONLY
Rx only
Net Wt. 3.5 g
(1/8 oz)
BAUSCH + LOMB
9733303
AB07534
Storage:
Store between 15°C to 25°C (59°F to 77°F). KEEP TIGHTLY CLOSED.
KEEP OUT OF REACH OF CHILDREN.
Distributed by:
Bausch & Lomb Americas Inc.
Bridgewater, NJ 08807 USA
Manufactured by:
Bausch & Lomb Incorporated
Tampa, FL 33637 USA
© 2023 Bausch & Lomb Incorporated or its affiliates
Revised: January 2023
9130705
(Folded)
9130605
(Flat)
Precautions (PRECAUTIONS)
As with other antibiotic preparations, prolonged use may result in overgrowth of nonsusceptible organisms, including fungi. Appropriate measures should be taken if this occurs.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Polymyxin B sulfate attacks gram-negative bacilli, including virtually all strains of Pseudomonas aeruginosaand Haemophilus influenzaespecies.
Bacitracin is active against most gram-positive bacilli and cocci including hemolytic streptococci.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:22.750479 · Updated: 2026-03-14T22:14:57.540523