These Highlights Do Not Include All The Information Needed To Use Trintellix®
1a5b68e2-14d0-419d-9ec6-1ca97145e838
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Antidepressants increased the risk of suicidal thoughts and behavior in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.1) ] . TRINTELLIX is not approved for use in pediatric patients [see Use in Specific Populations (8.4) ] .
Indications and Usage
TRINTELLIX is indicated for the treatment of major depressive disorder (MDD) in adults.
Dosage and Administration
The recommended starting dose is 10 mg administered orally once daily without regard to meals ( 2.1 ). The dose should then be increased to 20 mg/day, as tolerated ( 2.1 ). Consider 5 mg/day for patients who do not tolerate higher doses ( 2.1 ). TRINTELLIX can be discontinued abruptly. However, it is recommended that doses of 15 mg/day or 20 mg/day be reduced to 10 mg/day for one week prior to full discontinuation if possible ( 2.3 ). The maximum recommended dose is 10 mg/day in known CYP2D6 poor metabolizers ( 2.5 ).
Warnings and Precautions
Serotonin Syndrome : Increased risk when coadministered with other serotonergic agents, but also when taken alone. If it occurs, discontinue TRINTELLIX and serotonergic agents and initiate supportive measures ( 5.2 ). Increased Risk of Bleeding : Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs, other antiplatelet drugs, warfarin, or other drugs that affect coagulation may increase risk ( 5.3 ). Activation of Mania/Hypomania : Screen patients for bipolar disorder ( 5.4 ). Angle Closure Glaucoma: Angle closure glaucoma has occurred in patients with untreated anatomically narrow angles treated with antidepressants ( 5.6 ). Hyponatremia : Can occur in association with the syndrome of inappropriate antidiuretic hormone secretion (SIADH) ( 5.7 ). Sexual Dysfunction: TRINTELLIX may cause symptoms of sexual dysfunction ( 5.8 ).
Contraindications
Hypersensitivity to vortioxetine or any component of the formulation. Hypersensitivity reactions including anaphylaxis, angioedema, and urticaria have been reported in patients treated with TRINTELLIX [see Adverse Reactions (6.2) ] . The use of MAOIs intended to treat psychiatric disorders with TRINTELLIX or within 21 days of stopping treatment with TRINTELLIX is contraindicated because of an increased risk of serotonin syndrome. The use of TRINTELLIX within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated [see Dosage and Administration (2.4) , Warnings and Precautions (5.2) ] . Starting TRINTELLIX in a patient who is being treated with MAOIs such as linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome [see Warnings and Precautions (5.2) ] .
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label. Hypersensitivity [see Contraindications (4) ] Clinical Worsening and Suicide Risk [see Warnings and Precautions (5.1) ] Serotonin Syndrome [see Warnings and Precautions (5.2) ] Increased Risk of Bleeding [see Warnings and Precautions (5.3) ] Activation of Mania/Hypomania [see Warnings and Precautions (5.4) ] Discontinuation Syndrome [see Warnings and Precautions (5.5) ] Angle Closure Glaucoma [see Warnings and Precautions (5.6) ] Hyponatremia [see Warnings and Precautions (5.7) ]
Drug Interactions
Strong inhibitors of CYP2D6: Reduce TRINTELLIX dose by half when coadministered ( 2.5 , 7.1 ). Strong CYP Inducers: Consider dose increase of TRINTELLIX dose when coadministered for more than 14 days. The maximum recommended dose should not exceed 3 times the original dose ( 2.6 , 7.1 ).
Storage and Handling
TRINTELLIX tablets are available as follows: Features Strengths 5 mg 10 mg 20 mg Color pink yellow red Debossment "5" on one side of tablet "10" on one side of tablet "20" on one side of tablet "TL" on other side of tablet "TL" on other side of tablet "TL" on other side of tablet Presentations and NDC Codes Bottles of 30 64764-720-30 64764-730-30 64764-750-30 Bottles of 90 64764-720-90 64764-730-90 64764-750-90 Bottles of 500 64764-720-77 64764-730-77 64764-750-77
How Supplied
TRINTELLIX tablets are available as follows: Features Strengths 5 mg 10 mg 20 mg Color pink yellow red Debossment "5" on one side of tablet "10" on one side of tablet "20" on one side of tablet "TL" on other side of tablet "TL" on other side of tablet "TL" on other side of tablet Presentations and NDC Codes Bottles of 30 64764-720-30 64764-730-30 64764-750-30 Bottles of 90 64764-720-90 64764-730-90 64764-750-90 Bottles of 500 64764-720-77 64764-730-77 64764-750-77
Medication Information
Warnings and Precautions
Serotonin Syndrome : Increased risk when coadministered with other serotonergic agents, but also when taken alone. If it occurs, discontinue TRINTELLIX and serotonergic agents and initiate supportive measures ( 5.2 ). Increased Risk of Bleeding : Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs, other antiplatelet drugs, warfarin, or other drugs that affect coagulation may increase risk ( 5.3 ). Activation of Mania/Hypomania : Screen patients for bipolar disorder ( 5.4 ). Angle Closure Glaucoma: Angle closure glaucoma has occurred in patients with untreated anatomically narrow angles treated with antidepressants ( 5.6 ). Hyponatremia : Can occur in association with the syndrome of inappropriate antidiuretic hormone secretion (SIADH) ( 5.7 ). Sexual Dysfunction: TRINTELLIX may cause symptoms of sexual dysfunction ( 5.8 ).
Indications and Usage
TRINTELLIX is indicated for the treatment of major depressive disorder (MDD) in adults.
Dosage and Administration
The recommended starting dose is 10 mg administered orally once daily without regard to meals ( 2.1 ). The dose should then be increased to 20 mg/day, as tolerated ( 2.1 ). Consider 5 mg/day for patients who do not tolerate higher doses ( 2.1 ). TRINTELLIX can be discontinued abruptly. However, it is recommended that doses of 15 mg/day or 20 mg/day be reduced to 10 mg/day for one week prior to full discontinuation if possible ( 2.3 ). The maximum recommended dose is 10 mg/day in known CYP2D6 poor metabolizers ( 2.5 ).
Contraindications
Hypersensitivity to vortioxetine or any component of the formulation. Hypersensitivity reactions including anaphylaxis, angioedema, and urticaria have been reported in patients treated with TRINTELLIX [see Adverse Reactions (6.2) ] . The use of MAOIs intended to treat psychiatric disorders with TRINTELLIX or within 21 days of stopping treatment with TRINTELLIX is contraindicated because of an increased risk of serotonin syndrome. The use of TRINTELLIX within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated [see Dosage and Administration (2.4) , Warnings and Precautions (5.2) ] . Starting TRINTELLIX in a patient who is being treated with MAOIs such as linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome [see Warnings and Precautions (5.2) ] .
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label. Hypersensitivity [see Contraindications (4) ] Clinical Worsening and Suicide Risk [see Warnings and Precautions (5.1) ] Serotonin Syndrome [see Warnings and Precautions (5.2) ] Increased Risk of Bleeding [see Warnings and Precautions (5.3) ] Activation of Mania/Hypomania [see Warnings and Precautions (5.4) ] Discontinuation Syndrome [see Warnings and Precautions (5.5) ] Angle Closure Glaucoma [see Warnings and Precautions (5.6) ] Hyponatremia [see Warnings and Precautions (5.7) ]
Drug Interactions
Strong inhibitors of CYP2D6: Reduce TRINTELLIX dose by half when coadministered ( 2.5 , 7.1 ). Strong CYP Inducers: Consider dose increase of TRINTELLIX dose when coadministered for more than 14 days. The maximum recommended dose should not exceed 3 times the original dose ( 2.6 , 7.1 ).
Storage and Handling
TRINTELLIX tablets are available as follows: Features Strengths 5 mg 10 mg 20 mg Color pink yellow red Debossment "5" on one side of tablet "10" on one side of tablet "20" on one side of tablet "TL" on other side of tablet "TL" on other side of tablet "TL" on other side of tablet Presentations and NDC Codes Bottles of 30 64764-720-30 64764-730-30 64764-750-30 Bottles of 90 64764-720-90 64764-730-90 64764-750-90 Bottles of 500 64764-720-77 64764-730-77 64764-750-77
How Supplied
TRINTELLIX tablets are available as follows: Features Strengths 5 mg 10 mg 20 mg Color pink yellow red Debossment "5" on one side of tablet "10" on one side of tablet "20" on one side of tablet "TL" on other side of tablet "TL" on other side of tablet "TL" on other side of tablet Presentations and NDC Codes Bottles of 30 64764-720-30 64764-730-30 64764-750-30 Bottles of 90 64764-720-90 64764-730-90 64764-750-90 Bottles of 500 64764-720-77 64764-730-77 64764-750-77
Description
Antidepressants increased the risk of suicidal thoughts and behavior in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.1) ] . TRINTELLIX is not approved for use in pediatric patients [see Use in Specific Populations (8.4) ] .
Section 42229-5
Patient Exposure
TRINTELLIX was evaluated for safety in 5,852 patients (18 years to 88 years of age) diagnosed with MDD who participated in pre- and postmarketing clinical studies; 2,616 of those patients were exposed to TRINTELLIX in 6 to 8 week, placebo-controlled studies at doses ranging from 5 mg to 20 mg once daily; 204 patients were exposed to TRINTELLIX in a 24 to 64 week placebo-controlled maintenance study at doses of 5 mg to 10 mg once daily; and 429 patients were exposed to TRINTELLIX in a 32 week placebo-controlled maintenance study in the U.S. at doses of 5 mg, 10 mg, and 20 mg, once daily. Patients from the 6 to 8 week studies continued into 12-month open-label studies. A total of 2,586 patients were exposed to at least one dose of TRINTELLIX in open-label studies, 1,727 were exposed to TRINTELLIX for 6 months and 885 were exposed for at least 1 year.
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 2/2025 | ||
|
MEDICATION GUIDE
TRINTELLIX® (trin'-TELL-ix) (vortioxetine) Tablets |
|||
|
What is the most important information I should know about TRINTELLIX? |
|||
TRINTELLIX can cause serious side effects, including:
|
|||
| Call your healthcare provider or get emergency help right away if you have any of the following symptoms, especially if they are new, worse, or worry you: | |||
|
|
|
|
| What is TRINTELLIX? | |||
| TRINTELLIX is a prescription medicine used in adults to treat a certain type of depression called Major Depressive Disorder (MDD). TRINTELLIX has not been shown to be safe and effective for use in children. |
|||
Do not take TRINTELLIX if you:
|
|||
| Ask your healthcare provider or pharmacist if you are not sure if you take an MAOI, including the antibiotic linezolid or intravenous methylene blue. | |||
| Do not start taking an MAOI for at least 21 days after you stop treatment with TRINTELLIX. | |||
Before taking TRINTELLIX, tell your healthcare provider about all your medical conditions, including if you:
|
|||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||
| TRINTELLIX and some other medicines may affect each other causing possible serious side effects. TRINTELLIX may affect the way other medicines work and other medicines may affect the way TRINTELLIX works. | |||
Especially tell your healthcare provider if you take:
|
|||
| Ask your healthcare provider if you are not sure if you are taking any of these medicines. Your healthcare provider can tell you if it is safe to take TRINTELLIX with your other medicines. | |||
| Do not start or stop any other medicines during treatment with TRINTELLIX without talking to your healthcare provider first. Stopping TRINTELLIX suddenly may cause you to have serious side effects. See, "What are the possible side effects of TRINTELLIX?" | |||
| Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine. | |||
How should I take TRINTELLIX?
|
|||
|
What are the possible side effects of TRINTELLIX? |
|||
TRINTELLIX may cause serious side effects, including:
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
|
|
|
|
|||
|
|
||
| In more severe or more sudden cases, signs and symptoms include: | |||
|
|
||
|
|||
|
|
||
| Symptoms in females may include: | |||
|
|
||
| Talk to your healthcare provider if you develop any changes in your sexual function or if you have any questions or concerns about sexual problems during treatment with TRINTELLIX. There may be treatments your healthcare provider can suggest. | |||
|
The most common side effects of TRINTELLIX include nausea, constipation, and vomiting. These are not all the possible side effects of TRINTELLIX. |
|||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||
How should I store TRINTELLIX?
|
|||
| General information about the safe and effective use of TRINTELLIX. | |||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not take TRINTELLIX for a condition for which it was not prescribed. Do not give TRINTELLIX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about TRINTELLIX that is written for health professionals. | |||
|
What are the ingredients in TRINTELLIX?
Active ingredient: vortioxetine hydrobromide Inactive ingredients: mannitol, microcrystalline cellulose, hydroxypropyl cellulose, sodium starch glycolate, magnesium stearate and film coating consisting of hypromellose, titanium dioxide, polyethylene glycol 400, iron oxide red (5 mg and 20 mg), and iron oxide yellow (10 mg) Distributed and Marketed by: Takeda Pharmaceuticals America, Inc. Cambridge, MA 02142 TRINTELLIX is a trademark of H. Lundbeck A/S registered with the U.S. Patent and Trademark Office and used under license by Takeda Pharmaceuticals America, Inc. ©2025 Takeda Pharmaceuticals America, Inc. All rights reserved. For more information, go to www.TRINTELLIX.com or call 1-877-TAKEDA-7 (1-877-825-3327). |
Section 44425-7
Store at 77°F (25°C); excursions permitted to 59°F to 86°F (15°C to 30°C) [see USP Controlled Room Temperature].
10 Overdosage
There is limited clinical trial experience regarding human overdosage with TRINTELLIX. In premarketing clinical studies, cases of overdose were limited to patients who accidentally or intentionally consumed up to a maximum dose of 40 mg of TRINTELLIX. The maximum single dose tested was 75 mg in men. Ingestion of TRINTELLIX in the dose range of 40 to 75 mg was associated with increased rates of nausea, dizziness, diarrhea, abdominal discomfort, generalized pruritus, somnolence, and flushing.
There have been postmarketing reports of overdoses of TRINTELLIX. The most frequently reported symptoms with overdoses up to 80 mg (four times the maximum recommended daily dose) were nausea and vomiting. With overdoses greater than 80 mg, a case of serotonin syndrome in combination with another serotonergic drug, and a case of seizure, have been reported.
No specific antidotes for TRINTELLIX are known. Consider contacting the Poison Help Line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
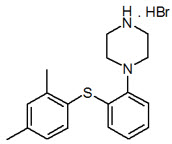
11 Description
TRINTELLIX is an immediate-release tablet for oral administration that contains the beta (β) polymorph of vortioxetine hydrobromide (HBr), an antidepressant. Vortioxetine HBr is known chemically as 1-[2-(2,4-Dimethyl-phenylsulfanyl)-phenyl]-piperazine, hydrobromide. The empirical formula is C18 H22 N2 S, HBr with a molecular weight of 379.36 g/mol. The structural formula is:
Vortioxetine HBr is a white to very slightly beige powder that is slightly soluble in water.
Each TRINTELLIX tablet contains 6.355 mg, 12.71 mg or 25.42 mg of vortioxetine HBr equivalent to 5 mg, 10 mg, or 20 mg of vortioxetine, respectively. The inactive ingredients in TRINTELLIX tablets include mannitol, microcrystalline cellulose, hydroxypropyl cellulose, sodium starch glycolate, magnesium stearate and film coating which consists of hypromellose, titanium dioxide, polyethylene glycol 400, iron oxide red (5 mg and 20 mg) and iron oxide yellow (10 mg).
5.7 Hyponatremia
Hyponatremia has occurred as a result of treatment with serotonergic drugs, including TRINTELLIX. In many cases, hyponatremia appears to be the result of the syndrome of inappropriate antidiuretic hormone secretion (SIADH). One case with serum sodium lower than 110 mmol/L was reported in a subject treated with TRINTELLIX in a premarketing clinical study. Elderly patients may be at greater risk of developing hyponatremia with a serotonergic antidepressant. Also, patients taking diuretics or who are otherwise volume-depleted can be at greater risk. Discontinuation of TRINTELLIX in patients with symptomatic hyponatremia and appropriate medical intervention should be instituted. Signs and symptoms of hyponatremia include headache, difficulty concentrating, memory impairment, confusion, weakness, and unsteadiness, which can lead to falls. More severe and/or acute cases have included hallucination, syncope, seizure, coma, respiratory arrest, and death.
8.4 Pediatric Use
The safety and effectiveness of TRINTELLIX have not been established in pediatric patients.
The safety and efficacy of TRINTELLIX were evaluated in two randomized, double-blind, placebo- and active-controlled 8-week studies in pediatric patients with MDD, one in patients 7 to 11 years of age (N=540 randomized) and one in patients 12 to 17 years of age (N=616 randomized). The primary efficacy endpoint for both studies was the change from baseline to week 8 in the Children's Depression Rating Scale-Revised (CDRS-R) total score. The CDRS-R assesses the severity of depression and change in depressive symptoms in children and adolescents with depression. TRINTELLIX was not superior to placebo in either study. Patients from the controlled 8-week studies were eligible to enroll in a 6-month open-label extension study (N=662 treated). Across the three studies, the most commonly observed adverse reactions to TRINTELLIX in pediatric patients 7 to 17 years of age were generally similar to those observed in adults [see Adverse Reactions (6)].
Antidepressants, such as TRINTELLIX, increase the risk of suicidal thoughts and behaviors in pediatric patients [see Boxed Warning and Warnings and Precautions (5.1)].
8.5 Geriatric Use
No dose adjustment is recommended on the basis of age (Figure 1). Results from a single-dose pharmacokinetic study in elderly (>65 years old) vs young (24 to 45 years old) subjects demonstrated that the pharmacokinetics were generally similar between the two age groups.
Of the 2,616 subjects in clinical studies of TRINTELLIX, 11% (286) were 65 and over, which included subjects from a placebo-controlled study specifically in elderly patients [see Clinical Studies (14)]. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients.
Serotonergic antidepressants have been associated with cases of clinically significant hyponatremia in elderly patients, who may be at greater risk for this adverse event [see Warnings and Precautions (5.7)].
14 Clinical Studies
The efficacy of TRINTELLIX in treatment for MDD was established in six, 6 to 8 week randomized, double-blind, placebo-controlled, fixed-dose studies (including one study in the elderly) and one maintenance study in adult inpatients and outpatients who met the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV-TR) criteria for MDD.
4 Contraindications
- Hypersensitivity to vortioxetine or any component of the formulation. Hypersensitivity reactions including anaphylaxis, angioedema, and urticaria have been reported in patients treated with TRINTELLIX [see Adverse Reactions (6.2)].
- The use of MAOIs intended to treat psychiatric disorders with TRINTELLIX or within 21 days of stopping treatment with TRINTELLIX is contraindicated because of an increased risk of serotonin syndrome. The use of TRINTELLIX within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated [see Dosage and Administration (2.4), Warnings and Precautions (5.2)].
Starting TRINTELLIX in a patient who is being treated with MAOIs such as linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome [see Warnings and Precautions (5.2)].
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label.
- Hypersensitivity [see Contraindications (4)]
- Clinical Worsening and Suicide Risk [see Warnings and Precautions (5.1)]
- Serotonin Syndrome [see Warnings and Precautions (5.2)]
- Increased Risk of Bleeding [see Warnings and Precautions (5.3)]
- Activation of Mania/Hypomania [see Warnings and Precautions (5.4)]
- Discontinuation Syndrome [see Warnings and Precautions (5.5)]
- Angle Closure Glaucoma [see Warnings and Precautions (5.6)]
- Hyponatremia [see Warnings and Precautions (5.7)]
7 Drug Interactions
12.2 Pharmacodynamics
Vortioxetine binds with high affinity to the human serotonin transporter (Ki=1.6 nM), but not to the norepinephrine (Ki=113 nM) or dopamine (Ki>1000 nM) transporters. Vortioxetine potently and selectively inhibits reuptake of serotonin (IC50=5.4 nM). Vortioxetine binds to 5-HT3 (Ki=3.7 nM), 5-HT1A (Ki=15 nM), 5-HT7 (Ki=19 nM), 5-HT1D (Ki=54 nM), and 5-HT1B (Ki=33 nM), receptors and is a 5-HT3, 5-HT1D, and 5-HT7 receptor antagonist, 5-HT1B receptor partial agonist, and 5-HT1A receptor agonist.
In humans, the mean 5-HT transporter occupancy, based on the results from two clinical PET studies using 5-HTT ligands ([11C]-MADAM or [11C]-DASB), was approximately 50% at 5 mg/day, 65% at 10 mg/day and approximately 80% at 20 mg/day in the regions of interest.
12.3 Pharmacokinetics
Vortioxetine pharmacological activity is due to the parent drug. The pharmacokinetics of vortioxetine (2.5 mg to 60 mg) are linear and dose-proportional when vortioxetine is administered once daily. The mean terminal half-life is approximately 66 hours, and steady-state plasma concentrations are typically achieved within two weeks of dosing.
2.1 Recommended Dosage
The recommended starting dose is 10 mg administered orally once daily without regard to meals. Dosage should then be increased to 20 mg/day, as tolerated. The efficacy and safety of doses above 20 mg/day have not been evaluated in controlled clinical trials. A dose decrease down to 5 mg/day may be considered for patients who do not tolerate higher doses [see Clinical Studies (14)].
5.2 Serotonin Syndrome
Serotonergic antidepressants, including TRINTELLIX, can precipitate serotonin syndrome, a potentially life-threatening condition. The risk is increased with concomitant use of other serotonergic drugs (including triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, meperidine, methadone, buspirone, amphetamines, and St. John's Wort) and with drugs that impair metabolism of serotonin, i.e., MAOIs [see Contraindications (4), Drug Interactions (7.1)]. Serotonin syndrome can also occur when these drugs are used alone.
Serotonin syndrome signs and symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea).
The concomitant use of TRINTELLIX with MAOIs is contraindicated. In addition, do not initiate TRINTELLIX in a patient being treated with MAOIs such as linezolid or intravenous methylene blue. No reports involved the administration of methylene blue by other routes (such as oral tablets or local tissue injection). If it is necessary to initiate treatment with an MAOI such as linezolid or intravenous methylene blue in a patient taking TRINTELLIX, discontinue TRINTELLIX before initiating treatment with the MAOI [see Contraindications (4), Drug Interactions (7.1)].
Monitor all patients taking TRINTELLIX for the emergence of serotonin syndrome. Discontinue treatment with TRINTELLIX and any concomitant serotonergic agents immediately if the above symptoms occur, and initiate supportive symptomatic treatment. If concomitant use of TRINTELLIX with other serotonergic drugs is clinically warranted, inform patients of the increased risk for serotonin syndrome and monitor for symptoms.
5.8 Sexual Dysfunction
Use of serotonergic antidepressants, including TRINTELLIX, may cause symptoms of sexual dysfunction [see Adverse Reactions (6.1)]. In male patients, serotonergic antidepressant use may result in ejaculatory delay or failure, decreased libido, and erectile dysfunction. In female patients, use may result in decreased libido and delayed or absent orgasm.
It is important for prescribers to inquire about sexual function prior to initiation of TRINTELLIX and to inquire specifically about changes in sexual function during treatment, because sexual function may not be spontaneously reported. When evaluating changes in sexual function, obtaining a detailed history (including timing of symptom onset) is important because sexual symptoms may have other causes, including the underlying psychiatric disorder. Discuss potential management strategies to support patients in making informed decisions about treatment.
1 Indications and Usage
TRINTELLIX is indicated for the treatment of major depressive disorder (MDD) in adults.
12.1 Mechanism of Action
The mechanism of the antidepressant effect of vortioxetine is not fully understood, but is thought to be related to its enhancement of serotonergic activity in the CNS through inhibition of the reuptake of serotonin (5-HT). It also has several other activities including 5-HT3 receptor antagonism and 5-HT1A receptor agonism. The contribution of these activities to vortioxetine's antidepressant effect has not been established.
5 Warnings and Precautions
- Serotonin Syndrome: Increased risk when coadministered with other serotonergic agents, but also when taken alone. If it occurs, discontinue TRINTELLIX and serotonergic agents and initiate supportive measures (5.2).
- Increased Risk of Bleeding: Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs, other antiplatelet drugs, warfarin, or other drugs that affect coagulation may increase risk (5.3).
- Activation of Mania/Hypomania: Screen patients for bipolar disorder (5.4).
- Angle Closure Glaucoma: Angle closure glaucoma has occurred in patients with untreated anatomically narrow angles treated with antidepressants (5.6).
- Hyponatremia: Can occur in association with the syndrome of inappropriate antidiuretic hormone secretion (SIADH) (5.7).
- Sexual Dysfunction: TRINTELLIX may cause symptoms of sexual dysfunction (5.8).
5.6 Angle Closure Glaucoma
The pupillary dilation that occurs following use of many antidepressant drugs, including TRINTELLIX, may trigger an angle closure attack in a patient with anatomically narrow angles who does not have a patent iridectomy.
2 Dosage and Administration
- The recommended starting dose is 10 mg administered orally once daily without regard to meals (2.1).
- The dose should then be increased to 20 mg/day, as tolerated (2.1).
- Consider 5 mg/day for patients who do not tolerate higher doses (2.1).
- TRINTELLIX can be discontinued abruptly. However, it is recommended that doses of 15 mg/day or 20 mg/day be reduced to 10 mg/day for one week prior to full discontinuation if possible (2.3).
- The maximum recommended dose is 10 mg/day in known CYP2D6 poor metabolizers (2.5).
2.3 Discontinuing Treatment
Although TRINTELLIX can be abruptly discontinued, in placebo-controlled trials patients experienced transient adverse reactions such as headache and muscle tension following abrupt discontinuation of TRINTELLIX 15 mg/day or 20 mg/day. It is recommended that the dose be decreased to 10 mg/day for one week before full discontinuation of TRINTELLIX 15 mg/day or 20 mg/day [see Warnings and Precautions (5.5) and Adverse Reactions (6)].
3 Dosage Forms and Strengths
TRINTELLIX is available as:
- 5 mg: pink, almond shaped biconvex film coated tablet, debossed with "5" on one side and "TL" on the other side
- 10 mg: yellow, almond shaped biconvex film coated tablet, debossed with "10" on one side and "TL" on the other side
- 20 mg: red, almond shaped biconvex film coated tablet, debossed with "20" on one side and "TL" on the other side
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of TRINTELLIX. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Endocrine disorders — hyperprolactinemia
Gastrointestinal System — acute pancreatitis
Immune system disorders — hypersensitivity reactions (including anaphylaxis and urticaria)
Metabolic disorders — weight gain
Nervous system disorders — seizure, headache
Psychiatric disorders — aggression, agitation, anger, hostility, irritability
Respiratory, thoracic and mediastinal disorders — anosmia, hyposmia
Skin and subcutaneous tissue disorders — rash, generalized rash, hyperhidrosis
8.6 Cyp2d6 Poor Metabolizers
Dosage reduction is recommended in patients known to be poor CYP2D6 metabolizers because these patients have higher vortioxetine plasma concentrations than extensive CYP2D6 metabolizers [see Dosage and Administration (2.5) and Clinical Pharmacology (12.3)].
5.5. Discontinuation Syndrome
Adverse reactions have been reported upon abrupt discontinuation of treatment with TRINTELLIX at doses of 15 mg/day and 20 mg/day [see Adverse Reactions (6.1)]. A gradual reduction in dosage rather than abrupt cessation is recommended whenever possible [see Dosage and Administration (2.3)].
Adverse reactions after discontinuation of serotonergic antidepressants, particularly after abrupt discontinuation include: nausea, sweating, dysphoric mood, irritability, agitation, dizziness, sensory disturbances (e.g., paresthesia, such as electric shock sensations), tremor, anxiety, confusion, headache, lethargy, emotional lability, insomnia, hypomania, tinnitus, and seizures.
8 Use in Specific Populations
Pregnancy: Third trimester use may increase risk for persistent pulmonary hypertension and withdrawal in the newborn (8.1).
5.3 Increased Risk of Bleeding
The use of drugs that interfere with serotonin reuptake inhibition, including TRINTELLIX, may increase the risk of bleeding events. Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), warfarin, and other anticoagulants may add to this risk. Case reports and epidemiological studies (case-control and cohort design) have demonstrated an association between use of drugs that interfere with serotonin reuptake and the occurrence of gastrointestinal bleeding. Based on data from the published observational studies, exposure to SSRIs or SNRIs, particularly in the month before delivery, has been associated with a less than 2-fold increase in the risk of postpartum hemorrhage [see Use in Specific Populations (8.1)]. Bleeding events related to drugs that inhibit serotonin reuptake have ranged from ecchymosis, hematoma, epistaxis, and petechiae to life-threatening hemorrhages.
Inform patients about the increased risk of bleeding when TRINTELLIX is coadministered with NSAIDs, aspirin, or other drugs that affect coagulation or bleeding. For patients taking warfarin, carefully monitor coagulation indices when initiating, titrating, or discontinuing TRINTELLIX [see Drug Interactions (7.1)].
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.4 Activation of Mania/hypomania
In patients with bipolar disorder, treating a depressive episode with TRINTELLIX or another antidepressant may precipitate a mixed/manic episode. Symptoms of mania/hypomania were reported in <0.1% of patients treated with TRINTELLIX in premarketing clinical studies. Prior to initiating treatment with TRINTELLIX, screen patients for any personal or family history of bipolar disorder, mania, or hypomania.
16 How Supplied/storage and Handling
TRINTELLIX tablets are available as follows:
| Features | Strengths | ||
|---|---|---|---|
| 5 mg | 10 mg | 20 mg | |
| Color | pink | yellow | red |
| Debossment | "5" on one side of tablet | "10" on one side of tablet | "20" on one side of tablet |
| "TL" on other side of tablet | "TL" on other side of tablet | "TL" on other side of tablet | |
| Presentations and NDC Codes | |||
| Bottles of 30 | 64764-720-30 | 64764-730-30 | 64764-750-30 |
| Bottles of 90 | 64764-720-90 | 64764-730-90 | 64764-750-90 |
| Bottles of 500 | 64764-720-77 | 64764-730-77 | 64764-750-77 |
Warning: Suicidal Thoughts and Behaviors
Antidepressants increased the risk of suicidal thoughts and behavior in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.1)]. TRINTELLIX is not approved for use in pediatric patients [see Use in Specific Populations (8.4)].
Principal Display Panel 5 Mg Tablet Bottle Label
NDC 64764-720-30
30 Tablets
Rx Only
Trintellix®
vortioxetine
tablets
5 mg*
Attention: Dispense with the
accompanying Medication Guide.
Takeda
Principal Display Panel 10 Mg Tablet Bottle Label
NDC 64764-730-30
30 Tablets
Rx Only
Trintellix®
vortioxetine
tablets
10 mg*
Attention: Dispense with the
accompanying Medication Guide.
Takeda
Principal Display Panel 20 Mg Tablet Bottle Label
NDC 64764-750-30
30 Tablets
Rx Only
Trintellix®
vortioxetine
tablets
20 mg*
Attention: Dispense with the
accompanying Medication Guide.
Takeda
2.2 Screen for Bipolar Disorder Prior to Starting Trintellix
Prior to initiating treatment with TRINTELLIX or another antidepressant, screen patients for personal or family history of bipolar disorder, mania, or hypomania [see Warnings and Precautions (5.4)].
2.6 Use of Trintellix in Patients Taking Strong Cyp Inducers
Consider increasing the dose of TRINTELLIX when a strong CYP inducer (e.g., rifampin, carbamazepine, or phenytoin) is coadministered for greater than 14 days. The maximum recommended dose should not exceed three times the original dose. The dose of TRINTELLIX should be reduced to the original level within 14 days, when the inducer is discontinued [see Drug Interactions (7.1)].
7.3 Interference With Urine Enzyme Immunoassays for Methadone
False positive results in urine enzyme immunoassays for methadone have been reported in patients who have taken vortioxetine. An alternative analytical technique (e.g., chromatographic methods) should be considered to confirm positive methadone urine drug screen results.
7.1 Drugs Having Clinically Important Interactions With Trintellix
| Monoamine Oxidase Inhibitors (MAOIs) | |
| Clinical Impact | The concomitant use of SSRIs and SNRIs including TRINTELLIX with MAOIs increases the risk of serotonin syndrome. |
| Intervention | Concomitant use of TRINTELLIX is contraindicated:
|
| Examples | selegiline, tranylcypromine, isocarboxazid, phenelzine, linezolid, methylene blue |
| Other Serotonergic Drugs | |
| Clinical Impact | Concomitant use of TRINTELLIX with other serotonergic drugs increases the risk of serotonin syndrome. |
| Intervention | Monitor for symptoms of serotonin syndrome when TRINTELLIX is used concomitantly with other drugs that may affect the serotonergic neurotransmitter systems. If serotonin syndrome occurs, consider discontinuation of TRINTELLIX and/or concomitant serotonergic drugs [see Warnings and Precautions (5.2)]. |
| Examples | Other SNRIs, SSRIs, triptans, tricyclic antidepressants, opioids, lithium, buspirone, amphetamines, tryptophan, and St. John's Wort |
| Strong Inhibitors of CYP2D6 | |
| Clinical Impact | Concomitant use of TRINTELLIX with strong CYP2D6 inhibitors increases plasma concentrations of vortioxetine. |
| Intervention | Reduce TRINTELLIX dose by half when a strong CYP2D6 inhibitor is coadministered [see Dosage and Administration (2.5)]. |
| Examples | bupropion, fluoxetine, paroxetine, quinidine |
| Strong CYP Inducers | |
| Clinical Impact | Concomitant use of TRINTELLIX with a strong CYP inducer decreases plasma concentrations of vortioxetine. |
| Intervention | Consider increasing the TRINTELLIX dose when a strong CYP inducer is coadministered. The maximum dose is not recommended to exceed three times the original dose [see Dosage and Administration (2.6)]. |
| Examples | rifampin, carbamazepine, phenytoin |
| Drugs that Interfere with Hemostasis (antiplatelets agents and anticoagulants) | |
| Clinical Impact | Concomitant use of TRINTELLIX with an antiplatelet or anticoagulant drug may potentiate the risk of bleeding. |
| Intervention | Inform patients of the increased risk of bleeding associated with the concomitant use of TRINTELLIX and antiplatelet agents and anticoagulants. For patients taking warfarin, carefully monitor the international normalized ratio [see Warnings and Precautions (5.3), Drug Interactions (7.2)]. |
| Examples | aspirin, clopidogrel, heparin, warfarin |
| Drugs Highly Bound to Plasma Protein | |
| Clinical Impact | TRINTELLIX is highly bound to plasma protein. The concomitant use of TRINTELLIX with another drug that is highly bound to plasma protein may increase free concentrations of TRINTELLIX or other tightly-bound drugs in plasma. |
| Intervention | Monitor for adverse reactions and reduce dosage of TRINTELLIX or other protein bound drugs as warranted [see Drug Interactions (7.2)]. |
| Examples | Warfarin |
5.1 Suicidal Thoughts and Behaviors in Adolescents and Young Adults
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients and 4,500 pediatric patients, the incidence of suicidal thoughts and behaviors in antidepressant-treated patients age 24 years and younger was greater than in placebo-treated patients. There was considerable variation in risk of suicidal thoughts and behaviors among drugs, but there was an increased risk identified in young patients for most drugs studied. There were differences in absolute risk of suicidal thoughts and behaviors across the different indications, with the highest incidence in patients with MDD. The drug-placebo differences in the number of cases of suicidal thoughts and behaviors per 1,000 patients treated are provided in Table 1.
| Age Range | Drug-Placebo Difference in Number of Patients with Suicidal Thoughts and Behaviors per 1000 Patients Treated |
|---|---|
| Increases Compared to Placebo | |
| <18 years old | 14 additional patients |
| 18 to 24 years old | 5 additional patients |
| Decreases Compared to Placebo | |
| 25 to 64 years old | 1 fewer patient |
| ≥65 years old | 6 fewer patients |
It is unknown whether the risk of suicidal thoughts and behaviors in adolescents and young adults extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with MDD that the use of antidepressants can delay the recurrence of depression and that depression itself is a risk factor for suicidal thoughts and behaviors.
Monitor all antidepressant-treated patients for all approved populations for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy, and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the healthcare provider. Consider changing the therapeutic regimen, including possibly discontinuing TRINTELLIX, in patients whose depression is persistently worse, or who are experiencing emergent suicidal thoughts and behaviors.
2.5 Use of Trintellix in Known Cyp2d6 Poor Metabolizers Or in Patients Taking Strong Cyp2d6 Inhibitors
The maximum recommended dose of TRINTELLIX is 10 mg/day in known CYP2D6 poor metabolizers. Reduce the dose of TRINTELLIX by one-half when patients are receiving a CYP2D6 strong inhibitor (e.g., bupropion, fluoxetine, paroxetine, or quinidine) concomitantly. The dose should be increased to the original level when the CYP2D6 inhibitor is discontinued [see Drug Interactions (7.1), Use in Specific Populations (8.6)].
2.4 Switching A Patient to Or From A Monoamine Oxidase Inhibitor (maoi) Intended to Treat Psychiatric Disorders
At least 14 days must elapse between discontinuation of a MAOI intended to treat psychiatric disorders and initiation of therapy with TRINTELLIX to avoid the risk of Serotonin Syndrome [see Warnings and Precautions (5.2)]. Conversely, at least 21 days must elapse after stopping TRINTELLIX before starting an MAOI intended to treat psychiatric disorders [see Contraindications (4)].
Structured Label Content
Section 42229-5 (42229-5)
Patient Exposure
TRINTELLIX was evaluated for safety in 5,852 patients (18 years to 88 years of age) diagnosed with MDD who participated in pre- and postmarketing clinical studies; 2,616 of those patients were exposed to TRINTELLIX in 6 to 8 week, placebo-controlled studies at doses ranging from 5 mg to 20 mg once daily; 204 patients were exposed to TRINTELLIX in a 24 to 64 week placebo-controlled maintenance study at doses of 5 mg to 10 mg once daily; and 429 patients were exposed to TRINTELLIX in a 32 week placebo-controlled maintenance study in the U.S. at doses of 5 mg, 10 mg, and 20 mg, once daily. Patients from the 6 to 8 week studies continued into 12-month open-label studies. A total of 2,586 patients were exposed to at least one dose of TRINTELLIX in open-label studies, 1,727 were exposed to TRINTELLIX for 6 months and 885 were exposed for at least 1 year.
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 2/2025 | ||
|
MEDICATION GUIDE
TRINTELLIX® (trin'-TELL-ix) (vortioxetine) Tablets |
|||
|
What is the most important information I should know about TRINTELLIX? |
|||
TRINTELLIX can cause serious side effects, including:
|
|||
| Call your healthcare provider or get emergency help right away if you have any of the following symptoms, especially if they are new, worse, or worry you: | |||
|
|
|
|
| What is TRINTELLIX? | |||
| TRINTELLIX is a prescription medicine used in adults to treat a certain type of depression called Major Depressive Disorder (MDD). TRINTELLIX has not been shown to be safe and effective for use in children. |
|||
Do not take TRINTELLIX if you:
|
|||
| Ask your healthcare provider or pharmacist if you are not sure if you take an MAOI, including the antibiotic linezolid or intravenous methylene blue. | |||
| Do not start taking an MAOI for at least 21 days after you stop treatment with TRINTELLIX. | |||
Before taking TRINTELLIX, tell your healthcare provider about all your medical conditions, including if you:
|
|||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||
| TRINTELLIX and some other medicines may affect each other causing possible serious side effects. TRINTELLIX may affect the way other medicines work and other medicines may affect the way TRINTELLIX works. | |||
Especially tell your healthcare provider if you take:
|
|||
| Ask your healthcare provider if you are not sure if you are taking any of these medicines. Your healthcare provider can tell you if it is safe to take TRINTELLIX with your other medicines. | |||
| Do not start or stop any other medicines during treatment with TRINTELLIX without talking to your healthcare provider first. Stopping TRINTELLIX suddenly may cause you to have serious side effects. See, "What are the possible side effects of TRINTELLIX?" | |||
| Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine. | |||
How should I take TRINTELLIX?
|
|||
|
What are the possible side effects of TRINTELLIX? |
|||
TRINTELLIX may cause serious side effects, including:
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
|
|
|
|
|||
|
|
||
| In more severe or more sudden cases, signs and symptoms include: | |||
|
|
||
|
|||
|
|
||
| Symptoms in females may include: | |||
|
|
||
| Talk to your healthcare provider if you develop any changes in your sexual function or if you have any questions or concerns about sexual problems during treatment with TRINTELLIX. There may be treatments your healthcare provider can suggest. | |||
|
The most common side effects of TRINTELLIX include nausea, constipation, and vomiting. These are not all the possible side effects of TRINTELLIX. |
|||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||
How should I store TRINTELLIX?
|
|||
| General information about the safe and effective use of TRINTELLIX. | |||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not take TRINTELLIX for a condition for which it was not prescribed. Do not give TRINTELLIX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about TRINTELLIX that is written for health professionals. | |||
|
What are the ingredients in TRINTELLIX?
Active ingredient: vortioxetine hydrobromide Inactive ingredients: mannitol, microcrystalline cellulose, hydroxypropyl cellulose, sodium starch glycolate, magnesium stearate and film coating consisting of hypromellose, titanium dioxide, polyethylene glycol 400, iron oxide red (5 mg and 20 mg), and iron oxide yellow (10 mg) Distributed and Marketed by: Takeda Pharmaceuticals America, Inc. Cambridge, MA 02142 TRINTELLIX is a trademark of H. Lundbeck A/S registered with the U.S. Patent and Trademark Office and used under license by Takeda Pharmaceuticals America, Inc. ©2025 Takeda Pharmaceuticals America, Inc. All rights reserved. For more information, go to www.TRINTELLIX.com or call 1-877-TAKEDA-7 (1-877-825-3327). |
Section 44425-7 (44425-7)
Store at 77°F (25°C); excursions permitted to 59°F to 86°F (15°C to 30°C) [see USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
There is limited clinical trial experience regarding human overdosage with TRINTELLIX. In premarketing clinical studies, cases of overdose were limited to patients who accidentally or intentionally consumed up to a maximum dose of 40 mg of TRINTELLIX. The maximum single dose tested was 75 mg in men. Ingestion of TRINTELLIX in the dose range of 40 to 75 mg was associated with increased rates of nausea, dizziness, diarrhea, abdominal discomfort, generalized pruritus, somnolence, and flushing.
There have been postmarketing reports of overdoses of TRINTELLIX. The most frequently reported symptoms with overdoses up to 80 mg (four times the maximum recommended daily dose) were nausea and vomiting. With overdoses greater than 80 mg, a case of serotonin syndrome in combination with another serotonergic drug, and a case of seizure, have been reported.
No specific antidotes for TRINTELLIX are known. Consider contacting the Poison Help Line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
11 Description (11 DESCRIPTION)
TRINTELLIX is an immediate-release tablet for oral administration that contains the beta (β) polymorph of vortioxetine hydrobromide (HBr), an antidepressant. Vortioxetine HBr is known chemically as 1-[2-(2,4-Dimethyl-phenylsulfanyl)-phenyl]-piperazine, hydrobromide. The empirical formula is C18 H22 N2 S, HBr with a molecular weight of 379.36 g/mol. The structural formula is:
Vortioxetine HBr is a white to very slightly beige powder that is slightly soluble in water.
Each TRINTELLIX tablet contains 6.355 mg, 12.71 mg or 25.42 mg of vortioxetine HBr equivalent to 5 mg, 10 mg, or 20 mg of vortioxetine, respectively. The inactive ingredients in TRINTELLIX tablets include mannitol, microcrystalline cellulose, hydroxypropyl cellulose, sodium starch glycolate, magnesium stearate and film coating which consists of hypromellose, titanium dioxide, polyethylene glycol 400, iron oxide red (5 mg and 20 mg) and iron oxide yellow (10 mg).
5.7 Hyponatremia
Hyponatremia has occurred as a result of treatment with serotonergic drugs, including TRINTELLIX. In many cases, hyponatremia appears to be the result of the syndrome of inappropriate antidiuretic hormone secretion (SIADH). One case with serum sodium lower than 110 mmol/L was reported in a subject treated with TRINTELLIX in a premarketing clinical study. Elderly patients may be at greater risk of developing hyponatremia with a serotonergic antidepressant. Also, patients taking diuretics or who are otherwise volume-depleted can be at greater risk. Discontinuation of TRINTELLIX in patients with symptomatic hyponatremia and appropriate medical intervention should be instituted. Signs and symptoms of hyponatremia include headache, difficulty concentrating, memory impairment, confusion, weakness, and unsteadiness, which can lead to falls. More severe and/or acute cases have included hallucination, syncope, seizure, coma, respiratory arrest, and death.
8.4 Pediatric Use
The safety and effectiveness of TRINTELLIX have not been established in pediatric patients.
The safety and efficacy of TRINTELLIX were evaluated in two randomized, double-blind, placebo- and active-controlled 8-week studies in pediatric patients with MDD, one in patients 7 to 11 years of age (N=540 randomized) and one in patients 12 to 17 years of age (N=616 randomized). The primary efficacy endpoint for both studies was the change from baseline to week 8 in the Children's Depression Rating Scale-Revised (CDRS-R) total score. The CDRS-R assesses the severity of depression and change in depressive symptoms in children and adolescents with depression. TRINTELLIX was not superior to placebo in either study. Patients from the controlled 8-week studies were eligible to enroll in a 6-month open-label extension study (N=662 treated). Across the three studies, the most commonly observed adverse reactions to TRINTELLIX in pediatric patients 7 to 17 years of age were generally similar to those observed in adults [see Adverse Reactions (6)].
Antidepressants, such as TRINTELLIX, increase the risk of suicidal thoughts and behaviors in pediatric patients [see Boxed Warning and Warnings and Precautions (5.1)].
8.5 Geriatric Use
No dose adjustment is recommended on the basis of age (Figure 1). Results from a single-dose pharmacokinetic study in elderly (>65 years old) vs young (24 to 45 years old) subjects demonstrated that the pharmacokinetics were generally similar between the two age groups.
Of the 2,616 subjects in clinical studies of TRINTELLIX, 11% (286) were 65 and over, which included subjects from a placebo-controlled study specifically in elderly patients [see Clinical Studies (14)]. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients.
Serotonergic antidepressants have been associated with cases of clinically significant hyponatremia in elderly patients, who may be at greater risk for this adverse event [see Warnings and Precautions (5.7)].
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of TRINTELLIX in treatment for MDD was established in six, 6 to 8 week randomized, double-blind, placebo-controlled, fixed-dose studies (including one study in the elderly) and one maintenance study in adult inpatients and outpatients who met the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV-TR) criteria for MDD.
4 Contraindications (4 CONTRAINDICATIONS)
- Hypersensitivity to vortioxetine or any component of the formulation. Hypersensitivity reactions including anaphylaxis, angioedema, and urticaria have been reported in patients treated with TRINTELLIX [see Adverse Reactions (6.2)].
- The use of MAOIs intended to treat psychiatric disorders with TRINTELLIX or within 21 days of stopping treatment with TRINTELLIX is contraindicated because of an increased risk of serotonin syndrome. The use of TRINTELLIX within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated [see Dosage and Administration (2.4), Warnings and Precautions (5.2)].
Starting TRINTELLIX in a patient who is being treated with MAOIs such as linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome [see Warnings and Precautions (5.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the label.
- Hypersensitivity [see Contraindications (4)]
- Clinical Worsening and Suicide Risk [see Warnings and Precautions (5.1)]
- Serotonin Syndrome [see Warnings and Precautions (5.2)]
- Increased Risk of Bleeding [see Warnings and Precautions (5.3)]
- Activation of Mania/Hypomania [see Warnings and Precautions (5.4)]
- Discontinuation Syndrome [see Warnings and Precautions (5.5)]
- Angle Closure Glaucoma [see Warnings and Precautions (5.6)]
- Hyponatremia [see Warnings and Precautions (5.7)]
7 Drug Interactions (7 DRUG INTERACTIONS)
12.2 Pharmacodynamics
Vortioxetine binds with high affinity to the human serotonin transporter (Ki=1.6 nM), but not to the norepinephrine (Ki=113 nM) or dopamine (Ki>1000 nM) transporters. Vortioxetine potently and selectively inhibits reuptake of serotonin (IC50=5.4 nM). Vortioxetine binds to 5-HT3 (Ki=3.7 nM), 5-HT1A (Ki=15 nM), 5-HT7 (Ki=19 nM), 5-HT1D (Ki=54 nM), and 5-HT1B (Ki=33 nM), receptors and is a 5-HT3, 5-HT1D, and 5-HT7 receptor antagonist, 5-HT1B receptor partial agonist, and 5-HT1A receptor agonist.
In humans, the mean 5-HT transporter occupancy, based on the results from two clinical PET studies using 5-HTT ligands ([11C]-MADAM or [11C]-DASB), was approximately 50% at 5 mg/day, 65% at 10 mg/day and approximately 80% at 20 mg/day in the regions of interest.
12.3 Pharmacokinetics
Vortioxetine pharmacological activity is due to the parent drug. The pharmacokinetics of vortioxetine (2.5 mg to 60 mg) are linear and dose-proportional when vortioxetine is administered once daily. The mean terminal half-life is approximately 66 hours, and steady-state plasma concentrations are typically achieved within two weeks of dosing.
2.1 Recommended Dosage
The recommended starting dose is 10 mg administered orally once daily without regard to meals. Dosage should then be increased to 20 mg/day, as tolerated. The efficacy and safety of doses above 20 mg/day have not been evaluated in controlled clinical trials. A dose decrease down to 5 mg/day may be considered for patients who do not tolerate higher doses [see Clinical Studies (14)].
5.2 Serotonin Syndrome
Serotonergic antidepressants, including TRINTELLIX, can precipitate serotonin syndrome, a potentially life-threatening condition. The risk is increased with concomitant use of other serotonergic drugs (including triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, meperidine, methadone, buspirone, amphetamines, and St. John's Wort) and with drugs that impair metabolism of serotonin, i.e., MAOIs [see Contraindications (4), Drug Interactions (7.1)]. Serotonin syndrome can also occur when these drugs are used alone.
Serotonin syndrome signs and symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea).
The concomitant use of TRINTELLIX with MAOIs is contraindicated. In addition, do not initiate TRINTELLIX in a patient being treated with MAOIs such as linezolid or intravenous methylene blue. No reports involved the administration of methylene blue by other routes (such as oral tablets or local tissue injection). If it is necessary to initiate treatment with an MAOI such as linezolid or intravenous methylene blue in a patient taking TRINTELLIX, discontinue TRINTELLIX before initiating treatment with the MAOI [see Contraindications (4), Drug Interactions (7.1)].
Monitor all patients taking TRINTELLIX for the emergence of serotonin syndrome. Discontinue treatment with TRINTELLIX and any concomitant serotonergic agents immediately if the above symptoms occur, and initiate supportive symptomatic treatment. If concomitant use of TRINTELLIX with other serotonergic drugs is clinically warranted, inform patients of the increased risk for serotonin syndrome and monitor for symptoms.
5.8 Sexual Dysfunction
Use of serotonergic antidepressants, including TRINTELLIX, may cause symptoms of sexual dysfunction [see Adverse Reactions (6.1)]. In male patients, serotonergic antidepressant use may result in ejaculatory delay or failure, decreased libido, and erectile dysfunction. In female patients, use may result in decreased libido and delayed or absent orgasm.
It is important for prescribers to inquire about sexual function prior to initiation of TRINTELLIX and to inquire specifically about changes in sexual function during treatment, because sexual function may not be spontaneously reported. When evaluating changes in sexual function, obtaining a detailed history (including timing of symptom onset) is important because sexual symptoms may have other causes, including the underlying psychiatric disorder. Discuss potential management strategies to support patients in making informed decisions about treatment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
TRINTELLIX is indicated for the treatment of major depressive disorder (MDD) in adults.
12.1 Mechanism of Action
The mechanism of the antidepressant effect of vortioxetine is not fully understood, but is thought to be related to its enhancement of serotonergic activity in the CNS through inhibition of the reuptake of serotonin (5-HT). It also has several other activities including 5-HT3 receptor antagonism and 5-HT1A receptor agonism. The contribution of these activities to vortioxetine's antidepressant effect has not been established.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Serotonin Syndrome: Increased risk when coadministered with other serotonergic agents, but also when taken alone. If it occurs, discontinue TRINTELLIX and serotonergic agents and initiate supportive measures (5.2).
- Increased Risk of Bleeding: Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs, other antiplatelet drugs, warfarin, or other drugs that affect coagulation may increase risk (5.3).
- Activation of Mania/Hypomania: Screen patients for bipolar disorder (5.4).
- Angle Closure Glaucoma: Angle closure glaucoma has occurred in patients with untreated anatomically narrow angles treated with antidepressants (5.6).
- Hyponatremia: Can occur in association with the syndrome of inappropriate antidiuretic hormone secretion (SIADH) (5.7).
- Sexual Dysfunction: TRINTELLIX may cause symptoms of sexual dysfunction (5.8).
5.6 Angle Closure Glaucoma
The pupillary dilation that occurs following use of many antidepressant drugs, including TRINTELLIX, may trigger an angle closure attack in a patient with anatomically narrow angles who does not have a patent iridectomy.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- The recommended starting dose is 10 mg administered orally once daily without regard to meals (2.1).
- The dose should then be increased to 20 mg/day, as tolerated (2.1).
- Consider 5 mg/day for patients who do not tolerate higher doses (2.1).
- TRINTELLIX can be discontinued abruptly. However, it is recommended that doses of 15 mg/day or 20 mg/day be reduced to 10 mg/day for one week prior to full discontinuation if possible (2.3).
- The maximum recommended dose is 10 mg/day in known CYP2D6 poor metabolizers (2.5).
2.3 Discontinuing Treatment
Although TRINTELLIX can be abruptly discontinued, in placebo-controlled trials patients experienced transient adverse reactions such as headache and muscle tension following abrupt discontinuation of TRINTELLIX 15 mg/day or 20 mg/day. It is recommended that the dose be decreased to 10 mg/day for one week before full discontinuation of TRINTELLIX 15 mg/day or 20 mg/day [see Warnings and Precautions (5.5) and Adverse Reactions (6)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
TRINTELLIX is available as:
- 5 mg: pink, almond shaped biconvex film coated tablet, debossed with "5" on one side and "TL" on the other side
- 10 mg: yellow, almond shaped biconvex film coated tablet, debossed with "10" on one side and "TL" on the other side
- 20 mg: red, almond shaped biconvex film coated tablet, debossed with "20" on one side and "TL" on the other side
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of TRINTELLIX. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Endocrine disorders — hyperprolactinemia
Gastrointestinal System — acute pancreatitis
Immune system disorders — hypersensitivity reactions (including anaphylaxis and urticaria)
Metabolic disorders — weight gain
Nervous system disorders — seizure, headache
Psychiatric disorders — aggression, agitation, anger, hostility, irritability
Respiratory, thoracic and mediastinal disorders — anosmia, hyposmia
Skin and subcutaneous tissue disorders — rash, generalized rash, hyperhidrosis
8.6 Cyp2d6 Poor Metabolizers (8.6 CYP2D6 Poor Metabolizers)
Dosage reduction is recommended in patients known to be poor CYP2D6 metabolizers because these patients have higher vortioxetine plasma concentrations than extensive CYP2D6 metabolizers [see Dosage and Administration (2.5) and Clinical Pharmacology (12.3)].
5.5. Discontinuation Syndrome
Adverse reactions have been reported upon abrupt discontinuation of treatment with TRINTELLIX at doses of 15 mg/day and 20 mg/day [see Adverse Reactions (6.1)]. A gradual reduction in dosage rather than abrupt cessation is recommended whenever possible [see Dosage and Administration (2.3)].
Adverse reactions after discontinuation of serotonergic antidepressants, particularly after abrupt discontinuation include: nausea, sweating, dysphoric mood, irritability, agitation, dizziness, sensory disturbances (e.g., paresthesia, such as electric shock sensations), tremor, anxiety, confusion, headache, lethargy, emotional lability, insomnia, hypomania, tinnitus, and seizures.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: Third trimester use may increase risk for persistent pulmonary hypertension and withdrawal in the newborn (8.1).
5.3 Increased Risk of Bleeding
The use of drugs that interfere with serotonin reuptake inhibition, including TRINTELLIX, may increase the risk of bleeding events. Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), warfarin, and other anticoagulants may add to this risk. Case reports and epidemiological studies (case-control and cohort design) have demonstrated an association between use of drugs that interfere with serotonin reuptake and the occurrence of gastrointestinal bleeding. Based on data from the published observational studies, exposure to SSRIs or SNRIs, particularly in the month before delivery, has been associated with a less than 2-fold increase in the risk of postpartum hemorrhage [see Use in Specific Populations (8.1)]. Bleeding events related to drugs that inhibit serotonin reuptake have ranged from ecchymosis, hematoma, epistaxis, and petechiae to life-threatening hemorrhages.
Inform patients about the increased risk of bleeding when TRINTELLIX is coadministered with NSAIDs, aspirin, or other drugs that affect coagulation or bleeding. For patients taking warfarin, carefully monitor coagulation indices when initiating, titrating, or discontinuing TRINTELLIX [see Drug Interactions (7.1)].
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.4 Activation of Mania/hypomania (5.4 Activation of Mania/Hypomania)
In patients with bipolar disorder, treating a depressive episode with TRINTELLIX or another antidepressant may precipitate a mixed/manic episode. Symptoms of mania/hypomania were reported in <0.1% of patients treated with TRINTELLIX in premarketing clinical studies. Prior to initiating treatment with TRINTELLIX, screen patients for any personal or family history of bipolar disorder, mania, or hypomania.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
TRINTELLIX tablets are available as follows:
| Features | Strengths | ||
|---|---|---|---|
| 5 mg | 10 mg | 20 mg | |
| Color | pink | yellow | red |
| Debossment | "5" on one side of tablet | "10" on one side of tablet | "20" on one side of tablet |
| "TL" on other side of tablet | "TL" on other side of tablet | "TL" on other side of tablet | |
| Presentations and NDC Codes | |||
| Bottles of 30 | 64764-720-30 | 64764-730-30 | 64764-750-30 |
| Bottles of 90 | 64764-720-90 | 64764-730-90 | 64764-750-90 |
| Bottles of 500 | 64764-720-77 | 64764-730-77 | 64764-750-77 |
Warning: Suicidal Thoughts and Behaviors (WARNING: SUICIDAL THOUGHTS AND BEHAVIORS)
Antidepressants increased the risk of suicidal thoughts and behavior in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.1)]. TRINTELLIX is not approved for use in pediatric patients [see Use in Specific Populations (8.4)].
Principal Display Panel 5 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 5 mg Tablet Bottle Label)
NDC 64764-720-30
30 Tablets
Rx Only
Trintellix®
vortioxetine
tablets
5 mg*
Attention: Dispense with the
accompanying Medication Guide.
Takeda
Principal Display Panel 10 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg Tablet Bottle Label)
NDC 64764-730-30
30 Tablets
Rx Only
Trintellix®
vortioxetine
tablets
10 mg*
Attention: Dispense with the
accompanying Medication Guide.
Takeda
Principal Display Panel 20 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 20 mg Tablet Bottle Label)
NDC 64764-750-30
30 Tablets
Rx Only
Trintellix®
vortioxetine
tablets
20 mg*
Attention: Dispense with the
accompanying Medication Guide.
Takeda
2.2 Screen for Bipolar Disorder Prior to Starting Trintellix (2.2 Screen for Bipolar Disorder Prior to Starting TRINTELLIX)
Prior to initiating treatment with TRINTELLIX or another antidepressant, screen patients for personal or family history of bipolar disorder, mania, or hypomania [see Warnings and Precautions (5.4)].
2.6 Use of Trintellix in Patients Taking Strong Cyp Inducers (2.6 Use of TRINTELLIX in Patients Taking Strong CYP Inducers)
Consider increasing the dose of TRINTELLIX when a strong CYP inducer (e.g., rifampin, carbamazepine, or phenytoin) is coadministered for greater than 14 days. The maximum recommended dose should not exceed three times the original dose. The dose of TRINTELLIX should be reduced to the original level within 14 days, when the inducer is discontinued [see Drug Interactions (7.1)].
7.3 Interference With Urine Enzyme Immunoassays for Methadone (7.3 Interference with Urine Enzyme Immunoassays for Methadone)
False positive results in urine enzyme immunoassays for methadone have been reported in patients who have taken vortioxetine. An alternative analytical technique (e.g., chromatographic methods) should be considered to confirm positive methadone urine drug screen results.
7.1 Drugs Having Clinically Important Interactions With Trintellix (7.1 Drugs Having Clinically Important Interactions with TRINTELLIX)
| Monoamine Oxidase Inhibitors (MAOIs) | |
| Clinical Impact | The concomitant use of SSRIs and SNRIs including TRINTELLIX with MAOIs increases the risk of serotonin syndrome. |
| Intervention | Concomitant use of TRINTELLIX is contraindicated:
|
| Examples | selegiline, tranylcypromine, isocarboxazid, phenelzine, linezolid, methylene blue |
| Other Serotonergic Drugs | |
| Clinical Impact | Concomitant use of TRINTELLIX with other serotonergic drugs increases the risk of serotonin syndrome. |
| Intervention | Monitor for symptoms of serotonin syndrome when TRINTELLIX is used concomitantly with other drugs that may affect the serotonergic neurotransmitter systems. If serotonin syndrome occurs, consider discontinuation of TRINTELLIX and/or concomitant serotonergic drugs [see Warnings and Precautions (5.2)]. |
| Examples | Other SNRIs, SSRIs, triptans, tricyclic antidepressants, opioids, lithium, buspirone, amphetamines, tryptophan, and St. John's Wort |
| Strong Inhibitors of CYP2D6 | |
| Clinical Impact | Concomitant use of TRINTELLIX with strong CYP2D6 inhibitors increases plasma concentrations of vortioxetine. |
| Intervention | Reduce TRINTELLIX dose by half when a strong CYP2D6 inhibitor is coadministered [see Dosage and Administration (2.5)]. |
| Examples | bupropion, fluoxetine, paroxetine, quinidine |
| Strong CYP Inducers | |
| Clinical Impact | Concomitant use of TRINTELLIX with a strong CYP inducer decreases plasma concentrations of vortioxetine. |
| Intervention | Consider increasing the TRINTELLIX dose when a strong CYP inducer is coadministered. The maximum dose is not recommended to exceed three times the original dose [see Dosage and Administration (2.6)]. |
| Examples | rifampin, carbamazepine, phenytoin |
| Drugs that Interfere with Hemostasis (antiplatelets agents and anticoagulants) | |
| Clinical Impact | Concomitant use of TRINTELLIX with an antiplatelet or anticoagulant drug may potentiate the risk of bleeding. |
| Intervention | Inform patients of the increased risk of bleeding associated with the concomitant use of TRINTELLIX and antiplatelet agents and anticoagulants. For patients taking warfarin, carefully monitor the international normalized ratio [see Warnings and Precautions (5.3), Drug Interactions (7.2)]. |
| Examples | aspirin, clopidogrel, heparin, warfarin |
| Drugs Highly Bound to Plasma Protein | |
| Clinical Impact | TRINTELLIX is highly bound to plasma protein. The concomitant use of TRINTELLIX with another drug that is highly bound to plasma protein may increase free concentrations of TRINTELLIX or other tightly-bound drugs in plasma. |
| Intervention | Monitor for adverse reactions and reduce dosage of TRINTELLIX or other protein bound drugs as warranted [see Drug Interactions (7.2)]. |
| Examples | Warfarin |
5.1 Suicidal Thoughts and Behaviors in Adolescents and Young Adults
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients and 4,500 pediatric patients, the incidence of suicidal thoughts and behaviors in antidepressant-treated patients age 24 years and younger was greater than in placebo-treated patients. There was considerable variation in risk of suicidal thoughts and behaviors among drugs, but there was an increased risk identified in young patients for most drugs studied. There were differences in absolute risk of suicidal thoughts and behaviors across the different indications, with the highest incidence in patients with MDD. The drug-placebo differences in the number of cases of suicidal thoughts and behaviors per 1,000 patients treated are provided in Table 1.
| Age Range | Drug-Placebo Difference in Number of Patients with Suicidal Thoughts and Behaviors per 1000 Patients Treated |
|---|---|
| Increases Compared to Placebo | |
| <18 years old | 14 additional patients |
| 18 to 24 years old | 5 additional patients |
| Decreases Compared to Placebo | |
| 25 to 64 years old | 1 fewer patient |
| ≥65 years old | 6 fewer patients |
It is unknown whether the risk of suicidal thoughts and behaviors in adolescents and young adults extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with MDD that the use of antidepressants can delay the recurrence of depression and that depression itself is a risk factor for suicidal thoughts and behaviors.
Monitor all antidepressant-treated patients for all approved populations for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy, and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the healthcare provider. Consider changing the therapeutic regimen, including possibly discontinuing TRINTELLIX, in patients whose depression is persistently worse, or who are experiencing emergent suicidal thoughts and behaviors.
2.5 Use of Trintellix in Known Cyp2d6 Poor Metabolizers Or in Patients Taking Strong Cyp2d6 Inhibitors (2.5 Use of TRINTELLIX in Known CYP2D6 Poor Metabolizers or in Patients Taking Strong CYP2D6 Inhibitors)
The maximum recommended dose of TRINTELLIX is 10 mg/day in known CYP2D6 poor metabolizers. Reduce the dose of TRINTELLIX by one-half when patients are receiving a CYP2D6 strong inhibitor (e.g., bupropion, fluoxetine, paroxetine, or quinidine) concomitantly. The dose should be increased to the original level when the CYP2D6 inhibitor is discontinued [see Drug Interactions (7.1), Use in Specific Populations (8.6)].
2.4 Switching A Patient to Or From A Monoamine Oxidase Inhibitor (maoi) Intended to Treat Psychiatric Disorders (2.4 Switching a Patient to or From a Monoamine Oxidase Inhibitor (MAOI) Intended to Treat Psychiatric Disorders)
At least 14 days must elapse between discontinuation of a MAOI intended to treat psychiatric disorders and initiation of therapy with TRINTELLIX to avoid the risk of Serotonin Syndrome [see Warnings and Precautions (5.2)]. Conversely, at least 21 days must elapse after stopping TRINTELLIX before starting an MAOI intended to treat psychiatric disorders [see Contraindications (4)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:05.308704 · Updated: 2026-03-14T22:23:58.586609