These Highlights Do Not Include All The Information Needed To Use Perampanel Tablets Safely And Effectively. See Full Prescribing Information For Perampanel Tablets.
1b0458ea-e718-4d48-ae55-ad9b4d863810
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: SERIOUS PSYCHIATRIC AND BEHAVIORAL REACTIONS Serious or life-threatening psychiatric and behavioral adverse reactions including aggression, hostility, irritability, anger, and homicidal ideation and threats have been reported in patients taking perampanel ( 5.1). These reactions occurred in patients with and without prior psychiatric history, prior aggressive behavior, or concomitant use of medications associated with hostility and aggression ( 5.1). Advise patients and caregivers to contact a healthcare provider immediately if any of these reactions or changes in mood, behavior, or personality that are not typical for the patient are observed while taking perampanel or after discontinuing perampanel ( 5.1). Closely monitor patients particularly during the titration period and at higher doses ( 5.1). Perampanel should be reduced if these symptoms occur and should be discontinued immediately if symptoms are severe or are worsening ( 5.1).
Indications and Usage
Perampanel tablets, a non-competitive AMPA glutamate receptor antagonist, is indicated for: Treatment of partial-onset seizures with or without secondarily generalized seizures in patients with epilepsy 4 years of age and older ( 1.1 ) Adjunctive therapy in the treatment of primary generalized tonic-clonic seizures in patients with epilepsy 12 years of age and older ( 1.2 )
Dosage and Administration
Dosing in the absence of moderate or strong CYP3A4 inducers Starting dose: 2 mg once daily orally at bedtime ( 2.1 , 2.2 ) May increase dose based on clinical response and tolerability by increments of 2 mg once daily no more frequently than at weekly intervals ( 2.1 , 2.2 ) Recommended maintenance dose in monotherapy or adjunctive therapy for partial-onset seizures: 8 mg to 12 mg once daily at bedtime ( 2.1 ) Recommended maintenance dose in adjunctive therapy for primary generalized tonic-clonic seizures: 8 mg once daily at bedtime ( 2.2 ) Dosing in the presence of concomitant moderate or strong CYP3A4 inducers : see section 2.3 Specific Populations Mild and Moderate Hepatic Impairment: Maximum recommended daily dose is 6 mg (mild) and 4 mg (moderate) once daily at bedtime ( 2.4 ) Severe Hepatic Impairment: Not recommended ( 2.4 ) Severe Renal Impairment or on Hemodialysis: Not recommended ( 2.5 ) Elderly: Increase dose no more frequently than every 2 weeks ( 2.6 )
Warnings and Precautions
Suicidal Behavior and Ideation: Monitor for suicidal thoughts or behavior ( 5.2 ) Neurologic Effects: Monitor for dizziness, gait disturbance, somnolence, and fatigue ( 5.3 ) Patients should use caution when driving or operating machinery ( 5.3 ) Falls: Monitor for falls and injuries ( 5.4 ) Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multi-Organ Hypersensitivity: Discontinue if no alternate etiology ( 5.5 ) Withdrawal of Antiepileptic Drugs: In patients with epilepsy, there may be an increase in seizure frequency ( 5.6 )
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling: Serious Psychiatric and Behavioral Reactions [see Warnings and Precautions (5.1) ] Suicidal Behavior and Ideation [see Warnings and Precautions (5.2) ] Neurologic Effects [see Warnings and Precautions (5.3) ] Falls [see Warnings and Precautions (5.4) ] Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [see Warnings and Precautions (5.5) ]
Drug Interactions
Contraceptives: 12 mg once daily may decrease the effectiveness of hormonal contraceptives containing levonorgestrel ( 7.1 ) Moderate and Strong CYP3A4 Inducers (including carbamazepine, oxcarbazepine, and phenytoin): increase clearance of perampanel and decrease perampanel plasma concentrations. When moderate or strong CYP3A4 inducers are introduced or withdrawn, monitor patients closely. Dose adjustment of perampanel tablets may be necessary ( 2.3 , 7.2 )
How Supplied
Perampanel Tablets 2 mg are orange, round, film-coated tablets debossed with "2" on one side and plain on the other side. They are supplied as follows: Blisters of (2 × 20) Total 40 tablets NDC 51672-4204-3 Bottles of 30 NDC 51672-4204-6 4 mg are red, round, film-coated tablets debossed with "T 4" on one side and plain on the other side. They are supplied as follows: Blisters of (3 × 10) Total 30 tablets NDC 51672-4205-2 Bottles of 30 NDC 51672-4205-6 6 mg are pink, round, film-coated tablets debossed with "T 6" on one side and plain on the other side. They are supplied as follows: Blisters of (3 × 10) Total 30 tablets NDC 51672-4206-2 Bottles of 30 NDC 51672-4206-6 8 mg are dark pink, round, film-coated tablets debossed with "T 8" on one side and plain on the other side. They are supplied as follows: Blisters of (3 × 10) Total 30 tablets NDC 51672-4207-2 Bottles of 30 NDC 51672-4207-6 10 mg are green, round, film-coated tablets debossed with "T 10" on one side and plain on the other side. They are supplied as follows: Blisters of (3 × 10) Total 30 tablets NDC 51672-4208-2 Bottles of 30 NDC 51672-4208-6 12 mg are blue, round, film-coated tablets debossed with "T 12" on one side and plain on the other side. They are supplied as follows: Blisters of (3 × 10) Total 30 tablets NDC 51672-4209-2 Bottles of 30 NDC 51672-4209-6
Medication Information
Recent Major Changes
WARNING: SERIOUS PSYCHIATRIC AND BEHAVIORAL REACTIONS
- Serious or life-threatening psychiatric and behavioral adverse reactions including aggression, hostility, irritability, anger, and homicidal ideation and threats have been reported in patients taking perampanel ( 5.1).
- These reactions occurred in patients with and without prior psychiatric history, prior aggressive behavior, or concomitant use of medications associated with hostility and aggression ( 5.1).
- Advise patients and caregivers to contact a healthcare provider immediately if any of these reactions or changes in mood, behavior, or personality that are not typical for the patient are observed while taking perampanel or after discontinuing perampanel ( 5.1).
- Closely monitor patients particularly during the titration period and at higher doses ( 5.1).
- Perampanel should be reduced if these symptoms occur and should be discontinued immediately if symptoms are severe or are worsening ( 5.1).
Warnings and Precautions
Suicidal Behavior and Ideation: Monitor for suicidal thoughts or behavior ( 5.2 ) Neurologic Effects: Monitor for dizziness, gait disturbance, somnolence, and fatigue ( 5.3 ) Patients should use caution when driving or operating machinery ( 5.3 ) Falls: Monitor for falls and injuries ( 5.4 ) Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multi-Organ Hypersensitivity: Discontinue if no alternate etiology ( 5.5 ) Withdrawal of Antiepileptic Drugs: In patients with epilepsy, there may be an increase in seizure frequency ( 5.6 )
Indications and Usage
Perampanel tablets, a non-competitive AMPA glutamate receptor antagonist, is indicated for: Treatment of partial-onset seizures with or without secondarily generalized seizures in patients with epilepsy 4 years of age and older ( 1.1 ) Adjunctive therapy in the treatment of primary generalized tonic-clonic seizures in patients with epilepsy 12 years of age and older ( 1.2 )
Dosage and Administration
Dosing in the absence of moderate or strong CYP3A4 inducers Starting dose: 2 mg once daily orally at bedtime ( 2.1 , 2.2 ) May increase dose based on clinical response and tolerability by increments of 2 mg once daily no more frequently than at weekly intervals ( 2.1 , 2.2 ) Recommended maintenance dose in monotherapy or adjunctive therapy for partial-onset seizures: 8 mg to 12 mg once daily at bedtime ( 2.1 ) Recommended maintenance dose in adjunctive therapy for primary generalized tonic-clonic seizures: 8 mg once daily at bedtime ( 2.2 ) Dosing in the presence of concomitant moderate or strong CYP3A4 inducers : see section 2.3 Specific Populations Mild and Moderate Hepatic Impairment: Maximum recommended daily dose is 6 mg (mild) and 4 mg (moderate) once daily at bedtime ( 2.4 ) Severe Hepatic Impairment: Not recommended ( 2.4 ) Severe Renal Impairment or on Hemodialysis: Not recommended ( 2.5 ) Elderly: Increase dose no more frequently than every 2 weeks ( 2.6 )
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling: Serious Psychiatric and Behavioral Reactions [see Warnings and Precautions (5.1) ] Suicidal Behavior and Ideation [see Warnings and Precautions (5.2) ] Neurologic Effects [see Warnings and Precautions (5.3) ] Falls [see Warnings and Precautions (5.4) ] Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [see Warnings and Precautions (5.5) ]
Drug Interactions
Contraceptives: 12 mg once daily may decrease the effectiveness of hormonal contraceptives containing levonorgestrel ( 7.1 ) Moderate and Strong CYP3A4 Inducers (including carbamazepine, oxcarbazepine, and phenytoin): increase clearance of perampanel and decrease perampanel plasma concentrations. When moderate or strong CYP3A4 inducers are introduced or withdrawn, monitor patients closely. Dose adjustment of perampanel tablets may be necessary ( 2.3 , 7.2 )
How Supplied
Perampanel Tablets 2 mg are orange, round, film-coated tablets debossed with "2" on one side and plain on the other side. They are supplied as follows: Blisters of (2 × 20) Total 40 tablets NDC 51672-4204-3 Bottles of 30 NDC 51672-4204-6 4 mg are red, round, film-coated tablets debossed with "T 4" on one side and plain on the other side. They are supplied as follows: Blisters of (3 × 10) Total 30 tablets NDC 51672-4205-2 Bottles of 30 NDC 51672-4205-6 6 mg are pink, round, film-coated tablets debossed with "T 6" on one side and plain on the other side. They are supplied as follows: Blisters of (3 × 10) Total 30 tablets NDC 51672-4206-2 Bottles of 30 NDC 51672-4206-6 8 mg are dark pink, round, film-coated tablets debossed with "T 8" on one side and plain on the other side. They are supplied as follows: Blisters of (3 × 10) Total 30 tablets NDC 51672-4207-2 Bottles of 30 NDC 51672-4207-6 10 mg are green, round, film-coated tablets debossed with "T 10" on one side and plain on the other side. They are supplied as follows: Blisters of (3 × 10) Total 30 tablets NDC 51672-4208-2 Bottles of 30 NDC 51672-4208-6 12 mg are blue, round, film-coated tablets debossed with "T 12" on one side and plain on the other side. They are supplied as follows: Blisters of (3 × 10) Total 30 tablets NDC 51672-4209-2 Bottles of 30 NDC 51672-4209-6
Description
WARNING: SERIOUS PSYCHIATRIC AND BEHAVIORAL REACTIONS Serious or life-threatening psychiatric and behavioral adverse reactions including aggression, hostility, irritability, anger, and homicidal ideation and threats have been reported in patients taking perampanel ( 5.1). These reactions occurred in patients with and without prior psychiatric history, prior aggressive behavior, or concomitant use of medications associated with hostility and aggression ( 5.1). Advise patients and caregivers to contact a healthcare provider immediately if any of these reactions or changes in mood, behavior, or personality that are not typical for the patient are observed while taking perampanel or after discontinuing perampanel ( 5.1). Closely monitor patients particularly during the titration period and at higher doses ( 5.1). Perampanel should be reduced if these symptoms occur and should be discontinued immediately if symptoms are severe or are worsening ( 5.1).
Section 42229-5
Monotherapy or Adjunctive Therapy
The recommended starting dosage of perampanel in adults and pediatric patients 4 years of age and older is 2 mg once daily taken orally at bedtime. Increase dosage no more frequently than at weekly intervals by increments of 2 mg once daily based on individual clinical response and tolerability.
The recommended maintenance dose range is 8 mg to 12 mg once daily, although some patients may respond to a dose of 4 mg daily. A dose of 12 mg once daily resulted in somewhat greater reductions in seizure rates than the dose of 8 mg once daily, but with a substantial increase in adverse reactions.
Dosage adjustment is recommended with concomitant use of moderate or strong CYP3A4 enzyme inducing drugs, which include certain antiepileptic drugs (AEDs) [see Dosage and Administration (2.3)] .
Section 42231-1
| MEDICATION GUIDE
Perampanel (per AM pa nel) Tablets, for oral use CIII |
|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration
Revised: April 2025 5258281-0425-00 |
|
What is the most important information I should know about perampanel tablets? |
1.
Perampanel tablets may cause mental (psychiatric) problems, including:
|
| 2.
Like other antiepileptic drugs, perampanel tablets may cause suicidal thoughts or actions in a very small number of people, about 1 in 500.
Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you:
|
| Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes. |
| How can I watch for early symptoms of suicidal thoughts and actions? |
|
| Call your healthcare provider between visits as needed, especially if you are worried about symptoms. |
| Do not stop perampanel tablets without first talking with a healthcare provider.Stopping perampanel tablets suddenly can cause serious problems. Stopping perampanel tablets suddenly can cause you to have seizures more often. |
| What are perampanel tablets? |
| Perampanel tablets are a prescription medicine used: |
|
| Perampanel tablets are a controlled substance (CIII) because it can be abused or lead to drug dependence. Keep your perampanel tablets in a safe place to protect it from theft. Never give your perampanel tablets to anyone else because it may harm them. Selling or giving away this medicine is against the law. |
| It is not known if perampanel tablets are safe and effective for partial onset seizures in children under 4 years of age or for primary generalized tonic-clonic seizures in patients under 12 years of age. |
| Before taking perampanel tablets, tell your healthcare provider about all of your medical conditions, including if you: |
|
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. |
| Taking perampanel with certain other medicines can cause side effects or reduce the benefit of either drug. Especially tell your healthcare provider if you take: |
|
| How should I take perampanel tablets? |
|
|
What should I avoid while taking perampanel tablets? |
|
| What are the possible side effects of perampanel tablets? |
| See " What is the most important information I should know about perampanel tablets?" |
| Perampanel may cause other serious side effects, including: |
|
The most common side effects of perampanel include:
|
| These are not all of the possible side effects of perampanel. For more information ask your healthcare provider or pharmacist. |
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
| How should I store perampanel tablets? |
|
| Keep perampanel tablets and all medicines out of the reach of children. |
| General information about the safe and effective use of perampanel tablets. |
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use perampanel for a condition for which it was not prescribed. Do not give perampanel to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about perampanel tablets that is written for health professionals. |
| What are the ingredients in perampanel tablets? |
| Active ingredient:perampanel |
| Inactive ingredients:hypromellose, lactose monohydrate, low-substituted hydroxypropyl cellulose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, purified water, talc, and titanium dioxide. The 2 mg, 4 mg, 6 mg and 8 mg also contain ferric oxide red. The 2 mg and the 10 mg also contain ferric oxide yellow. The 8 mg, 10 mg and 12 mg also contain FD&C Blue No. 2 and the 8 mg contains FD&C Red No. 40. |
| Mfd. by: Taro Pharmaceutical Industries Ltd.
Haifa Bay, Israel 2624761 Dist. by: Sun Pharmaceutical Industries, Inc. Cranbury, NJ 08512 All trademarks are the property of their respective owners. For more information, call Sun Pharmaceutical Industries, Inc. at 1-866-923-4914. |
5.4 Falls
An increased risk of falls, in some cases leading to serious injuries including head injuries and bone fracture, occurred in patients being treated with perampanel (with and without concurrent seizures). In the controlled partial-onset seizure clinical trials, falls were reported in 5% and 10% of patients randomized to receive perampanel at doses of 8 mg and 12 mg per day, respectively, compared to 3% of placebo-treated patients. Falls were reported as serious and led to discontinuation more frequently in perampanel-treated patients than placebo-treated patients. Elderly patients had an increased risk of falls compared to younger adults and pediatric patients.
9.2 Abuse
Prescription drug abuse is the intentional non-therapeutic use of a drug, even once, for its rewarding psychological or physiological effects. Drug addiction, which develops after repeated drug abuse, is characterized by a strong desire to take a drug despite harmful consequences, difficulty in controlling its use, giving a higher priority to drug use than to obligations, increased tolerance, and sometimes physical withdrawal. Drug abuse and drug addiction are separate and distinct from physical dependence (for example, abuse may not be accompanied by physical dependence) [see Drug Abuse and Dependence (9.3)].
Studies of human abuse potential were performed to evaluate the abuse potential of perampanel tablets (8 mg, 24 mg, and 36 mg) as compared to alprazolam C-IV (1.5 mg and 3 mg), and oral ketamine C-III (100 mg) in recreational polydrug users. Supra-therapeutic doses of perampanel tablets 24 mg and 36 mg produced responses for "Euphoria" that were similar to ketamine 100 mg and alprazolam 3 mg. For "High," perampanel tablets 24 mg and 36 mg produced responses comparable to ketamine 100 mg and significantly higher than both doses of alprazolam on a visual analog scale (VAS). "Drug Liking," "Overall Drug Liking," and "Take Drug Again" for perampanel tablets were each statistically lower than ketamine 100 mg. In addition, for "Bad Drug Effects," perampanel tablets 24 mg and 36 mg produced responses significantly higher than ketamine 100 mg. For "Sedation," perampanel tablets 24 mg and 36 mg produced responses similar to alprazolam 3 mg and higher than ketamine 100 mg.
Additionally, on VAS measures related to dissociative phenomena such as "Floating," "Spaced Out," and "Detached," perampanel tablets at supra-therapeutic doses produced responses similar to ketamine 100 mg and greater than both doses of alprazolam tested. Of note, due to somnolence a number of subjects had missing data around T maxof perampanel. The above described data might represent an underestimate of perampanel's effects. The duration of effects of higher doses of perampanel on the majority of measures was much greater than alprazolam 3 mg and ketamine 100 mg.
In this study, the incidence of euphoria following perampanel administration 8 mg, 24 mg, and 36 mg was 37%, 46%, 46%, respectively, which was higher than alprazolam 3 mg (13%) but lower than ketamine 100 mg (89%).
16.2 Storage
Store at 20°C to 25°C (68°F to 77°F)[See USP Controlled Room Temperature].
10 Overdosage
The highest reported overdose of perampanel was 300 mg. Events reported after perampanel overdose include somnolence, stupor, coma, psychiatric or behavioral reactions, altered mental status, and dizziness or gait disturbances.
There is no available specific antidote to the overdose reactions of perampanel. In the event of overdose, standard medical practice for the management of any overdose should be used. An adequate airway, oxygenation, and ventilation should be ensured; monitoring of cardiac rhythm and vital sign measurement is recommended. A certified poison control center should be contacted for updated information on the management of overdose with perampanel. Due to its long half-life, the reactions caused by perampanel could be prolonged.
11 Description
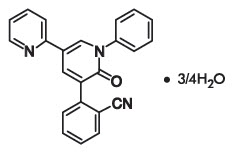
Perampanel tablets contain perampanel a non-competitive AMPA receptor antagonist as a 4:3 hydrate.
The chemical name of the active ingredient is 2-(1´,6´-dihydro-6´-oxo-1´-phenyl[2,3´-bipyridin]-5´-yl)-benzonitrile, hydrate (4:3).
The molecular formula is C 23H 15N 3O ∙ ¾H 2O, and the molecular weight is 362.90 (349.39 for anhydrous perampanel). It is a white to yellowish white powder. It is freely soluble in 1-methyl-2-pyrrolidinone, sparingly soluble in acetonitrile and acetone, slightly soluble in methanol, ethanol and ethyl acetate, very slightly soluble in 1-octanol and diethyl ether, and practically insoluble in heptane and water. The chemical structure of perampanel is:
Perampanel tablets are round, film-coated tablets containing 2 mg, 4 mg, 6 mg, 8 mg, 10 mg, or 12 mg of perampanel. Tablets contain the following inactive ingredients: hypromellose, lactose monohydrate, low-substituted hydroxypropyl cellulose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, purified water, talc, and titanium dioxide. The 2 mg, 4 mg, 6 mg and 8 mg also contain ferric oxide red. The 2 mg and the 10 mg also contain ferric oxide yellow. The 8 mg, 10 mg and 12 mg also contain FD&C Blue No. 2 and the 8 mg contains FD&C Red No. 40.
9.3 Dependence
Physical dependence is characterized by withdrawal symptoms after abrupt discontinuation or a significant dose reduction of a drug.
A nonclinical dependence study in rats demonstrated withdrawal symptoms, including hyperreactivity to handling, muscle rigidity, and decreases in food consumption and body weights.
Perampanel may cause dependence and withdrawal symptoms that may include anxiety, nervousness, irritability, fatigue, lethargy, asthenia, mood swings, and insomnia.
16.1 How Supplied
Perampanel Tablets
- 2 mg are orange, round, film-coated tablets debossed with "2" on one side and plain on the other side. They are supplied as follows:
Blisters of (2 × 20) Total 40 tablets NDC 51672-4204-3 Bottles of 30 NDC 51672-4204-6 - 4 mg are red, round, film-coated tablets debossed with "T 4" on one side and plain on the other side. They are supplied as follows:
Blisters of (3 × 10) Total 30 tablets NDC 51672-4205-2 Bottles of 30 NDC 51672-4205-6 - 6 mg are pink, round, film-coated tablets debossed with "T 6" on one side and plain on the other side. They are supplied as follows:
Blisters of (3 × 10) Total 30 tablets NDC 51672-4206-2 Bottles of 30 NDC 51672-4206-6 - 8 mg are dark pink, round, film-coated tablets debossed with "T 8" on one side and plain on the other side. They are supplied as follows:
Blisters of (3 × 10) Total 30 tablets NDC 51672-4207-2 Bottles of 30 NDC 51672-4207-6 - 10 mg are green, round, film-coated tablets debossed with "T 10" on one side and plain on the other side. They are supplied as follows:
Blisters of (3 × 10) Total 30 tablets NDC 51672-4208-2 Bottles of 30 NDC 51672-4208-6 - 12 mg are blue, round, film-coated tablets debossed with "T 12" on one side and plain on the other side. They are supplied as follows:
Blisters of (3 × 10) Total 30 tablets NDC 51672-4209-2 Bottles of 30 NDC 51672-4209-6
8.4 Pediatric Use
Safety and effectiveness of perampanel for the treatment of partial-onset seizures have been established in pediatric patients 4 years of age and older.
The safety and effectiveness of perampanel in patients 12 years of age and older was established by three randomized double-blind, placebo-controlled, multicenter studies, which included 72 pediatric patients between 12 and 16 years of age exposed to perampanel [see Clinical Pharmacology (12.3)and Clinical Studies (14.1)] . Use of perampanel for the treatment of partial-onset seizures in pediatric patients 4 years to less than 12 years of age is supported by evidence from adequate and well-controlled studies of perampanel in patients 12 years of age and older with partial onset seizures, pharmacokinetic data from adult and pediatric patients, and safety data in 225 pediatric patients 4 years to less than 12 years of age treated with perampanel [see Adverse Reactions (6.1)and Clinical Pharmacology (12.3)] .
The safety and efficacy of perampanel for the adjunctive therapy of primary generalized tonic-clonic seizures in pediatric patients 12 years of age and older was established in a single randomized double-blind, placebo-controlled, multicenter trial (n=164), which included 11 pediatric patients 12 to 16 years of age exposed to perampanel; an additional 6 patients were treated with perampanel in the open-label extension of the study [see Clinical Studies (14.2)] .
The safety and effectiveness of perampanel for the treatment of partial-onset seizures in pediatric patients less than 4 years of age or for the treatment of primary generalized tonic-clonic seizures in pediatric patients less than 12 years of age have not been established.
8.5 Geriatric Use
Clinical studies of perampanel did not include sufficient numbers of patients aged 65 and over to determine the safety and efficacy of perampanel in the elderly population. Because of increased likelihood for adverse reactions in the elderly, dosing titration should proceed slowly in patients aged 65 years and older [see Dosage and Administration (2.5)].
7.1 Contraceptives
With concomitant use, perampanel at a dose of 12 mg per day reduced levonorgestrel exposure by approximately 40% [see Clinical Pharmacology (12.3)] . Use of perampanel with contraceptives containing levonorgestrel may render them less effective. Additional non-hormonal forms of contraception are recommended [see Use in Specific Populations (8.3)] .
4 Contraindications
None.
6 Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling:
- Serious Psychiatric and Behavioral Reactions [see Warnings and Precautions (5.1)]
- Suicidal Behavior and Ideation [see Warnings and Precautions (5.2)]
- Neurologic Effects [see Warnings and Precautions (5.3)]
- Falls [see Warnings and Precautions (5.4)]
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [see Warnings and Precautions (5.5)]
7 Drug Interactions
- Contraceptives: 12 mg once daily may decrease the effectiveness of hormonal contraceptives containing levonorgestrel ( 7.1)
- Moderate and Strong CYP3A4 Inducers (including carbamazepine, oxcarbazepine, and phenytoin): increase clearance of perampanel and decrease perampanel plasma concentrations. When moderate or strong CYP3A4 inducers are introduced or withdrawn, monitor patients closely. Dose adjustment of perampanel tablets may be necessary ( 2.3, 7.2)
8.7 Renal Impairment
Dose adjustment is not required in patients with mild renal impairment. Perampanel should be used with caution in patients with moderate renal impairment, and slower titration may be considered. Use in patients with severe renal impairment or patients undergoing hemodialysis is not recommended [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
Pharmacokinetics of perampanel are similar in healthy subjects, patients with partial-onset seizures, and patients with primary generalized tonic-clonic seizures. The half-life of perampanel is about 105 hours, so that steady state is reached in about 2 to 3 weeks. AUC of perampanel increased in a dose-proportional manner after single-dose administration of 0.2 mg to 12 mg tablets and after multiple-dose administration of 1 mg to 12 mg tablets once daily.
Perampanel oral suspension has comparable bioavailability to perampanel tablets under steady state. Both formulations may be used interchangeably.
The pharmacokinetics of perampanel are similar when used as monotherapy or as adjunctive therapy for the treatment of partial-onset seizures (in the absence of concomitant moderate or strong CYP3A4 inducers).
8.6 Hepatic Impairment
Use of perampanel in patients with severe hepatic impairment is not recommended, and dosage adjustments are recommended in patients with mild or moderate hepatic impairment [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
1 Indications and Usage
Perampanel tablets, a non-competitive AMPA glutamate receptor antagonist, is indicated for:
12.1 Mechanism of Action
Perampanel is a non-competitive antagonist of the ionotropic α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) glutamate receptor on post-synaptic neurons. Glutamate is the primary excitatory neurotransmitter in the central nervous system and is implicated in a number of neurological disorders caused by neuronal over excitation.
The precise mechanism by which perampanel exerts its antiepileptic effects in humans is unknown.
9.1 Controlled Substance
Perampanel tablets contain perampanel and are listed as a Schedule III controlled substance.
1.1 Partial Onset Seizures
Perampanel tablets are indicated for the treatment of partial-onset seizures with or without secondarily generalized seizures in patients with epilepsy 4 years of age and older.
5 Warnings and Precautions
- Suicidal Behavior and Ideation: Monitor for suicidal thoughts or behavior ( 5.2)
- Neurologic Effects: Monitor for dizziness, gait disturbance, somnolence, and fatigue (
5.3)
Patients should use caution when driving or operating machinery ( 5.3) - Falls: Monitor for falls and injuries ( 5.4)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multi-Organ Hypersensitivity: Discontinue if no alternate etiology ( 5.5)
- Withdrawal of Antiepileptic Drugs: In patients with epilepsy, there may be an increase in seizure frequency ( 5.6)
14.1 Partial Onset Seizures
The efficacy of perampanel in partial-onset seizures, with or without secondary generalization, was studied in patients who were not adequately controlled with 1 to 3 concomitant AEDs in 3 randomized, double-blind, placebo-controlled, multicenter trials (Studies 1, 2, and 3) in adult and pediatric patients (12 years of age and older). All trials had an initial 6-week Baseline Period, during which patients were required to have more than five seizures in order to be randomized. The Baseline Period was followed by a 19-week Treatment Period consisting of a 6-week Titration Phase and a 13-week Maintenance Phase. Patients in these 3 trials had a mean duration of epilepsy of approximately 21 years and a median baseline seizure frequency ranging from 9 to 14 seizures per 28 days. During the trials, more than 85% of patients were taking 2 to 3 concomitant AEDs with or without concurrent vagal nerve stimulation, and approximately 50% were on at least one AED known to induce CYP3A4, an enzyme critical to the metabolism of perampanel (i.e., carbamazepine, oxcarbazepine, or phenytoin), resulting in a significant reduction in perampanel's serum concentration [see Drug Interactions (7.2), Clinical Pharmacology (12.3)].
Each study evaluated placebo and multiple perampanel dosages (see Figure 1). During the Titration period in all 3 trials, patients on perampanel received an initial 2 mg once daily dose, which was subsequently increased in weekly increments of 2 mg per day to the final dose. Patients experiencing intolerable adverse reactions were permitted to have their dose reduced to the previously tolerated dose.
The primary endpoint in Studies 1, 2, and 3 was the percent change in seizure frequency per 28 days during the Treatment Period as compared to the Baseline Period. The criterion for statistical significance was p<0.05. A statistically significant decrease in seizure rate was observed at doses of 4 mg to 12 mg per day. Dose response was apparent at 4 mg to 8 mg with little additional reduction in frequency at 12 mg per day.
Figure 1. Median Percent Reduction in Seizure Frequency per 28 Days from Baseline to Treatment Period
*Statistically significant as compared to placebo based on ANCOVA with treatment and pooled country as factors and the ranked baseline seizure frequency per 28 days as a covariate.
Tables 4 and 5 present an analysis combining data from all 3 studies, grouping patients based upon whether or not concomitant enzyme-inducing AEDs (carbamazepine, oxcarbazepine, or phenytoin) were used. The analysis revealed a substantially reduced effect in the presence of inducers.
| Without Enzyme-Inducing AEDs | With Enzyme-Inducing AEDs | |||
|---|---|---|---|---|
| Placebo
% |
Perampanel
% |
Placebo
% |
Perampanel
% |
|
| 2 mg/day | 16 | 23 | 14 | 16 |
| 4 mg/day | 16 | 22 | 14 | 33 |
| 8 mg/day | 19 | 45 | 12 | 24 |
| 12 mg/day | 19 | 54 | 9 | 22 |
| Without Enzyme-Inducing AEDs | With Enzyme-Inducing AEDs | |||
|---|---|---|---|---|
| Placebo
% |
Perampanel
% |
Placebo
% |
Perampanel
% |
|
| 2 mg/day | 19 | 26 | 18 | 20 |
| 4 mg/day | 19 | 35 | 18 | 26 |
| 8 mg/day | 17 | 45 | 19 | 32 |
| 12 mg/day | 15 | 54 | 21 | 33 |
Figure 2 shows the proportion of patients with different percent reductions during the maintenance phase over baseline across all three trials.
Patients in whom the seizure frequency increased are shown at left as "worse." Patients in whom the seizure frequency decreased are shown in the remaining four categories.
Figure 2. Proportion of Patients Exhibiting Different Percent Reductions During the Maintenance Phase Over Baseline Across All Three Trials
The percentages of patients with a 50% or greater reduction in seizure frequency were 19%, 29%, 35%, 35% for placebo, 4 mg, 8 mg, and 12 mg, respectively.
2 Dosage and Administration
Dosing in the absence of moderate or strong CYP3A4 inducers
- Starting dose: 2 mg once daily orally at bedtime ( 2.1, 2.2)
- May increase dose based on clinical response and tolerability by increments of 2 mg once daily no more frequently than at weekly intervals ( 2.1, 2.2)
- Recommended maintenance dose in monotherapy or adjunctive therapy for partial-onset seizures: 8 mg to 12 mg once daily at bedtime ( 2.1)
- Recommended maintenance dose in adjunctive therapy for primary generalized tonic-clonic seizures: 8 mg once daily at bedtime ( 2.2)
Dosing in the presence of concomitant moderate or strong CYP3A4 inducers: see section 2.3
Specific Populations
- Mild and Moderate Hepatic Impairment: Maximum recommended daily dose is 6 mg (mild) and 4 mg (moderate) once daily at bedtime ( 2.4)
- Severe Hepatic Impairment: Not recommended ( 2.4)
- Severe Renal Impairment or on Hemodialysis: Not recommended ( 2.5)
- Elderly: Increase dose no more frequently than every 2 weeks ( 2.6)
3 Dosage Forms and Strengths
- 2 mg tablets: orange, round, debossed with "2" on one side and plain on the other.
- 4 mg tablets: red, round, debossed with "T 4" on one side and plain on the other.
- 6 mg tablets: pink, round, debossed with "T 6" on one side and plain on the other.
- 8 mg tablets: dark pink, round, debossed with "T 8" on one side and plain on the other.
- 10 mg tablets: green, round, debossed with "T 10" on one side and plain on the other.
- 12 mg tablets: blue, round, debossed with "T 12" on one side and plain on the other.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of perampanel. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Dermatologic: Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) [see Warnings and Precautions (5.5)]
Psychiatric: Acute psychosis, hallucinations, delusions, paranoia, delirium, confusional state, disorientation, memory impairment [see Warnings and Precautions (5.1)] .
8 Use in Specific Populations
Pregnancy: Based on animal data, may cause fetal harm ( 8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.2 Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including perampanel, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% CI: 1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as 1 week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5 to 100 years) in the clinical trials analyzed.
Table 1 shows absolute and relative risk by indication for all evaluated AEDs.
| Indication | Placebo Patients with Events per 1000 Patients | Drug Patients with Events per 1000 patients | Relative Risk:
Incidence of Events in drug Patients/Incidence in Placebo Patients |
Risk Difference:
Additional Drug Patients with Events per 1000 Patients |
|---|---|---|---|---|
| Epilepsy | 1.0 | 3.4 | 3.5 | 2.4 |
| Psychiatric | 5.7 | 8.5 | 1.5 | 2.9 |
| Other | 1.0 | 1.8 | 1.9 | 0.9 |
| Total | 2.4 | 4.3 | 1.8 | 1.9 |
The relative risk for suicidal thoughts or behavior was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
Anyone considering prescribing perampanel or any other AED must balance the risk of suicidal thoughts or behavior with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
5.6 Withdrawal of Antiepileptic Drugs
There is the potential of increased seizure frequency in patients with seizure disorders when antiepileptic drugs are withdrawn abruptly. Perampanel has a half-life of approximately 105 hours so that even after abrupt cessation, blood levels fall gradually. In epilepsy clinical trials perampanel was withdrawn without down-titration. Although a small number of patients exhibited seizures following discontinuation, the data were not sufficient to allow any recommendations regarding appropriate withdrawal regimens. A gradual withdrawal is generally recommended with antiepileptic drugs, but if withdrawal is a response to adverse events, prompt withdrawal can be considered.
7.3 Alcohol and Other Cns Depressants
The concomitant use of perampanel and CNS depressants including alcohol may increase CNS depression. A pharmacodynamic interaction study in healthy subjects found that the effects of perampanel on complex tasks such as driving ability were additive or supra-additive to the impairment effects of alcohol [see Clinical Pharmacology (12.3)]. Multiple dosing of perampanel 12 mg per day also enhanced the effects of alcohol to interfere with vigilance and alertness, and increased levels of anger, confusion, and depression. These effects may also be seen when perampanel is used in combination with other CNS depressants. Care should be taken when administering perampanel with these agents. Patients should limit activity until they have experience with concomitant use of CNS depressants (e.g., benzodiazepines, narcotics, barbiturates, sedating antihistamines). Advise patients not to drive or operate machinery until they have gained sufficient experience on perampanel to gauge whether it adversely affects these activities.
7.2 Moderate and Strong Cyp3a4 Inducers
The concomitant use of known moderate and strong CYP3A4 inducers including carbamazepine, phenytoin, or oxcarbazepine with perampanel decreased the plasma levels of perampanel by approximately 50% to 67% [see Clinical Pharmacology (12.3)] . The starting doses for perampanel should be increased in the presence of moderate or strong CYP3A4 inducers [see Dosage and Administration (2.3)] .
When these moderate or strong CYP3A4 inducers are introduced or withdrawn from a patient's treatment regimen, the patient should be closely monitored for clinical response and tolerability. Dose adjustment of perampanel may be necessary [see Dosage and Administration (2.3)].
2.6 Dosage Information for Elderly Patients
In elderly patients, increase dosage no more frequently than every 2 weeks during titration [see Use in Specific Populations (8.5)].
1.2 Primary Generalized Tonic Clonic Seizures
Perampanel tablets are indicated as adjunctive therapy for the treatment of primary generalized tonic-clonic seizures in patients with epilepsy 12 years of age and older.
5.1 Serious Psychiatric and Behavioral Reactions
In the controlled partial-onset seizure clinical trials, hostility- and aggression-related adverse reactions occurred in 12% and 20% of patients randomized to receive perampanel at doses of 8 mg and 12 mg per day, respectively, compared to 6% of patients in the placebo group. These effects were dose-related and generally appeared within the first 6 weeks of treatment, although new events continued to be observed through more than 37 weeks. Perampanel-treated patients experienced more hostility- and aggression-related adverse reactions that were serious, severe, and led to dose reduction, interruption, and discontinuation more frequently than placebo-treated patients.
In general, in placebo-controlled partial-onset seizure clinical trials, neuropsychiatric events were reported more frequently in patients being treated with perampanel than in patients taking placebo. These events included irritability, aggression, anger, and anxiety, which occurred in 2% or greater of perampanel-treated patients and twice as frequently as in placebo-treated patients. Other symptoms that occurred with perampanel and were more common than with placebo included belligerence, affect lability, agitation, and physical assault. Some of these events were reported as serious and life-threatening. Homicidal ideation and/or threat were exhibited in 0.1% of 4,368 perampanel-treated patients in controlled and open label trials, including non-epilepsy trials. Homicidal ideation and/or threat have also been reported postmarketing in patients treated with perampanel.
In the partial-onset seizure clinical trials, these events occurred in patients with and without prior psychiatric history, prior aggressive behavior, or concomitant use of medications associated with hostility and aggression. Some patients experienced worsening of their pre-existing psychiatric conditions. Patients with active psychotic disorders and unstable recurrent affective disorders were excluded from the clinical trials. The combination of alcohol and perampanel significantly worsened mood and increased anger. Patients taking perampanel should avoid the use of alcohol [see Drug Interactions (7.3)].
Similar serious psychiatric and behavioral events were observed in the primary generalized tonic-clonic seizure clinical trial.
In healthy volunteers taking perampanel, observed psychiatric events included paranoia, euphoric mood, agitation, anger, mental status changes, and disorientation/confusional state.
In the non-epilepsy trials, psychiatric events that occurred in perampanel-treated patients more often than placebo-treated patients included disorientation, delusion, and paranoia.
In the postmarketing setting, there have been reports of psychosis (acute psychosis, hallucinations, delusions, paranoia) and delirium (delirium, confusional state, disorientation, memory impairment) in patients treated with perampanel [see Adverse Reactions (6.2)] .
Patients, their caregivers, and families should be informed that perampanel may increase the risk of psychiatric events. Patients should be monitored during treatment and for at least 1 month after the last dose of perampanel, and especially when taking higher doses and during the initial few weeks of drug therapy (titration period) or at other times of dose increases. Dose of perampanel should be reduced if these symptoms occur. Permanently discontinue perampanel for persistent severe or worsening psychiatric symptoms or behaviors and refer for psychiatric evaluation.
Principal Display Panel 2 Mg Tablet Bottle Label
NDC 51672- 4204-6
30 Tablets
Perampanel
Tablets
2 mg
CIII
ATTENTION PHARMACIST:
Dispense the accompanying
Medication Guide to each patient.
Rx only
Principal Display Panel 4 Mg Tablet Bottle Label
NDC 51672- 4205-6
30 Tablets
Perampanel
Tablets
4 mg
CIII
ATTENTION PHARMACIST:
Dispense the accompanying
Medication Guide to each patient.
Rx only
Principal Display Panel 6 Mg Tablet Bottle Label
NDC 51672- 4206-6
30 Tablets
Perampanel
Tablets
6 mg
CIII
ATTENTION PHARMACIST:
Dispense the accompanying
Medication Guide to each patient.
Rx only
Principal Display Panel 8 Mg Tablet Bottle Label
NDC 51672- 4207-6
30 Tablets
Perampanel
Tablets
8 mg
CIII
ATTENTION PHARMACIST:
Dispense the accompanying
Medication Guide to each patient.
Rx only
Principal Display Panel 10 Mg Tablet Bottle Label
NDC 51672- 4208-6
30 Tablets
Perampanel
Tablets
10 mg
CIII
ATTENTION PHARMACIST:
Dispense the accompanying
Medication Guide to each patient.
Rx only
Principal Display Panel 12 Mg Tablet Bottle Label
NDC 51672- 4209-6
30 Tablets
Perampanel
Tablets
12 mg
CIII
ATTENTION PHARMACIST:
Dispense the accompanying
Medication Guide to each patient.
Rx only
14.2 Primary Generalized Tonic Clonic (pgtc) Seizures
The efficacy of perampanel as adjunctive therapy in patients 12 years of age and older with idiopathic generalized epilepsy experiencing primary generalized tonic-clonic seizures was established in one multicenter, randomized, double-blind, placebo-controlled study (Study 4), conducted at 78 sites in 16 countries. Eligible patients on a stable dose of 1 to 3 AEDs experiencing at least 3 primary generalized tonic-clonic seizures during the 8-week baseline period were randomized to either perampanel or placebo. Efficacy was analyzed in 162 patients (perampanel N=81, placebo N=81) who received medication and at least one post-treatment seizure assessment. Patients were titrated over 4 weeks up to a dose of 8 mg per day or the highest tolerated dose and treated for an additional 13 weeks on the last dose level achieved at the end of the titration period. The total treatment period was 17 weeks. Study drug was given once per day.
The primary endpoint was the percent change from baseline in primary generalized tonic-clonic seizure frequency per 28 days during the treatment period as compared to the baseline period. The criterion for statistical significance was p<0.05. Table 6 shows the results of this study. A statistically significant decrease in seizure rate was observed with perampanel compared to placebo.
| Placebo
(N=81) |
Perampanel 8 mg
(N=81) |
|
|---|---|---|
| Percent Reduction During Treatment | 38 | 76
P-value compared to placebo: <0.0001. Statistically significant as compared to placebo based on ANCOVA with treatment and pooled country as factors and the ranked baseline seizure frequency per 28 days as a covariate.
|
Figure 3 shows the proportion of patients with different percent reductions during the maintenance phase over baseline in primary generalized tonic-clonic seizure frequency. Patients in whom the seizure frequency increased are shown at left as "worse." Patients in whom the seizure frequency decreased are shown in the remaining four categories.
Figure 3. Proportion of Patients Exhibiting Different Percent Reductions During the Maintenance Phase Over Baseline in Primary Generalized Tonic-Clonic Seizure Frequency
2.4 Dosage Adjustment in Patients With Hepatic Impairment
In patients with mild and moderate hepatic impairment, the starting dose of perampanel is 2 mg once daily. Increase dosage by increments of 2 mg once daily no more frequently than every 2 weeks. The maximum recommended daily dose is 6 mg for patients with mild hepatic impairment and 4 mg for patients with moderate hepatic impairment. Perampanel is not recommended for use in patients with severe hepatic impairment [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
2.5 Dosage Information for Patients With Renal Impairment
Perampanel tablets can be used in patients with moderate renal impairment with close monitoring. A slower titration may be considered, based on clinical response and tolerability. Perampanel tablets are not recommended in patients with severe renal impairment or patients undergoing hemodialysis [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
2.3 Dosage Modifications With Concomitant Use of Moderate Or Strong Cyp3a4 Enzyme Inducers
Moderate and strong CYP3A4 inducers, including enzyme-inducing AEDs such as phenytoin, carbamazepine, and oxcarbazepine, cause a reduction in perampanel plasma levels [see Drug Interactions (7.2), Clinical Pharmacology (12.3)] . Therefore, in adults and pediatric patients 4 years of age and older receiving these concomitant enzyme-inducing drugs, the recommended starting dosage of perampanel is 4 mg once daily taken orally at bedtime.
Increase dosage by increments of 2 mg once daily based on individual clinical response and tolerability, no more frequently than at weekly intervals. A maintenance dose has not been established in clinical trials. The highest dose studied in patients on concomitant enzyme-inducing AEDs was 12 mg once daily.
When moderate or strong CYP3A4 inducers are introduced or withdrawn from a patient's treatment regimen, the patient should be closely monitored for clinical response and tolerability. Dose adjustment of perampanel may be necessary.
5.5 Drug Reaction With Eosinophilia and Systemic Symptoms (dress)/multiorgan Hypersensitivity
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), also known as Multiorgan hypersensitivity, has been reported in patients taking antiepileptic drugs, including perampanel. DRESS may be fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling, in association with other organ system involvement, such as hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis sometimes resembling an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its expression, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, the patient should be evaluated immediately. Perampanel should be discontinued if an alternative etiology for the signs or symptoms cannot be established.
Structured Label Content
Recent Major Changes (34066-1)
WARNING: SERIOUS PSYCHIATRIC AND BEHAVIORAL REACTIONS
- Serious or life-threatening psychiatric and behavioral adverse reactions including aggression, hostility, irritability, anger, and homicidal ideation and threats have been reported in patients taking perampanel ( 5.1).
- These reactions occurred in patients with and without prior psychiatric history, prior aggressive behavior, or concomitant use of medications associated with hostility and aggression ( 5.1).
- Advise patients and caregivers to contact a healthcare provider immediately if any of these reactions or changes in mood, behavior, or personality that are not typical for the patient are observed while taking perampanel or after discontinuing perampanel ( 5.1).
- Closely monitor patients particularly during the titration period and at higher doses ( 5.1).
- Perampanel should be reduced if these symptoms occur and should be discontinued immediately if symptoms are severe or are worsening ( 5.1).
Section 42229-5 (42229-5)
Monotherapy or Adjunctive Therapy
The recommended starting dosage of perampanel in adults and pediatric patients 4 years of age and older is 2 mg once daily taken orally at bedtime. Increase dosage no more frequently than at weekly intervals by increments of 2 mg once daily based on individual clinical response and tolerability.
The recommended maintenance dose range is 8 mg to 12 mg once daily, although some patients may respond to a dose of 4 mg daily. A dose of 12 mg once daily resulted in somewhat greater reductions in seizure rates than the dose of 8 mg once daily, but with a substantial increase in adverse reactions.
Dosage adjustment is recommended with concomitant use of moderate or strong CYP3A4 enzyme inducing drugs, which include certain antiepileptic drugs (AEDs) [see Dosage and Administration (2.3)] .
Section 42231-1 (42231-1)
| MEDICATION GUIDE
Perampanel (per AM pa nel) Tablets, for oral use CIII |
|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration
Revised: April 2025 5258281-0425-00 |
|
What is the most important information I should know about perampanel tablets? |
1.
Perampanel tablets may cause mental (psychiatric) problems, including:
|
| 2.
Like other antiepileptic drugs, perampanel tablets may cause suicidal thoughts or actions in a very small number of people, about 1 in 500.
Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you:
|
| Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes. |
| How can I watch for early symptoms of suicidal thoughts and actions? |
|
| Call your healthcare provider between visits as needed, especially if you are worried about symptoms. |
| Do not stop perampanel tablets without first talking with a healthcare provider.Stopping perampanel tablets suddenly can cause serious problems. Stopping perampanel tablets suddenly can cause you to have seizures more often. |
| What are perampanel tablets? |
| Perampanel tablets are a prescription medicine used: |
|
| Perampanel tablets are a controlled substance (CIII) because it can be abused or lead to drug dependence. Keep your perampanel tablets in a safe place to protect it from theft. Never give your perampanel tablets to anyone else because it may harm them. Selling or giving away this medicine is against the law. |
| It is not known if perampanel tablets are safe and effective for partial onset seizures in children under 4 years of age or for primary generalized tonic-clonic seizures in patients under 12 years of age. |
| Before taking perampanel tablets, tell your healthcare provider about all of your medical conditions, including if you: |
|
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. |
| Taking perampanel with certain other medicines can cause side effects or reduce the benefit of either drug. Especially tell your healthcare provider if you take: |
|
| How should I take perampanel tablets? |
|
|
What should I avoid while taking perampanel tablets? |
|
| What are the possible side effects of perampanel tablets? |
| See " What is the most important information I should know about perampanel tablets?" |
| Perampanel may cause other serious side effects, including: |
|
The most common side effects of perampanel include:
|
| These are not all of the possible side effects of perampanel. For more information ask your healthcare provider or pharmacist. |
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
| How should I store perampanel tablets? |
|
| Keep perampanel tablets and all medicines out of the reach of children. |
| General information about the safe and effective use of perampanel tablets. |
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use perampanel for a condition for which it was not prescribed. Do not give perampanel to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about perampanel tablets that is written for health professionals. |
| What are the ingredients in perampanel tablets? |
| Active ingredient:perampanel |
| Inactive ingredients:hypromellose, lactose monohydrate, low-substituted hydroxypropyl cellulose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, purified water, talc, and titanium dioxide. The 2 mg, 4 mg, 6 mg and 8 mg also contain ferric oxide red. The 2 mg and the 10 mg also contain ferric oxide yellow. The 8 mg, 10 mg and 12 mg also contain FD&C Blue No. 2 and the 8 mg contains FD&C Red No. 40. |
| Mfd. by: Taro Pharmaceutical Industries Ltd.
Haifa Bay, Israel 2624761 Dist. by: Sun Pharmaceutical Industries, Inc. Cranbury, NJ 08512 All trademarks are the property of their respective owners. For more information, call Sun Pharmaceutical Industries, Inc. at 1-866-923-4914. |
5.4 Falls
An increased risk of falls, in some cases leading to serious injuries including head injuries and bone fracture, occurred in patients being treated with perampanel (with and without concurrent seizures). In the controlled partial-onset seizure clinical trials, falls were reported in 5% and 10% of patients randomized to receive perampanel at doses of 8 mg and 12 mg per day, respectively, compared to 3% of placebo-treated patients. Falls were reported as serious and led to discontinuation more frequently in perampanel-treated patients than placebo-treated patients. Elderly patients had an increased risk of falls compared to younger adults and pediatric patients.
9.2 Abuse
Prescription drug abuse is the intentional non-therapeutic use of a drug, even once, for its rewarding psychological or physiological effects. Drug addiction, which develops after repeated drug abuse, is characterized by a strong desire to take a drug despite harmful consequences, difficulty in controlling its use, giving a higher priority to drug use than to obligations, increased tolerance, and sometimes physical withdrawal. Drug abuse and drug addiction are separate and distinct from physical dependence (for example, abuse may not be accompanied by physical dependence) [see Drug Abuse and Dependence (9.3)].
Studies of human abuse potential were performed to evaluate the abuse potential of perampanel tablets (8 mg, 24 mg, and 36 mg) as compared to alprazolam C-IV (1.5 mg and 3 mg), and oral ketamine C-III (100 mg) in recreational polydrug users. Supra-therapeutic doses of perampanel tablets 24 mg and 36 mg produced responses for "Euphoria" that were similar to ketamine 100 mg and alprazolam 3 mg. For "High," perampanel tablets 24 mg and 36 mg produced responses comparable to ketamine 100 mg and significantly higher than both doses of alprazolam on a visual analog scale (VAS). "Drug Liking," "Overall Drug Liking," and "Take Drug Again" for perampanel tablets were each statistically lower than ketamine 100 mg. In addition, for "Bad Drug Effects," perampanel tablets 24 mg and 36 mg produced responses significantly higher than ketamine 100 mg. For "Sedation," perampanel tablets 24 mg and 36 mg produced responses similar to alprazolam 3 mg and higher than ketamine 100 mg.
Additionally, on VAS measures related to dissociative phenomena such as "Floating," "Spaced Out," and "Detached," perampanel tablets at supra-therapeutic doses produced responses similar to ketamine 100 mg and greater than both doses of alprazolam tested. Of note, due to somnolence a number of subjects had missing data around T maxof perampanel. The above described data might represent an underestimate of perampanel's effects. The duration of effects of higher doses of perampanel on the majority of measures was much greater than alprazolam 3 mg and ketamine 100 mg.
In this study, the incidence of euphoria following perampanel administration 8 mg, 24 mg, and 36 mg was 37%, 46%, 46%, respectively, which was higher than alprazolam 3 mg (13%) but lower than ketamine 100 mg (89%).
16.2 Storage
Store at 20°C to 25°C (68°F to 77°F)[See USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
The highest reported overdose of perampanel was 300 mg. Events reported after perampanel overdose include somnolence, stupor, coma, psychiatric or behavioral reactions, altered mental status, and dizziness or gait disturbances.
There is no available specific antidote to the overdose reactions of perampanel. In the event of overdose, standard medical practice for the management of any overdose should be used. An adequate airway, oxygenation, and ventilation should be ensured; monitoring of cardiac rhythm and vital sign measurement is recommended. A certified poison control center should be contacted for updated information on the management of overdose with perampanel. Due to its long half-life, the reactions caused by perampanel could be prolonged.
11 Description (11 DESCRIPTION)
Perampanel tablets contain perampanel a non-competitive AMPA receptor antagonist as a 4:3 hydrate.
The chemical name of the active ingredient is 2-(1´,6´-dihydro-6´-oxo-1´-phenyl[2,3´-bipyridin]-5´-yl)-benzonitrile, hydrate (4:3).
The molecular formula is C 23H 15N 3O ∙ ¾H 2O, and the molecular weight is 362.90 (349.39 for anhydrous perampanel). It is a white to yellowish white powder. It is freely soluble in 1-methyl-2-pyrrolidinone, sparingly soluble in acetonitrile and acetone, slightly soluble in methanol, ethanol and ethyl acetate, very slightly soluble in 1-octanol and diethyl ether, and practically insoluble in heptane and water. The chemical structure of perampanel is:
Perampanel tablets are round, film-coated tablets containing 2 mg, 4 mg, 6 mg, 8 mg, 10 mg, or 12 mg of perampanel. Tablets contain the following inactive ingredients: hypromellose, lactose monohydrate, low-substituted hydroxypropyl cellulose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, purified water, talc, and titanium dioxide. The 2 mg, 4 mg, 6 mg and 8 mg also contain ferric oxide red. The 2 mg and the 10 mg also contain ferric oxide yellow. The 8 mg, 10 mg and 12 mg also contain FD&C Blue No. 2 and the 8 mg contains FD&C Red No. 40.
9.3 Dependence
Physical dependence is characterized by withdrawal symptoms after abrupt discontinuation or a significant dose reduction of a drug.
A nonclinical dependence study in rats demonstrated withdrawal symptoms, including hyperreactivity to handling, muscle rigidity, and decreases in food consumption and body weights.
Perampanel may cause dependence and withdrawal symptoms that may include anxiety, nervousness, irritability, fatigue, lethargy, asthenia, mood swings, and insomnia.
16.1 How Supplied
Perampanel Tablets
- 2 mg are orange, round, film-coated tablets debossed with "2" on one side and plain on the other side. They are supplied as follows:
Blisters of (2 × 20) Total 40 tablets NDC 51672-4204-3 Bottles of 30 NDC 51672-4204-6 - 4 mg are red, round, film-coated tablets debossed with "T 4" on one side and plain on the other side. They are supplied as follows:
Blisters of (3 × 10) Total 30 tablets NDC 51672-4205-2 Bottles of 30 NDC 51672-4205-6 - 6 mg are pink, round, film-coated tablets debossed with "T 6" on one side and plain on the other side. They are supplied as follows:
Blisters of (3 × 10) Total 30 tablets NDC 51672-4206-2 Bottles of 30 NDC 51672-4206-6 - 8 mg are dark pink, round, film-coated tablets debossed with "T 8" on one side and plain on the other side. They are supplied as follows:
Blisters of (3 × 10) Total 30 tablets NDC 51672-4207-2 Bottles of 30 NDC 51672-4207-6 - 10 mg are green, round, film-coated tablets debossed with "T 10" on one side and plain on the other side. They are supplied as follows:
Blisters of (3 × 10) Total 30 tablets NDC 51672-4208-2 Bottles of 30 NDC 51672-4208-6 - 12 mg are blue, round, film-coated tablets debossed with "T 12" on one side and plain on the other side. They are supplied as follows:
Blisters of (3 × 10) Total 30 tablets NDC 51672-4209-2 Bottles of 30 NDC 51672-4209-6
8.4 Pediatric Use
Safety and effectiveness of perampanel for the treatment of partial-onset seizures have been established in pediatric patients 4 years of age and older.
The safety and effectiveness of perampanel in patients 12 years of age and older was established by three randomized double-blind, placebo-controlled, multicenter studies, which included 72 pediatric patients between 12 and 16 years of age exposed to perampanel [see Clinical Pharmacology (12.3)and Clinical Studies (14.1)] . Use of perampanel for the treatment of partial-onset seizures in pediatric patients 4 years to less than 12 years of age is supported by evidence from adequate and well-controlled studies of perampanel in patients 12 years of age and older with partial onset seizures, pharmacokinetic data from adult and pediatric patients, and safety data in 225 pediatric patients 4 years to less than 12 years of age treated with perampanel [see Adverse Reactions (6.1)and Clinical Pharmacology (12.3)] .
The safety and efficacy of perampanel for the adjunctive therapy of primary generalized tonic-clonic seizures in pediatric patients 12 years of age and older was established in a single randomized double-blind, placebo-controlled, multicenter trial (n=164), which included 11 pediatric patients 12 to 16 years of age exposed to perampanel; an additional 6 patients were treated with perampanel in the open-label extension of the study [see Clinical Studies (14.2)] .
The safety and effectiveness of perampanel for the treatment of partial-onset seizures in pediatric patients less than 4 years of age or for the treatment of primary generalized tonic-clonic seizures in pediatric patients less than 12 years of age have not been established.
8.5 Geriatric Use
Clinical studies of perampanel did not include sufficient numbers of patients aged 65 and over to determine the safety and efficacy of perampanel in the elderly population. Because of increased likelihood for adverse reactions in the elderly, dosing titration should proceed slowly in patients aged 65 years and older [see Dosage and Administration (2.5)].
7.1 Contraceptives
With concomitant use, perampanel at a dose of 12 mg per day reduced levonorgestrel exposure by approximately 40% [see Clinical Pharmacology (12.3)] . Use of perampanel with contraceptives containing levonorgestrel may render them less effective. Additional non-hormonal forms of contraception are recommended [see Use in Specific Populations (8.3)] .
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described below and elsewhere in the labeling:
- Serious Psychiatric and Behavioral Reactions [see Warnings and Precautions (5.1)]
- Suicidal Behavior and Ideation [see Warnings and Precautions (5.2)]
- Neurologic Effects [see Warnings and Precautions (5.3)]
- Falls [see Warnings and Precautions (5.4)]
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [see Warnings and Precautions (5.5)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Contraceptives: 12 mg once daily may decrease the effectiveness of hormonal contraceptives containing levonorgestrel ( 7.1)
- Moderate and Strong CYP3A4 Inducers (including carbamazepine, oxcarbazepine, and phenytoin): increase clearance of perampanel and decrease perampanel plasma concentrations. When moderate or strong CYP3A4 inducers are introduced or withdrawn, monitor patients closely. Dose adjustment of perampanel tablets may be necessary ( 2.3, 7.2)
8.7 Renal Impairment
Dose adjustment is not required in patients with mild renal impairment. Perampanel should be used with caution in patients with moderate renal impairment, and slower titration may be considered. Use in patients with severe renal impairment or patients undergoing hemodialysis is not recommended [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
Pharmacokinetics of perampanel are similar in healthy subjects, patients with partial-onset seizures, and patients with primary generalized tonic-clonic seizures. The half-life of perampanel is about 105 hours, so that steady state is reached in about 2 to 3 weeks. AUC of perampanel increased in a dose-proportional manner after single-dose administration of 0.2 mg to 12 mg tablets and after multiple-dose administration of 1 mg to 12 mg tablets once daily.
Perampanel oral suspension has comparable bioavailability to perampanel tablets under steady state. Both formulations may be used interchangeably.
The pharmacokinetics of perampanel are similar when used as monotherapy or as adjunctive therapy for the treatment of partial-onset seizures (in the absence of concomitant moderate or strong CYP3A4 inducers).
8.6 Hepatic Impairment
Use of perampanel in patients with severe hepatic impairment is not recommended, and dosage adjustments are recommended in patients with mild or moderate hepatic impairment [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Perampanel tablets, a non-competitive AMPA glutamate receptor antagonist, is indicated for:
12.1 Mechanism of Action
Perampanel is a non-competitive antagonist of the ionotropic α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) glutamate receptor on post-synaptic neurons. Glutamate is the primary excitatory neurotransmitter in the central nervous system and is implicated in a number of neurological disorders caused by neuronal over excitation.
The precise mechanism by which perampanel exerts its antiepileptic effects in humans is unknown.
9.1 Controlled Substance
Perampanel tablets contain perampanel and are listed as a Schedule III controlled substance.
1.1 Partial Onset Seizures (1.1 Partial-Onset Seizures)
Perampanel tablets are indicated for the treatment of partial-onset seizures with or without secondarily generalized seizures in patients with epilepsy 4 years of age and older.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Suicidal Behavior and Ideation: Monitor for suicidal thoughts or behavior ( 5.2)
- Neurologic Effects: Monitor for dizziness, gait disturbance, somnolence, and fatigue (
5.3)
Patients should use caution when driving or operating machinery ( 5.3) - Falls: Monitor for falls and injuries ( 5.4)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multi-Organ Hypersensitivity: Discontinue if no alternate etiology ( 5.5)
- Withdrawal of Antiepileptic Drugs: In patients with epilepsy, there may be an increase in seizure frequency ( 5.6)
14.1 Partial Onset Seizures (14.1 Partial-Onset Seizures)
The efficacy of perampanel in partial-onset seizures, with or without secondary generalization, was studied in patients who were not adequately controlled with 1 to 3 concomitant AEDs in 3 randomized, double-blind, placebo-controlled, multicenter trials (Studies 1, 2, and 3) in adult and pediatric patients (12 years of age and older). All trials had an initial 6-week Baseline Period, during which patients were required to have more than five seizures in order to be randomized. The Baseline Period was followed by a 19-week Treatment Period consisting of a 6-week Titration Phase and a 13-week Maintenance Phase. Patients in these 3 trials had a mean duration of epilepsy of approximately 21 years and a median baseline seizure frequency ranging from 9 to 14 seizures per 28 days. During the trials, more than 85% of patients were taking 2 to 3 concomitant AEDs with or without concurrent vagal nerve stimulation, and approximately 50% were on at least one AED known to induce CYP3A4, an enzyme critical to the metabolism of perampanel (i.e., carbamazepine, oxcarbazepine, or phenytoin), resulting in a significant reduction in perampanel's serum concentration [see Drug Interactions (7.2), Clinical Pharmacology (12.3)].
Each study evaluated placebo and multiple perampanel dosages (see Figure 1). During the Titration period in all 3 trials, patients on perampanel received an initial 2 mg once daily dose, which was subsequently increased in weekly increments of 2 mg per day to the final dose. Patients experiencing intolerable adverse reactions were permitted to have their dose reduced to the previously tolerated dose.
The primary endpoint in Studies 1, 2, and 3 was the percent change in seizure frequency per 28 days during the Treatment Period as compared to the Baseline Period. The criterion for statistical significance was p<0.05. A statistically significant decrease in seizure rate was observed at doses of 4 mg to 12 mg per day. Dose response was apparent at 4 mg to 8 mg with little additional reduction in frequency at 12 mg per day.
Figure 1. Median Percent Reduction in Seizure Frequency per 28 Days from Baseline to Treatment Period
*Statistically significant as compared to placebo based on ANCOVA with treatment and pooled country as factors and the ranked baseline seizure frequency per 28 days as a covariate.
Tables 4 and 5 present an analysis combining data from all 3 studies, grouping patients based upon whether or not concomitant enzyme-inducing AEDs (carbamazepine, oxcarbazepine, or phenytoin) were used. The analysis revealed a substantially reduced effect in the presence of inducers.
| Without Enzyme-Inducing AEDs | With Enzyme-Inducing AEDs | |||
|---|---|---|---|---|
| Placebo
% |
Perampanel
% |
Placebo
% |
Perampanel
% |
|
| 2 mg/day | 16 | 23 | 14 | 16 |
| 4 mg/day | 16 | 22 | 14 | 33 |
| 8 mg/day | 19 | 45 | 12 | 24 |
| 12 mg/day | 19 | 54 | 9 | 22 |
| Without Enzyme-Inducing AEDs | With Enzyme-Inducing AEDs | |||
|---|---|---|---|---|
| Placebo
% |
Perampanel
% |
Placebo
% |
Perampanel
% |
|
| 2 mg/day | 19 | 26 | 18 | 20 |
| 4 mg/day | 19 | 35 | 18 | 26 |
| 8 mg/day | 17 | 45 | 19 | 32 |
| 12 mg/day | 15 | 54 | 21 | 33 |
Figure 2 shows the proportion of patients with different percent reductions during the maintenance phase over baseline across all three trials.
Patients in whom the seizure frequency increased are shown at left as "worse." Patients in whom the seizure frequency decreased are shown in the remaining four categories.
Figure 2. Proportion of Patients Exhibiting Different Percent Reductions During the Maintenance Phase Over Baseline Across All Three Trials
The percentages of patients with a 50% or greater reduction in seizure frequency were 19%, 29%, 35%, 35% for placebo, 4 mg, 8 mg, and 12 mg, respectively.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Dosing in the absence of moderate or strong CYP3A4 inducers
- Starting dose: 2 mg once daily orally at bedtime ( 2.1, 2.2)
- May increase dose based on clinical response and tolerability by increments of 2 mg once daily no more frequently than at weekly intervals ( 2.1, 2.2)
- Recommended maintenance dose in monotherapy or adjunctive therapy for partial-onset seizures: 8 mg to 12 mg once daily at bedtime ( 2.1)
- Recommended maintenance dose in adjunctive therapy for primary generalized tonic-clonic seizures: 8 mg once daily at bedtime ( 2.2)
Dosing in the presence of concomitant moderate or strong CYP3A4 inducers: see section 2.3
Specific Populations
- Mild and Moderate Hepatic Impairment: Maximum recommended daily dose is 6 mg (mild) and 4 mg (moderate) once daily at bedtime ( 2.4)
- Severe Hepatic Impairment: Not recommended ( 2.4)
- Severe Renal Impairment or on Hemodialysis: Not recommended ( 2.5)
- Elderly: Increase dose no more frequently than every 2 weeks ( 2.6)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- 2 mg tablets: orange, round, debossed with "2" on one side and plain on the other.
- 4 mg tablets: red, round, debossed with "T 4" on one side and plain on the other.
- 6 mg tablets: pink, round, debossed with "T 6" on one side and plain on the other.
- 8 mg tablets: dark pink, round, debossed with "T 8" on one side and plain on the other.
- 10 mg tablets: green, round, debossed with "T 10" on one side and plain on the other.
- 12 mg tablets: blue, round, debossed with "T 12" on one side and plain on the other.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of perampanel. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Dermatologic: Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) [see Warnings and Precautions (5.5)]
Psychiatric: Acute psychosis, hallucinations, delusions, paranoia, delirium, confusional state, disorientation, memory impairment [see Warnings and Precautions (5.1)] .
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: Based on animal data, may cause fetal harm ( 8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.2 Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including perampanel, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% CI: 1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as 1 week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5 to 100 years) in the clinical trials analyzed.
Table 1 shows absolute and relative risk by indication for all evaluated AEDs.
| Indication | Placebo Patients with Events per 1000 Patients | Drug Patients with Events per 1000 patients | Relative Risk:
Incidence of Events in drug Patients/Incidence in Placebo Patients |
Risk Difference:
Additional Drug Patients with Events per 1000 Patients |
|---|---|---|---|---|
| Epilepsy | 1.0 | 3.4 | 3.5 | 2.4 |
| Psychiatric | 5.7 | 8.5 | 1.5 | 2.9 |
| Other | 1.0 | 1.8 | 1.9 | 0.9 |
| Total | 2.4 | 4.3 | 1.8 | 1.9 |
The relative risk for suicidal thoughts or behavior was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
Anyone considering prescribing perampanel or any other AED must balance the risk of suicidal thoughts or behavior with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
5.6 Withdrawal of Antiepileptic Drugs
There is the potential of increased seizure frequency in patients with seizure disorders when antiepileptic drugs are withdrawn abruptly. Perampanel has a half-life of approximately 105 hours so that even after abrupt cessation, blood levels fall gradually. In epilepsy clinical trials perampanel was withdrawn without down-titration. Although a small number of patients exhibited seizures following discontinuation, the data were not sufficient to allow any recommendations regarding appropriate withdrawal regimens. A gradual withdrawal is generally recommended with antiepileptic drugs, but if withdrawal is a response to adverse events, prompt withdrawal can be considered.
7.3 Alcohol and Other Cns Depressants (7.3 Alcohol and Other CNS Depressants)
The concomitant use of perampanel and CNS depressants including alcohol may increase CNS depression. A pharmacodynamic interaction study in healthy subjects found that the effects of perampanel on complex tasks such as driving ability were additive or supra-additive to the impairment effects of alcohol [see Clinical Pharmacology (12.3)]. Multiple dosing of perampanel 12 mg per day also enhanced the effects of alcohol to interfere with vigilance and alertness, and increased levels of anger, confusion, and depression. These effects may also be seen when perampanel is used in combination with other CNS depressants. Care should be taken when administering perampanel with these agents. Patients should limit activity until they have experience with concomitant use of CNS depressants (e.g., benzodiazepines, narcotics, barbiturates, sedating antihistamines). Advise patients not to drive or operate machinery until they have gained sufficient experience on perampanel to gauge whether it adversely affects these activities.
7.2 Moderate and Strong Cyp3a4 Inducers (7.2 Moderate and Strong CYP3A4 Inducers)
The concomitant use of known moderate and strong CYP3A4 inducers including carbamazepine, phenytoin, or oxcarbazepine with perampanel decreased the plasma levels of perampanel by approximately 50% to 67% [see Clinical Pharmacology (12.3)] . The starting doses for perampanel should be increased in the presence of moderate or strong CYP3A4 inducers [see Dosage and Administration (2.3)] .
When these moderate or strong CYP3A4 inducers are introduced or withdrawn from a patient's treatment regimen, the patient should be closely monitored for clinical response and tolerability. Dose adjustment of perampanel may be necessary [see Dosage and Administration (2.3)].
2.6 Dosage Information for Elderly Patients
In elderly patients, increase dosage no more frequently than every 2 weeks during titration [see Use in Specific Populations (8.5)].
1.2 Primary Generalized Tonic Clonic Seizures (1.2 Primary Generalized Tonic-Clonic Seizures)
Perampanel tablets are indicated as adjunctive therapy for the treatment of primary generalized tonic-clonic seizures in patients with epilepsy 12 years of age and older.
5.1 Serious Psychiatric and Behavioral Reactions
In the controlled partial-onset seizure clinical trials, hostility- and aggression-related adverse reactions occurred in 12% and 20% of patients randomized to receive perampanel at doses of 8 mg and 12 mg per day, respectively, compared to 6% of patients in the placebo group. These effects were dose-related and generally appeared within the first 6 weeks of treatment, although new events continued to be observed through more than 37 weeks. Perampanel-treated patients experienced more hostility- and aggression-related adverse reactions that were serious, severe, and led to dose reduction, interruption, and discontinuation more frequently than placebo-treated patients.
In general, in placebo-controlled partial-onset seizure clinical trials, neuropsychiatric events were reported more frequently in patients being treated with perampanel than in patients taking placebo. These events included irritability, aggression, anger, and anxiety, which occurred in 2% or greater of perampanel-treated patients and twice as frequently as in placebo-treated patients. Other symptoms that occurred with perampanel and were more common than with placebo included belligerence, affect lability, agitation, and physical assault. Some of these events were reported as serious and life-threatening. Homicidal ideation and/or threat were exhibited in 0.1% of 4,368 perampanel-treated patients in controlled and open label trials, including non-epilepsy trials. Homicidal ideation and/or threat have also been reported postmarketing in patients treated with perampanel.
In the partial-onset seizure clinical trials, these events occurred in patients with and without prior psychiatric history, prior aggressive behavior, or concomitant use of medications associated with hostility and aggression. Some patients experienced worsening of their pre-existing psychiatric conditions. Patients with active psychotic disorders and unstable recurrent affective disorders were excluded from the clinical trials. The combination of alcohol and perampanel significantly worsened mood and increased anger. Patients taking perampanel should avoid the use of alcohol [see Drug Interactions (7.3)].
Similar serious psychiatric and behavioral events were observed in the primary generalized tonic-clonic seizure clinical trial.
In healthy volunteers taking perampanel, observed psychiatric events included paranoia, euphoric mood, agitation, anger, mental status changes, and disorientation/confusional state.
In the non-epilepsy trials, psychiatric events that occurred in perampanel-treated patients more often than placebo-treated patients included disorientation, delusion, and paranoia.
In the postmarketing setting, there have been reports of psychosis (acute psychosis, hallucinations, delusions, paranoia) and delirium (delirium, confusional state, disorientation, memory impairment) in patients treated with perampanel [see Adverse Reactions (6.2)] .
Patients, their caregivers, and families should be informed that perampanel may increase the risk of psychiatric events. Patients should be monitored during treatment and for at least 1 month after the last dose of perampanel, and especially when taking higher doses and during the initial few weeks of drug therapy (titration period) or at other times of dose increases. Dose of perampanel should be reduced if these symptoms occur. Permanently discontinue perampanel for persistent severe or worsening psychiatric symptoms or behaviors and refer for psychiatric evaluation.
Principal Display Panel 2 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 2 mg Tablet Bottle Label)
NDC 51672- 4204-6
30 Tablets
Perampanel
Tablets
2 mg
CIII
ATTENTION PHARMACIST:
Dispense the accompanying
Medication Guide to each patient.
Rx only
Principal Display Panel 4 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 4 mg Tablet Bottle Label)
NDC 51672- 4205-6
30 Tablets
Perampanel
Tablets
4 mg
CIII
ATTENTION PHARMACIST:
Dispense the accompanying
Medication Guide to each patient.
Rx only
Principal Display Panel 6 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 6 mg Tablet Bottle Label)
NDC 51672- 4206-6
30 Tablets
Perampanel
Tablets
6 mg
CIII
ATTENTION PHARMACIST:
Dispense the accompanying
Medication Guide to each patient.
Rx only
Principal Display Panel 8 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 8 mg Tablet Bottle Label)
NDC 51672- 4207-6
30 Tablets
Perampanel
Tablets
8 mg
CIII
ATTENTION PHARMACIST:
Dispense the accompanying
Medication Guide to each patient.
Rx only
Principal Display Panel 10 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg Tablet Bottle Label)
NDC 51672- 4208-6
30 Tablets
Perampanel
Tablets
10 mg
CIII
ATTENTION PHARMACIST:
Dispense the accompanying
Medication Guide to each patient.
Rx only
Principal Display Panel 12 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 12 mg Tablet Bottle Label)
NDC 51672- 4209-6
30 Tablets
Perampanel
Tablets
12 mg
CIII
ATTENTION PHARMACIST:
Dispense the accompanying
Medication Guide to each patient.
Rx only
14.2 Primary Generalized Tonic Clonic (pgtc) Seizures (14.2 Primary Generalized Tonic-Clonic (PGTC) Seizures)
The efficacy of perampanel as adjunctive therapy in patients 12 years of age and older with idiopathic generalized epilepsy experiencing primary generalized tonic-clonic seizures was established in one multicenter, randomized, double-blind, placebo-controlled study (Study 4), conducted at 78 sites in 16 countries. Eligible patients on a stable dose of 1 to 3 AEDs experiencing at least 3 primary generalized tonic-clonic seizures during the 8-week baseline period were randomized to either perampanel or placebo. Efficacy was analyzed in 162 patients (perampanel N=81, placebo N=81) who received medication and at least one post-treatment seizure assessment. Patients were titrated over 4 weeks up to a dose of 8 mg per day or the highest tolerated dose and treated for an additional 13 weeks on the last dose level achieved at the end of the titration period. The total treatment period was 17 weeks. Study drug was given once per day.
The primary endpoint was the percent change from baseline in primary generalized tonic-clonic seizure frequency per 28 days during the treatment period as compared to the baseline period. The criterion for statistical significance was p<0.05. Table 6 shows the results of this study. A statistically significant decrease in seizure rate was observed with perampanel compared to placebo.
| Placebo
(N=81) |
Perampanel 8 mg
(N=81) |
|
|---|---|---|
| Percent Reduction During Treatment | 38 | 76
P-value compared to placebo: <0.0001. Statistically significant as compared to placebo based on ANCOVA with treatment and pooled country as factors and the ranked baseline seizure frequency per 28 days as a covariate.
|
Figure 3 shows the proportion of patients with different percent reductions during the maintenance phase over baseline in primary generalized tonic-clonic seizure frequency. Patients in whom the seizure frequency increased are shown at left as "worse." Patients in whom the seizure frequency decreased are shown in the remaining four categories.
Figure 3. Proportion of Patients Exhibiting Different Percent Reductions During the Maintenance Phase Over Baseline in Primary Generalized Tonic-Clonic Seizure Frequency
2.4 Dosage Adjustment in Patients With Hepatic Impairment (2.4 Dosage Adjustment in Patients with Hepatic Impairment)
In patients with mild and moderate hepatic impairment, the starting dose of perampanel is 2 mg once daily. Increase dosage by increments of 2 mg once daily no more frequently than every 2 weeks. The maximum recommended daily dose is 6 mg for patients with mild hepatic impairment and 4 mg for patients with moderate hepatic impairment. Perampanel is not recommended for use in patients with severe hepatic impairment [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
2.5 Dosage Information for Patients With Renal Impairment (2.5 Dosage Information for Patients with Renal Impairment)
Perampanel tablets can be used in patients with moderate renal impairment with close monitoring. A slower titration may be considered, based on clinical response and tolerability. Perampanel tablets are not recommended in patients with severe renal impairment or patients undergoing hemodialysis [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
2.3 Dosage Modifications With Concomitant Use of Moderate Or Strong Cyp3a4 Enzyme Inducers (2.3 Dosage Modifications with Concomitant Use of Moderate or Strong CYP3A4 Enzyme Inducers)
Moderate and strong CYP3A4 inducers, including enzyme-inducing AEDs such as phenytoin, carbamazepine, and oxcarbazepine, cause a reduction in perampanel plasma levels [see Drug Interactions (7.2), Clinical Pharmacology (12.3)] . Therefore, in adults and pediatric patients 4 years of age and older receiving these concomitant enzyme-inducing drugs, the recommended starting dosage of perampanel is 4 mg once daily taken orally at bedtime.
Increase dosage by increments of 2 mg once daily based on individual clinical response and tolerability, no more frequently than at weekly intervals. A maintenance dose has not been established in clinical trials. The highest dose studied in patients on concomitant enzyme-inducing AEDs was 12 mg once daily.
When moderate or strong CYP3A4 inducers are introduced or withdrawn from a patient's treatment regimen, the patient should be closely monitored for clinical response and tolerability. Dose adjustment of perampanel may be necessary.
5.5 Drug Reaction With Eosinophilia and Systemic Symptoms (dress)/multiorgan Hypersensitivity (5.5 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity)
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), also known as Multiorgan hypersensitivity, has been reported in patients taking antiepileptic drugs, including perampanel. DRESS may be fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling, in association with other organ system involvement, such as hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis sometimes resembling an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its expression, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, the patient should be evaluated immediately. Perampanel should be discontinued if an alternative etiology for the signs or symptoms cannot be established.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:28.065436 · Updated: 2026-03-14T22:40:31.369897