These Highlights Do Not Include All The Information Needed To Use Nubeqa Safely And Effectively. See Full Prescribing Information For Nubeqa.
1a7cb212-56e4-4b9d-a73d-bfee7fe4735e
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 6/2025 Dosage and Administration ( 2.1 ) 6/2025 Dosage and Administration ( 2.1 ) X/XXXX Warnings and Precautions ( 5.1 , 5.2 ) 6/2025
Indications and Usage
NUBEQA is an androgen receptor inhibitor indicated for the treatment of adult patients with: non-metastatic castration-resistant prostate cancer (nmCRPC). ( 1 ) metastatic castration-sensitive prostate cancer (mCSPC). ( 1 ) metastatic castration-sensitive prostate cancer (mCSPC) in combination with docetaxel. ( 1 )
Dosage and Administration
Recommended Dosage : NUBEQA 600 mg (two 300 mg tablets) administered orally twice daily. Swallow tablets whole. Take NUBEQA with food. ( 2.1 ) For patients with mCSPC treated with NUBEQA in combination with docetaxel, administer the first cycle of docetaxel within 6 weeks after the start of NUBEQA treatment. ( 2.1 ) Patients should also receive a gonadotropin-releasing hormone (GnRH) agonist or antagonist concurrently or have had bilateral orchiectomy. ( 2.1 )
Warnings and Precautions
Ischemic Heart Disease : Optimize management of cardiovascular risk factors. Monitor for signs and symptoms of coronary artery disease. Discontinue NUBEQA for Grade 3-4 events. ( 5.1 ) Seizure : Consider discontinuation of NUBEQA in patients who develop a seizure during treatment. ( 5.2 ) Embryo-Fetal Toxicity : NUBEQA can cause fetal harm and loss of pregnancy. Advise males with female partners of reproductive potential to use effective contraception. ( 5.3 , 8.1 , 8.3 )
Contraindications
None.
Adverse Reactions
In nmCRPC and mCSPC : The most common adverse reactions (>10% with a ≥2% increase over placebo), including laboratory test abnormalities, are increased AST, decreased neutrophil count, increased bilirubin, fatigue, and increased ALT. ( 6.1 ) In mCSPC in combination with docetaxel : The most common adverse reactions (≥10% with a ≥2% increase over placebo) are constipation, rash, decreased appetite, hemorrhage, increased weight, and hypertension. The most common laboratory test abnormalities (≥30%) are anemia, hyperglycemia, decreased lymphocyte count, decreased neutrophil count, increased AST, increased ALT, and hypocalcemia. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Bayer HealthCare Pharmaceuticals Inc. at 1-888-842-2937 or FDA at 1-800-FDA-1088 or WWW.FDA.GOV/MEDWATCH.
Drug Interactions
Combined P-gp and Strong or Moderate CYP3A Inducers : Avoid concomitant use. ( 7.1 ) Combined P-gp and Strong CYP3A Inhibitors : Monitor patients more frequently for NUBEQA adverse reactions. ( 7.1 ) BCRP Substrates : Avoid concomitant use with drugs that are BCRP substrates where possible. If used together, monitor patients more frequently for adverse reactions and consider dose reduction of the BCRP substrate drug. ( 7.2 ) OATP1B1 and OATP1B3 Substrates : Concomitant use of NUBEQA may increase the plasma concentrations of OATP1B1 or OATP1B3 substrates. If used together, monitor patients more frequently for adverse reactions and consider dose reduction of these drugs. ( 7.2 )
Medication Information
Warnings and Precautions
Ischemic Heart Disease : Optimize management of cardiovascular risk factors. Monitor for signs and symptoms of coronary artery disease. Discontinue NUBEQA for Grade 3-4 events. ( 5.1 ) Seizure : Consider discontinuation of NUBEQA in patients who develop a seizure during treatment. ( 5.2 ) Embryo-Fetal Toxicity : NUBEQA can cause fetal harm and loss of pregnancy. Advise males with female partners of reproductive potential to use effective contraception. ( 5.3 , 8.1 , 8.3 )
Indications and Usage
NUBEQA is an androgen receptor inhibitor indicated for the treatment of adult patients with: non-metastatic castration-resistant prostate cancer (nmCRPC). ( 1 ) metastatic castration-sensitive prostate cancer (mCSPC). ( 1 ) metastatic castration-sensitive prostate cancer (mCSPC) in combination with docetaxel. ( 1 )
Dosage and Administration
Recommended Dosage : NUBEQA 600 mg (two 300 mg tablets) administered orally twice daily. Swallow tablets whole. Take NUBEQA with food. ( 2.1 ) For patients with mCSPC treated with NUBEQA in combination with docetaxel, administer the first cycle of docetaxel within 6 weeks after the start of NUBEQA treatment. ( 2.1 ) Patients should also receive a gonadotropin-releasing hormone (GnRH) agonist or antagonist concurrently or have had bilateral orchiectomy. ( 2.1 )
Contraindications
None.
Adverse Reactions
In nmCRPC and mCSPC : The most common adverse reactions (>10% with a ≥2% increase over placebo), including laboratory test abnormalities, are increased AST, decreased neutrophil count, increased bilirubin, fatigue, and increased ALT. ( 6.1 ) In mCSPC in combination with docetaxel : The most common adverse reactions (≥10% with a ≥2% increase over placebo) are constipation, rash, decreased appetite, hemorrhage, increased weight, and hypertension. The most common laboratory test abnormalities (≥30%) are anemia, hyperglycemia, decreased lymphocyte count, decreased neutrophil count, increased AST, increased ALT, and hypocalcemia. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Bayer HealthCare Pharmaceuticals Inc. at 1-888-842-2937 or FDA at 1-800-FDA-1088 or WWW.FDA.GOV/MEDWATCH.
Drug Interactions
Combined P-gp and Strong or Moderate CYP3A Inducers : Avoid concomitant use. ( 7.1 ) Combined P-gp and Strong CYP3A Inhibitors : Monitor patients more frequently for NUBEQA adverse reactions. ( 7.1 ) BCRP Substrates : Avoid concomitant use with drugs that are BCRP substrates where possible. If used together, monitor patients more frequently for adverse reactions and consider dose reduction of the BCRP substrate drug. ( 7.2 ) OATP1B1 and OATP1B3 Substrates : Concomitant use of NUBEQA may increase the plasma concentrations of OATP1B1 or OATP1B3 substrates. If used together, monitor patients more frequently for adverse reactions and consider dose reduction of these drugs. ( 7.2 )
Description
Indications and Usage ( 1 ) 6/2025 Dosage and Administration ( 2.1 ) 6/2025 Dosage and Administration ( 2.1 ) X/XXXX Warnings and Precautions ( 5.1 , 5.2 ) 6/2025
Section 42229-5
NUBEQA is indicated for the treatment of adult patients with:
- non-metastatic castration resistant prostate cancer (nmCRPC)
- metastatic castration-sensitive prostate cancer (mCSPC)
- metastatic castration-sensitive prostate cancer (mCSPC) in combination with docetaxel.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: X/202X |
|
PATIENT INFORMATION NUBEQA® (NOO-bə-kə) (darolutamide) tablets |
|
|
What is NUBEQA?
NUBEQA is a prescription medicine used to treat adults with prostate cancer:
|
|
Before taking NUBEQA, tell your healthcare provider about all your medical conditions, including if you:
You should not start or stop any medicine before you talk with the healthcare provider that prescribed NUBEQA. Know the medicines you take. Keep a list of them with you to show to your healthcare provider and pharmacist when you get a new medicine. |
|
How should I take NUBEQA?
|
|
|
What are the possible side effects of NUBEQA?
NUBEQA may cause serious side effects, including:
|
|
| The most common side effects of NUBEQA include: | |
|
|
|
The most common side effects of NUBEQA in combination with docetaxel include: |
|
|
|
| NUBEQA may cause fertility problems in males, which may affect the ability to father children. Talk to your healthcare provider if you have concerns about fertility. These are not all the possible side effects of NUBEQA. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store NUBEQA?
|
|
|
General information about the safe and effective use of NUBEQA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use NUBEQA for a condition for which it was not prescribed. Do not give NUBEQA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about NUBEQA that is written for health professionals. |
|
|
What are the ingredients in NUBEQA? Active ingredient: darolutamide Inactive ingredients: calcium hydrogen phosphate, croscarmellose sodium, lactose monohydrate, magnesium stearate, povidone K 30, hypromellose 15 cP, macrogol 3350, and titanium dioxide. Manufactured by: Orion Corporation, Orion Pharma, FI-02101 Espoo, Finland Manufactured for: Bayer HealthCare Pharmaceuticals Inc., Whippany, NJ 07981 USA © 2019 Bayer HealthCare Pharmaceuticals Inc. For more information, call Bayer HealthCare Pharmaceuticals Inc. at Bayer at 1-888-842-2937 or go to www.NUBEQA-us.com |
Section 43683-2
Section 44425-7
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature].
Keep the bottle tightly closed after first opening.
5.2 Seizure
Seizure occurred in patients receiving NUBEQA.
In a pooled analysis of ARAMIS and ARANOTE, Grade 1-3 seizure occurred in 0.2% of patients receiving NUBEQA. Seizure occurred from 261 to 665 days after initiation of NUBEQA.
In ARASENS, seizure occurred in 0.8% of patients receiving NUBEQA with docetaxel, including two Grade 3 events. Seizure occurred from 38 to 1754 days after initiation of NUBEQA.
It is unknown whether anti-epileptic medications will prevent seizures with NUBEQA. Advise patients of the risk of developing a seizure while receiving NUBEQA and of engaging in any activity where sudden loss of consciousness could cause harm to themselves or others. Consider discontinuation of NUBEQA in patients who develop a seizure during treatment.
10 Overdosage
There is no known specific antidote for darolutamide overdose. The highest dose of NUBEQA studied clinically was 900 mg twice daily, equivalent to a total daily dose of 1800 mg. No dose limiting toxicities were observed with this dose.
Considering the saturable absorption and the absence of evidence for acute toxicity, an intake of a higher than recommended dose of darolutamide is not expected to lead to systemic toxicity in patients with intact hepatic and renal function [see Clinical Pharmacology (12.3)].
In the event of intake of a higher than recommended dose in patients with severe renal impairment or moderate hepatic impairment, if there is suspicion of toxicity, interrupt NUBEQA treatment and undertake general supportive measures until clinical toxicity has been diminished or resolved. If there is no suspicion of toxicity, NUBEQA treatment can be continued with the next dose as scheduled.
11 Description
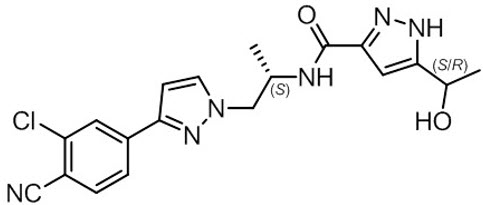
NUBEQA is an androgen receptor inhibitor. The chemical name is N-{(2S)-1-[3-(3-chloro-4-cyanophenyl)-1H-pyrazol-1-yl]propan-2-yl}-5-(1-hydroxyethyl)-1H-pyrazole-3-carboxamide.
The molecular weight is 398.85 and the molecular formula is C19H19Cl N6O2. The structural formula is:
Darolutamide is an optically active with a specific rotation value [α]20 D= 72.2 o*mL/(dm*g), white to greyish- or yellowish white crystalline powder, that is soluble in tetrahydrofuran, but practically insoluble in aqueous medium. Darolutamide has a pKa of 11.75.
NUBEQA (darolutamide) is supplied as film-coated tablets containing 300 mg of darolutamide for oral use. The inactive ingredients of the tablet are: calcium hydrogen phosphate, croscarmellose sodium, lactose monohydrate, magnesium stearate, povidone K 30, hypromellose 15 cP, macrogol 3350, and titanium dioxide.
8.4 Pediatric Use
Safety and effectiveness of NUBEQA in pediatric patients have not been established.
8.5 Geriatric Use
Of the 954 patients who received NUBEQA in ARAMIS, 88% of patients were 65 years and over, and 49% were 75 years and over. Of the 445 patients who received NUBEQA in ARANOTE, 74% of patients were 65 years and over, and 30% were 75 years and over. Of the 652 patients who received NUBEQA in ARASENS, 63% of patients were 65 years and over, and 16% were 75 years and over. No overall differences in safety or efficacy were observed between these patients and younger patients in both studies.
4 Contraindications
None.
6 Adverse Reactions
In nmCRPC and mCSPC: The most common adverse reactions (>10% with a ≥2% increase over placebo), including laboratory test abnormalities, are increased AST, decreased neutrophil count, increased bilirubin, fatigue, and increased ALT. (6.1)
In mCSPC in combination with docetaxel: The most common adverse reactions (≥10% with a ≥2% increase over placebo) are constipation, rash, decreased appetite, hemorrhage, increased weight, and hypertension.
The most common laboratory test abnormalities (≥30%) are anemia, hyperglycemia, decreased lymphocyte count, decreased neutrophil count, increased AST, increased ALT, and hypocalcemia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Bayer HealthCare Pharmaceuticals Inc. at 1-888-842-2937 or FDA at 1-800-FDA-1088 or WWW.FDA.GOV/MEDWATCH.
7 Drug Interactions
- Combined P-gp and Strong or Moderate CYP3A Inducers: Avoid concomitant use. (7.1)
- Combined P-gp and Strong CYP3A Inhibitors: Monitor patients more frequently for NUBEQA adverse reactions. (7.1 )
- BCRP Substrates: Avoid concomitant use with drugs that are BCRP substrates where possible. If used together, monitor patients more frequently for adverse reactions and consider dose reduction of the BCRP substrate drug. (7.2 )
- OATP1B1 and OATP1B3 Substrates: Concomitant use of NUBEQA may increase the plasma concentrations of OATP1B1 or OATP1B3 substrates. If used together, monitor patients more frequently for adverse reactions and consider dose reduction of these drugs. (7.2 )
8.6 Renal Impairment
Patients with severe renal impairment (eGFR 15–29 mL/min/1.73 m2) who are not receiving hemodialysis have a higher exposure to NUBEQA and reduction of the dose is recommended [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)]. No dose reduction is needed for patients with mild or moderate renal impairment (eGFR 30-89 mL/min/1.73 m2). The effect of end stage renal disease (eGFR ≤15 mL/min/1.73 m2) on darolutamide pharmacokinetics is unknown.
12.2 Pharmacodynamics
PSA reduction was observed in CRPC patients receiving darolutamide doses of 100-900 mg twice a day, reaching a plateau of PSA reduction at the 600 mg twice daily dose.
Twice daily dosing of 600 mg darolutamide in nmCRPC patients resulted in undetectable PSA levels in 24% of patients at 12 months compared to 0.4% of patients in the placebo arm.
Twice daily dosing of 600 mg darolutamide in mCSPC patients resulted in undetectable PSA levels (<0.2 ng/mL) in 63% of patients compared to 19% of patients in the placebo arm.
Twice daily dosing of 600 mg darolutamide in combination with docetaxel in mCSPC patients resulted in undetectable PSA levels in 60% of patients at 12 months compared to 26% of patients in the placebo with docetaxel arm.
12.3 Pharmacokinetics
Following administration of 600 mg twice daily, darolutamide mean (%CV) steady-state peak plasma concentration (Cmax) is 4.79 mg/L (30.9%) and area under the plasma concentration-time curve from time 0 to 12 hours (AUC12h) is 52.82 h∙µg/mL (33.9%). Steady-state is reached 2–5 days after repeated dosing with food, with an approximate 2-fold accumulation.
The exposure (Cmax and AUC12) of the darolutamide and the active metabolite ketodarolutamide increase in a nearly dose-proportional manner in the dose range of 100 to 700 mg (0.17 to 1.17 times the approved recommended dosage). No further increase in darolutamide exposure was observed at 900 mg twice daily (1.5 times the approved recommended dosage).
2.1 Recommended Dosage
The recommended dose of NUBEQA is 600 mg (two 300 mg tablets) taken orally, twice daily, with food [see Clinical Pharmacology (12.3)].
Continue treatment until disease progression or unacceptable toxicity occurs.
Patients receiving NUBEQA should also receive a gonadotropin-releasing hormone (GnRH) agonist or antagonist concurrently or have had a bilateral orchiectomy.
When used in combination with docetaxel for mCSPC, administer the first of 6 cycles of docetaxel within 6 weeks after the start of NUBEQA treatment. Refer to docetaxel prescribing information for additional dosing information, including dosage modifications. Treatment with NUBEQA may be continued until disease progression or unacceptable toxicity, even if a cycle of docetaxel is delayed, interrupted, or discontinued [see Dosage and Administration (2.2)].
Advise patients to swallow tablets whole with food, to take any missed dose as soon as they remember. A missed dose may be taken together with the next scheduled dose. Thereafter, resume the regularly scheduled twice daily dosing.
8.7 Hepatic Impairment
Patients with moderate hepatic impairment (Child-Pugh Class B) have a higher exposure to NUBEQA and reduction of the dose is recommended [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)]. No dose reduction is needed for patients with mild hepatic impairment. The effect of severe hepatic impairment (Child-Pugh C) on darolutamide pharmacokinetics is unknown.
1 Indications and Usage
NUBEQA is an androgen receptor inhibitor indicated for the treatment of adult patients with:
12.1 Mechanism of Action
Darolutamide is an androgen receptor (AR) inhibitor. Darolutamide competitively inhibits androgen binding, AR nuclear translocation, and AR-mediated transcription. A major metabolite, ketodarolutamide, exhibited similar in vitro activity to darolutamide. In addition, darolutamide functioned as a progesterone receptor (PR) antagonist in vitro (approximately 1% activity compared to AR). Darolutamide decreased prostate cancer cell proliferation in vitro and tumor volume in mouse xenograft models of prostate cancer.
5.3 Embryo Fetal Toxicity
The safety and efficacy of NUBEQA have not been established in females. Based on its mechanism of action, NUBEQA can cause fetal harm and loss of pregnancy when administered to a pregnant female [see Clinical Pharmacology (12.1)].
Advise males with female partners of reproductive potential to use effective contraception during treatment and for 1 week after the last dose of NUBEQA [see Use in Specific Populations (8.1, 8.3)].
2.2 Dosage Modification
If a patient experiences a greater than or equal to Grade 3 or an intolerable adverse reaction, withhold NUBEQA or reduce dosage to 300 mg twice daily until symptoms improve. NUBEQA may be resumed at a dose of 600 mg twice daily, when adverse reaction returns to baseline [see Adverse Reactions (6.1)]. Dosage reduction below 300 mg twice daily is not recommended.
For patients who experience ischemic heart disease or seizure, additional dose modifications may be required [see Warnings and Precautions (5.1 and 5.2)].
5 Warnings and Precautions
- Ischemic Heart Disease: Optimize management of cardiovascular risk factors. Monitor for signs and symptoms of coronary artery disease. Discontinue NUBEQA for Grade 3-4 events. (5.1)
- Seizure: Consider discontinuation of NUBEQA in patients who develop a seizure during treatment. (5.2)
- Embryo-Fetal Toxicity: NUBEQA can cause fetal harm and loss of pregnancy. Advise males with female partners of reproductive potential to use effective contraception. (5.3, 8.1, 8.3)
5.1 Ischemic Heart Disease
Ischemic heart disease, including fatal cases, occurred in patients receiving NUBEQA.
In a pooled analysis of ARAMIS and ARANOTE, ischemic heart disease occurred in 3.4% of patients receiving NUBEQA and 2.2% receiving placebo, including Grade 3-4 events in 1.4% and 0.3%, respectively. Ischemic events led to death in 0.4% of patients receiving NUBEQA and 0.4% receiving placebo.
In ARASENS, ischemic heart disease occurred in 3.2% of patients receiving NUBEQA with docetaxel and 2% receiving placebo with docetaxel, including Grade 3-4 events in 1.3% and 1.1%, respectively. Ischemic events led to death in 0.3% of patients receiving NUBEQA with docetaxel and 0% receiving placebo with docetaxel.
Monitor for signs and symptoms of ischemic heart disease. Optimize management of cardiovascular risk factors, such as hypertension, diabetes, or dyslipidemia. Discontinue NUBEQA for Grade 3-4 ischemic heart disease.
2 Dosage and Administration
Recommended Dosage: NUBEQA 600 mg (two 300 mg tablets) administered orally twice daily. Swallow tablets whole. Take NUBEQA with food. (2.1)
For patients with mCSPC treated with NUBEQA in combination with docetaxel, administer the first cycle of docetaxel within 6 weeks after the start of NUBEQA treatment. (2.1)
Patients should also receive a gonadotropin-releasing hormone (GnRH) agonist or antagonist concurrently or have had bilateral orchiectomy. (2.1)
3 Dosage Forms and Strengths
Tablets (300 mg): white to off-white oval film-coated tablets marked with "300" on one side and "Bayer" on the other.
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The pooled data in WARNINGS and PRECAUTIONS reflect two randomized clinical trials [ARAMIS, ARANOTE] in patients with nmCRPC (N = 954) and mCSPC (N = 445) treated with NUBEQA. In these trials, the median duration of treatment was 18.2 months (range 0.03 to 44.3) for patients who received NUBEQA [see Clinical Studies (14) ]. In this pooled safety population, the most common adverse reactions (>10% with a ≥2% increase over placebo), including laboratory test abnormalities were increased AST, decreased neutrophil count, increased bilirubin, fatigue and increased ALT.
The safety of NUBEQA in combination with docetaxel in mCSPC is based on data from 1302 patients of whom 652 received at least one dose of NUBEQA in the ARASENS study [see Clinical Studies (14)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information)
Package/label Principal Display Panel
50419-395-01
Rx Only
NUBEQA
(darolutamide) tablets
300 mg
120 film-coated tablets
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Oral administration of darolutamide to male and female RasH2 transgenic mice for 26 weeks did not show carcinogenic potential at doses up to 1000 mg/kg/day.
Darolutamide was clastogenic in an in vitro chromosome aberration assay in human peripheral blood lymphocytes. Darolutamide did not induce mutations in the bacterial reverse mutation (Ames) assay and was not genotoxic in the in vivo combined bone marrow micronucleus assay and the Comet assay in the liver and duodenum of the rat.
Fertility studies in animals have not been conducted with darolutamide. In repeat-dose toxicity studies in male rats (up to 26 weeks) and dogs (up to 39 weeks), tubular dilatation of testes, hypospermia, and atrophy of seminal vesicles, testes, prostate gland and epididymides were observed at doses ≥ 100 mg/kg/day in rats (0.6 times the human exposure based on AUC) and ≥ 50 mg/kg/day in dogs (approximately 1 times the human exposure based on AUC).
2.3 Recommended Dosage in Patients With Severe Renal Impairment
For patients with severe renal impairment (eGFR 15–29 mL/min/1.73 m2) not receiving hemodialysis, the recommended dose of NUBEQA is 300 mg twice daily [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
2.4 Recommended Dosage in Patients With Moderate Hepatic Impairment
For patients with moderate hepatic impairment (Child-Pugh Class B), the recommended dose of NUBEQA is 300 mg twice daily [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
Structured Label Content
Section 42229-5 (42229-5)
NUBEQA is indicated for the treatment of adult patients with:
- non-metastatic castration resistant prostate cancer (nmCRPC)
- metastatic castration-sensitive prostate cancer (mCSPC)
- metastatic castration-sensitive prostate cancer (mCSPC) in combination with docetaxel.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: X/202X |
|
PATIENT INFORMATION NUBEQA® (NOO-bə-kə) (darolutamide) tablets |
|
|
What is NUBEQA?
NUBEQA is a prescription medicine used to treat adults with prostate cancer:
|
|
Before taking NUBEQA, tell your healthcare provider about all your medical conditions, including if you:
You should not start or stop any medicine before you talk with the healthcare provider that prescribed NUBEQA. Know the medicines you take. Keep a list of them with you to show to your healthcare provider and pharmacist when you get a new medicine. |
|
How should I take NUBEQA?
|
|
|
What are the possible side effects of NUBEQA?
NUBEQA may cause serious side effects, including:
|
|
| The most common side effects of NUBEQA include: | |
|
|
|
The most common side effects of NUBEQA in combination with docetaxel include: |
|
|
|
| NUBEQA may cause fertility problems in males, which may affect the ability to father children. Talk to your healthcare provider if you have concerns about fertility. These are not all the possible side effects of NUBEQA. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store NUBEQA?
|
|
|
General information about the safe and effective use of NUBEQA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use NUBEQA for a condition for which it was not prescribed. Do not give NUBEQA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about NUBEQA that is written for health professionals. |
|
|
What are the ingredients in NUBEQA? Active ingredient: darolutamide Inactive ingredients: calcium hydrogen phosphate, croscarmellose sodium, lactose monohydrate, magnesium stearate, povidone K 30, hypromellose 15 cP, macrogol 3350, and titanium dioxide. Manufactured by: Orion Corporation, Orion Pharma, FI-02101 Espoo, Finland Manufactured for: Bayer HealthCare Pharmaceuticals Inc., Whippany, NJ 07981 USA © 2019 Bayer HealthCare Pharmaceuticals Inc. For more information, call Bayer HealthCare Pharmaceuticals Inc. at Bayer at 1-888-842-2937 or go to www.NUBEQA-us.com |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature].
Keep the bottle tightly closed after first opening.
5.2 Seizure
Seizure occurred in patients receiving NUBEQA.
In a pooled analysis of ARAMIS and ARANOTE, Grade 1-3 seizure occurred in 0.2% of patients receiving NUBEQA. Seizure occurred from 261 to 665 days after initiation of NUBEQA.
In ARASENS, seizure occurred in 0.8% of patients receiving NUBEQA with docetaxel, including two Grade 3 events. Seizure occurred from 38 to 1754 days after initiation of NUBEQA.
It is unknown whether anti-epileptic medications will prevent seizures with NUBEQA. Advise patients of the risk of developing a seizure while receiving NUBEQA and of engaging in any activity where sudden loss of consciousness could cause harm to themselves or others. Consider discontinuation of NUBEQA in patients who develop a seizure during treatment.
10 Overdosage (10 OVERDOSAGE)
There is no known specific antidote for darolutamide overdose. The highest dose of NUBEQA studied clinically was 900 mg twice daily, equivalent to a total daily dose of 1800 mg. No dose limiting toxicities were observed with this dose.
Considering the saturable absorption and the absence of evidence for acute toxicity, an intake of a higher than recommended dose of darolutamide is not expected to lead to systemic toxicity in patients with intact hepatic and renal function [see Clinical Pharmacology (12.3)].
In the event of intake of a higher than recommended dose in patients with severe renal impairment or moderate hepatic impairment, if there is suspicion of toxicity, interrupt NUBEQA treatment and undertake general supportive measures until clinical toxicity has been diminished or resolved. If there is no suspicion of toxicity, NUBEQA treatment can be continued with the next dose as scheduled.
11 Description (11 DESCRIPTION)
NUBEQA is an androgen receptor inhibitor. The chemical name is N-{(2S)-1-[3-(3-chloro-4-cyanophenyl)-1H-pyrazol-1-yl]propan-2-yl}-5-(1-hydroxyethyl)-1H-pyrazole-3-carboxamide.
The molecular weight is 398.85 and the molecular formula is C19H19Cl N6O2. The structural formula is:
Darolutamide is an optically active with a specific rotation value [α]20 D= 72.2 o*mL/(dm*g), white to greyish- or yellowish white crystalline powder, that is soluble in tetrahydrofuran, but practically insoluble in aqueous medium. Darolutamide has a pKa of 11.75.
NUBEQA (darolutamide) is supplied as film-coated tablets containing 300 mg of darolutamide for oral use. The inactive ingredients of the tablet are: calcium hydrogen phosphate, croscarmellose sodium, lactose monohydrate, magnesium stearate, povidone K 30, hypromellose 15 cP, macrogol 3350, and titanium dioxide.
8.4 Pediatric Use
Safety and effectiveness of NUBEQA in pediatric patients have not been established.
8.5 Geriatric Use
Of the 954 patients who received NUBEQA in ARAMIS, 88% of patients were 65 years and over, and 49% were 75 years and over. Of the 445 patients who received NUBEQA in ARANOTE, 74% of patients were 65 years and over, and 30% were 75 years and over. Of the 652 patients who received NUBEQA in ARASENS, 63% of patients were 65 years and over, and 16% were 75 years and over. No overall differences in safety or efficacy were observed between these patients and younger patients in both studies.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
In nmCRPC and mCSPC: The most common adverse reactions (>10% with a ≥2% increase over placebo), including laboratory test abnormalities, are increased AST, decreased neutrophil count, increased bilirubin, fatigue, and increased ALT. (6.1)
In mCSPC in combination with docetaxel: The most common adverse reactions (≥10% with a ≥2% increase over placebo) are constipation, rash, decreased appetite, hemorrhage, increased weight, and hypertension.
The most common laboratory test abnormalities (≥30%) are anemia, hyperglycemia, decreased lymphocyte count, decreased neutrophil count, increased AST, increased ALT, and hypocalcemia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Bayer HealthCare Pharmaceuticals Inc. at 1-888-842-2937 or FDA at 1-800-FDA-1088 or WWW.FDA.GOV/MEDWATCH.
7 Drug Interactions (7 DRUG INTERACTIONS)
- Combined P-gp and Strong or Moderate CYP3A Inducers: Avoid concomitant use. (7.1)
- Combined P-gp and Strong CYP3A Inhibitors: Monitor patients more frequently for NUBEQA adverse reactions. (7.1 )
- BCRP Substrates: Avoid concomitant use with drugs that are BCRP substrates where possible. If used together, monitor patients more frequently for adverse reactions and consider dose reduction of the BCRP substrate drug. (7.2 )
- OATP1B1 and OATP1B3 Substrates: Concomitant use of NUBEQA may increase the plasma concentrations of OATP1B1 or OATP1B3 substrates. If used together, monitor patients more frequently for adverse reactions and consider dose reduction of these drugs. (7.2 )
8.6 Renal Impairment
Patients with severe renal impairment (eGFR 15–29 mL/min/1.73 m2) who are not receiving hemodialysis have a higher exposure to NUBEQA and reduction of the dose is recommended [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)]. No dose reduction is needed for patients with mild or moderate renal impairment (eGFR 30-89 mL/min/1.73 m2). The effect of end stage renal disease (eGFR ≤15 mL/min/1.73 m2) on darolutamide pharmacokinetics is unknown.
12.2 Pharmacodynamics
PSA reduction was observed in CRPC patients receiving darolutamide doses of 100-900 mg twice a day, reaching a plateau of PSA reduction at the 600 mg twice daily dose.
Twice daily dosing of 600 mg darolutamide in nmCRPC patients resulted in undetectable PSA levels in 24% of patients at 12 months compared to 0.4% of patients in the placebo arm.
Twice daily dosing of 600 mg darolutamide in mCSPC patients resulted in undetectable PSA levels (<0.2 ng/mL) in 63% of patients compared to 19% of patients in the placebo arm.
Twice daily dosing of 600 mg darolutamide in combination with docetaxel in mCSPC patients resulted in undetectable PSA levels in 60% of patients at 12 months compared to 26% of patients in the placebo with docetaxel arm.
12.3 Pharmacokinetics
Following administration of 600 mg twice daily, darolutamide mean (%CV) steady-state peak plasma concentration (Cmax) is 4.79 mg/L (30.9%) and area under the plasma concentration-time curve from time 0 to 12 hours (AUC12h) is 52.82 h∙µg/mL (33.9%). Steady-state is reached 2–5 days after repeated dosing with food, with an approximate 2-fold accumulation.
The exposure (Cmax and AUC12) of the darolutamide and the active metabolite ketodarolutamide increase in a nearly dose-proportional manner in the dose range of 100 to 700 mg (0.17 to 1.17 times the approved recommended dosage). No further increase in darolutamide exposure was observed at 900 mg twice daily (1.5 times the approved recommended dosage).
2.1 Recommended Dosage
The recommended dose of NUBEQA is 600 mg (two 300 mg tablets) taken orally, twice daily, with food [see Clinical Pharmacology (12.3)].
Continue treatment until disease progression or unacceptable toxicity occurs.
Patients receiving NUBEQA should also receive a gonadotropin-releasing hormone (GnRH) agonist or antagonist concurrently or have had a bilateral orchiectomy.
When used in combination with docetaxel for mCSPC, administer the first of 6 cycles of docetaxel within 6 weeks after the start of NUBEQA treatment. Refer to docetaxel prescribing information for additional dosing information, including dosage modifications. Treatment with NUBEQA may be continued until disease progression or unacceptable toxicity, even if a cycle of docetaxel is delayed, interrupted, or discontinued [see Dosage and Administration (2.2)].
Advise patients to swallow tablets whole with food, to take any missed dose as soon as they remember. A missed dose may be taken together with the next scheduled dose. Thereafter, resume the regularly scheduled twice daily dosing.
8.7 Hepatic Impairment
Patients with moderate hepatic impairment (Child-Pugh Class B) have a higher exposure to NUBEQA and reduction of the dose is recommended [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)]. No dose reduction is needed for patients with mild hepatic impairment. The effect of severe hepatic impairment (Child-Pugh C) on darolutamide pharmacokinetics is unknown.
1 Indications and Usage (1 INDICATIONS AND USAGE)
NUBEQA is an androgen receptor inhibitor indicated for the treatment of adult patients with:
12.1 Mechanism of Action
Darolutamide is an androgen receptor (AR) inhibitor. Darolutamide competitively inhibits androgen binding, AR nuclear translocation, and AR-mediated transcription. A major metabolite, ketodarolutamide, exhibited similar in vitro activity to darolutamide. In addition, darolutamide functioned as a progesterone receptor (PR) antagonist in vitro (approximately 1% activity compared to AR). Darolutamide decreased prostate cancer cell proliferation in vitro and tumor volume in mouse xenograft models of prostate cancer.
5.3 Embryo Fetal Toxicity (5.3 Embryo-Fetal Toxicity)
The safety and efficacy of NUBEQA have not been established in females. Based on its mechanism of action, NUBEQA can cause fetal harm and loss of pregnancy when administered to a pregnant female [see Clinical Pharmacology (12.1)].
Advise males with female partners of reproductive potential to use effective contraception during treatment and for 1 week after the last dose of NUBEQA [see Use in Specific Populations (8.1, 8.3)].
2.2 Dosage Modification
If a patient experiences a greater than or equal to Grade 3 or an intolerable adverse reaction, withhold NUBEQA or reduce dosage to 300 mg twice daily until symptoms improve. NUBEQA may be resumed at a dose of 600 mg twice daily, when adverse reaction returns to baseline [see Adverse Reactions (6.1)]. Dosage reduction below 300 mg twice daily is not recommended.
For patients who experience ischemic heart disease or seizure, additional dose modifications may be required [see Warnings and Precautions (5.1 and 5.2)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Ischemic Heart Disease: Optimize management of cardiovascular risk factors. Monitor for signs and symptoms of coronary artery disease. Discontinue NUBEQA for Grade 3-4 events. (5.1)
- Seizure: Consider discontinuation of NUBEQA in patients who develop a seizure during treatment. (5.2)
- Embryo-Fetal Toxicity: NUBEQA can cause fetal harm and loss of pregnancy. Advise males with female partners of reproductive potential to use effective contraception. (5.3, 8.1, 8.3)
5.1 Ischemic Heart Disease
Ischemic heart disease, including fatal cases, occurred in patients receiving NUBEQA.
In a pooled analysis of ARAMIS and ARANOTE, ischemic heart disease occurred in 3.4% of patients receiving NUBEQA and 2.2% receiving placebo, including Grade 3-4 events in 1.4% and 0.3%, respectively. Ischemic events led to death in 0.4% of patients receiving NUBEQA and 0.4% receiving placebo.
In ARASENS, ischemic heart disease occurred in 3.2% of patients receiving NUBEQA with docetaxel and 2% receiving placebo with docetaxel, including Grade 3-4 events in 1.3% and 1.1%, respectively. Ischemic events led to death in 0.3% of patients receiving NUBEQA with docetaxel and 0% receiving placebo with docetaxel.
Monitor for signs and symptoms of ischemic heart disease. Optimize management of cardiovascular risk factors, such as hypertension, diabetes, or dyslipidemia. Discontinue NUBEQA for Grade 3-4 ischemic heart disease.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Recommended Dosage: NUBEQA 600 mg (two 300 mg tablets) administered orally twice daily. Swallow tablets whole. Take NUBEQA with food. (2.1)
For patients with mCSPC treated with NUBEQA in combination with docetaxel, administer the first cycle of docetaxel within 6 weeks after the start of NUBEQA treatment. (2.1)
Patients should also receive a gonadotropin-releasing hormone (GnRH) agonist or antagonist concurrently or have had bilateral orchiectomy. (2.1)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets (300 mg): white to off-white oval film-coated tablets marked with "300" on one side and "Bayer" on the other.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The pooled data in WARNINGS and PRECAUTIONS reflect two randomized clinical trials [ARAMIS, ARANOTE] in patients with nmCRPC (N = 954) and mCSPC (N = 445) treated with NUBEQA. In these trials, the median duration of treatment was 18.2 months (range 0.03 to 44.3) for patients who received NUBEQA [see Clinical Studies (14) ]. In this pooled safety population, the most common adverse reactions (>10% with a ≥2% increase over placebo), including laboratory test abnormalities were increased AST, decreased neutrophil count, increased bilirubin, fatigue and increased ALT.
The safety of NUBEQA in combination with docetaxel in mCSPC is based on data from 1302 patients of whom 652 received at least one dose of NUBEQA in the ARASENS study [see Clinical Studies (14)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information)
Package/label Principal Display Panel (PACKAGE/LABEL PRINCIPAL DISPLAY PANEL)
50419-395-01
Rx Only
NUBEQA
(darolutamide) tablets
300 mg
120 film-coated tablets
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Oral administration of darolutamide to male and female RasH2 transgenic mice for 26 weeks did not show carcinogenic potential at doses up to 1000 mg/kg/day.
Darolutamide was clastogenic in an in vitro chromosome aberration assay in human peripheral blood lymphocytes. Darolutamide did not induce mutations in the bacterial reverse mutation (Ames) assay and was not genotoxic in the in vivo combined bone marrow micronucleus assay and the Comet assay in the liver and duodenum of the rat.
Fertility studies in animals have not been conducted with darolutamide. In repeat-dose toxicity studies in male rats (up to 26 weeks) and dogs (up to 39 weeks), tubular dilatation of testes, hypospermia, and atrophy of seminal vesicles, testes, prostate gland and epididymides were observed at doses ≥ 100 mg/kg/day in rats (0.6 times the human exposure based on AUC) and ≥ 50 mg/kg/day in dogs (approximately 1 times the human exposure based on AUC).
2.3 Recommended Dosage in Patients With Severe Renal Impairment (2.3 Recommended Dosage in Patients with Severe Renal Impairment)
For patients with severe renal impairment (eGFR 15–29 mL/min/1.73 m2) not receiving hemodialysis, the recommended dose of NUBEQA is 300 mg twice daily [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
2.4 Recommended Dosage in Patients With Moderate Hepatic Impairment (2.4 Recommended Dosage in Patients with Moderate Hepatic Impairment)
For patients with moderate hepatic impairment (Child-Pugh Class B), the recommended dose of NUBEQA is 300 mg twice daily [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:54.771017 · Updated: 2026-03-14T22:51:27.803574