Inpefa
1a46614e-05f6-421a-b6f4-d6f8760d643a
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
INPEFA is indicated to reduce the risk of cardiovascular death, hospitalization for heart failure, and urgent heart failure visit in adults with: heart failure or type 2 diabetes mellitus, chronic kidney disease, and other cardiovascular risk factors
Dosage and Administration
Correct volume status before starting INPEFA at 200 mg daily and titrate to 400 mg as tolerated. ( 2.2 ) In patients with decompensated heart failure, begin dosing when patients are hemodynamically stable. ( 2.1 ) Withhold INPEFA at least 3 days, if possible, prior to major surgery or procedures associated with prolonged fasting. ( 2.3 )
Contraindications
INPEFA is contraindicated in patients with a history of serious hypersensitivity reaction to INPEFA.
Warnings and Precautions
Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients with type 1 diabetes mellitus and consider ketone monitoring in others at risk for ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue INPEFA if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting. ( 5.1 ) Volume Depletion: Before initiating, correct volume status. Monitor for signs and symptoms of hypotension during therapy. ( 5.2 ) Urosepsis and Pyelonephritis: Monitor for signs and symptoms during therapy and treat promptly. ( 5.3 ) Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues: Lower dose of insulin or insulin secretagogue may be required. ( 5.4 ) Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) : Monitor for pain, tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. Discontinue INPEFA and treat urgently. ( 5.5 ) Genital Mycotic Infections : Monitor and treat as appropriate. ( 5.6 )
Adverse Reactions
The following important adverse reactions are described elsewhere in the labeling: Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis [see Warnings and Precautions ( 5.1 )] Volume Depletion [see Warnings and Precautions ( 5.2 )] Urosepsis and Pyelonephritis [see Warnings and Precautions ( 5.3 )] Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues [see Warnings and Precautions ( 5.4 )] Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) [see Warnings and Precautions ( 5.5 )] Genital Mycotic Infections [see Warnings and Precautions ( 5.6 )]
Drug Interactions
Digoxin : Monitor digoxin levels. ( 7.1 ) Uridine 5'-diphospho-glucuronosyltransferase Inducers (e.g., rifampin): Sotagliflozin exposure is reduced. Consider monitoring of clinical status. ( 7.2 ) Lithium: Monitor serum lithium concentrations. ( 7.3 )
How Supplied
INPEFA tablets are oval and film-coated. Printed Tablets Debossed Tablets Strength (mg) Color Printing Bottle/30 Deboss Bottle/30 Blister/30 (3 x 10) 200 Blue LX200 on one side NDC 70183-220-30 LEX on one side, 200 on other side NDC 70183-221-30 NDC 70183-221-31 400 Yellow -- -- LEX on one side, 400 on other side NDC 70183-241-30 --
Storage and Handling
INPEFA tablets are oval and film-coated. Printed Tablets Debossed Tablets Strength (mg) Color Printing Bottle/30 Deboss Bottle/30 Blister/30 (3 x 10) 200 Blue LX200 on one side NDC 70183-220-30 LEX on one side, 200 on other side NDC 70183-221-30 NDC 70183-221-31 400 Yellow -- -- LEX on one side, 400 on other side NDC 70183-241-30 --
Description
INPEFA is indicated to reduce the risk of cardiovascular death, hospitalization for heart failure, and urgent heart failure visit in adults with: heart failure or type 2 diabetes mellitus, chronic kidney disease, and other cardiovascular risk factors
Medication Information
Warnings and Precautions
Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients with type 1 diabetes mellitus and consider ketone monitoring in others at risk for ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue INPEFA if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting. ( 5.1 ) Volume Depletion: Before initiating, correct volume status. Monitor for signs and symptoms of hypotension during therapy. ( 5.2 ) Urosepsis and Pyelonephritis: Monitor for signs and symptoms during therapy and treat promptly. ( 5.3 ) Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues: Lower dose of insulin or insulin secretagogue may be required. ( 5.4 ) Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) : Monitor for pain, tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. Discontinue INPEFA and treat urgently. ( 5.5 ) Genital Mycotic Infections : Monitor and treat as appropriate. ( 5.6 )
Indications and Usage
INPEFA is indicated to reduce the risk of cardiovascular death, hospitalization for heart failure, and urgent heart failure visit in adults with: heart failure or type 2 diabetes mellitus, chronic kidney disease, and other cardiovascular risk factors
Dosage and Administration
Correct volume status before starting INPEFA at 200 mg daily and titrate to 400 mg as tolerated. ( 2.2 ) In patients with decompensated heart failure, begin dosing when patients are hemodynamically stable. ( 2.1 ) Withhold INPEFA at least 3 days, if possible, prior to major surgery or procedures associated with prolonged fasting. ( 2.3 )
Contraindications
INPEFA is contraindicated in patients with a history of serious hypersensitivity reaction to INPEFA.
Adverse Reactions
The following important adverse reactions are described elsewhere in the labeling: Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis [see Warnings and Precautions ( 5.1 )] Volume Depletion [see Warnings and Precautions ( 5.2 )] Urosepsis and Pyelonephritis [see Warnings and Precautions ( 5.3 )] Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues [see Warnings and Precautions ( 5.4 )] Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) [see Warnings and Precautions ( 5.5 )] Genital Mycotic Infections [see Warnings and Precautions ( 5.6 )]
Drug Interactions
Digoxin : Monitor digoxin levels. ( 7.1 ) Uridine 5'-diphospho-glucuronosyltransferase Inducers (e.g., rifampin): Sotagliflozin exposure is reduced. Consider monitoring of clinical status. ( 7.2 ) Lithium: Monitor serum lithium concentrations. ( 7.3 )
Storage and Handling
INPEFA tablets are oval and film-coated. Printed Tablets Debossed Tablets Strength (mg) Color Printing Bottle/30 Deboss Bottle/30 Blister/30 (3 x 10) 200 Blue LX200 on one side NDC 70183-220-30 LEX on one side, 200 on other side NDC 70183-221-30 NDC 70183-221-31 400 Yellow -- -- LEX on one side, 400 on other side NDC 70183-241-30 --
How Supplied
INPEFA tablets are oval and film-coated. Printed Tablets Debossed Tablets Strength (mg) Color Printing Bottle/30 Deboss Bottle/30 Blister/30 (3 x 10) 200 Blue LX200 on one side NDC 70183-220-30 LEX on one side, 200 on other side NDC 70183-221-30 NDC 70183-221-31 400 Yellow -- -- LEX on one side, 400 on other side NDC 70183-241-30 --
Description
INPEFA is indicated to reduce the risk of cardiovascular death, hospitalization for heart failure, and urgent heart failure visit in adults with: heart failure or type 2 diabetes mellitus, chronic kidney disease, and other cardiovascular risk factors
Section 42229-5
Changes in Laboratory Test Values During Treatment
Section 42231-1
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Issued 01/2024 |
|||||||
|
MEDICATION GUIDE
INPEFA® (in peh' fah) (sotagliflozin) tablets, for oral use |
||||||||
|
What is the most important information I should know about INPEFA?
|
||||||||
| See “What are the possible side effects of INPEFA?” for more information about side effects. | ||||||||
|
What is INPEFA?
INPEFA is a prescription medicine used to reduce the risk of death due to heart problems (cardiovascular death), hospitalization for heart failure, and urgent visits to the doctor for heart failure in adults with:
|
||||||||
| It is not known if INPEFA is safe and effective in children under 18 years of age. | ||||||||
Do not take INPEFA if you:
|
||||||||
Before taking INPEFA, tell your healthcare provider about all of your medical conditions, including if you:
|
||||||||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. INPEFA may affect the way other medicines work, and other medicines may affect how INPEFA works. | ||||||||
How should I take INPEFA?
|
||||||||
|
What are the possible side effects of INPEFA?
|
||||||||
|
Talk to your healthcare provider about what to do if you get symptoms of a yeast infection of the vagina or penis. Your healthcare provider may suggest you use an over-the-counter antifungal medicine. Talk to your healthcare provider right away if you use an over-the-counter antifungal medication and your symptoms do not go away. The most common side effects of INPEFA include:
These are not all the possible side effects of INPEFA. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||||||
How should I store INPEFA?
|
||||||||
| Keep INPEFA and all medicines out of the reach of children. | ||||||||
|
General information about the safe and effective use of INPEFA.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use INPEFA for a condition for which it is not prescribed. Do not give INPEFA to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about INPEFA that is written for health professionals. |
||||||||
|
What are the ingredients in INPEFA?
Active ingredient: sotagliflozin. Inactive ingredients: The core of the tablet contains: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose, and talc. The film coating for the 200 mg tablet contains: indigo carmine aluminum lake, polyethylene glycol, polyvinyl alcohol (partly hydrolyzed), talc, and titanium dioxide. The film coating for the 400 mg tablet contains: hypromellose, lactose monohydrate, titanium dioxide, triacetin, and yellow iron oxide. For tablets that are printed and not debossed, the ink contains: ammonium hydroxide, black iron oxide, isopropyl alcohol, N-butyl alcohol, propylene glycol, and shellac. Manufactured for: Lexicon Pharmaceuticals, Inc. The Woodlands, TX, 77381. INPEFA is a registered trademark of Lexicon Pharmaceuticals, Inc. For more information about INPEFA, go to www.lexpharma.com or call 1-855-330-2573. |
Section 44425-7
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature].
Section 51945-4
Principal Display Panel – 30 Count 200 mg Bottle Label
NDC 70183-220-30
XXXXXXXX
inpefa™
sotagliflozin tablets
Rx only
200 mg
Dispense with Enclosed
Medication Guide
30 Tablets for oral use
Lexicon
pharmaceuticals
7.1 Digoxin
There is an increase in the exposure of digoxin when coadministered with INPEFA 400 mg. Patients taking INPEFA with concomitant digoxin should be monitored appropriately [see Clinical Pharmacology (12.3)].
7.3 Lithium
Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. Monitor serum lithium concentration more frequently during INPEFA initiation and dosage changes.
10 Overdosage
There were no confirmed reports of symptomatic overdose with sotagliflozin during the clinical development program of INPEFA.
In the event of an overdose with INPEFA, contact the Poison Control Center. Employ the usual supportive measures as dictated by the patient's clinical status.
The removal of sotagliflozin by hemodialysis has not been studied.
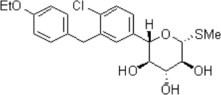
11 Description
INPEFA tablets for oral administration contain sotagliflozin, a sodium-glucose cotransporter 2 (SGLT2) inhibitor.
The chemical name of sotagliflozin is (2S,3R,4R,5S,6R)-2-(4-chloro-3-(4-ethoxybenzyl)phenyl)-6-(methylthio)tetrahydro-2H-pyran-3,4,5-triol.
Its molecular formula is C21H25ClO5S and the molecular weight is 424.94. The structural formula is:
Sotagliflozin is a white to off-white solid. It is practically insoluble in water.
Each film-coated tablet of INPEFA contains 200 mg or 400 mg of sotagliflozin and the following inactive ingredients. The core of the tablet contains colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose, and talc. The film coating for the 200 mg tablet contains: indigo carmine aluminum lake, polyethylene glycol, polyvinyl alcohol (partly hydrolyzed), talc, and titanium dioxide. The film coating for the 400 mg tablet contains: hypromellose, lactose monohydrate, titanium dioxide, triacetin, and yellow iron oxide. The 200 mg printed tablet also includes black ink which contains: ammonium hydroxide, black iron oxide, isopropyl alcohol, N-butyl alcohol, propylene glycol, and shellac.
14.2 Scored Study
The SCORED (Effects of Sotagliflozin on Cardiovascular and Renal Events in Patients with Type 2 Diabetes Mellitus, Cardiovascular Risk Factors and Moderately Impaired Renal Function) study (NCT03315143) was a randomized, double-blind, placebo-controlled, parallel-group, multicenter study in patients with type 2 diabetes mellitus (A1C > 7%), chronic kidney disease (eGFR 25 to 60 mL/min/1.73 m2), and additional cardiovascular risk factors, such as a history of heart failure, obesity, dyslipidemia, hypertension, or elevated cardiac and inflammatory biomarkers, to determine if INPEFA reduces the risk of total occurrence of cardiovascular death, hospitalization for heart failure, and urgent heart failure visit. Of the 10,584 randomized patients, 5,292 were randomized to INPEFA and 5,292 to placebo.
The dose of study drug was to be up-titrated from 200 mg to 400 mg sotagliflozin or matching placebo as soon as 4 weeks or up until 6 months after initiation of treatment. The dose was increased to 400 mg once daily for 3,934 patients (74%) in the INPEFA group and for 3,987 patients (75%) in the placebo group. The median time to up-titration was 29 days. Up-titration was to be performed based on the judgment of the investigator, who considered whether the patient's clinical condition was satisfactory, whether the drug was well tolerated, and whether AEs typical of SGLT2 inhibitors had occurred, such as those associated with volume depletion.
At baseline, median age was 68 years, 45% were female, 82% were White, 3% Black or African American, 6% Asian, and 4% American Indian or Native Alaskan. Median A1C was 8.3%, median BMI was 32 kg/m2, median eGFR was 45 mL/min/1.73 m2 (8% with eGFR < 30, 44% with eGFR 30 to < 45, and 48% with eGFR ≥ 45 mL/min/1.73 m2), and median urinary albumin-to-creatinine ratio (UACR) was 82 mg/g (32% with UACR ≥ 300 mg/g). A history of heart failure was present in 31%, prior myocardial infarction had occurred in 20%, a prior cerebrovascular event had occurred in 9%, and coronary revascularization had been performed in 22% of patients prior to study entry.
At baseline, 97% were treated with at least one antihyperglycemic medication, including 56% with a biguanide, 64% with insulin, 27% with a sulfonylurea, 20% with a DPP4 inhibitor, and 6% with a GLP-1 receptor agonist. At baseline, 88% were treated with inhibitors of the renin-angiotensin-aldosterone system, 14% with a beta blocker, 42% with a calcium channel blocker, 35% with a loop diuretic, and 30% with another diuretic.
INPEFA was superior to placebo in reducing the risk of the primary composite endpoint (HR 0.75 [95% CI 0.63, 0.88]; p < 0.001) (Table 3).
| Primary Endpoint a | Event Rates (per 100 Patient-years) |
Hazard Ratio
(95% CI) |
|
|---|---|---|---|
|
INPEFA
N = 5,292 |
Placebo
N = 5,292 |
||
|
a Based on investigator-reported events in all randomized patients, analyzed according to the treatment group allocated by randomization. |
|||
|
b Predefined primary endpoint. |
|||
|
c Predefined secondary endpoint and tested with multiplicity control. |
|||
|
d Time-to-event analysis was performed; event rates are percentages of patients with events. |
|||
| Total occurrence of cardiovascular death, hospitalization for heart failure, and urgent heart failure visitb | 5.6 | 7.5 | 0.75 (0.63, 0.88) p < 0.001 |
| Primary Endpoint Components | |||
| Cardiovascular deathc,d | 2.9 | 3.2 | 0.90 (0.73, 1.12) |
| Hospitalization for heart failure | 2.8 | 4.2 | 0.66 (0.53, 0.82) |
| Urgent heart failure visit | 0.7 | 0.9 | 0.73 (0.48, 1.11) |
| Secondary Endpoint c | |||
| Hospitalization for heart failure or urgent heart failure visitc | 3.5 | 5.1 | 0.67 (0.55, 0.82) p = 0.0001 |
Figure 5 displays a cumulative events plot of the primary composite endpoint. The INPEFA and placebo event curves separated early and continued to diverge over the study period following randomization.
Figure 5 Primary Composite Endpoint (Total Occurrences of Cardiovascular Death, Hospitalization for Heart Failure, and Urgent Heart Failure Visit) Over Time in the SCORED Study
Primary composite endpoint was based on investigator-reported events in all randomized patients, analyzed according to the treatment group allocated by randomization.
The results of the primary composite endpoint were generally consistent across prespecified subgroups, including history of cardiovascular disease, history of heart failure, and screening LVEF < 50 or ≥ 50% (Figure 6).
Figure 6 Treatment Effect for Primary Composite Endpoint (Total Occurrences of Cardiovascular Death, Hospitalization for Heart Failure, and Urgent Heart Failure Visit) Subgroup Analysis (SCORED Study)
CVD = cardiovascular disease; eGFR = estimated glomerular filtration rate; LVEF = left ventricular ejection fraction; HF = heart failure; PY = patient-years
Primary composite endpoint was based on investigator-reported events in all randomized patients, analyzed according to the treatment group allocated by randomization.
Note: The figure above presents effects in various subgroups all of which are baseline characteristics and all of which were pre-specified. The 95% confidence limits that are shown do not take into account how many comparisons were made, nor do they reflect the effect of a particular factor after adjustment for all other factors. Apparent homogeneity or heterogeneity among groups should not be over-interpreted.
8.4 Pediatric Use
The safety and effectiveness of INPEFA in pediatric patients under 18 years of age have not been established.
8.5 Geriatric Use
No INPEFA dosage change is recommended based on age.
In the SOLOIST study, a total of 241 (40%) patients treated with INPEFA were between 65 and < 75 years of age, and 174 (29%) were ≥ 75 years of age. In the SCORED study, a total of 2,470 (47%) patients treated with INPEFA were between 65 and < 75 years of age, and 1,240 (23%) were ≥ 75 years of age.
No overall differences in efficacy were detected between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Elderly patients may be at increased risk for volume depletion adverse reactions, including hypotension. In patients ≥ 65 years of age, a higher proportion of patients treated with INPEFA had adverse reactions of volume depletion [see Warnings and Precautions (5.2) and Adverse Reactions (6.1)].
14.1 Soloist Study
The SOLOIST (Effects of Sotagliflozin on Clinical Outcomes in Hemodynamically Stable Patients with Type 2 Diabetes Post Worsening Heart Failure) study (NCT03521934) was a randomized, double-blind, placebo-controlled, parallel-group, multicenter study in patients with type 2 diabetes mellitus who had been admitted to the hospital, a heart failure unit, infusion center, or emergency department for worsening heart failure to determine if INPEFA reduces the risk of total occurrence of cardiovascular death, hospitalization for heart failure, and urgent heart failure visit. Patients were randomized to treatment if they met the following criteria for clinical stability: no need for oxygen therapy, a systolic blood pressure of at least 100 mmHg, no need for intravenous inotropic or vasodilator therapy (excluding nitrates) and transitioned from intravenous to oral diuretic therapy.
Of the 1,222 randomized patients, 608 were randomized to INPEFA and 614 to placebo. Assigned treatment was initiated in the hospital or within a median of 2 days following hospital discharge.
The dose of study drug was to be up-titrated from 200 mg to 400 mg sotagliflozin or matching placebo as soon as 2 weeks or up until 8 months after initiation of treatment. The dose was increased to 400 mg once daily for 336 patients (56%) in the sotagliflozin group and 325 patients (53%) in the placebo group. The median time to up-titration was 16 days. Up-titration was to be performed based on the judgment of the investigator, who considered whether the patient's clinical condition was satisfactory, whether the drug was well tolerated, and whether AEs typical of SGLT2 inhibitors had occurred, such as those associated with volume depletion.
At baseline, median age was 70 years, 34% were female, 93% were White and 4% were Black or African American. Median A1C was 7.1%, median body mass index (BMI) was 31 kg/m2, and median eGFR was 50 mL/min/1.73 m2. Median left ventricular ejection fraction (LVEF) was 35% (79% with LVEF < 50%), median N-terminal pro B-type natriuretic peptide (NT-proBNP) was 1806 pg/mL, and the median Kansas City Cardiomyopathy Questionnaire-12 (KCCQ-12) score was 41.
At baseline, 86% were treated with at least one antihyperglycemic medication, including 52% with a biguanide, 36% with insulin, 19% with a sulfonylurea, 16% with a dipeptidyl peptidase-4 (DPP4) inhibitor, and 3% with a glucagon-like peptide 1 (GLP-1) receptor agonist. At baseline, 91% were treated with inhibitors of the renin-angiotensin-aldosterone system, 92% with a beta blocker, 95% with a loop diuretic, and 10% with another diuretic.
INPEFA was superior to placebo in reducing the risk of the primary composite endpoint (Hazard Ratio [HR] 0.67 [95% confidence interval (CI) 0.53, 0.85]; p = 0.001). (Table 2).
| Primary Endpoint a | Event Rates per 100 Patient-years |
Hazard Ratio
(95% CI) |
|
|---|---|---|---|
|
INPEFA
N = 608 |
Placebo
N = 614 |
||
|
a Based on investigator-reported events in all randomized patients, analyzed according to the treatment group allocated by randomization. |
|||
|
b Predefined primary endpoint. |
|||
|
c Predefined secondary endpoint and tested with multiplicity control. |
|||
|
d Time-to-event analysis was performed; event rates are percentages of patients with events. |
|||
| Total occurrence of cardiovascular death, hospitalization for heart failure, and urgent heart failure visitb | 51.3 | 76.4 | 0.67 (0.53, 0.85) p = 0.001 |
| Primary Endpoint Components | |||
| Cardiovascular deathc,d | 8.4 | 9.4 | 0.84 (0.58, 1.23) |
| Hospitalization for heart failure | 33.7 | 51.9 | 0.65 (0.49, 0.87) |
| Urgent heart failure visit | 6.9 | 12.1 | 0.60 (0.34, 1.06) |
| Secondary Endpoint c | |||
| Hospitalization for heart failure and urgent heart failure visitc | 40.6 | 63.9 | 0.64 (0.50, 0.84) p = 0.0009 |
Figure 3 displays a cumulative events plot of the primary composite endpoint. The INPEFA and placebo event curves diverged early and remained separated over the study period.
Figure 3 Primary Composite Endpoint (Total Occurrences of Cardiovascular Death, Hospitalization for Heart Failure, and Urgent Heart Failure Visit) Over Time in the SOLOIST Study
Primary composite endpoint was based on investigator-reported events in all randomized patients, analyzed according to the treatment group allocated by randomization.
The results of the primary composite endpoint were generally consistent across prespecified subgroups (Figure 4), including screening LVEF < 50 or ≥ 50% and timing of first dose (post-discharge versus prior to discharge).
Figure 4 Treatment Effect for Primary Composite Endpoint (Total Occurrences of Cardiovascular Death, Hospitalization for Heart Failure, and Urgent Heart Failure Visit) Subgroup Analysis (SOLOIST Study)
HF = heart failure; LVEF = left ventricular ejection fraction; MRA-mineralocorticoid receptor antagonist; NT-proBNP = N-terminal pro B-type natriuretic peptide; PY = patient-years
Primary composite endpoint was based on investigator-reported events in all randomized patients, analyzed according to the treatment group allocated by randomization.
Discharge may have been from hospital or urgent treatment facility where urgent heart failure visit occurred.
Note: The figure above presents effects in various subgroups all of which are baseline characteristics and all of which were pre-specified. The 95% confidence limits that are shown do not take into account how many comparisons were made, nor do they reflect the effect of a particular factor after adjustment for all other factors. Apparent homogeneity or heterogeneity among groups should not be over-interpreted.
4 Contraindications
INPEFA is contraindicated in patients with a history of serious hypersensitivity reaction to INPEFA.
6 Adverse Reactions
The following important adverse reactions are described elsewhere in the labeling:
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis [see Warnings and Precautions (5.1)]
- Volume Depletion [see Warnings and Precautions (5.2)]
- Urosepsis and Pyelonephritis [see Warnings and Precautions (5.3)]
- Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues [see Warnings and Precautions (5.4)]
- Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) [see Warnings and Precautions (5.5)]
- Genital Mycotic Infections [see Warnings and Precautions (5.6)]
7 Drug Interactions
5.2 Volume Depletion
INPEFA can cause intravascular volume depletion which may sometimes manifest as symptomatic hypotension or acute transient changes in creatinine. There have been postmarketing reports of acute kidney injury, some requiring hospitalization and dialysis, in patients with type 2 diabetes mellitus receiving SGLT2 inhibitors. Patients with impaired renal function (eGFR < 60 mL/min/1.73 m2), elderly patients, or patients on loop diuretics may be at increased risk for volume depletion or hypotension [see Adverse Reactions (6.1) and Use in Specific Populations (8.5, 8.6)]. Before initiating INPEFA in patients with one or more of these characteristics, assess volume status and renal function. Monitor for signs and symptoms of hypotension, and renal function after initiating therapy.
8.6 Renal Impairment
INPEFA was evaluated in 5,292 patients with chronic kidney disease (eGFR 25 to 60 mL/min/1.73 m2) in the SCORED study and in 426 patients with heart failure with eGFR < 60 mL/min/1.73 m2 in the SOLOIST study. The safety profile of INPEFA across eGFR subgroups in these studies was consistent with the known safety profile. There was an increase in volume-related adverse events (e.g., hypotension, dizziness) in patients with eGFR < 30 mL/min/1.73 m2 relative to the overall safety population [see Warnings and Precautions (5.2) and Adverse Reactions (6.1)].
Efficacy and safety studies with INPEFA did not enroll patients with an eGFR less than 25 mL/min/1.73 m2 or on dialysis. After starting therapy in these studies, patients were discontinued if eGFR fell below 15 mL/min/1.73 m2 or were initiated on chronic dialysis.
12.3 Pharmacokinetics
Plasma Cmax and AUC of sotagliflozin increased in a dose-proportional manner in the therapeutic dose range of 200 mg to 400 mg once daily. Accumulation of sotagliflozin was observed with an approximate 50-100% increase in both Cmax and area under the concentration-time curve from 0 to 24 hours (AUC0-24h) at steady state verses the first day of dosing.
2.2 Recommended Dosage
The recommended starting dose of INPEFA is 200 mg orally once daily not more than one hour before the first meal of the day.
Up-titrate after at least 2 weeks to 400 mg orally once daily as tolerated [see Clinical Studies (14)]. Down-titrate to 200 mg as necessary [see Adverse Reactions (6.1), Warnings and Precautions (5) and Use in Specific Populations (8.6)].
Swallow tablets whole. Do not cut, crush, or chew tablets.
If a dose of INPEFA is missed by more than 6 hours, take the next dose as prescribed the next day.
8.7 Hepatic Impairment
In a clinical pharmacology study in patients with hepatic impairment, the exposure in mild hepatic impairment was not increased, but was approximately 3-fold as high in moderate and approximately 6-fold as high in severely hepatic-impaired subjects compared to subjects with normal hepatic function [see Clinical Pharmacology (12.3)].
No dosage adjustment is necessary in patients with mild hepatic impairment.
The safety and efficacy of INPEFA have not been established in patients with moderate or severe hepatic impairment [see Clinical Pharmacology (12.3)]. INPEFA is not recommended in patients with moderate or severe hepatic impairment.
1 Indications and Usage
INPEFA is indicated to reduce the risk of cardiovascular death, hospitalization for heart failure, and urgent heart failure visit in adults with:
- heart failure or
- type 2 diabetes mellitus, chronic kidney disease, and other cardiovascular risk factors
12.1 Mechanism of Action
Sotagliflozin is an inhibitor of SGLT2 and SGLT1. Inhibiting SGLT2 reduces renal reabsorption of glucose and sodium which may influence several physiological functions such as lowering both pre-and afterload of the heart and downregulating sympathetic activity. Inhibiting SGLT1 reduces intestinal absorption of glucose and sodium which likely contributes to diarrhea. The mechanism for sotagliflozin's cardiovascular benefits has not been established.
5 Warnings and Precautions
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients with type 1 diabetes mellitus and consider ketone monitoring in others at risk for ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue INPEFA if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting. (5.1)
- Volume Depletion: Before initiating, correct volume status. Monitor for signs and symptoms of hypotension during therapy. (5.2)
- Urosepsis and Pyelonephritis: Monitor for signs and symptoms during therapy and treat promptly. (5.3)
- Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues: Lower dose of insulin or insulin secretagogue may be required. (5.4)
- Necrotizing Fasciitis of the Perineum (Fournier's Gangrene): Monitor for pain, tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. Discontinue INPEFA and treat urgently. (5.5)
- Genital Mycotic Infections: Monitor and treat as appropriate. (5.6)
2 Dosage and Administration
Correct volume status before starting INPEFA at 200 mg daily and titrate to 400 mg as tolerated. (2.2) In patients with decompensated heart failure, begin dosing when patients are hemodynamically stable. (2.1)
Withhold INPEFA at least 3 days, if possible, prior to major surgery or procedures associated with prolonged fasting. (2.3)
3 Dosage Forms and Strengths
INPEFA 200 mg printed tablet is an oval, blue, film-coated tablet imprinted in black ink with “LX200” on one side.
INPEFA 200 mg debossed tablet is an oval, blue, film-coated tablet debossed with “LEX” on one side and “200” on the other side.
INPEFA 400 mg debossed tablet is an oval, yellow, film-coated tablet debossed with “LEX” on one side and “400” on the other side.
8 Use in Specific Populations
Pregnancy: Advise females of the potential risk to a fetus especially during the second and third trimesters. (8.1)
Lactation: INPEFA is not recommended when breastfeeding. (8.2)
Geriatrics: Higher incidence of adverse reactions related to volume depletion. (5.2, 8.5)
Renal Impairment: Higher incidence of adverse reactions related to volume depletion. (5.2, 8.6)
5.6 Genital Mycotic Infections
INPEFA increases the risk of genital mycotic infections. Patients with a history of genital mycotic infections were more likely to develop genital mycotic infections [see Adverse Reactions (6.1)]. Monitor and treat appropriately.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In the phase 3 (SOLOIST [see Clinical Studies (14.1)] and SCORED [see Clinical Studies (14.2)]) placebo-controlled trials, 5,896 subjects received INPEFA.
In the SOLOIST study, 336 patients (56%) reached the 400 mg dose. In the SCORED study, 3,934 patients (74%) reached the 400 mg dose.
In the SOLOIST study, 5.6% of patients in the INPEFA group and 5.4% of patients in the placebo group discontinued therapy due to adverse events (AEs). In the SCORED study, 5.0% of patients in the INPEFA group and 4.5% of patients in the placebo group discontinued therapy due to AEs.
| Adverse Reaction |
SOLOIST
N = 1,216 |
SCORED
N = 10,577 |
||
|
Placebo (%)
N = 611 |
INPEFA (%)
N = 605 |
Placebo (%)
N = 5,286 |
INPEFA (%)
N = 5,291 |
|
| Urinary tract infection | 7.2 | 8.6 | 11.0 | 11.5 |
| Volume depletion | 8.8 | 9.3 | 4.0 | 5.2 |
| Diarrhea | 4.1 | 6.9 | 6.0 | 8.4 |
| Hypoglycemia | 2.8 | 4.3 | 7.9 | 7.7 |
| Dizziness | 2.5 | 2.6 | 2.8 | 3.3 |
| Genital mycotic infection | 0.2 | 0.8 | 0.9 | 2.4 |
5.7 Positive Urine Glucose Test
Monitoring glucose levels with urine glucose tests is not recommended as SGLT2 inhibition increases urinary glucose excretion and will lead to positive urine glucose tests. Use alternative methods to monitor glucose levels.
5.3 Urosepsis and Pyelonephritis
Treatment with SGLT2 inhibitors, including INPEFA, increases the risk for urinary tract infections. Serious urinary tract infections including urosepsis and pyelonephritis requiring hospitalization have been reported. Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated [see Adverse Reactions (6.1)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.1 Prior to Initiation of Inpefa
Assess volume status and, if necessary, correct volume depletion prior to initiation of INPEFA [see Warnings and Precautions (5.2) and Use in Specific Populations (8.5, 8.6)].
Assess renal function prior to initiation of INPEFA and then as clinically indicated [see Warnings and Precautions (5.2)].
For patients with decompensated heart failure, dosing may begin as soon as the patient is hemodynamically stable, including during hospitalization or urgent outpatient treatment or immediately upon discharge.
16 How Supplied/storage and Handling
INPEFA tablets are oval and film-coated.
| Printed Tablets | Debossed Tablets | |||||
| Strength (mg) | Color | Printing | Bottle/30 | Deboss | Bottle/30 |
Blister/30 (3 x 10) |
| 200 | Blue | LX200 on one side |
NDC 70183-220-30 | LEX on one side, 200 on other side | NDC 70183-221-30 | NDC 70183-221-31 |
| 400 | Yellow | -- | -- | LEX on one side, 400 on other side | NDC 70183-241-30 | -- |
2.3 Temporary Interruption for Surgery
5.8 Interference With 1,5 Anhydroglucitol (1,5 Ag) Assay
Monitoring glucose levels with 1,5-AG assay is not recommended as measurements of 1,5-AG are unreliable in assessing glucose levels in patients taking SGLT2 inhibitors. Use alternative methods to monitor glucose levels.
7.2 Uridine 5' Diphospho Glucuronosyltransferase (ugt) Inducer
Glucuronidation by UGT1A9, to form the 3-O-glucuronide, was identified as a major metabolic pathway for sotagliflozin. The coadministration of rifampicin, an inducer of UGTs, with a single dose of 400 mg sotagliflozin resulted in a decrease in the exposure to sotagliflozin. This decrease in exposure to sotagliflozin may decrease efficacy [see Clinical Pharmacology (12.3)].
5.5 Necrotizing Fasciitis of the Perineum (fournier's Gangrene)
Reports of necrotizing fasciitis of the perineum (Fournier's Gangrene), a rare but serious and life-threatening necrotizing infection requiring urgent surgical intervention, have been identified in postmarketing surveillance in patients with diabetes mellitus receiving SGLT2 inhibitors. Cases have been reported in both females and males. Serious outcomes have included hospitalization, multiple surgeries, and death.
Patients treated with INPEFA presenting with pain, tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, should be assessed for necrotizing fasciitis. If suspected, start treatment immediately with broad-spectrum antibiotics and, if necessary, surgical debridement. Discontinue INPEFA, closely monitor blood glucose levels, and provide appropriate alternative therapy for heart failure.
5.4 Hypoglycemia With Concomitant Use With Insulin and Insulin Secretagogues
Insulin and insulin secretagogues are known to cause hypoglycemia. INPEFA may increase the risk of hypoglycemia when combined with insulin or an insulin secretagogue [see Adverse Reactions (6.1)]. Therefore, a lower dose of insulin or insulin secretagogue may be required to minimize the risk of hypoglycemia when these agents are used in combination with INPEFA.
5.1 Diabetic Ketoacidosis in Patients With Type 1 Diabetes Mellitus and Other Ketoacidosis
In patients with type 1 diabetes mellitus, INPEFA significantly increases the risk of diabetic ketoacidosis, a life-threatening event, beyond the background rate. In placebo-controlled trials of patients with type 1 diabetes mellitus, the risk of ketoacidosis was markedly increased in patients who received sodium glucose cotransporter 2 (SGLT2) inhibitors compared to patients who received placebo; this risk may be greater with higher doses of INPEFA. INPEFA is not indicated for glycemic control.
Type 2 diabetes mellitus and pancreatic disorders (e.g., history of pancreatitis or pancreatic surgery) are also risk factors for ketoacidosis. There have been postmarketing reports of fatal events of ketoacidosis in patients with type 2 diabetes using SGLT2 inhibitors.
Precipitating conditions for diabetic ketoacidosis or other ketoacidosis include acute febrile illness, reduced caloric intake, ketogenic diet, surgery, insulin dose reduction, volume depletion, and alcohol abuse.
Signs and symptoms are consistent with dehydration and severe metabolic acidosis and include nausea, vomiting, abdominal pain, generalized malaise, and shortness of breath. Blood glucose levels at presentation may be below those typically expected for diabetic ketoacidosis (e.g., less than 250 mg/dL). Ketoacidosis and glucosuria may persist longer than typically expected. Urinary glucose excretion persists for 3 days after discontinuing INPEFA [see Clinical Pharmacology (12.3)]; however, there have been postmarketing reports of ketoacidosis and glucosuria lasting greater than 6 days and some up to 2 weeks after discontinuation of SGLT2 inhibitors.
Consider ketone monitoring in patients with type 1 diabetes mellitus and consider ketone monitoring in others at risk for ketoacidosis if indicated by the clinical situation. Assess for ketoacidosis regardless of presenting blood glucose levels in patients who present with signs and symptoms consistent with severe metabolic acidosis. If ketoacidosis is suspected, discontinue INPEFA, promptly evaluate, and treat ketoacidosis, if confirmed. Monitor patients for resolution of ketoacidosis before restarting INPEFA.
Withhold INPEFA, if possible, in temporary clinical situations that could predispose patients to ketoacidosis. Resume INPEFA when the patient is clinically stable and has resumed oral intake [see Dosage and Administration (2.3)].
Educate all patients on the signs and symptoms of ketoacidosis and instruct patients to discontinue INPEFA and seek medical attention immediately if signs and symptoms occur.
Structured Label Content
Section 42229-5 (42229-5)
Changes in Laboratory Test Values During Treatment
Section 42231-1 (42231-1)
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Issued 01/2024 |
|||||||
|
MEDICATION GUIDE
INPEFA® (in peh' fah) (sotagliflozin) tablets, for oral use |
||||||||
|
What is the most important information I should know about INPEFA?
|
||||||||
| See “What are the possible side effects of INPEFA?” for more information about side effects. | ||||||||
|
What is INPEFA?
INPEFA is a prescription medicine used to reduce the risk of death due to heart problems (cardiovascular death), hospitalization for heart failure, and urgent visits to the doctor for heart failure in adults with:
|
||||||||
| It is not known if INPEFA is safe and effective in children under 18 years of age. | ||||||||
Do not take INPEFA if you:
|
||||||||
Before taking INPEFA, tell your healthcare provider about all of your medical conditions, including if you:
|
||||||||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. INPEFA may affect the way other medicines work, and other medicines may affect how INPEFA works. | ||||||||
How should I take INPEFA?
|
||||||||
|
What are the possible side effects of INPEFA?
|
||||||||
|
Talk to your healthcare provider about what to do if you get symptoms of a yeast infection of the vagina or penis. Your healthcare provider may suggest you use an over-the-counter antifungal medicine. Talk to your healthcare provider right away if you use an over-the-counter antifungal medication and your symptoms do not go away. The most common side effects of INPEFA include:
These are not all the possible side effects of INPEFA. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||||||
How should I store INPEFA?
|
||||||||
| Keep INPEFA and all medicines out of the reach of children. | ||||||||
|
General information about the safe and effective use of INPEFA.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use INPEFA for a condition for which it is not prescribed. Do not give INPEFA to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about INPEFA that is written for health professionals. |
||||||||
|
What are the ingredients in INPEFA?
Active ingredient: sotagliflozin. Inactive ingredients: The core of the tablet contains: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose, and talc. The film coating for the 200 mg tablet contains: indigo carmine aluminum lake, polyethylene glycol, polyvinyl alcohol (partly hydrolyzed), talc, and titanium dioxide. The film coating for the 400 mg tablet contains: hypromellose, lactose monohydrate, titanium dioxide, triacetin, and yellow iron oxide. For tablets that are printed and not debossed, the ink contains: ammonium hydroxide, black iron oxide, isopropyl alcohol, N-butyl alcohol, propylene glycol, and shellac. Manufactured for: Lexicon Pharmaceuticals, Inc. The Woodlands, TX, 77381. INPEFA is a registered trademark of Lexicon Pharmaceuticals, Inc. For more information about INPEFA, go to www.lexpharma.com or call 1-855-330-2573. |
Section 44425-7 (44425-7)
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature].
Section 51945-4 (51945-4)
Principal Display Panel – 30 Count 200 mg Bottle Label
NDC 70183-220-30
XXXXXXXX
inpefa™
sotagliflozin tablets
Rx only
200 mg
Dispense with Enclosed
Medication Guide
30 Tablets for oral use
Lexicon
pharmaceuticals
7.1 Digoxin
There is an increase in the exposure of digoxin when coadministered with INPEFA 400 mg. Patients taking INPEFA with concomitant digoxin should be monitored appropriately [see Clinical Pharmacology (12.3)].
7.3 Lithium
Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. Monitor serum lithium concentration more frequently during INPEFA initiation and dosage changes.
10 Overdosage (10 OVERDOSAGE)
There were no confirmed reports of symptomatic overdose with sotagliflozin during the clinical development program of INPEFA.
In the event of an overdose with INPEFA, contact the Poison Control Center. Employ the usual supportive measures as dictated by the patient's clinical status.
The removal of sotagliflozin by hemodialysis has not been studied.
11 Description (11 DESCRIPTION)
INPEFA tablets for oral administration contain sotagliflozin, a sodium-glucose cotransporter 2 (SGLT2) inhibitor.
The chemical name of sotagliflozin is (2S,3R,4R,5S,6R)-2-(4-chloro-3-(4-ethoxybenzyl)phenyl)-6-(methylthio)tetrahydro-2H-pyran-3,4,5-triol.
Its molecular formula is C21H25ClO5S and the molecular weight is 424.94. The structural formula is:
Sotagliflozin is a white to off-white solid. It is practically insoluble in water.
Each film-coated tablet of INPEFA contains 200 mg or 400 mg of sotagliflozin and the following inactive ingredients. The core of the tablet contains colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose, and talc. The film coating for the 200 mg tablet contains: indigo carmine aluminum lake, polyethylene glycol, polyvinyl alcohol (partly hydrolyzed), talc, and titanium dioxide. The film coating for the 400 mg tablet contains: hypromellose, lactose monohydrate, titanium dioxide, triacetin, and yellow iron oxide. The 200 mg printed tablet also includes black ink which contains: ammonium hydroxide, black iron oxide, isopropyl alcohol, N-butyl alcohol, propylene glycol, and shellac.
14.2 Scored Study (14.2 SCORED Study)
The SCORED (Effects of Sotagliflozin on Cardiovascular and Renal Events in Patients with Type 2 Diabetes Mellitus, Cardiovascular Risk Factors and Moderately Impaired Renal Function) study (NCT03315143) was a randomized, double-blind, placebo-controlled, parallel-group, multicenter study in patients with type 2 diabetes mellitus (A1C > 7%), chronic kidney disease (eGFR 25 to 60 mL/min/1.73 m2), and additional cardiovascular risk factors, such as a history of heart failure, obesity, dyslipidemia, hypertension, or elevated cardiac and inflammatory biomarkers, to determine if INPEFA reduces the risk of total occurrence of cardiovascular death, hospitalization for heart failure, and urgent heart failure visit. Of the 10,584 randomized patients, 5,292 were randomized to INPEFA and 5,292 to placebo.
The dose of study drug was to be up-titrated from 200 mg to 400 mg sotagliflozin or matching placebo as soon as 4 weeks or up until 6 months after initiation of treatment. The dose was increased to 400 mg once daily for 3,934 patients (74%) in the INPEFA group and for 3,987 patients (75%) in the placebo group. The median time to up-titration was 29 days. Up-titration was to be performed based on the judgment of the investigator, who considered whether the patient's clinical condition was satisfactory, whether the drug was well tolerated, and whether AEs typical of SGLT2 inhibitors had occurred, such as those associated with volume depletion.
At baseline, median age was 68 years, 45% were female, 82% were White, 3% Black or African American, 6% Asian, and 4% American Indian or Native Alaskan. Median A1C was 8.3%, median BMI was 32 kg/m2, median eGFR was 45 mL/min/1.73 m2 (8% with eGFR < 30, 44% with eGFR 30 to < 45, and 48% with eGFR ≥ 45 mL/min/1.73 m2), and median urinary albumin-to-creatinine ratio (UACR) was 82 mg/g (32% with UACR ≥ 300 mg/g). A history of heart failure was present in 31%, prior myocardial infarction had occurred in 20%, a prior cerebrovascular event had occurred in 9%, and coronary revascularization had been performed in 22% of patients prior to study entry.
At baseline, 97% were treated with at least one antihyperglycemic medication, including 56% with a biguanide, 64% with insulin, 27% with a sulfonylurea, 20% with a DPP4 inhibitor, and 6% with a GLP-1 receptor agonist. At baseline, 88% were treated with inhibitors of the renin-angiotensin-aldosterone system, 14% with a beta blocker, 42% with a calcium channel blocker, 35% with a loop diuretic, and 30% with another diuretic.
INPEFA was superior to placebo in reducing the risk of the primary composite endpoint (HR 0.75 [95% CI 0.63, 0.88]; p < 0.001) (Table 3).
| Primary Endpoint a | Event Rates (per 100 Patient-years) |
Hazard Ratio
(95% CI) |
|
|---|---|---|---|
|
INPEFA
N = 5,292 |
Placebo
N = 5,292 |
||
|
a Based on investigator-reported events in all randomized patients, analyzed according to the treatment group allocated by randomization. |
|||
|
b Predefined primary endpoint. |
|||
|
c Predefined secondary endpoint and tested with multiplicity control. |
|||
|
d Time-to-event analysis was performed; event rates are percentages of patients with events. |
|||
| Total occurrence of cardiovascular death, hospitalization for heart failure, and urgent heart failure visitb | 5.6 | 7.5 | 0.75 (0.63, 0.88) p < 0.001 |
| Primary Endpoint Components | |||
| Cardiovascular deathc,d | 2.9 | 3.2 | 0.90 (0.73, 1.12) |
| Hospitalization for heart failure | 2.8 | 4.2 | 0.66 (0.53, 0.82) |
| Urgent heart failure visit | 0.7 | 0.9 | 0.73 (0.48, 1.11) |
| Secondary Endpoint c | |||
| Hospitalization for heart failure or urgent heart failure visitc | 3.5 | 5.1 | 0.67 (0.55, 0.82) p = 0.0001 |
Figure 5 displays a cumulative events plot of the primary composite endpoint. The INPEFA and placebo event curves separated early and continued to diverge over the study period following randomization.
Figure 5 Primary Composite Endpoint (Total Occurrences of Cardiovascular Death, Hospitalization for Heart Failure, and Urgent Heart Failure Visit) Over Time in the SCORED Study
Primary composite endpoint was based on investigator-reported events in all randomized patients, analyzed according to the treatment group allocated by randomization.
The results of the primary composite endpoint were generally consistent across prespecified subgroups, including history of cardiovascular disease, history of heart failure, and screening LVEF < 50 or ≥ 50% (Figure 6).
Figure 6 Treatment Effect for Primary Composite Endpoint (Total Occurrences of Cardiovascular Death, Hospitalization for Heart Failure, and Urgent Heart Failure Visit) Subgroup Analysis (SCORED Study)
CVD = cardiovascular disease; eGFR = estimated glomerular filtration rate; LVEF = left ventricular ejection fraction; HF = heart failure; PY = patient-years
Primary composite endpoint was based on investigator-reported events in all randomized patients, analyzed according to the treatment group allocated by randomization.
Note: The figure above presents effects in various subgroups all of which are baseline characteristics and all of which were pre-specified. The 95% confidence limits that are shown do not take into account how many comparisons were made, nor do they reflect the effect of a particular factor after adjustment for all other factors. Apparent homogeneity or heterogeneity among groups should not be over-interpreted.
8.4 Pediatric Use
The safety and effectiveness of INPEFA in pediatric patients under 18 years of age have not been established.
8.5 Geriatric Use
No INPEFA dosage change is recommended based on age.
In the SOLOIST study, a total of 241 (40%) patients treated with INPEFA were between 65 and < 75 years of age, and 174 (29%) were ≥ 75 years of age. In the SCORED study, a total of 2,470 (47%) patients treated with INPEFA were between 65 and < 75 years of age, and 1,240 (23%) were ≥ 75 years of age.
No overall differences in efficacy were detected between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Elderly patients may be at increased risk for volume depletion adverse reactions, including hypotension. In patients ≥ 65 years of age, a higher proportion of patients treated with INPEFA had adverse reactions of volume depletion [see Warnings and Precautions (5.2) and Adverse Reactions (6.1)].
14.1 Soloist Study (14.1 SOLOIST Study)
The SOLOIST (Effects of Sotagliflozin on Clinical Outcomes in Hemodynamically Stable Patients with Type 2 Diabetes Post Worsening Heart Failure) study (NCT03521934) was a randomized, double-blind, placebo-controlled, parallel-group, multicenter study in patients with type 2 diabetes mellitus who had been admitted to the hospital, a heart failure unit, infusion center, or emergency department for worsening heart failure to determine if INPEFA reduces the risk of total occurrence of cardiovascular death, hospitalization for heart failure, and urgent heart failure visit. Patients were randomized to treatment if they met the following criteria for clinical stability: no need for oxygen therapy, a systolic blood pressure of at least 100 mmHg, no need for intravenous inotropic or vasodilator therapy (excluding nitrates) and transitioned from intravenous to oral diuretic therapy.
Of the 1,222 randomized patients, 608 were randomized to INPEFA and 614 to placebo. Assigned treatment was initiated in the hospital or within a median of 2 days following hospital discharge.
The dose of study drug was to be up-titrated from 200 mg to 400 mg sotagliflozin or matching placebo as soon as 2 weeks or up until 8 months after initiation of treatment. The dose was increased to 400 mg once daily for 336 patients (56%) in the sotagliflozin group and 325 patients (53%) in the placebo group. The median time to up-titration was 16 days. Up-titration was to be performed based on the judgment of the investigator, who considered whether the patient's clinical condition was satisfactory, whether the drug was well tolerated, and whether AEs typical of SGLT2 inhibitors had occurred, such as those associated with volume depletion.
At baseline, median age was 70 years, 34% were female, 93% were White and 4% were Black or African American. Median A1C was 7.1%, median body mass index (BMI) was 31 kg/m2, and median eGFR was 50 mL/min/1.73 m2. Median left ventricular ejection fraction (LVEF) was 35% (79% with LVEF < 50%), median N-terminal pro B-type natriuretic peptide (NT-proBNP) was 1806 pg/mL, and the median Kansas City Cardiomyopathy Questionnaire-12 (KCCQ-12) score was 41.
At baseline, 86% were treated with at least one antihyperglycemic medication, including 52% with a biguanide, 36% with insulin, 19% with a sulfonylurea, 16% with a dipeptidyl peptidase-4 (DPP4) inhibitor, and 3% with a glucagon-like peptide 1 (GLP-1) receptor agonist. At baseline, 91% were treated with inhibitors of the renin-angiotensin-aldosterone system, 92% with a beta blocker, 95% with a loop diuretic, and 10% with another diuretic.
INPEFA was superior to placebo in reducing the risk of the primary composite endpoint (Hazard Ratio [HR] 0.67 [95% confidence interval (CI) 0.53, 0.85]; p = 0.001). (Table 2).
| Primary Endpoint a | Event Rates per 100 Patient-years |
Hazard Ratio
(95% CI) |
|
|---|---|---|---|
|
INPEFA
N = 608 |
Placebo
N = 614 |
||
|
a Based on investigator-reported events in all randomized patients, analyzed according to the treatment group allocated by randomization. |
|||
|
b Predefined primary endpoint. |
|||
|
c Predefined secondary endpoint and tested with multiplicity control. |
|||
|
d Time-to-event analysis was performed; event rates are percentages of patients with events. |
|||
| Total occurrence of cardiovascular death, hospitalization for heart failure, and urgent heart failure visitb | 51.3 | 76.4 | 0.67 (0.53, 0.85) p = 0.001 |
| Primary Endpoint Components | |||
| Cardiovascular deathc,d | 8.4 | 9.4 | 0.84 (0.58, 1.23) |
| Hospitalization for heart failure | 33.7 | 51.9 | 0.65 (0.49, 0.87) |
| Urgent heart failure visit | 6.9 | 12.1 | 0.60 (0.34, 1.06) |
| Secondary Endpoint c | |||
| Hospitalization for heart failure and urgent heart failure visitc | 40.6 | 63.9 | 0.64 (0.50, 0.84) p = 0.0009 |
Figure 3 displays a cumulative events plot of the primary composite endpoint. The INPEFA and placebo event curves diverged early and remained separated over the study period.
Figure 3 Primary Composite Endpoint (Total Occurrences of Cardiovascular Death, Hospitalization for Heart Failure, and Urgent Heart Failure Visit) Over Time in the SOLOIST Study
Primary composite endpoint was based on investigator-reported events in all randomized patients, analyzed according to the treatment group allocated by randomization.
The results of the primary composite endpoint were generally consistent across prespecified subgroups (Figure 4), including screening LVEF < 50 or ≥ 50% and timing of first dose (post-discharge versus prior to discharge).
Figure 4 Treatment Effect for Primary Composite Endpoint (Total Occurrences of Cardiovascular Death, Hospitalization for Heart Failure, and Urgent Heart Failure Visit) Subgroup Analysis (SOLOIST Study)
HF = heart failure; LVEF = left ventricular ejection fraction; MRA-mineralocorticoid receptor antagonist; NT-proBNP = N-terminal pro B-type natriuretic peptide; PY = patient-years
Primary composite endpoint was based on investigator-reported events in all randomized patients, analyzed according to the treatment group allocated by randomization.
Discharge may have been from hospital or urgent treatment facility where urgent heart failure visit occurred.
Note: The figure above presents effects in various subgroups all of which are baseline characteristics and all of which were pre-specified. The 95% confidence limits that are shown do not take into account how many comparisons were made, nor do they reflect the effect of a particular factor after adjustment for all other factors. Apparent homogeneity or heterogeneity among groups should not be over-interpreted.
4 Contraindications (4 CONTRAINDICATIONS)
INPEFA is contraindicated in patients with a history of serious hypersensitivity reaction to INPEFA.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following important adverse reactions are described elsewhere in the labeling:
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis [see Warnings and Precautions (5.1)]
- Volume Depletion [see Warnings and Precautions (5.2)]
- Urosepsis and Pyelonephritis [see Warnings and Precautions (5.3)]
- Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues [see Warnings and Precautions (5.4)]
- Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) [see Warnings and Precautions (5.5)]
- Genital Mycotic Infections [see Warnings and Precautions (5.6)]
7 Drug Interactions (7 DRUG INTERACTIONS)
5.2 Volume Depletion
INPEFA can cause intravascular volume depletion which may sometimes manifest as symptomatic hypotension or acute transient changes in creatinine. There have been postmarketing reports of acute kidney injury, some requiring hospitalization and dialysis, in patients with type 2 diabetes mellitus receiving SGLT2 inhibitors. Patients with impaired renal function (eGFR < 60 mL/min/1.73 m2), elderly patients, or patients on loop diuretics may be at increased risk for volume depletion or hypotension [see Adverse Reactions (6.1) and Use in Specific Populations (8.5, 8.6)]. Before initiating INPEFA in patients with one or more of these characteristics, assess volume status and renal function. Monitor for signs and symptoms of hypotension, and renal function after initiating therapy.
8.6 Renal Impairment
INPEFA was evaluated in 5,292 patients with chronic kidney disease (eGFR 25 to 60 mL/min/1.73 m2) in the SCORED study and in 426 patients with heart failure with eGFR < 60 mL/min/1.73 m2 in the SOLOIST study. The safety profile of INPEFA across eGFR subgroups in these studies was consistent with the known safety profile. There was an increase in volume-related adverse events (e.g., hypotension, dizziness) in patients with eGFR < 30 mL/min/1.73 m2 relative to the overall safety population [see Warnings and Precautions (5.2) and Adverse Reactions (6.1)].
Efficacy and safety studies with INPEFA did not enroll patients with an eGFR less than 25 mL/min/1.73 m2 or on dialysis. After starting therapy in these studies, patients were discontinued if eGFR fell below 15 mL/min/1.73 m2 or were initiated on chronic dialysis.
12.3 Pharmacokinetics
Plasma Cmax and AUC of sotagliflozin increased in a dose-proportional manner in the therapeutic dose range of 200 mg to 400 mg once daily. Accumulation of sotagliflozin was observed with an approximate 50-100% increase in both Cmax and area under the concentration-time curve from 0 to 24 hours (AUC0-24h) at steady state verses the first day of dosing.
2.2 Recommended Dosage
The recommended starting dose of INPEFA is 200 mg orally once daily not more than one hour before the first meal of the day.
Up-titrate after at least 2 weeks to 400 mg orally once daily as tolerated [see Clinical Studies (14)]. Down-titrate to 200 mg as necessary [see Adverse Reactions (6.1), Warnings and Precautions (5) and Use in Specific Populations (8.6)].
Swallow tablets whole. Do not cut, crush, or chew tablets.
If a dose of INPEFA is missed by more than 6 hours, take the next dose as prescribed the next day.
8.7 Hepatic Impairment
In a clinical pharmacology study in patients with hepatic impairment, the exposure in mild hepatic impairment was not increased, but was approximately 3-fold as high in moderate and approximately 6-fold as high in severely hepatic-impaired subjects compared to subjects with normal hepatic function [see Clinical Pharmacology (12.3)].
No dosage adjustment is necessary in patients with mild hepatic impairment.
The safety and efficacy of INPEFA have not been established in patients with moderate or severe hepatic impairment [see Clinical Pharmacology (12.3)]. INPEFA is not recommended in patients with moderate or severe hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
INPEFA is indicated to reduce the risk of cardiovascular death, hospitalization for heart failure, and urgent heart failure visit in adults with:
- heart failure or
- type 2 diabetes mellitus, chronic kidney disease, and other cardiovascular risk factors
12.1 Mechanism of Action
Sotagliflozin is an inhibitor of SGLT2 and SGLT1. Inhibiting SGLT2 reduces renal reabsorption of glucose and sodium which may influence several physiological functions such as lowering both pre-and afterload of the heart and downregulating sympathetic activity. Inhibiting SGLT1 reduces intestinal absorption of glucose and sodium which likely contributes to diarrhea. The mechanism for sotagliflozin's cardiovascular benefits has not been established.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients with type 1 diabetes mellitus and consider ketone monitoring in others at risk for ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue INPEFA if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting. (5.1)
- Volume Depletion: Before initiating, correct volume status. Monitor for signs and symptoms of hypotension during therapy. (5.2)
- Urosepsis and Pyelonephritis: Monitor for signs and symptoms during therapy and treat promptly. (5.3)
- Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues: Lower dose of insulin or insulin secretagogue may be required. (5.4)
- Necrotizing Fasciitis of the Perineum (Fournier's Gangrene): Monitor for pain, tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. Discontinue INPEFA and treat urgently. (5.5)
- Genital Mycotic Infections: Monitor and treat as appropriate. (5.6)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Correct volume status before starting INPEFA at 200 mg daily and titrate to 400 mg as tolerated. (2.2) In patients with decompensated heart failure, begin dosing when patients are hemodynamically stable. (2.1)
Withhold INPEFA at least 3 days, if possible, prior to major surgery or procedures associated with prolonged fasting. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
INPEFA 200 mg printed tablet is an oval, blue, film-coated tablet imprinted in black ink with “LX200” on one side.
INPEFA 200 mg debossed tablet is an oval, blue, film-coated tablet debossed with “LEX” on one side and “200” on the other side.
INPEFA 400 mg debossed tablet is an oval, yellow, film-coated tablet debossed with “LEX” on one side and “400” on the other side.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: Advise females of the potential risk to a fetus especially during the second and third trimesters. (8.1)
Lactation: INPEFA is not recommended when breastfeeding. (8.2)
Geriatrics: Higher incidence of adverse reactions related to volume depletion. (5.2, 8.5)
Renal Impairment: Higher incidence of adverse reactions related to volume depletion. (5.2, 8.6)
5.6 Genital Mycotic Infections
INPEFA increases the risk of genital mycotic infections. Patients with a history of genital mycotic infections were more likely to develop genital mycotic infections [see Adverse Reactions (6.1)]. Monitor and treat appropriately.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In the phase 3 (SOLOIST [see Clinical Studies (14.1)] and SCORED [see Clinical Studies (14.2)]) placebo-controlled trials, 5,896 subjects received INPEFA.
In the SOLOIST study, 336 patients (56%) reached the 400 mg dose. In the SCORED study, 3,934 patients (74%) reached the 400 mg dose.
In the SOLOIST study, 5.6% of patients in the INPEFA group and 5.4% of patients in the placebo group discontinued therapy due to adverse events (AEs). In the SCORED study, 5.0% of patients in the INPEFA group and 4.5% of patients in the placebo group discontinued therapy due to AEs.
| Adverse Reaction |
SOLOIST
N = 1,216 |
SCORED
N = 10,577 |
||
|
Placebo (%)
N = 611 |
INPEFA (%)
N = 605 |
Placebo (%)
N = 5,286 |
INPEFA (%)
N = 5,291 |
|
| Urinary tract infection | 7.2 | 8.6 | 11.0 | 11.5 |
| Volume depletion | 8.8 | 9.3 | 4.0 | 5.2 |
| Diarrhea | 4.1 | 6.9 | 6.0 | 8.4 |
| Hypoglycemia | 2.8 | 4.3 | 7.9 | 7.7 |
| Dizziness | 2.5 | 2.6 | 2.8 | 3.3 |
| Genital mycotic infection | 0.2 | 0.8 | 0.9 | 2.4 |
5.7 Positive Urine Glucose Test
Monitoring glucose levels with urine glucose tests is not recommended as SGLT2 inhibition increases urinary glucose excretion and will lead to positive urine glucose tests. Use alternative methods to monitor glucose levels.
5.3 Urosepsis and Pyelonephritis
Treatment with SGLT2 inhibitors, including INPEFA, increases the risk for urinary tract infections. Serious urinary tract infections including urosepsis and pyelonephritis requiring hospitalization have been reported. Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated [see Adverse Reactions (6.1)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.1 Prior to Initiation of Inpefa (2.1 Prior to Initiation of INPEFA)
Assess volume status and, if necessary, correct volume depletion prior to initiation of INPEFA [see Warnings and Precautions (5.2) and Use in Specific Populations (8.5, 8.6)].
Assess renal function prior to initiation of INPEFA and then as clinically indicated [see Warnings and Precautions (5.2)].
For patients with decompensated heart failure, dosing may begin as soon as the patient is hemodynamically stable, including during hospitalization or urgent outpatient treatment or immediately upon discharge.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
INPEFA tablets are oval and film-coated.
| Printed Tablets | Debossed Tablets | |||||
| Strength (mg) | Color | Printing | Bottle/30 | Deboss | Bottle/30 |
Blister/30 (3 x 10) |
| 200 | Blue | LX200 on one side |
NDC 70183-220-30 | LEX on one side, 200 on other side | NDC 70183-221-30 | NDC 70183-221-31 |
| 400 | Yellow | -- | -- | LEX on one side, 400 on other side | NDC 70183-241-30 | -- |
2.3 Temporary Interruption for Surgery
5.8 Interference With 1,5 Anhydroglucitol (1,5 Ag) Assay (5.8 Interference with 1,5-anhydroglucitol (1,5-AG) Assay)
Monitoring glucose levels with 1,5-AG assay is not recommended as measurements of 1,5-AG are unreliable in assessing glucose levels in patients taking SGLT2 inhibitors. Use alternative methods to monitor glucose levels.
7.2 Uridine 5' Diphospho Glucuronosyltransferase (ugt) Inducer (7.2 Uridine 5'-diphospho-glucuronosyltransferase (UGT) Inducer)
Glucuronidation by UGT1A9, to form the 3-O-glucuronide, was identified as a major metabolic pathway for sotagliflozin. The coadministration of rifampicin, an inducer of UGTs, with a single dose of 400 mg sotagliflozin resulted in a decrease in the exposure to sotagliflozin. This decrease in exposure to sotagliflozin may decrease efficacy [see Clinical Pharmacology (12.3)].
5.5 Necrotizing Fasciitis of the Perineum (fournier's Gangrene) (5.5 Necrotizing Fasciitis of the Perineum (Fournier's Gangrene))
Reports of necrotizing fasciitis of the perineum (Fournier's Gangrene), a rare but serious and life-threatening necrotizing infection requiring urgent surgical intervention, have been identified in postmarketing surveillance in patients with diabetes mellitus receiving SGLT2 inhibitors. Cases have been reported in both females and males. Serious outcomes have included hospitalization, multiple surgeries, and death.
Patients treated with INPEFA presenting with pain, tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, should be assessed for necrotizing fasciitis. If suspected, start treatment immediately with broad-spectrum antibiotics and, if necessary, surgical debridement. Discontinue INPEFA, closely monitor blood glucose levels, and provide appropriate alternative therapy for heart failure.
5.4 Hypoglycemia With Concomitant Use With Insulin and Insulin Secretagogues (5.4 Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues)
Insulin and insulin secretagogues are known to cause hypoglycemia. INPEFA may increase the risk of hypoglycemia when combined with insulin or an insulin secretagogue [see Adverse Reactions (6.1)]. Therefore, a lower dose of insulin or insulin secretagogue may be required to minimize the risk of hypoglycemia when these agents are used in combination with INPEFA.
5.1 Diabetic Ketoacidosis in Patients With Type 1 Diabetes Mellitus and Other Ketoacidosis (5.1 Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis)
In patients with type 1 diabetes mellitus, INPEFA significantly increases the risk of diabetic ketoacidosis, a life-threatening event, beyond the background rate. In placebo-controlled trials of patients with type 1 diabetes mellitus, the risk of ketoacidosis was markedly increased in patients who received sodium glucose cotransporter 2 (SGLT2) inhibitors compared to patients who received placebo; this risk may be greater with higher doses of INPEFA. INPEFA is not indicated for glycemic control.
Type 2 diabetes mellitus and pancreatic disorders (e.g., history of pancreatitis or pancreatic surgery) are also risk factors for ketoacidosis. There have been postmarketing reports of fatal events of ketoacidosis in patients with type 2 diabetes using SGLT2 inhibitors.
Precipitating conditions for diabetic ketoacidosis or other ketoacidosis include acute febrile illness, reduced caloric intake, ketogenic diet, surgery, insulin dose reduction, volume depletion, and alcohol abuse.
Signs and symptoms are consistent with dehydration and severe metabolic acidosis and include nausea, vomiting, abdominal pain, generalized malaise, and shortness of breath. Blood glucose levels at presentation may be below those typically expected for diabetic ketoacidosis (e.g., less than 250 mg/dL). Ketoacidosis and glucosuria may persist longer than typically expected. Urinary glucose excretion persists for 3 days after discontinuing INPEFA [see Clinical Pharmacology (12.3)]; however, there have been postmarketing reports of ketoacidosis and glucosuria lasting greater than 6 days and some up to 2 weeks after discontinuation of SGLT2 inhibitors.
Consider ketone monitoring in patients with type 1 diabetes mellitus and consider ketone monitoring in others at risk for ketoacidosis if indicated by the clinical situation. Assess for ketoacidosis regardless of presenting blood glucose levels in patients who present with signs and symptoms consistent with severe metabolic acidosis. If ketoacidosis is suspected, discontinue INPEFA, promptly evaluate, and treat ketoacidosis, if confirmed. Monitor patients for resolution of ketoacidosis before restarting INPEFA.
Withhold INPEFA, if possible, in temporary clinical situations that could predispose patients to ketoacidosis. Resume INPEFA when the patient is clinically stable and has resumed oral intake [see Dosage and Administration (2.3)].
Educate all patients on the signs and symptoms of ketoacidosis and instruct patients to discontinue INPEFA and seek medical attention immediately if signs and symptoms occur.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:37.239562 · Updated: 2026-03-14T22:44:14.787449