Clōsys®
1a994f24-99ee-4b27-aab0-e816931a22c8
34390-5
HUMAN OTC DRUG LABEL
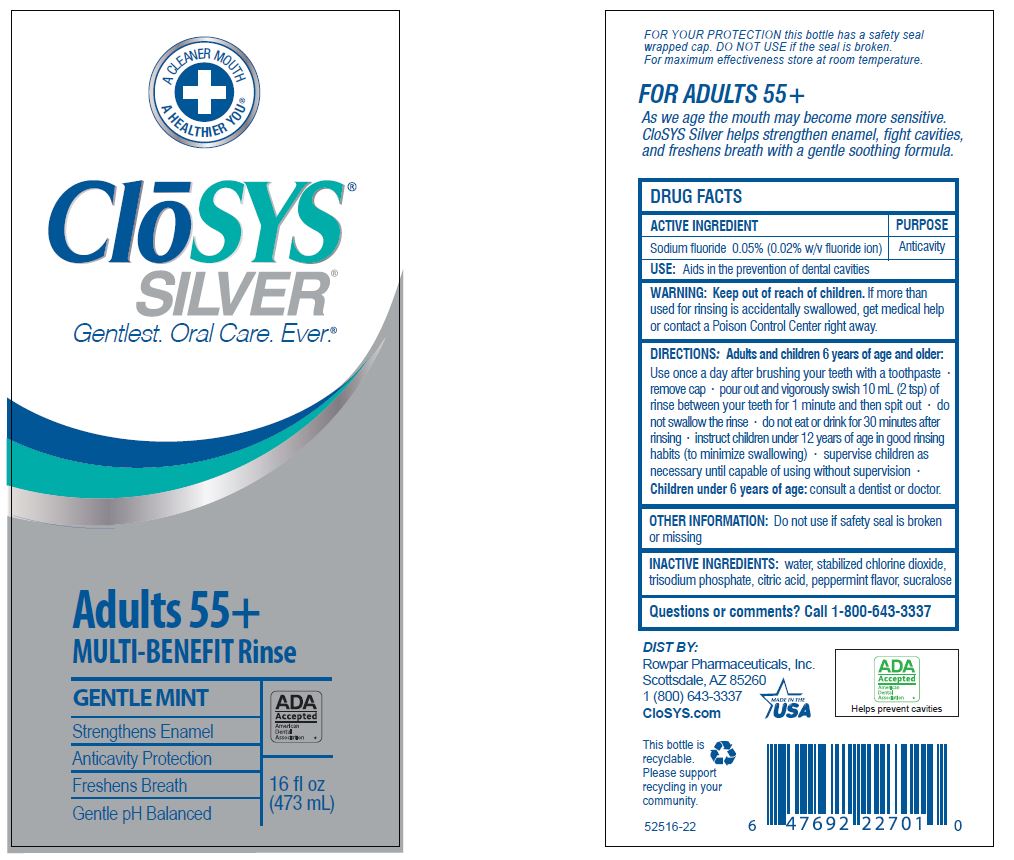
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Sodium fluoride 0.05% (0.02% w/v fluoride ion)
Purpose
Anticavity
Medication Information
Purpose
Anticavity
Description
Sodium fluoride 0.05% (0.02% w/v fluoride ion)
Use:
Aids in the prevention of dental cavities
Section 42229-5
Section 50565-1
Keep out of reach of children. If more than used for rinsing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Section 53413-1
Questions or comments? Call 1-800-643-3337
Packaging
473ml Inner Label
473 mL Carton
100 ml Label
Active Ingredient
Sodium fluoride 0.05% (0.02% w/v fluoride ion)
Other Information:
Do not use if safety seal is broken or missing
Inactive Ingredients:
water, stabilized chlorine dioxide, trisodium phosphate, citric acid, peppermint flavor, sucralose
Directions: Adults and Children 6 Years of Age and Older:
Use once a day after brushing your teeth with a toothpaste · remove cap · pour out and vigorously swish 10 ml (2 tsp) of rinse between your teeth for 1 minute and then spit out · do not swallow the rinse · do not eat or drink for 30 minutes after rinsing • instruct children under 12 years of age in good rinsing habits (to minimize swallowing) · supervise children as necessary until capable of using without supervision ·
Children under 6 years of age: consult a dentist or doctor.
Structured Label Content
Use: (USE:)
Aids in the prevention of dental cavities
Section 42229-5 (42229-5)
Section 50565-1 (50565-1)
Keep out of reach of children. If more than used for rinsing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Section 53413-1 (53413-1)
Questions or comments? Call 1-800-643-3337
Purpose (PURPOSE)
Anticavity
Packaging
473ml Inner Label
473 mL Carton
100 ml Label
Active Ingredient (ACTIVE INGREDIENT)
Sodium fluoride 0.05% (0.02% w/v fluoride ion)
Other Information: (OTHER INFORMATION:)
Do not use if safety seal is broken or missing
Inactive Ingredients: (INACTIVE INGREDIENTS:)
water, stabilized chlorine dioxide, trisodium phosphate, citric acid, peppermint flavor, sucralose
Directions: Adults and Children 6 Years of Age and Older: (DIRECTIONS: Adults and children 6 years of age and older:)
Use once a day after brushing your teeth with a toothpaste · remove cap · pour out and vigorously swish 10 ml (2 tsp) of rinse between your teeth for 1 minute and then spit out · do not swallow the rinse · do not eat or drink for 30 minutes after rinsing • instruct children under 12 years of age in good rinsing habits (to minimize swallowing) · supervise children as necessary until capable of using without supervision ·
Children under 6 years of age: consult a dentist or doctor.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:53.377986 · Updated: 2026-03-14T23:11:51.346073